1. Introduction
The trend to miniaturize electronic components leads to an increased power density and thus to new requirements of thermal management. Especially in the field of power electronics, cooling plays a key role. Currently, the power density dissipation of semiconductors is about 100 W/cm
. Modern wide bandgap semiconductors such as SiC or GaN will raise the heat flux density above 1000 W/cm
[
1,
2]. These large heat fluxes are exceptionally well handled by two-phase cooling systems.
An established two-phase cooling system is the conventional heat pipe. They are considered to be state of the art in electronic devices and are available in different designs and shapes [
3]. However, the cooling potential of a conventional heat pipe is limited by the mass flow of the wick. To handle higher heat fluxes, Akachi [
4] developed and patented the first oscillating heat pipe in 1990. An oscillating heat pipe (OHP), also called pulsating heat pipe (PHP), is a passive two-phase system for heat spreading. Besides the tubular OHP shape from Ackachi, there are also flat plate OHPs [
5]. Currently, various OHP shapes are available. Czajkowski et al. [
6] presented a flower shape oscillating heat pipe for rotating systems. Also multi-layers three-dimensional OHPs have been experimentally investigated by Qu et al. [
7]. Regardless of the shape, all OHPs have the same working principle. The first step to put an OHP into operation is to evacuate the channels. The OHP is then partially filled with a working liquid so that a vapor and a liquid phase is formed. After the filling process, both phases are in a thermodynamic equilibrium. A temperature difference between the evaporator and condenser leads to a thermodynamic disequilibrium. Due to the phase change and the pressure difference, the fluid starts to oscillate [
8]. The OHP operates reliably as long as there is a continuous oscillating plug flow.
To describe the proper function of an OHP, Drolen and Smoot [
9] developed a model that is based on the following parameters as operating limits:
Channel size
Vapor inertia limit
Heat flux limit
Sonic limit
Viscous limit
The OHP can only operate when the vapor bubbles in the micro channel are separated by the liquid plugs. The fluid inside the channel has to consist of altering liquid and vapor parts. This formation depends on the surface tension. The smaller the diameter, the more dominates the surface tension, and the gravitational force can be neglected. Depending on the thermophysical properties of the fluid, there is a different threshold value for a micro channel diameter [
10,
11,
12]. Regarding the maximum channel size, there is a vast body of work in the literature. In the area of PHPs, there are various threshold values which have been established experimentally. The threshold can be described by different numbers. The
number [
13] is a dimensionless number to define the micro channel threshold and is given by Equation (
1).
Here,
d is the diameter of the channel,
g is the gravitational force,
represents the surface tension,
is the liquid density and
the vapor density. The dimensionless
number describes the ratio of hydrostatic and capillary pressure. The
number [
2,
14] is defined by a unit of length and describes the threshold value above which the surface tension dominates. If the diameter is below the
number, the gravitational force can be neglected [
15]. The
number is given by Equation (
2).
The dimensionless Eötvös number
is very similar to the
number. Depending on the researcher, there are different definitions of the Eötvös number [
2,
14,
16,
17]. Khandekar and Groll [
11] define the the
number by Equation (
3).
Table 1 shows different thresholds which are standardized by the
number [
2]. All definitions have in common that they include the density
and the surface tension
. Given that both parameters are dependent on the temperature, the diameter is also a function of temperature
. For higher temperatures, the maximum diameter for micro channel flow becomes smaller, since the surface tension decreases at higher temperatures.
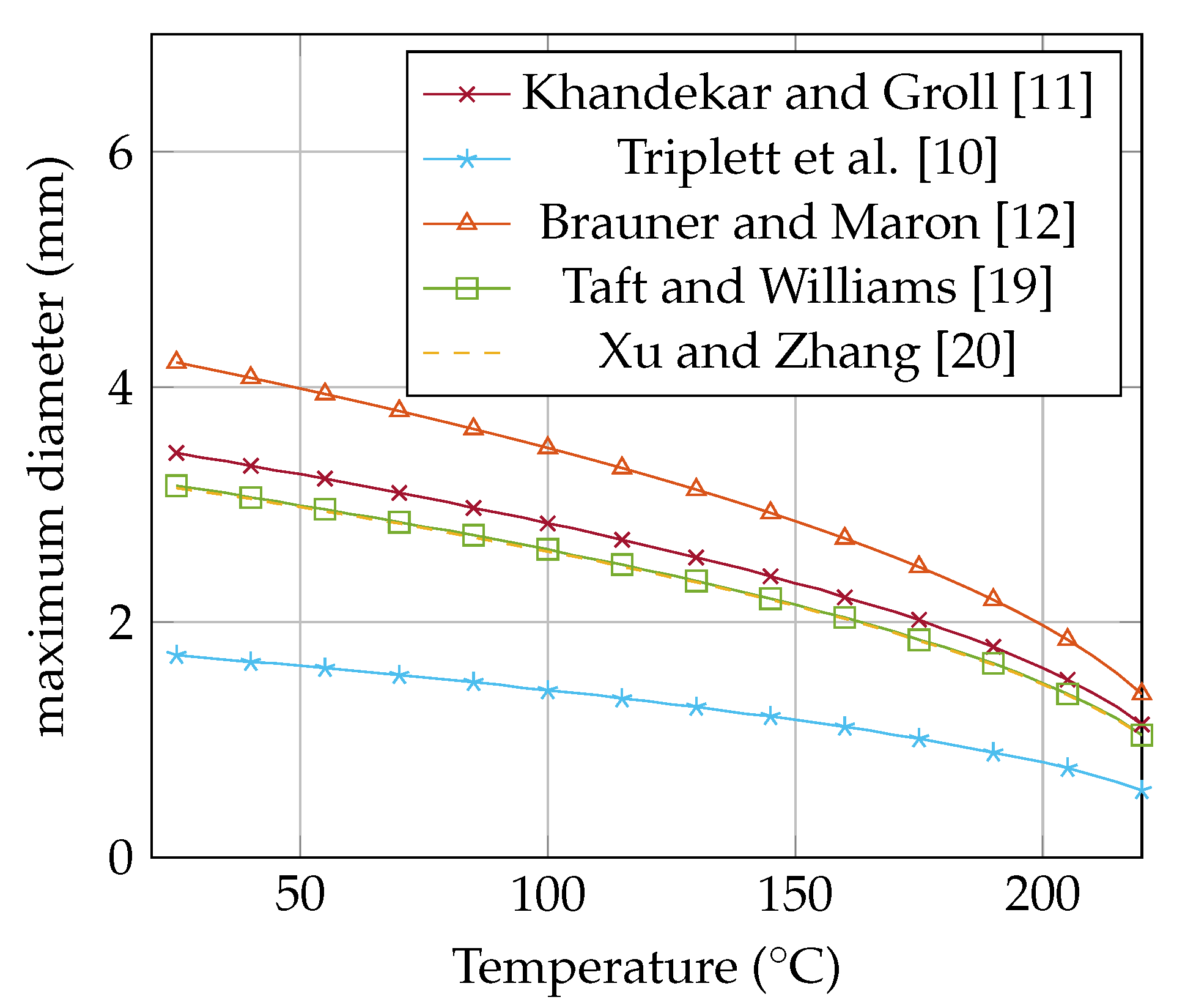
Figure 1 shows the maximum diameter in dependence on the temperature of acetone. The values are calculated with the equations of
Table 1 and the thermodynamic fluid properties from REFPROP [
18]. The calculated diameter is the maximum permissible diameter for a plug flow.
All threshold values tend towards zero, since the surface tension at the critical point becomes zero, which becomes apparent when the two-phases disappear. Taft and Williams [
19] set the Bo number of 0.85 for the maximum radius. That leads to a normalized definition of
. Also, Ma [
5] is using this definition to predict the maximum radius.
Table 1.
Comparison of the threshold diameter for water at 25 .
Table 1.
Comparison of the threshold diameter for water at 25 .
| Name | Equation | Normalized | d (mm) |
|---|
| Khandekar and Groll [11] | | | 3.44 |
| Triplett et al. [10] | | | 1.72 |
| Brauner and Maron [12] | | | 4.21 |
| Xu and Zhang [20] | | | 3.14 |
| Taft and Williams [19] | | | 3.16 |
The other limits, namely vapor inertia limit, heat flux limit and viscous limit have in common that the evaporation fraction plays a major role and dominates the limits. Drolen and Smoot [
9] derive equations for these specific limits. All these limits lead to a dryout of the OHP. In case of dryout, the two-phase flow changes from slug to churn flow, thus the heat transfer deteriorates rapidly. Bastakoti et al. [
21] states in a literature review that the dryout occurs especially when there is a low filling ratio and a high heat input. The filling ratio (FR) plays a key role for OHPs. It is not only decisive for reliable operation of OHPs, but also for the start up and the dryout limit. Zhang and Faghri [
8] showed that an OHP operates reliably with a filling ratio of 20% to 80%. Also Yin et al. [
22] derived a formulation for the theoretical maximum filling ratio of 80%. Liu et al. [
6] showed that each fluid has an individual optimal filling ratio for the start up process.
Shi and Pan [
23] investigated the influence of the filling ratio during the start up process. Therefore, they had an experimental setup for a flat plate OHP with 40 channels. The 1 × 1
channels were charged with acetone. They measured the thermal resistance for various filling ratios and increased the heat input from 23 W to 210 W. With a filling ratio of 50%, the dryout limit was at 80 W heat input. While a filling ratio of 70% leads to a dryout at about 110 W. The thermal performance improves with increased heat input until the OHP dries out and the heat transport abruptly subsides.
Winkler et al. [
24] tested a 20 channel OHP made of copper with a 1.5 × 1.5
cross section filled with acetone. They measured a constant filling ratio of 50% and increased the heat input from 0 W to 180 W and investigated the influence of the condenser sizes. The experiment with a 1.5 × 1.5
OHP reaches the dryout limit above 125 W heat input.
As shown above, the optimal OHP design strongly depends on the filling ratio [
5,
8,
22]. On the one hand, the filling ratio has an impact to the start up behavior, on the other hand it influences the thermal resistance [
23]. The operating limits are dominated by the dryout limit, which is also a function of the filling ratio [
9]. Therefore, the filling ratio plays a key role in the optimization of the OHP design. At the optimal operation point, there is a maximum heat transfer and a minimal thermal resistance. Due to different heat inputs, geometries and working fluids in previous studies [
8,
23,
24] it is not possible to predict the dryout limit and to find an optimal filling ratio. When a dryout takes place, the flow pattern changes to annular flow and the heat transfer deteriorates rapidly. There are different approaches to explain the dryout phenomena itself [
5,
25,
26]. However there is no empirical value that indicates the dryout limit of OHPs. With the present study, we aim to predict the dryout limit in dependence of the filling ratio. Our theoretical approach is based on the change of the filling ratio as a function of the operating temperature. This effect has already been published in [
5,
9], but it is assumed in all studies that this effect is negligible. Firstly, we derive an equation to calculate the vapor quality
x for OHPs as a function of the operation temperature. Therefore, it is possible to calculate the change of the filling ratio. The vapor quality
x is used to describe the dryout threshold of OHPs. Secondly, the dryout threshold of
x = 0.006 is measured experimentally. As soon as it exceeds this threshold value, the OHP dries out. Finally, this approach also validates the previous studies in the literature with their different boundary conditions and merges the findings into one unified theory.
2. Experimental Setup
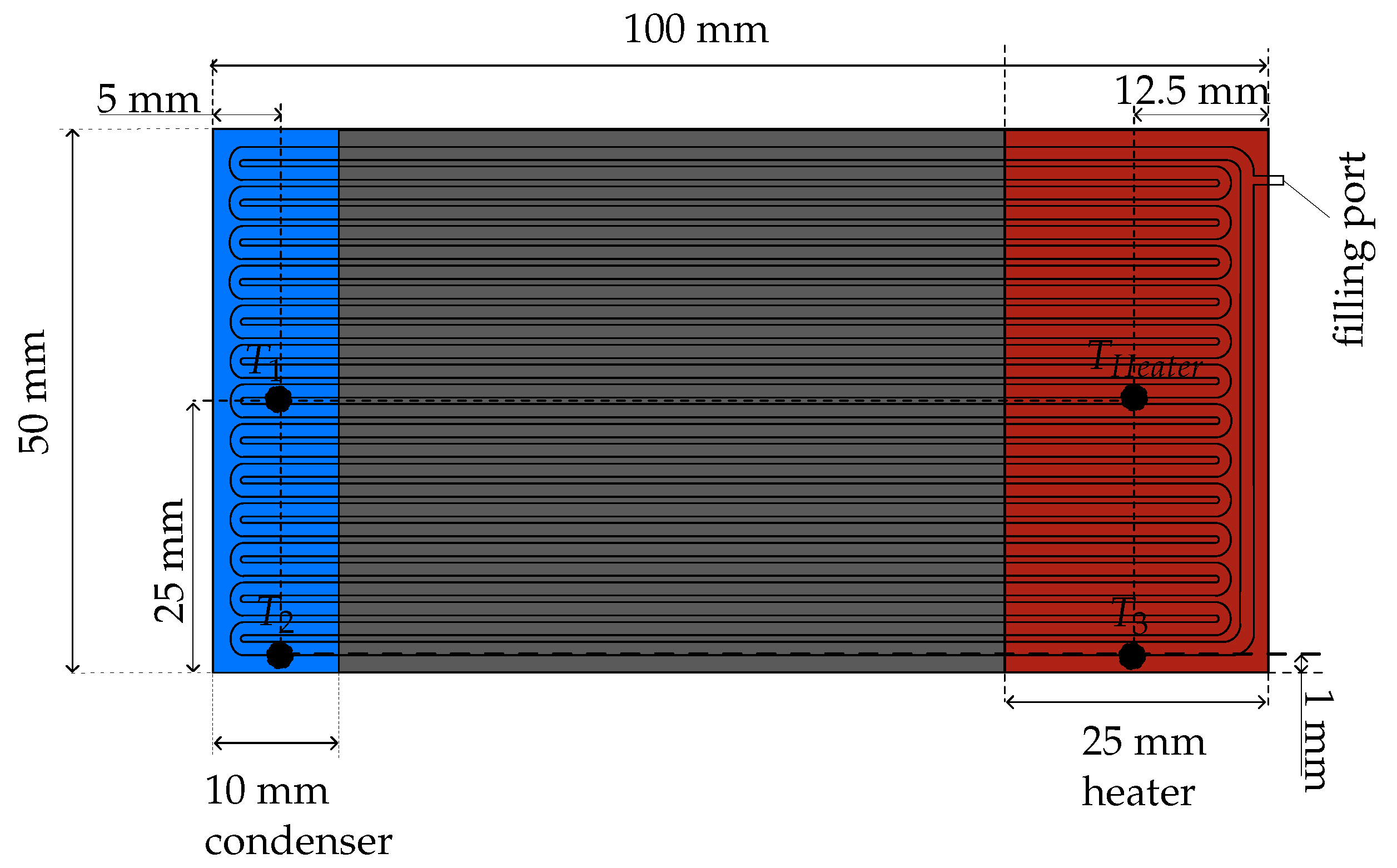
The OHP has an overall volume of 150 × 50 × 2 . It is made of copper and has 26 channels with a cross section of 1 × 1 . The distance between the channels is set to 0.5 . To manufacture the OHP, the channels are first milled into the lower half of the copper plate, then it is brazed to a copper cover. The OHP was manufactured by the Fraunhofer IPM.
Figure 2 shows a schematic representation of the OHP with the meandering channel design. The heater (on the right side in
Figure 2) has the dimensions of 50
× 25
× 2.5
, which results in a heater area of 50 × 25
. The condenser (on the left side in
Figure 2) is realized by a water heat sink made of aluminum and has the dimensions of 50
× 10
× 10
. The contact area between condenser and OHP is 50 × 10
.
The temperature of the heater
is measured by the integrated thermocouple of the heater Ultramic600. Two thermocouples type K (from TC Ltd., London, UK) are used to measure the temperature of the condenser.
is located in the center between the OHP and the heat sink.
is placed on the side of the OHP. Experimental studies have shown that OHPs work reliably with a filling ratio between 50% and 60% [
21,
27]. Therefore, a filling ratio of 53 % was chosen for our experiment. Acetone was used as a working fluid. The fluid was thoroughly degassed prior to filling. The experiment was investigated in horizontal orientation.
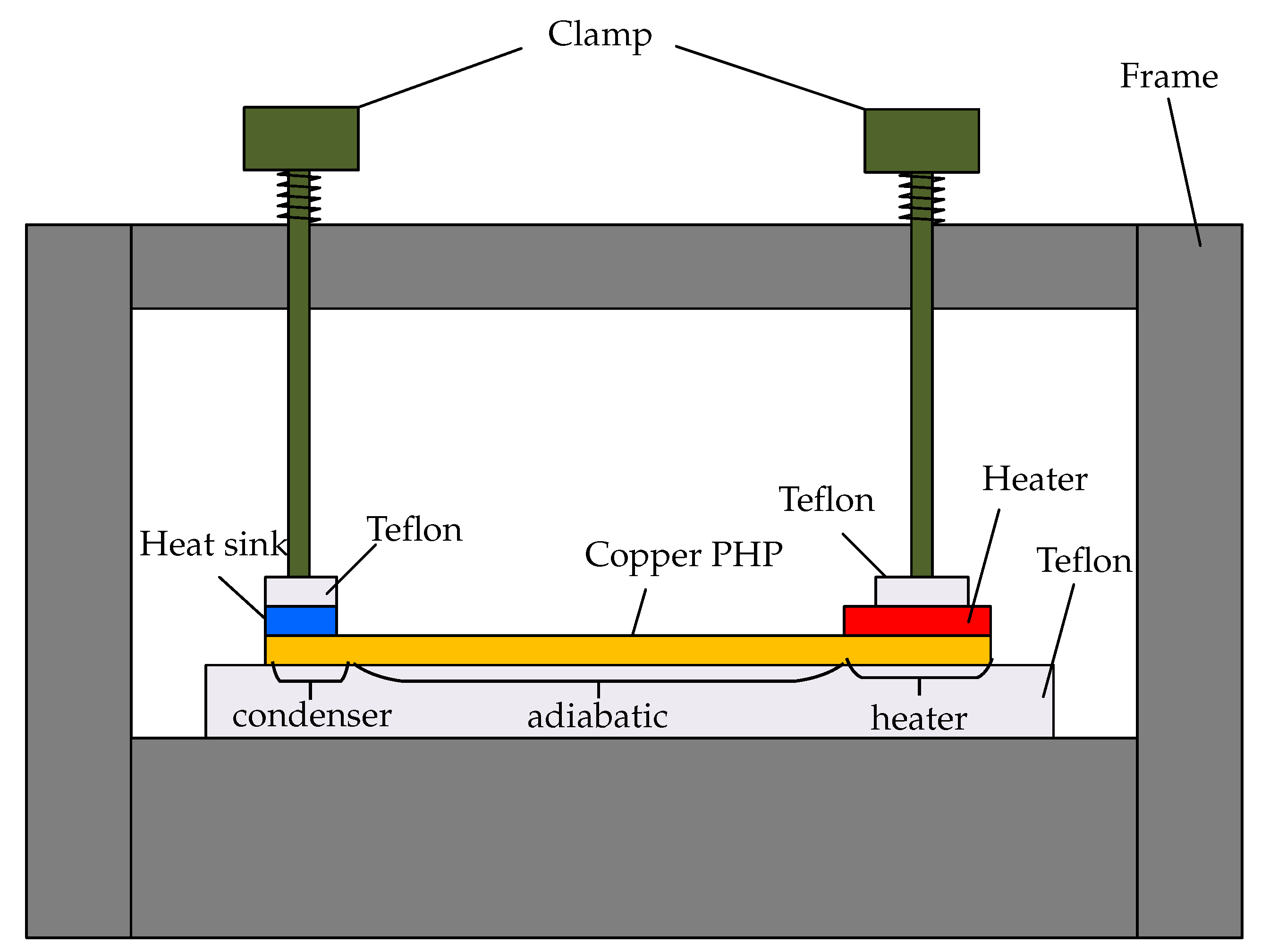
The experimental setup is shown in
Figure 3. It consists of an OHP module with an evaporator, an adiabatic and a condenser section. For the experiment, Watlow ceramic heater Ultramic600 has been used as a heat source, which was supplied with a DC current power supply unit (Xantrex XKW 150-7). The cooling water is provided by a circulating chiller (Lauda Alpha A6). The section between evaporator and condenser is insulated by a Teflon plate from below and by wool from above. Both heater and heat sink are fixed by clamps. Due to the isolation, it is assumed that the heat flux through the OHP is equal to the electrical power (
). The temperature of the condenser
is the average of
and
. The thermal resistance is calculated by Equation (
4):
The accuracy in the measurement is
for the temperature, ±0.1 V for the voltage and ±0.01 A for the current of the power source. At 15 W, there is a temperature difference of 14.2 K (±3 K) between the heater and the condenser. This leads to a measurement accuracy of 21% for the temperature at 15 W. The temperature difference at 90 W is 39.4 K (±3 K), that results in an accuracy of 8%. The measurement accuracy for the power source is 45 V (±0.1 V)
0.34 A (±0.01 A) = 15.3 W (±3.16%) for the lowest power input. At the highest power level there is an accuracy of 119 V (±0.1 V)
0.76 A (±0.01 A) = 90.44 W (±1.40%). Calculating the thermal resistant
R with Equation (
4) results in an accuracy range from 24.3% at 15 W to 9.0% at 90 W.
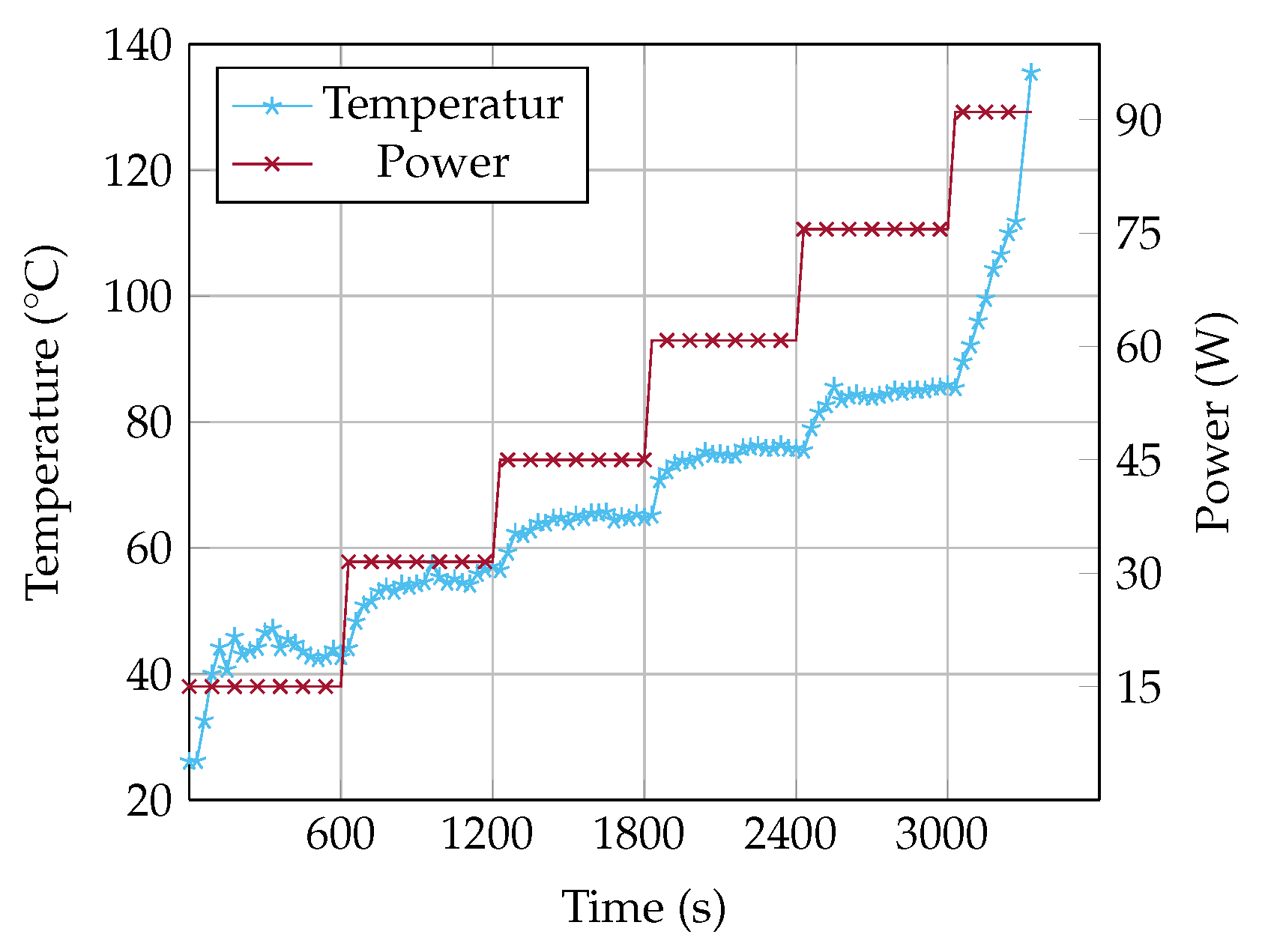
Results of the Measurement
Figure 4 shows the temperature of the heater as a function of increasing heat input. The temperature fluctuation at a heat input of 15 W results from the start up operation condition of the OHP. During the start up, there is no continuous flow, the temperature and thereby also the pressure is increasing until the fluid starts to oscillate. As soon as the fluid oscillates, the temperature and the pressure at the heater decreases and the fluid stops to oscillate. The heat input is not high enough for a continuous oscillation. Above a heat input of 30 W, the fluid oscillates continuously and the OHP operates reliably. Up to 75 W, the temperature rises continuously with each power level. At a heat input of 90 W, the temperature rises from 85
to 140
. This rapid rise in temperature is due to the dryout. As soon as heat can no longer be transferred, the temperature rises rapidly. For safety regulations, the experiment is limited to a maximum temperature of 140
.
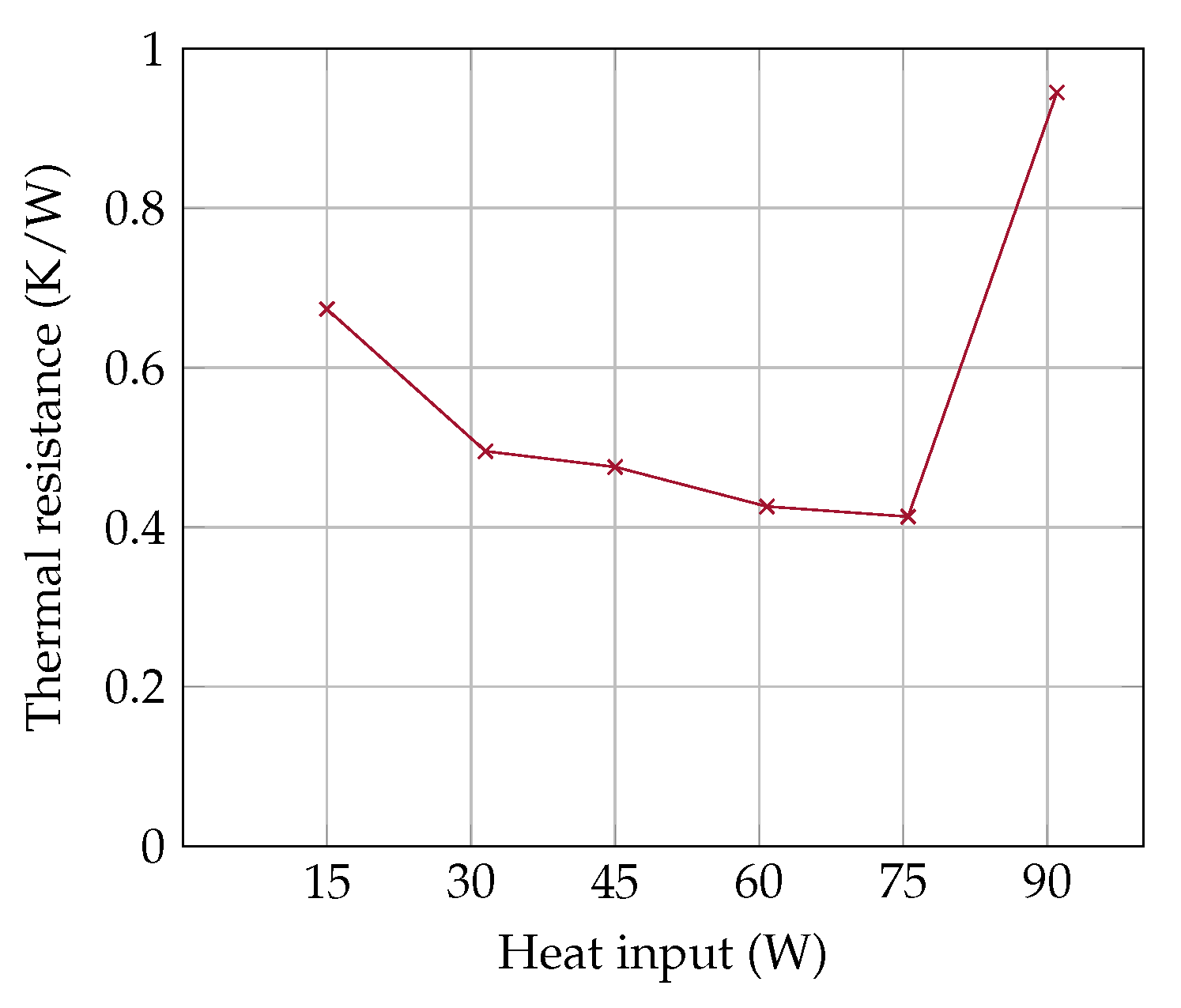
The thermal resistance
of the OHP is shown in
Figure 5. At 15 W, there is no continuous fluid flow, which results in a high thermal resistance of 0.7 (K/W). Between 30 W and 75 W the OHP works reliably and the
improves continuously up to 0.41 (K/W) with increasing heat input. Above 75 W, the OHP dries out and the thermal resistance deteriorates rapidly above 0.9 (K/W).
3. Thermodynamic Considerations on the Working Principle of OHPs
To predict the dryout limit, an equation for the vapor quality is derived in
Section 4. For this derivation, we assume that the filling ratio is a function of the temperature. When an OHP gets filled with acetone at 25
temperature with a filling rate of 35% and operates at a temperature of 175
, the filling ratio changes to 43%. In the following, the relation between the filling ratio and the vapor quality is illustrated in a PT- and PV-diagram.
There are various ways to define the filling ratio of a two-phase system. In the field of OHPs, the filling ratio
is typically defined as the ratio of liquid volume to the total volume. The liquid volume is a function of the temperature. Therefore, Equation (
5) refers to the filling ratio
at the filling temperature (
).
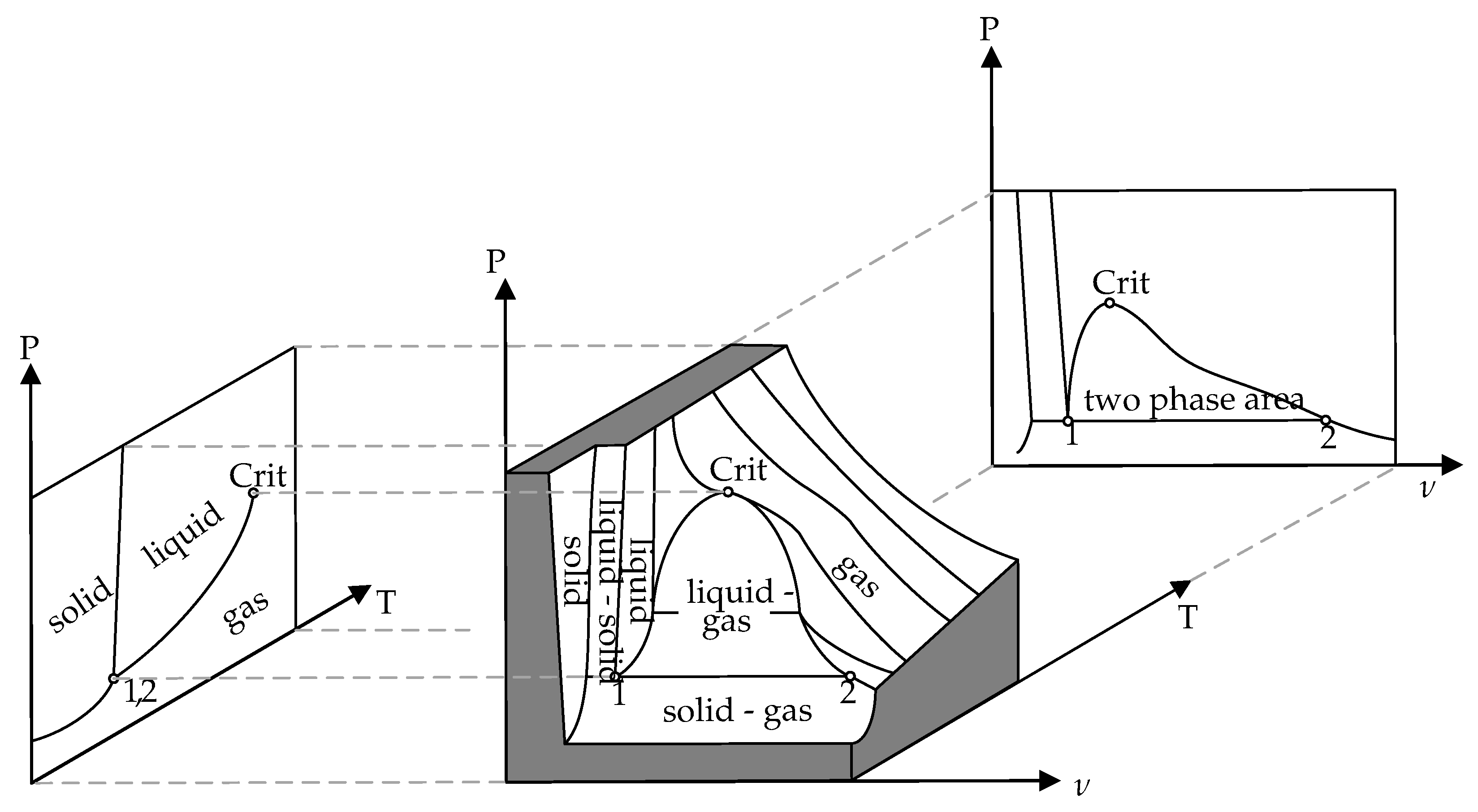
Both phases are in a thermodynamic equilibrium, so the pressure is only a function of the temperature. Every point is on the vapor pressure curve and can be calculated with the relation of Clausius-Clapeyron [
28]. On the left side of
Figure 6, there is a PT-diagram with a vapor pressure curve for a pure fluid. A vapor-liquid system which is in a thermodynamic equilibrium can be located at any point on the vapor pressure curve between the triple point and the critical point. However, the PT-diagram contains no information about the change of the filling ratio, which occurs when the temperature rises. The PV-diagram, on the right side in
Figure 6, has a two-phase area where the vapor quality
x is given. All the information about the change of state is illustrated in the PVT-diagram. The PV- and PT-diagrams are only different planes in the PVT-diagram. For the design of an OHP, it is important to know how the ratio of vapor to liquid is changing during the operation.
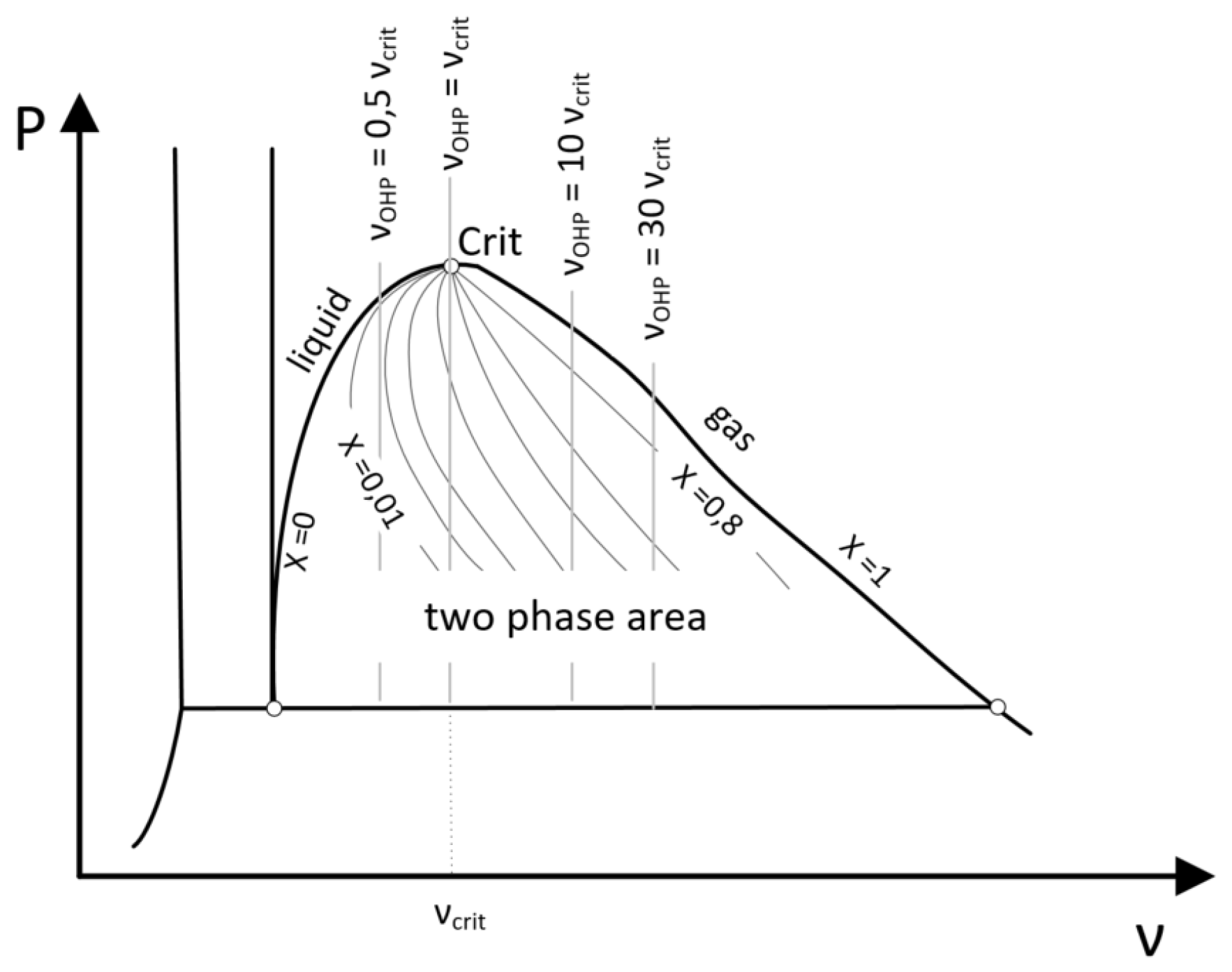
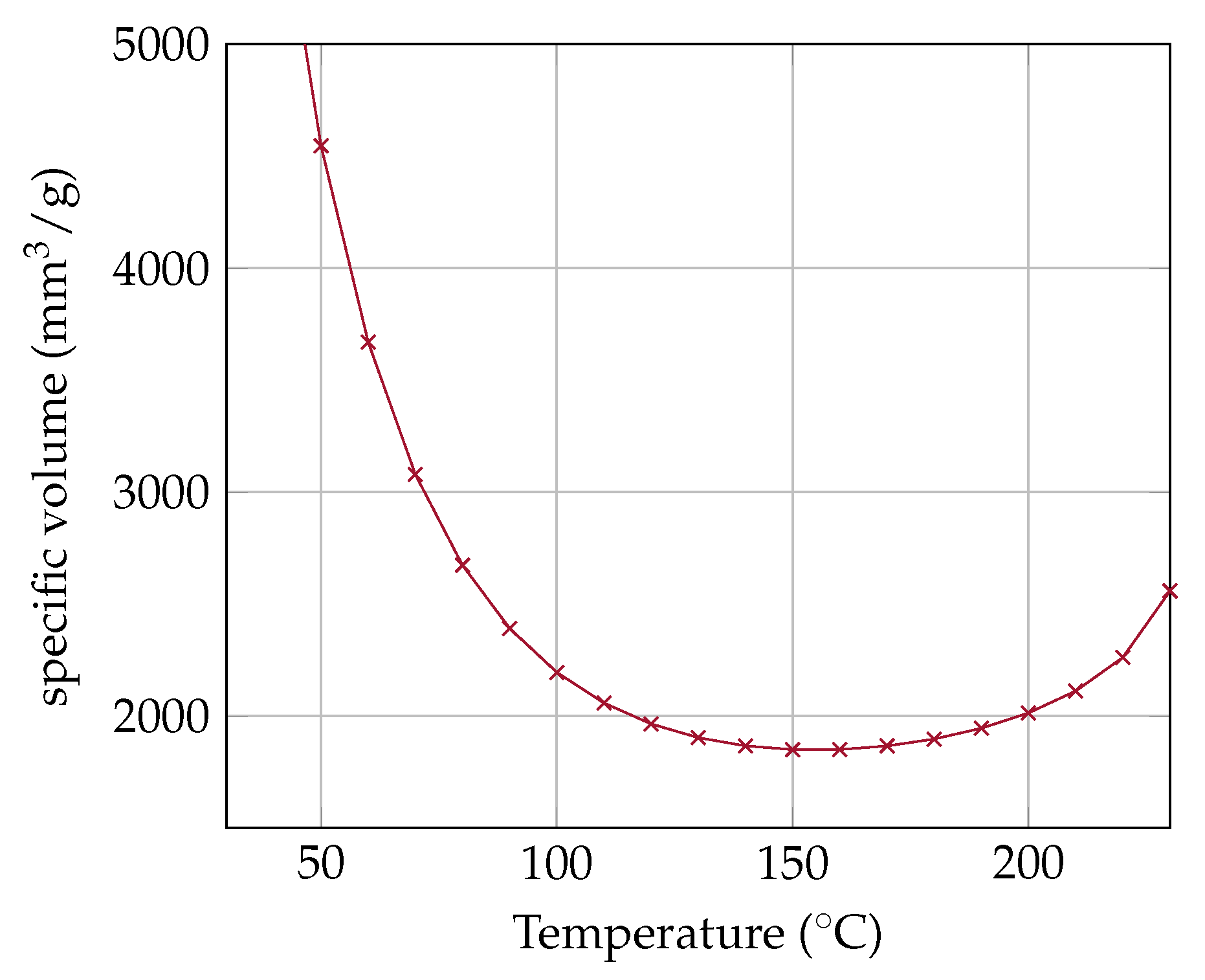
Since the OHP has a closed volume, an isochoric state of change can be assumed for the entire system. The specific volume of the OHP is given by Equation (
6).
Figure 7 illustrates some isochoric states of change in a PV-diagram. The isochoric curve can be described as a ratio of the specific volume of the system to the critical volume of the fluid. In general, the isochoric curves in
Figure 7 can be classified into three categories:
: the entire fluid can become liquid
: there are always two-phases, because the isochoric is equal to the vapor pressure curve
: the entire fluid can become gaseous
In
Section 4, an analytical model is presented to determine the dryout threshold. The approach is based on the influence of vapor quality on the flow pattern. If the vapor quality exceeds a certain limit, the flow pattern changes from slug flow to annular flow and the OHP dries out. This effect can be illustrated by calculating the vapor quality along the specific isochoric curve.
4. Model to Determine the Dryout Threshold
By applying Equation (7), it is possible to calculate the thermodynamic state of the vapor quality
x as a function of the temperature (
T).
The vapor quality
x is the ratio of the vapor mass at saturated temperature
to the total mass
of the fluid. The total mass
is the sum of the vapor
and the liquid mass
. Equation (
8) shows another way to calculate the vapor quality in dependence of the specific volume of the OHP
, the specific liquid volume
and the specific vapor volume
of the working fluid.
Here
is the saturated specific volume of the liquid and
of the vapor phase. The specific volume of the OHP
is the ratio of the total OHP volume
and the fluid mass
, which is expressed by Equation (
9). It shows that the specific volume of the OHP
is independent of the temperature and remains constant after filling the filling process.
When the total mass of the fluid
is known, the vapor quality
x can be calculated by Equation (
8). In many studies, only the filling ratio of the OHP is given. Therefore, the filling ratio must be converted into the specific volume of the OHP
. This is possible by the following steps which lead to Equation (
15) in dependence of the filling temperature (FT). Rearranging Equation (
7) shows a relation between the liquid mass
and the vapor quality
x which is represented by Equation (
10).
Equations (
9) and (
10) lead to (
11).
The liquid volume
is also given by Equation (
12).
Combining Equations (
5), (
12) and (
11) lead to Equation (
13). The filling ratio
is a function of the specific volume
and the vapor quality
x.
Substituting Equation (
8) in (
13) leads to Equation (
14), the filling ratio is solely a function of the specific volume. Since the specific volume at filling temperature is calculated, the temperature (
T) in Equation (
8) is set to the filling temperature (
).
The specific OHP volume can be calculated by rearranging Equation (
14) to Equation (
15).
To calculate the vapor quality in dependence of the specific volume and the filling ratio, it is necessary to consider the reference temperature of Equations (
8) and (
13). The effect on an increasing and decreasing vapor quality is only traceable along the isochoric curve. Hence, the specific volume
in Equation (
15) is constant and refers to the filling temperature (
). However, the values in Equation (
8) refer to the operation temperature (
T). Considering the reference temperatures and combining (
8) and (
15) leads to Equation (
16).
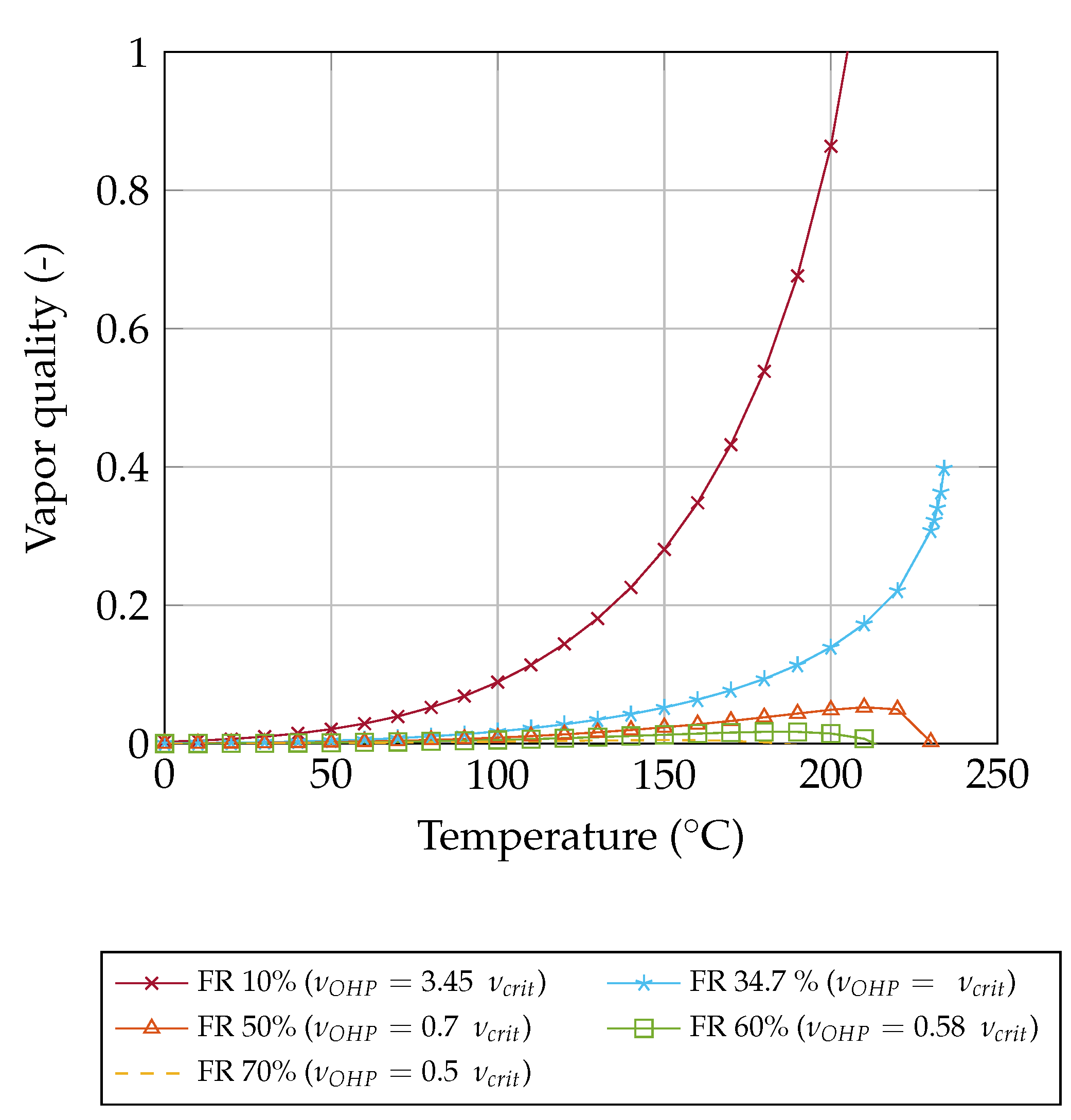
Figure 8 illustrates the vapor quality (Equation (
16)) of acetone for different filling ratios in general. For
, the entire OHP becomes gaseous at 210
. For
, the vapor quality is increasing and end at the critical point. For
the vapor quality first increases and then decreases as the temperature rises.
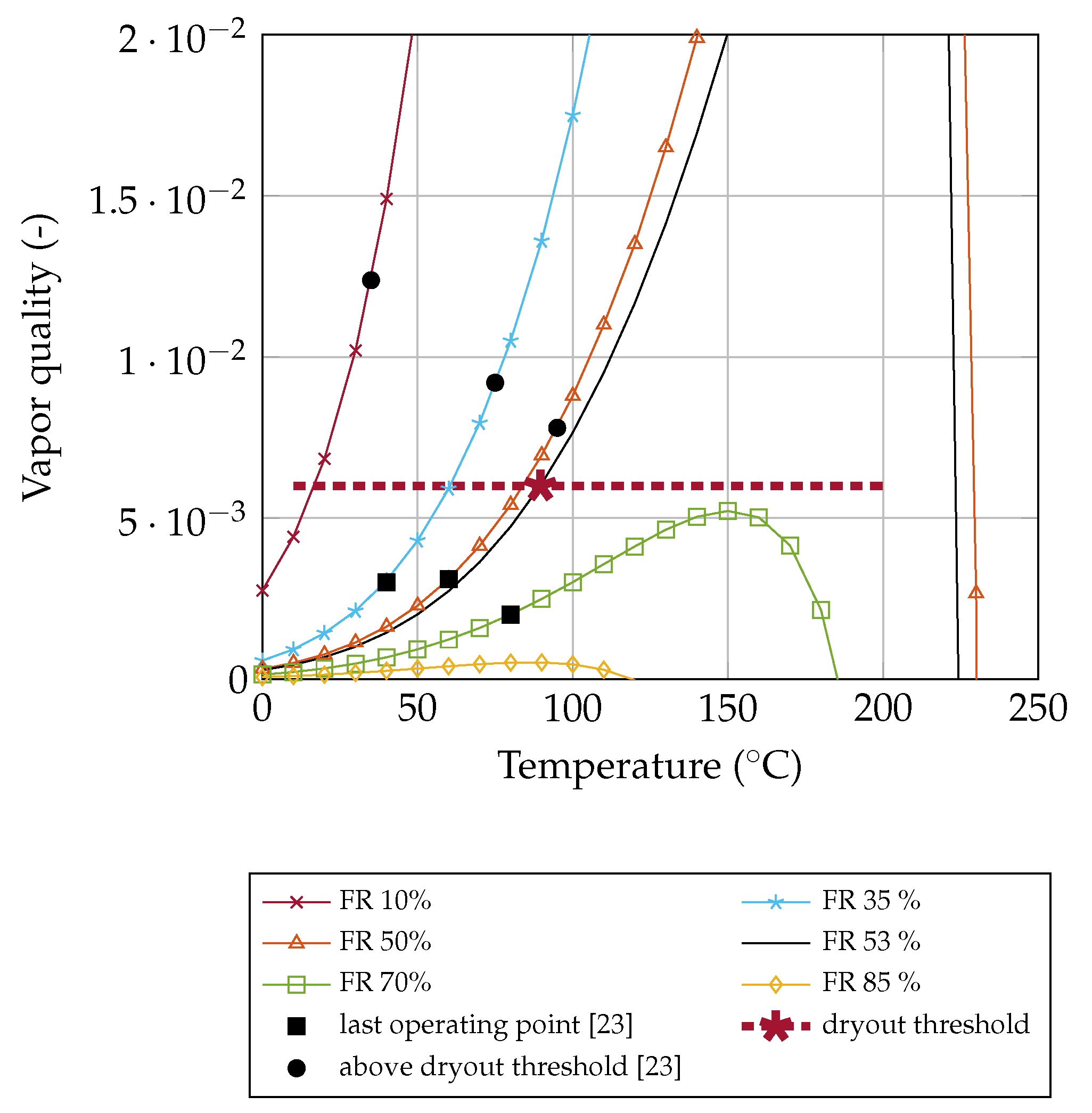
Determination of the Dryout Threshold
Our experimental data shows a dryout at a temperature of 89.6
with a filling ratio of 53%. Using Equation (
16) leads to a vapor quality of
x = 0.006. Above this threshold, the flow changes from slug flow to annular flow and the heat transfer decreases abruptly (dryout). According to the literature, this limit is generally valid for OHPs filled with acetone. Regardless of the filling ratio, the dryout takes place at a vapor quality threshold of
x = 0.006.
In the field of convective flow boiling in micro channels, the flow pattern is a function of the vapor quality
x [
16,
29]. There are flow pattern maps which illustrate the relationship between the vapor quality and the flow pattern [
14,
30]. Depending on the vapor quality
x, the flow changes from slug flow to annular flow. These flow pattern maps have not been used in OHP research so far for two main reasons. Firstly, the vapor quality
x is in the region of interest in the range from
x = 0.1 to 1 for convective flow boiling. Secondly, it has been assumed that the ratio of vapor to liquid remains almost constant and can therefore be neglected in the field of OHPs [
9]. However, our results show that the vapor quality also has an influence on the flow pattern for
.
Shi and Pan [
23] investigated the start up process of OHPs. Therefore, they had an experimental setup for a flat plate OHP with 40 channels. The 1 × 1
channels were charged with acetone. They measured the thermal resistance for various filling ratios and increased the heat input from 23 W to 210 W.
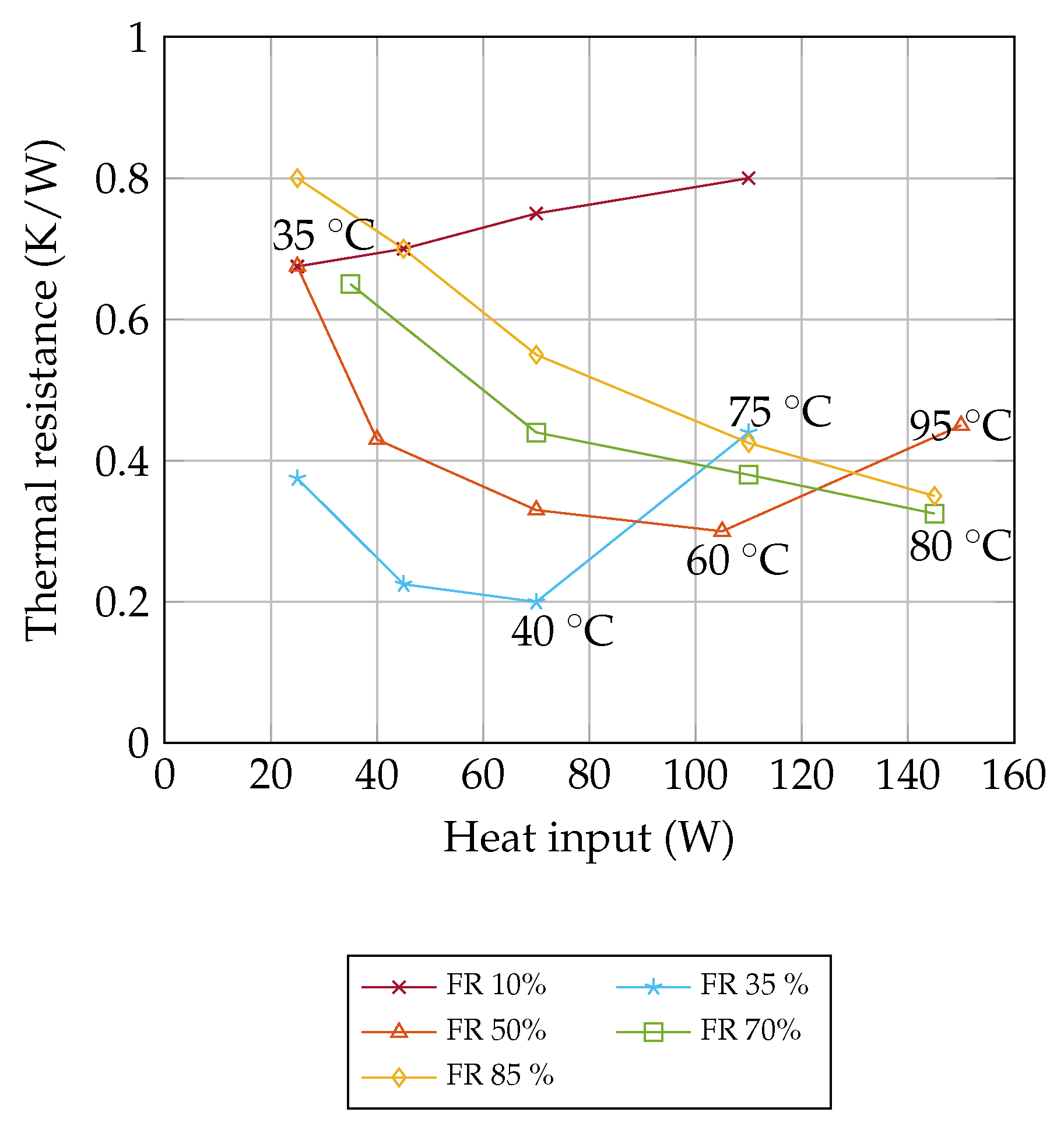
Figure 9 shows the experimental data from Shi and Pan [
23]. By applying Equation (
16), the vapor quality
x is calculated for the experimental data. The filling ratio of 35 % has a comparatively low thermal resistance of 0.2 K/W at a heat input of 70 W. Between 70 W and 110 W, the thermal resistance increases rapidly from 0.2 K/W to 0.42 K/W and the temperature rises from 40
to 75
. For these temperatures and a filling ratio of 35%, the vapor quality
x is calculated and drawn in
Figure 10. According to the same principle, the vapor quality for the filling ratio 50% at 60
and 95
as well as the filling ratio of 70% at 80
is shown in
Figure 10. For all filling ratios, the performance decreases rapidly at the same vapor quality threshold
x = 0.006. The filling ratio of 10% is too low for the OHP, the thermal resistance above 0.7 K/W and the vapor quality x is 0.0123 at 35
. The high vapor quality indicates that there is an annular flow and the OHP is not operating reliably.
In the following, the results of our compact analytical model are additionally validated with the data from Winkler et al. [
24]. They tested an OHP made of copper with a 1.5 × 1.5
cross section. The OHP has 20 channels and is charged with 50% acetone. They increased the heat input stepwise from 0 W to 180 W and measured the temperature of the heater. The OHP dries out at a heat input of approximately 125 W and a temperature of 93
. When calculating the vapor quality according to our method for their measured data, the dryout takes place at a value of
x = 0.006. This is the same value as found for the study by Shi and Pan [
23] (see above) and in our measurement.
Our approach is valid for a heater that has a contact patch across the entire width of the channels. If the heater is smaller, the dryout is limited locally [
21]. In this case, there would be no immediate failure of the electronic devices. The dryout limit also depends on the orientation position. Our experiments were conducted horizontally since the dryout takes places at a lower heat input compared to a vertical orientation [
24]. For applied power electronics, it is important to consider the worst-case orientation to avoid a burnout of the electronics components.
6. Conclusions and Outlook
A theory to determine the dryout threshold for OHPs was derived through a thermodynamic consideration. The vapor quality
x strongly influences the flow pattern. When the vapor quality
x overruns the limit of
x = 0.006, the plug flow merges into an annular flow and the OHP dries out. The filling ratio at charging temperature is different from the filling ratio at operating temperature. This gap between these filling ratios can be calculated by the vapor quality
x. First, it is necessary to calculate the specific volume of the OHP
at filling temperature. The vapor quality
x is calculated along these specific isochoric curves
. This leads to Equation (
16). Due to our experimental results, we set the vapor quality threshold for acetone to
x = 0.006. Therefore, it is possible to determine the dryout limit for each filling ratio and to find the optimal filling ratio for each operating point. The results show that there is a uniform threshold value, which has additionally been validated by data from the literature. With that approach, it is possible to design an OHP with an optimized filling ratio and, hence, optimize its cooling abilities.
Given the fact that the filling ratio is a crucial parameter in optimizing the OHP design, the compact theory presented in this paper will contribute to a better understanding of OHPs and its design limits.
In a next step, our theory enables determining the threshold values for other fluids. We hypothesize that there is a different threshold value for each fluid. Furthermore, the vapor quality also describes the start up process. The gradient of the vapor quality curve is responsible for the start up. For higher gradients the OHP starts to operate more rapidly. Thus, the theory will be applicable both for an optimal filling ratio and for evaluating the start up process.