Feedstock-Dependent Phosphate Recovery in a Pilot-Scale Hydrothermal Liquefaction Bio-Crude Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Hydrothermal Liquefaction and its Products
2.2. Phosphate Recovery.
2.2.1. Solubility Experiments
2.2.2. Struvite Production
- (1)
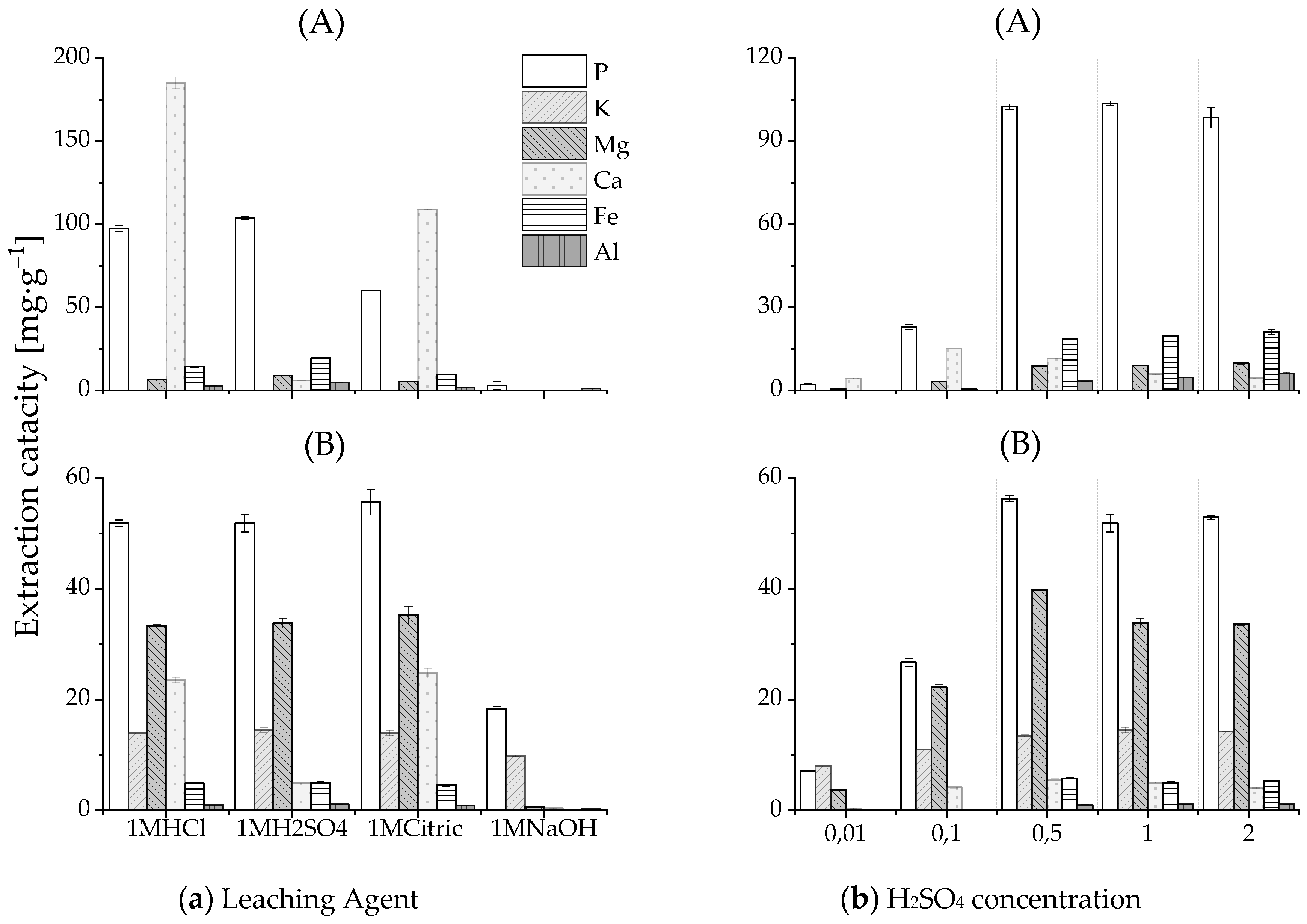
- 5 g of the dry HTL solid phase was suspended in 50 mL Milli-Q water to ensure pumpability at future large scales, and to reproduce the average output moisture condition from the HTL pilot plant. The slurry was subsequently leached with 1 molar sulfuric acid at pH 2 for 2 h (SI Analytics, TitroLine 7000)
- (2)
- 5 mL of phosphate-rich leachate that was separated from the solid residual via filtration was mixed with the HTL liquid phase, which was high in ammonium ions, in a 1-to-6 ratio to guarantee an oversaturation state for struvite crystallization [37].
- (3)
- After the addition of a previously estimated amount of MgCl2·6H2O to test the underdose and overdose scenarios of Mg2+, the mixture was stirred for 2 h at constant pH 9 that was adjusted by 1M NaOH (SI Analytics, TitroLine 7000).
- (4)
- Finally, the solid that precipitated was filtered with 0.13 µm filter paper and dried at 35 °C for future analysis. The HTL liquid phase from the SPR was also directly subjected to the struvite precipitation through supplementation of the magnesium source and maintained constant pH 9 for 2 h. Each trial was performed in duplicate.
2.3. Analyses
2.3.1. Solid and Oil Phase
2.3.2. Liquid Phase
3. Results and Discussion
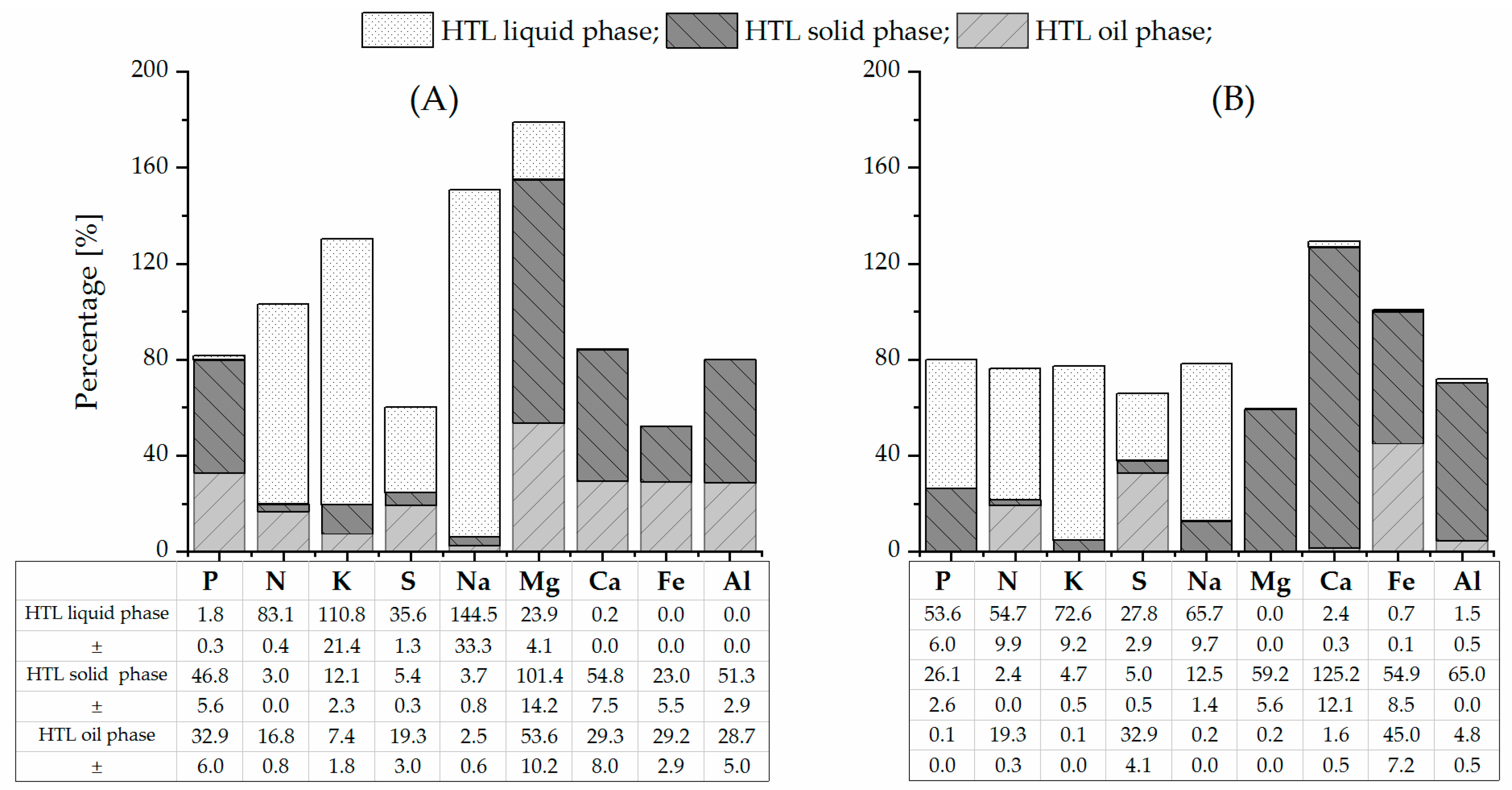
3.1. Nutrient Distribution in Feedstocks and Between HTL Products
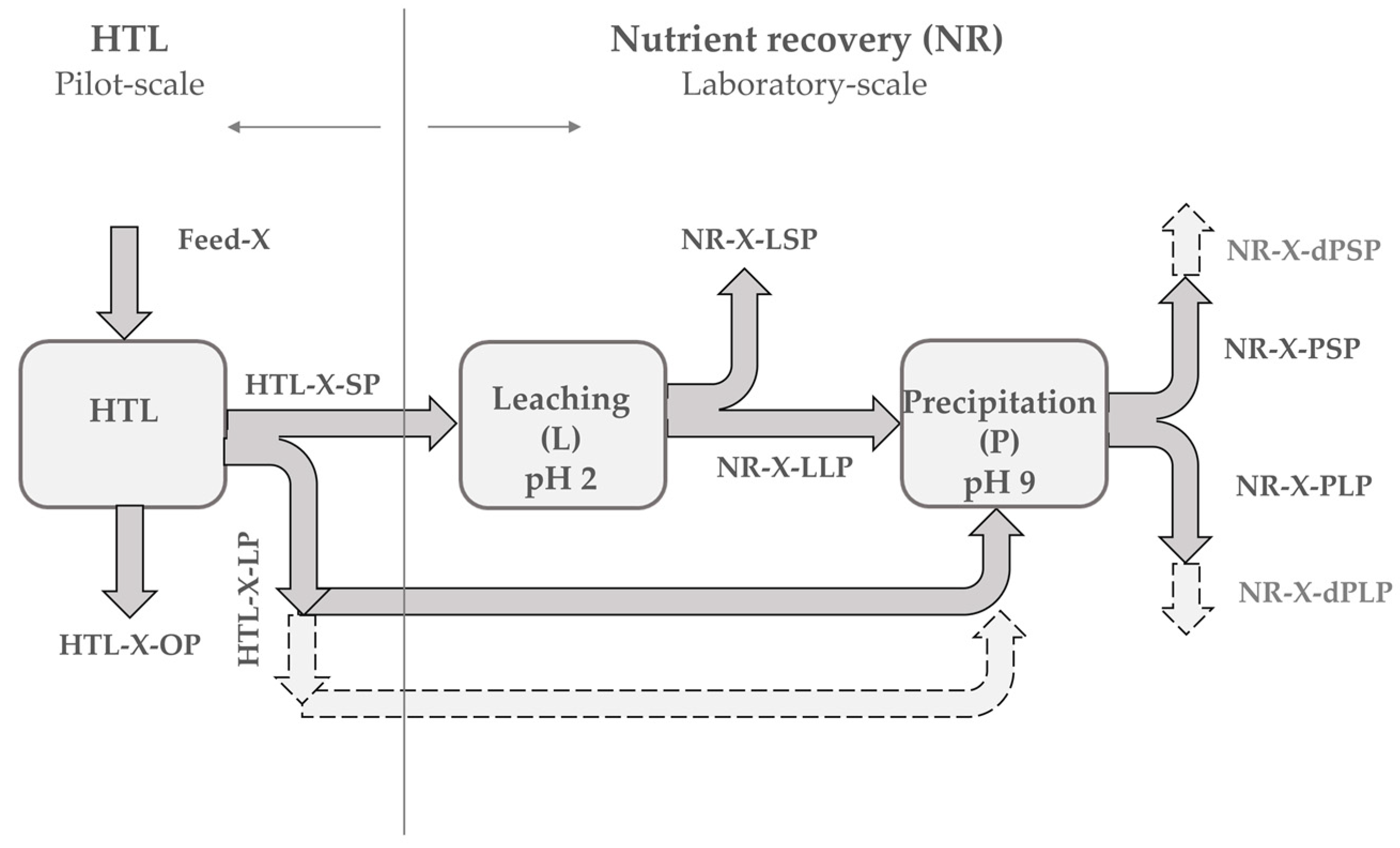
3.2. Strategy of Phosphate Recovery
3.3. Phosphate Recovery
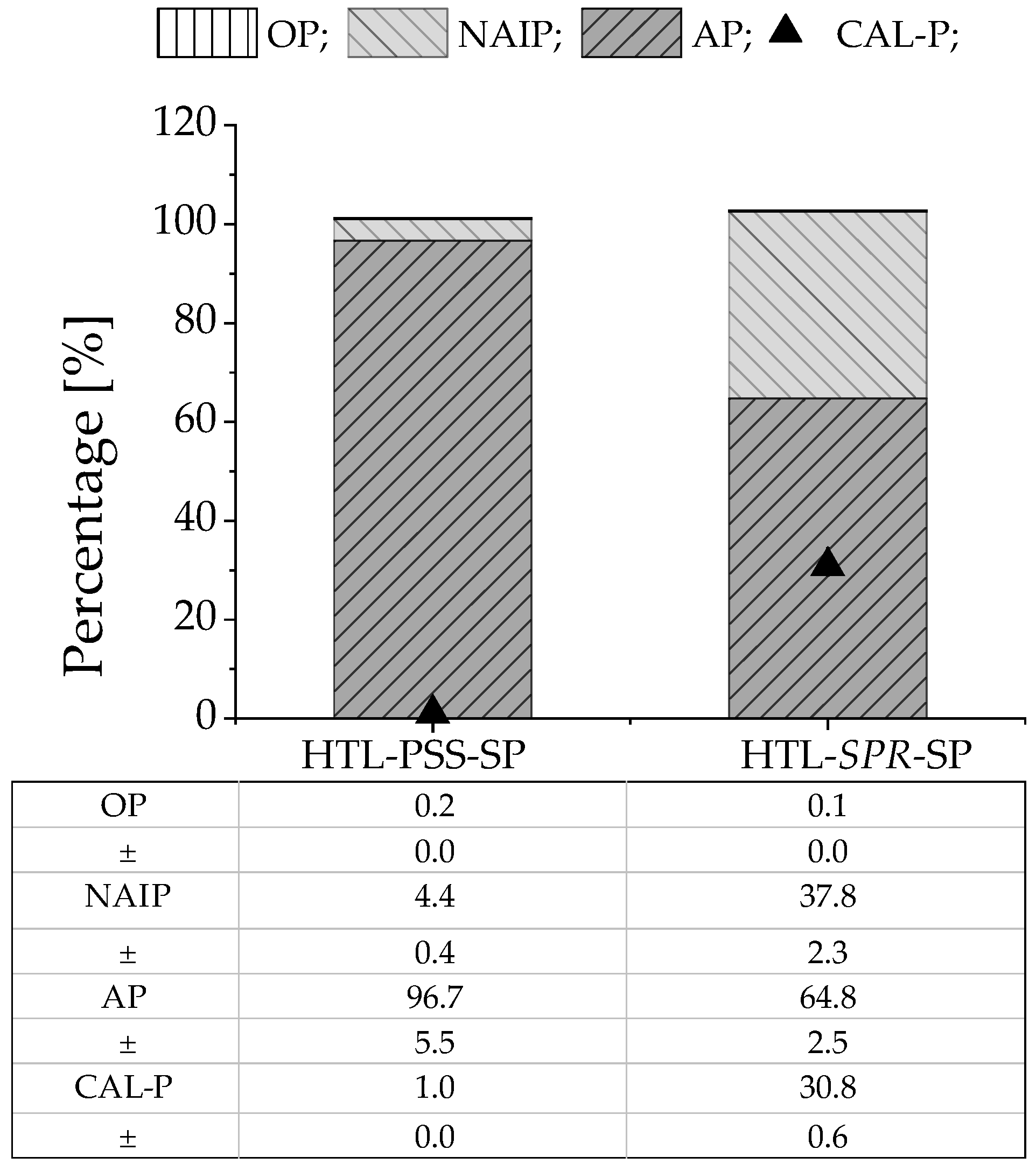
3.3.1. Release of Phosphate from the HTL Solid Phase
3.3.2. Phosphate Precipitation in the Form of Struvite
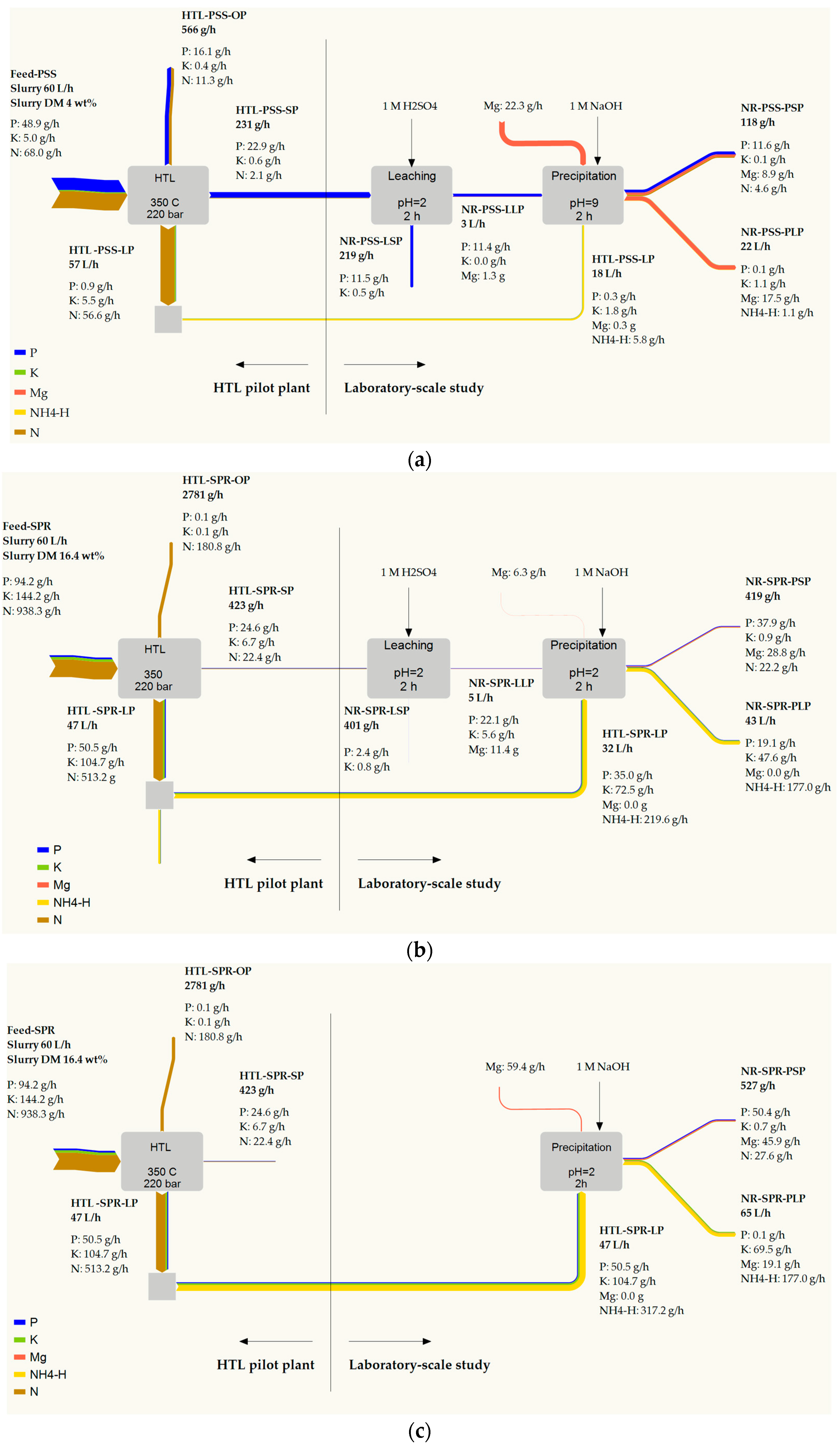
3.3.3. Overall Consideration of Process and Mass Flow of Macronutrients
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jong, S.D.; Hoefnagels, R.; Faaij, A.; Slade, R.; Mawhood, R.; Junginger, M. The feasibility of short-term production strategies for renewable jet fuels—A comprehensive techno-economic comparison. Biofuels Bioprod. Bioref. 2015, 9, 778–800. [Google Scholar] [CrossRef]
- Biller, P.; Roth, A. Hydrothermal Liquefaction: A Promising Pathway Towards Renewable Jet Fuel. In Biokerosene: Status and Prospects; Kaltschmitt, M., Neuling, U., Eds.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 607–635. ISBN 978-3-662-53065-8. [Google Scholar]
- Peterson, A.A.; Vogel, F.; Lachance, R.P.; Fröling, M.; Michael, J.; Antal, J.R.; Tester, J.W. Thermochemical biofuel production in hydrothermal media: A review of sub- and supercritical water technologies. Energy Environ. Sci. 2008, 1, 32–65. [Google Scholar] [CrossRef]
- Savage, P.E.; Levine, R.B.; Huelsman, C.M. Hydrothermal Processing of Biomass. In Thermochemical Conversion of Biomass to Liquid Fuels and Chemicals; Crocker, M., Ed.; RSC Publishing: Cambridge, UK, 2010; Chapter 8; pp. 192–221. ISBN 978-1-84973-035-8. [Google Scholar]
- Castello, D.; Haider, M.S.; Rosendahl, L. Catalytic upgrading of hydrothermal liquefaction biocrudes: Different challenges for different feedstocks. Renew. Energy 2019, 141, 420–430. [Google Scholar] [CrossRef]
- Ekpo, U.; Ross, A.B.; Camargo-Valero, M.A.; Williams, P.T. A comparison of product yields and inorganic content in process streams following thermal hydrolysis and hydrothermal processing of microalgae, manure and digestate. Bioresour. Technol. 2016, 200, 951–960. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.C.; Biller, P.; Ross, A.B.; Schmidt, A.J.; Jones, S.B. Hydrothermal liquefaction of biomass: Developments from batch to continuous process. Bioresour. Technol. 2015, 178, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.C. Review of recent reports on process technology for thermochemical conversion of whole algae to liquid fuels. Algal Res. 2016, 13, 255–263. [Google Scholar] [CrossRef]
- López Barreiro, D.; Prins, W.; Ronsse, F.; Brilman, W. Hydrothermal liquefaction (HTL) of microalgae for biofuel production: State of the art review and future prospects. Biomass Bioenergy 2013, 53, 113–127. [Google Scholar] [CrossRef]
- Castello, D.; Pedersen, T.; Rosendahl, L. Continuous Hydrothermal Liquefaction of Biomass: A Critical Review. Energies 2018, 11, 3165. [Google Scholar] [CrossRef]
- Edmundson, S.; Huesemann, M.; Kruk, R.; Lemmon, T.; Billing, J.; Schmidt, A.; Anderson, D. Phosphorus and nitrogen recycle following algal bio-crude production via continuous hydrothermal liquefaction. Algal Res. 2017, 26, 415–421. [Google Scholar] [CrossRef]
- McGinn, P.J.; Park, K.C.; Robertson, G.; Scoles, L.; Ma, W.; Singh, D. Strategies for recovery and recycling of nutrients from municipal sewage treatment effluent and hydrothermal liquefaction wastewaters for the growth of the microalga Scenedesmus sp. AMDD. Algal Res. 2019, 38, 101418. [Google Scholar] [CrossRef]
- Suzuki, A.; Nakamura, T.; Yokoyama, S.-Y.; Ogi, T.; Koguchi, K. Conversion of sewage sludge to heavy oil by direct thermochemical liquefaction. J. Chem. Eng. Jpn. JCEJ 1988, 21, 288–293. [Google Scholar] [CrossRef][Green Version]
- Vardon, D.R.; Sharma, B.K.; Scott, J.; Yu, G.; Wang, Z.; Schideman, L.; Zhang, Y.; Strathmann, T.J. Chemical properties of biocrude oil from the hydrothermal liquefaction of Spirulina algae, swine manure, and digested anaerobic sludge. Bioresour. Technol. 2011, 102, 8295–8303. [Google Scholar] [CrossRef] [PubMed]
- Marrone, P.A.; Elliott, D.C.; Billing, J.M.; Hallen, R.T.; Hart, T.R.; Kadota, P.; Moeller, J.C.; Randel, M.A.; Schmidt, A.J. Bench-Scale Evaluation of Hydrothermal Processing Technology for Conversion of Wastewater Solids to Fuels. Water Environ. Res. 2018, 90, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Peccia, J.; Westerhoff, P. We Should Expect More out of Our Sewage Sludge. Environ. Sci. Technol. 2015, 49, 8271–8276. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-J.; Yuan, X.-Z. The migration and transformation behaviors of heavy metals during the hydrothermal treatment of sewage sludge. Bioresour. Technol. 2016, 200, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.; Yuan, X.; Leng, L.; Huang, H.; Jiang, L.; Wang, H.; Chen, X.; Zeng, G. The comparison of the migration and transformation behavior of heavy metals during pyrolysis and liquefaction of municipal sewage sludge, paper mill sludge, and slaughterhouse sludge. Bioresour. Technol. 2015, 198, 16–22. [Google Scholar] [CrossRef]
- Becker, G.C.; Wüst, D.; Köhler, H.; Lautenbach, A.; Kruse, A. Novel approach of phosphate-reclamation as struvite from sewage sludge by utilising hydrothermal carbonization. J. Environ. Manag. 2019, 238, 119–125. [Google Scholar] [CrossRef]
- Yu, Y.; Lei, Z.; Yuan, T.; Jiang, Y.; Chen, N.; Feng, C.; Shimizu, K.; Zhang, Z. Simultaneous phosphorus and nitrogen recovery from anaerobically digested sludge using a hybrid system coupling hydrothermal pretreatment with MAP precipitation. Bioresour. Technol. 2017, 243, 634–640. [Google Scholar] [CrossRef]
- Zhao, X.; Becker, G.C.; Faweya, N.; Rodriguez Correa, C.; Yang, S.; Xie, X.; Kruse, A. Fertilizer and activated carbon production by hydrothermal carbonization of digestate. Biomass Convers. Biorefin. 2018, 8, 423–436. [Google Scholar] [CrossRef]
- Zhao, X.; Stökle, K.; Becker, G.C.; Zimmermann, M.; Kruse, A. Hydrothermal carbonization of Spirulina platensis and Chlorella vulgaris combined with protein isolation and struvite production. Bioresour. Technol. Rep. 2019, 6, 159–167. [Google Scholar] [CrossRef]
- Bauer, S.; Cheng, F.; Colosi, L. Evaluating the Impacts of ACP Management on the Energy Performance of Hydrothermal Liquefaction via Nutrient Recovery. Energies 2019, 12, 729. [Google Scholar] [CrossRef]
- Dyhrman, S.T. Nutrients and Their Acquisition: Phosphorus Physiology in Microalgae. In The Physiology of Microalgae; Beardall, J., Raven, J.A., Borowitzka, M.A., Eds.; Springer: Cham, Switzerland, 2016; pp. 155–183. ISBN 978-3-319-24945-2. [Google Scholar]
- Valdez, P.J.; Nelson, M.C.; Wang, H.Y.; Lin, X.N.; Savage, P.E. Hydrothermal liquefaction of Nannochloropsis sp.: Systematic study of process variables and analysis of the product fractions. Biomass Bioenergy 2012, 46, 317–331. [Google Scholar] [CrossRef]
- Jena, U.; Vaidyanathan, N.; Chinnasamy, S.; Das, K.C. Evaluation of microalgae cultivation using recovered aqueous co-product from thermochemical liquefaction of algal biomass. Bioresour. Technol. 2011, 102, 3380–3387. [Google Scholar] [CrossRef] [PubMed]
- Bagnoud-Velásquez, M.; Schmid-Staiger, U.; Peng, G.; Vogel, F.; Ludwig, C. First developments towards closing the nutrient cycle in a biofuel production process. Algal Res. 2015, 8, 76–82. [Google Scholar] [CrossRef]
- Barbera, E.; Bertucco, A.; Kumar, S. Nutrients recovery and recycling in algae processing for biofuels production. Renew. Sustain. Energy Rev. 2018, 90, 28–42. [Google Scholar] [CrossRef]
- López Barreiro, D.; Bauer, M.; Hornung, U.; Posten, C.; Kruse, A.; Prins, W. Cultivation of microalgae with recovered nutrients after hydrothermal liquefaction. Algal Res. 2015, 9, 99–106. [Google Scholar] [CrossRef]
- Leng, L.; Li, J.; Wen, Z.; Zhou, W. Use of microalgae to recycle nutrients in aqueous phase derived from hydrothermal liquefaction process. Bioresour. Technol. 2018, 256, 529–542. [Google Scholar] [CrossRef]
- Gu, Y.; Zhang, X.; Deal, B.; Han, L. Biological systems for treatment and valorization of wastewater generated from hydrothermal liquefaction of biomass and systems thinking: A review. Bioresour. Technol. 2019, 278, 329–345. [Google Scholar] [CrossRef]
- Biller, P.; Ross, A.B.; Skill, S.C.; Lea-Langton, A.; Balasundaram, B.; Hall, C.; Riley, R.; Llewellyn, C.A. Nutrient recycling of aqueous phase for microalgae cultivation from the hydrothermal liquefaction process. Algal Res. 2012, 1, 70–76. [Google Scholar] [CrossRef]
- Garcia Alba, L.; Torri, C.; Fabbri, D.; Kersten, S.R.A.; Brilman, D.W.F. Microalgae growth on the aqueous phase from Hydrothermal Liquefaction of the same microalgae. Chem. Eng. J. 2013, 228, 214–223. [Google Scholar] [CrossRef]
- Shanmugam, S.R.; Adhikari, S.; Shakya, R. Nutrient removal and energy production from aqueous phase of bio-oil generated via hydrothermal liquefaction of algae. Bioresour. Technol. 2017, 230, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Dai, H.; Wu, Y.; Peng, Y.; Lu, X. A comprehensive review of phosphorus recovery from wastewater by crystallization processes. Chemosphere 2018, 197, 768–781. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; He, X.; Deng, Y.; Tsang, D.C.W.; Jiang, R.; Becker, G.C.; Kruse, A. Phosphorus recovered from digestate by hydrothermal processes with struvite crystallization and its potential as a fertilizer. Sci. Total Environ. 2019, 698, 134240. [Google Scholar] [CrossRef]
- Le Corre, K.S.; Valsami-Jones, E.; Hobbs, P.; Parsons, S.A. Phosphorus Recovery from Wastewater by Struvite Crystallization: A Review. Crit. Rev. Environ. Sci. Technol. 2009, 39, 433–477. [Google Scholar] [CrossRef]
- Anastasakis, K.; Biller, P.; Madsen, R.; Glasius, M.; Johannsen, I. Continuous Hydrothermal Liquefaction of Biomass in a Novel Pilot Plant with Heat Recovery and Hydraulic Oscillation. Energies 2018, 11, 2695. [Google Scholar] [CrossRef]
- Ruban, V.; López-Sánchez, J.F.; Pardo, P.; Rauret, G.; Muntau, H.; Quevauviller, P. Harmonized protocol and certified reference material for the determination of extractable contents of phosphorus in freshwater sediments—A synthesis of recent works. Fresenius J. Anal. Chem. 2001, 370, 224–228. [Google Scholar] [CrossRef] [PubMed]
- González Medeiros, J.J.; Pérez Cid, B.; Fernández Gómez, E. Analytical phosphorus fractionation in sewage sludge and sediment samples. Anal. Bioanal. Chem. 2005, 381, 873–878. [Google Scholar] [CrossRef]
- Schüller, H. Die CAL-Methode, eine neue Methode zur Bestimmung des pflanzenverfügbaren Phosphates in Böden. Z. Pflanzenernaehr. Bodenk. 1969, 123, 48–63. [Google Scholar] [CrossRef]
- Thun, R.; Hoffmann, G. Die Untersuchung von Böden, 4., neubearb. u. erw. Aufl; VDLUFA-Verl.: Darmstadt, Germany, 2012; ISBN 9783941273139. [Google Scholar]
- Ovsyannikova, E.; Arauzo, P.J.; Becker, G.С.; Kruse, A. Experimental and thermodynamic studies of phosphate behavior during the hydrothermal carbonization of sewage sludge. Sci. Total Environ. 2019, 692, 147–156. [Google Scholar] [CrossRef]
- Jena, U.; Das, K.C.; Kastner, J.R. Effect of operating conditions of thermochemical liquefaction on biocrude production from Spirulina platensis. Bioresour. Technol. 2011, 102, 6221–6229. [Google Scholar] [CrossRef]
- Haider, M.; Castello, D.; Michalski, K.; Pedersen, T.; Rosendahl, L. Catalytic Hydrotreatment of Microalgae Biocrude from Continuous Hydrothermal Liquefaction: Heteroatom Removal and Their Distribution in Distillation Cuts. Energies 2018, 11, 3360. [Google Scholar] [CrossRef]
- Lemoine, F.; Maupin, I.; Lemée, L.; Lavoie, J.-M.; Lemberton, J.-L.; Pouilloux, Y.; Pinard, L. Alternative fuel production by catalytic hydroliquefaction of solid municipal wastes, primary sludges and microalgae. Bioresour. Technol. 2013, 142, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Madsen, R.B.; Glasius, M. How Do Hydrothermal Liquefaction Conditions and Feedstock Type Influence Product Distribution and Elemental Composition? Ind. Eng. Chem. Res. 2019. [Google Scholar] [CrossRef]
- Juneja, A.; Ceballos, R.; Murthy, G. Effects of Environmental Factors and Nutrient Availability on the Biochemical Composition of Algae for Biofuels Production: A Review. Energies 2013, 6, 4607–4638. [Google Scholar] [CrossRef]
- Tabatabai, M.; Frankenberger, W. Chemical composition of sewage sludges in Iowa. Res. Bull. 1979, 36, 1. [Google Scholar]
- Biller, P.; Ross, A.B. Potential yields and properties of oil from the hydrothermal liquefaction of microalgae with different biochemical content. Bioresour. Technol. 2011, 102, 215–225. [Google Scholar] [CrossRef]
- Jarvis, J.M.; Sudasinghe, N.M.; Albrecht, K.O.; Schmidt, A.J.; Hallen, R.T.; Anderson, D.B.; Billing, J.M.; Schaub, T.M. Impact of iron porphyrin complexes when hydroprocessing algal HTL biocrude. Fuel 2016, 182, 411–418. [Google Scholar] [CrossRef]
- Jensen, C.U. PIUS—Hydrofaction(TM) Platform with Integrated Upgrading Step. Ph.d.-serien for Det Ingeniør- og Naturvidenskabelige Fakultet. Ph.D. Thesis, Aalborg Universitet, Aalborg, Denmark, 2018. [Google Scholar]
- Speight, J.G. Handbook of Petroleum Product Analysis, 2nd ed.; Wiley: Hoboken, NJ, USA, 2015; ISBN 9781118986370. [Google Scholar]
- Jensen, C.U.; Rosendahl, L.A.; Olofsson, G. Impact of nitrogenous alkaline agent on continuous HTL of lignocellulosic biomass and biocrude upgrading. Fuel Process. Technol. 2017, 159, 376–385. [Google Scholar] [CrossRef]
- Kubička, D.; Horáček, J. Deactivation of HDS catalysts in deoxygenation of vegetable oils. Appl. Catal. A Gen. 2011, 394, 9–17. [Google Scholar] [CrossRef]
- Jiang, J.; Savage, P.E. Metals and Other Elements in Biocrude from Fast and Isothermal Hydrothermal Liquefaction of Microalgae. Energy Fuels 2018, 32, 4118–4126. [Google Scholar] [CrossRef]
- Jiang, J.; Savage, P.E. Influence of process conditions and interventions on metals content in biocrude from hydrothermal liquefaction of microalgae. Algal Res. 2017, 26, 131–134. [Google Scholar] [CrossRef]
- Marchetti, A.; Maldonado, M.T. Iron. In The Physiology of Microalgae; Beardall, J., Raven, J.A., Borowitzka, M.A., Eds.; Springer: Cham, Switzerland, 2016; pp. 233–279. ISBN 978-3-319-24945-2. [Google Scholar]
- Jiang, J.; Savage, P.E. Using Solvents to Reduce the Metal Content in Crude Bio-oil from Hydrothermal Liquefaction of Microalgae. Ind. Eng. Chem. Res. 2019. [Google Scholar] [CrossRef]
- Agblevor, F.A.; Besler, S. Inorganic Compounds in Biomass Feedstocks. 1. Effect on the Quality of Fast Pyrolysis Oils. Energy Fuels 1996, 10, 293–298. [Google Scholar] [CrossRef]
- Elliott, D.C.; Hart, T.R.; Schmidt, A.J.; Neuenschwander, G.G.; Rotness, L.J.; Olarte, M.V.; Zacher, A.H.; Albrecht, K.O.; Hallen, R.T.; Holladay, J.E. Process development for hydrothermal liquefaction of algae feedstocks in a continuous-flow reactor. Algal Res. 2013, 2, 445–454. [Google Scholar] [CrossRef]
- Hable, R.D.; Alimoradi, S.; Sturm, B.S.M.; Stagg-Williams, S.M. Simultaneous solid and biocrude product transformations from the hydrothermal treatment of high pH-induced flocculated algae at varying Ca concentrations. Algal Res. 2019, 40, 101501. [Google Scholar] [CrossRef]
- Leusbrock, I.; Metz, S.J.; Rexwinkel, G.; Versteeg, G.F. The solubilities of phosphate and sulfate salts in supercritical water. J. Supercrit. Fluids 2010, 54, 1–8. [Google Scholar] [CrossRef]
- Roberts, G.W.; Sturm, B.S.M.; Hamdeh, U.; Stanton, G.E.; Rocha, A.; Kinsella, T.L.; Fortier, M.-O.P.; Sazdar, S.; Detamore, M.S.; Stagg-Williams, S.M. Promoting catalysis and high-value product streams by in situ hydroxyapatite crystallization during hydrothermal liquefaction of microalgae cultivated with reclaimed nutrients. Green Chem. 2015, 17, 2560–2569. [Google Scholar] [CrossRef]
- Riman, R.E.; Suchanek, W.L.; Byrappa, K.; Chen, C.-W.; Shuk, P.; Oakes, C.S. Solution synthesis of hydroxyapatite designer particulates. Solid State Ionics 2002, 151, 393–402. [Google Scholar] [CrossRef]
- Christensen, P.S.; Peng, G.; Vogel, F.; Iversen, B.B. Hydrothermal Liquefaction of the Microalgae Phaeodactylum tricornutum: Impact of Reaction Conditions on Product and Elemental Distribution. Energy Fuels 2014, 28, 5792–5803. [Google Scholar] [CrossRef]
- Anastasakis, K.; Ross, A.B. Hydrothermal liquefaction of the brown macro-alga Laminaria Saccharina: Effect of reaction conditions on product distribution and composition. Bioresour. Technol. 2011, 102, 4876–4883. [Google Scholar] [CrossRef]
- Kruse, A.; Koch, F.; Stelzl, K.; Wüst, D.; Zeller, M. Fate of Nitrogen during Hydrothermal Carbonization. Energy Fuels 2016, 30, 8037–8042. [Google Scholar] [CrossRef]
- Biller, P.; Johannsen, I.; Dos Passos, J.S.; Ottosen, L.D.M. Primary sewage sludge filtration using biomass filter aids and subsequent hydrothermal co-liquefaction. Water Res. 2018, 130, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Tang, Y. Speciation Dynamics of Phosphorus during (Hydro)Thermal Treatments of Sewage Sludge. Environ. Sci. Technol. 2015, 49, 14466–14474. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Fang, C.; Lu, X.; Jiang, R.; Tang, Y. Transformation of Phosphorus during (Hydro)thermal Treatments of Solid Biowastes: Reaction Mechanisms and Implications for P Reclamation and Recycling. Environ. Sci. Technol. 2017, 51, 10284–10298. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Ma, K.; Yu, T.; Bai, S.; Pei, D.; Bai, T.; Zhang, Q.; Yin, L.; Hu, Y.; Chen, D. Phosphorus Transformation in Hydrothermal Pretreatment and Steam Gasification of Sewage Sludge. Energy Fuels 2018, 32, 8545–8551. [Google Scholar] [CrossRef]
- Xu, Y.; Yang, F.; Zhang, L.; Wang, X.; Sun, Y.; Liu, Q.; Qian, G. Migration and transformation of phosphorus in municipal sludge by the hydrothermal treatment and its directional adjustment. Waste Manag. 2018, 81, 196–201. [Google Scholar] [CrossRef]
- Huang, R.; Fang, C.; Zhang, B.; Tang, Y. Transformations of Phosphorus Speciation during (Hydro)thermal Treatments of Animal Manures. Environ. Sci. Technol. 2018, 52, 3016–3026. [Google Scholar] [CrossRef]
- Yakaboylu, O.; Harinck, J.; Gerton Smit, K.G.; Jong, W. Supercritical water gasification of manure: A thermodynamic equilibrium modeling approach. Biomass Bioenergy 2013, 59, 253–263. [Google Scholar] [CrossRef]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry. Chemical Equilibria and Rates in Natural Waters, 3rd ed.; John Wiley & Sons Inc.: New York, NY, USA; Chichester, UK; Brisbane, Australia; Toronto, ON, Canada; Singapore, 1996; ISBN 9780471511854. [Google Scholar]
- Petzet, S.; Peplinski, B.; Cornel, P. On wet chemical phosphorus recovery from sewage sludge ash by acidic or alkaline leaching and an optimized combination of both. Water Res. 2012, 46, 3769–3780. [Google Scholar] [CrossRef]
- Heilmann, S.M.; Molde, J.S.; Timler, J.G.; Wood, B.M.; Mikula, A.L.; Vozhdayev, G.V.; Colosky, E.C.; Spokas, K.A.; Valentas, K.J. Phosphorus reclamation through hydrothermal carbonization of animal manures. Environ. Sci. Technol. 2014, 48, 10323–10329. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, S.; Ye, X.; Xiao, W. Effects of organic substances on struvite crystallization and recovery. Desalin. Water Treat. 2016, 57, 10924–10933. [Google Scholar] [CrossRef]
- Donatello, S.; Tong, D.; Cheeseman, C.R. Production of technical grade phosphoric acid from incinerator sewage sludge ash (ISSA). Waste Manag. 2010, 30, 1634–1642. [Google Scholar] [CrossRef] [PubMed]
- Franz, M. Phosphate fertilizer from sewage sludge ash (SSA). Waste Manag. 2008, 28, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Donatello, S. Characteristics of Incinerated Sewage Sludge Ashes: Potential for Phosphate Extraction and Re-Use as a Pozzolanic Material in Construction Products. Ph.D. Thesis, Imperial College London, London, UK, 2009. [Google Scholar]
- Li, B.; Huang, H.M.; Boiarkina, I.; Yu, W.; Huang, Y.F.; Wang, G.Q.; Young, B.R. Phosphorus recovery through struvite crystallisation: Recent developments in the understanding of operational factors. J. Environ. Manag. 2019, 248, 109254. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Kumar, S.; Kwag, J.-H.; Ra, C. Magnesium ammonium phosphate formation, recovery and its application as valuable resources: A review. J. Chem. Technol. Biotechnol. 2013, 88, 181–189. [Google Scholar] [CrossRef]
- Pastor, L.; Mangin, D.; Barat, R.; Seco, A. A pilot-scale study of struvite precipitation in a stirred tank reactor: Conditions influencing the process. Bioresour. Technol. 2008, 99, 6285–6291. [Google Scholar] [CrossRef]
- Stratful, I.; Scrimshaw, M.D.; Lester, J.N. Conditions influencing the precipitation of magnesium ammonium phosphate. Water Res. 2001, 35, 4191–4199. [Google Scholar] [CrossRef]
- Li, B.; Boiarkina, I.; Yu, W.; Huang, H.M.; Munir, T.; Wang, G.Q.; Young, B.R. Phosphorous recovery through struvite crystallization: Challenges for future design. Sci. Total Environ. 2019, 648, 1244–1256. [Google Scholar] [CrossRef]
- Jaffer, Y.; Clark, T.A.; Pearce, P.; Parsons, S.A. Potential phosphorus recovery by struvite formation. Water Res. 2002, 36, 1834–1842. [Google Scholar] [CrossRef]
- Ghyselbrecht, K.; Monballiu, A.; Somers, M.H.; Sigurnjak, I.; Meers, E.; Appels, L.; Meesschaert, B. Stripping and scrubbing of ammonium using common fractionating columns to prove ammonium inhibition during anaerobic digestion. Int. J. Energy Environ. Eng. 2018, 9, 447–455. [Google Scholar] [CrossRef]

| Sample ID | Ash | P | N | K | S | Na | Mg | Ca | Fe | Al |
|---|---|---|---|---|---|---|---|---|---|---|
| - | wt% | mg·g−1 | wt% | mg·g−1 | mg·g−1 | mg·g−1 | mg·g−1 | mg·g−1 | mg·g−1 | mg·g−1 |
| Feed-PSS a | 19.3 | 21.2 | 2.9 | 2.2 | 3.7 | 2.7 | 1.4 | 40.2 | 6.4 | 3.6 |
| ± | 0.6 | 2.5 | 0.0 | 0.4 | 0.1 | 0.6 | 0.2 | 4.7 | 0.4 | 0.2 |
| Feed-SPR a | 5.8 | 11.1 | 11.1 | 17.1 | 8.0 | 4.5 | 3.2 | 1.1 | 0.7 | 0.1 |
| ± | 0.0 | 1.1 | 0.0 | 1.7 | 0.7 | 0.5 | 0.3 | 0.1 | 0.1 | 0.0 |
| HTL-PSS-OP b | 28.4 c | 28.5 | 2.0 | 0.7 | 2.9 | 0.3 | 3.1 | 48.2 | 7.6 | 4.2 |
| ± | 3.9 | 0.1 | 0.1 | 0.4 | 0.0 | 0.4 | 11.8 | 0.6 | 0.7 | |
| HTL-SPR-OP b | 6.6 c | <0.1 | 6.5 | <0.1 | 8.3 | <0.1 | <0.1 | <0.1 | 0.9 | <0.1 |
| ± | 0.1 | 0.7 | 0.0 | |||||||
| HTL-PSS-SP a | 79.6 | 99.2 | 0.9 | 2.6 | 2.0 | 1.0 | 14.5 | 220.4 | 14.7 | 18.4 |
| ± | 0.2 | 1.8 | 0.0 | 0.1 | 0.1 | 0.0 | 0.1 | 15.4 | 3.4 | 0.0 |
| HTL-SPR-SP a | 32.0 | 58.2 | 5.3 | 15.9 | 7.8 | 11.4 | 38.4 | 22.6 | 7.2 | 1.8 |
| ± | 0.2 | 0.8 | 0.0 | 0.3 | 0.4 | 0.2 | 0.8 | 0.6 | 0.1 | 0.0 |
| wt% | mg·L−1 | wt% | mg·L−1 | mg·L−1 | mg·L−1 | mg·L−1 | mg·L−1 | mg·L−1 | mg·L−1 | |
| HTL-PSS-LP | NA d | 15.8 | 0.1 | 97.6 | 54.3 | 157.8 | 13.9 | 3.2 | <0.3 | <1.5 |
| ± | 1.4 | 0.0 | 5.4 | 0.4 | 8.5 | 1.4 | 0.1 | |||
| HTL-SPR-LP | NA d | 1082.4 | 1.1 | 2243.7 | 403.4 | 541.0 | 0.2 | 4.5 | 0.9 | <1.5 |
| ± | 58.0 | 0.2 | 176.6 | 22.5 | 53.7 | 0.1 | 0.5 | 0.1 |
| Sample ID | P | N | K | Mg | Ca | NH4-N |
|---|---|---|---|---|---|---|
| - | mg·g−1 | wt% | mg·g−1 | mg·g−1 | mg·g−1 | mg·g−1 |
| NR-PSS-LSP | 47.5 | NA a | 2.1 | 7.9 | 109.8 | - |
| ± | 3.0 | 0.1 | 0.2 | 5.8 | ||
| NR-SPR-LSP | 7.2 | NA a | 2.5 | 2.7 | 25.5 | - |
| ± | 0.6 | 0.1 | 0.4 | 0.1 | ||
| NR-PSS-PSP | 97.9 | 3.9 | 1.2 | 75.2 | 15.7 | - |
| ± | 8.2 | 0.0 | 0.2 | 6.0 | 2.2 | |
| NR-SPR-PSP | 90.0 | 5.3 | 2.2 | 68.8 | 7.7 | - |
| ± | 8.6 | 0.0 | 0.2 | 6.8 | 0.5 | |
| NR-SPR-dPSP | 95.6 | 5.2 | 1.4 | 87.1 | 0.2 | - |
| ± | 4.9 | 0.0 | 0.2 | 11.5 | 0.0 | |
| - | mg·L−1 | wt% | mg·L−1 | mg·L−1 | mg·L−1 | mg·L−1 |
| NR-PSS-LLP | 3890.1 | NA a | 5.0 | 466.3 | 1066.3 | NA a |
| ± | 146.9 | 0.0 | 21.7 | 7.5 | ||
| NR-SPR-LLP | 4309.1 | NA a | 1087.0 | 2232.2 | 440.6 | NA a |
| ± | 33.4 | 25.8 | 2.5 | 5.1 | ||
| NR-PSS-PLP | 5.2 | NA a | 51.4 | 846.0 | 119.9 | 53.5 |
| ± | 0.2 | 1.3 | 30.3 | 10.8 | 7.5 | |
| NR-SPR-PLP | 449.4 | NA a | 1100.6 | 0.1 | 9.4 | 4080.0 |
| ± | 44.2 | 119.7 | 0.0 | 0.6 | 510 | |
| NR-SPR-dPLP | 1.0 | NA a | 1063 | 292.2 | 3.8 | 44,650 |
| ± | 0.0 | 75.7 | 57.1 | 1.3 | 525 |
| X | Unit | PSS | SPR | dSP |
|---|---|---|---|---|
| Leaching | ||||
| HTL-X-SP/H2O | - | 1:10 | 1:10 | - |
| 1M H2SO4/HTL-X-SP | mL·g−1 | 2.7 ± 0.3 | 2.2 ± 0.1 | - |
| Extraction capacity of P | mgp·g−1sp | 49.4 ± 2.3 | 52.2 ± 0.1 | - |
| Release rate P | % | 49.9 ± 2.2 | 90.2 ± 0.8 | - |
| Precipitation | ||||
| Leachate/HTL-X-LP | - | 1:6 | 1:6 | - |
| NH4+:PO43− | mol·mol−1 | 1.1 | 8.4 | 13.9 |
| Mg2+:PO43− | mol·mol−1 | 2.6 | 0.4 | 1.5 |
| Mg2+:Ca2+ | mol·mol−1 | 12.3 | 11.8 | 467 |
| Recovery rate P | % | 99.0 ± 0.0 | 66.5 ± 2.9 | 99.9 ± 0.0 |
| Recovery rate Mg | % | 22.6 ± 3.0 | 99.9 ± 3.0 | 67.9 ± 4.3 |
| NH4-N recovery | % | 79.4 ± 2.8 | 19.4 ± 9.3 | 8.0 ± 4.4 |
| Masse precipitate/P in initial mix solution | g·g−1P | 11.1 ± 0.2 | 5.8 ± 0.1 | 9.3 ± 0.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ovsyannikova, E.; Kruse, A.; Becker, G.C. Feedstock-Dependent Phosphate Recovery in a Pilot-Scale Hydrothermal Liquefaction Bio-Crude Production. Energies 2020, 13, 379. https://doi.org/10.3390/en13020379
Ovsyannikova E, Kruse A, Becker GC. Feedstock-Dependent Phosphate Recovery in a Pilot-Scale Hydrothermal Liquefaction Bio-Crude Production. Energies. 2020; 13(2):379. https://doi.org/10.3390/en13020379
Chicago/Turabian StyleOvsyannikova, Ekaterina, Andrea Kruse, and Gero C. Becker. 2020. "Feedstock-Dependent Phosphate Recovery in a Pilot-Scale Hydrothermal Liquefaction Bio-Crude Production" Energies 13, no. 2: 379. https://doi.org/10.3390/en13020379
APA StyleOvsyannikova, E., Kruse, A., & Becker, G. C. (2020). Feedstock-Dependent Phosphate Recovery in a Pilot-Scale Hydrothermal Liquefaction Bio-Crude Production. Energies, 13(2), 379. https://doi.org/10.3390/en13020379






