Comparing Hydrogen Sulfide Removal Efficiency in a Field-Scale Digester Using Microaeration and Iron Filters
Abstract
1. Introduction
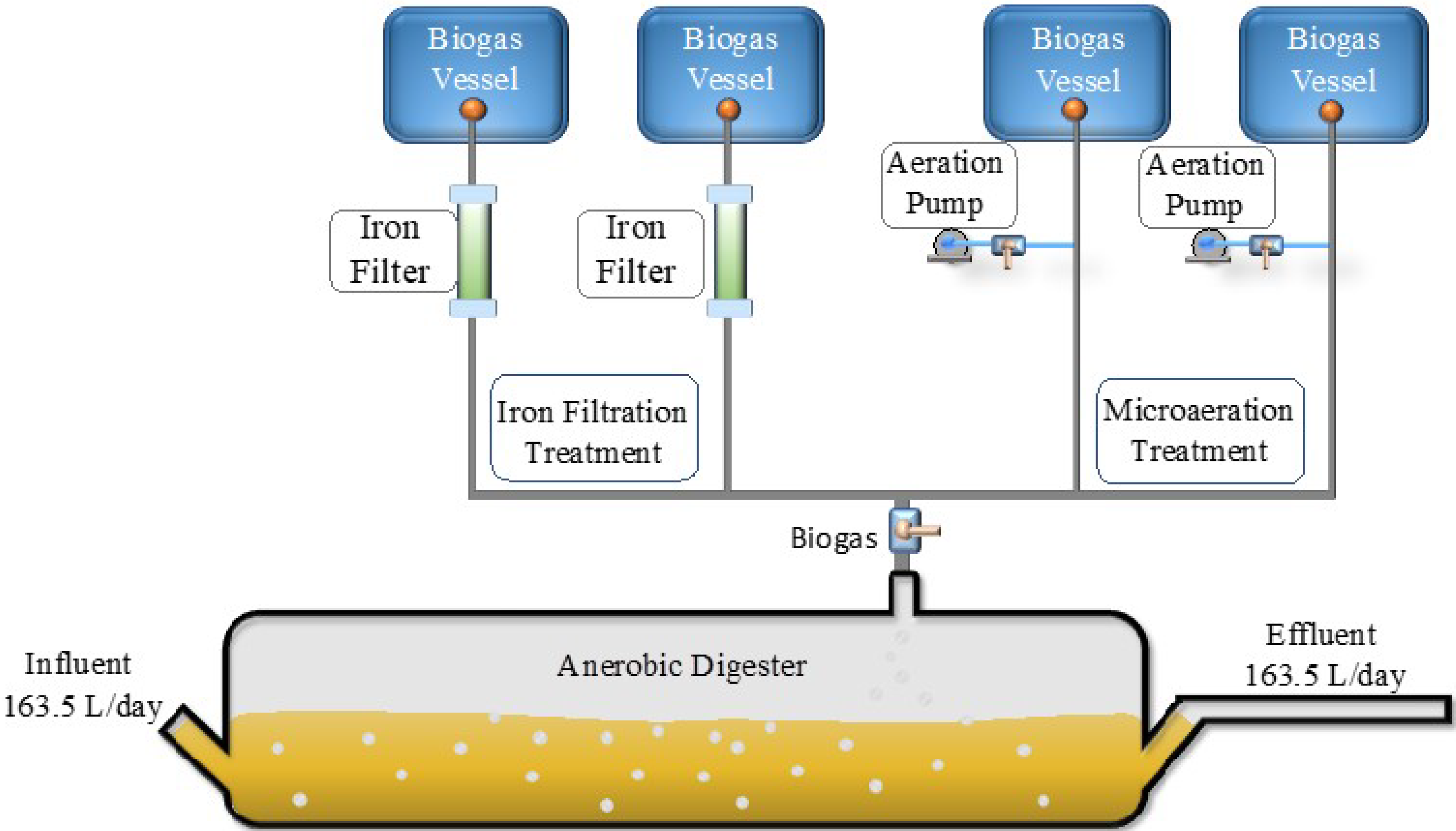
2. Materials and Methods
2.1. Digestion Reactor
2.2. Micro-Aeration
2.3. Sulfur-Oxidizing Bacteria (SOB) Observation and Isolation
2.4. Iron Filter
2.5. Statistical Methods
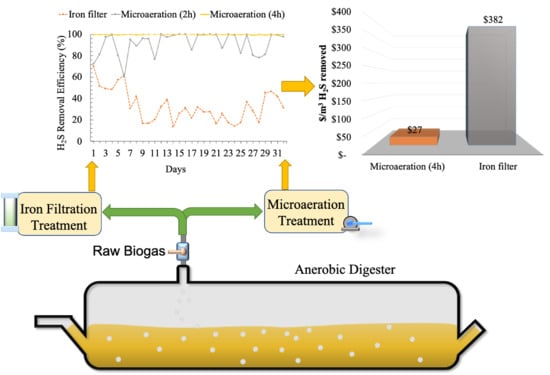
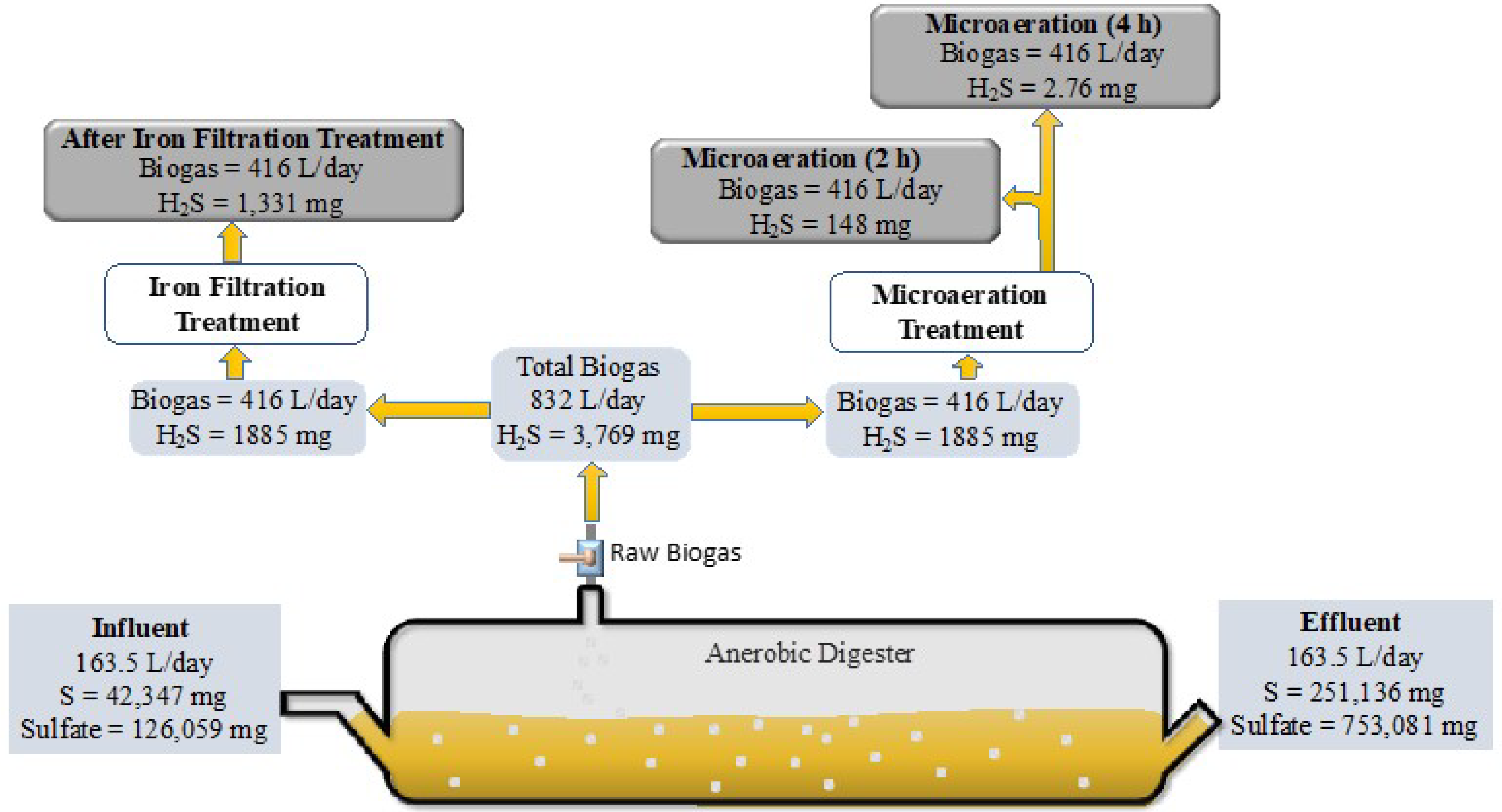
3. Results and Discussion
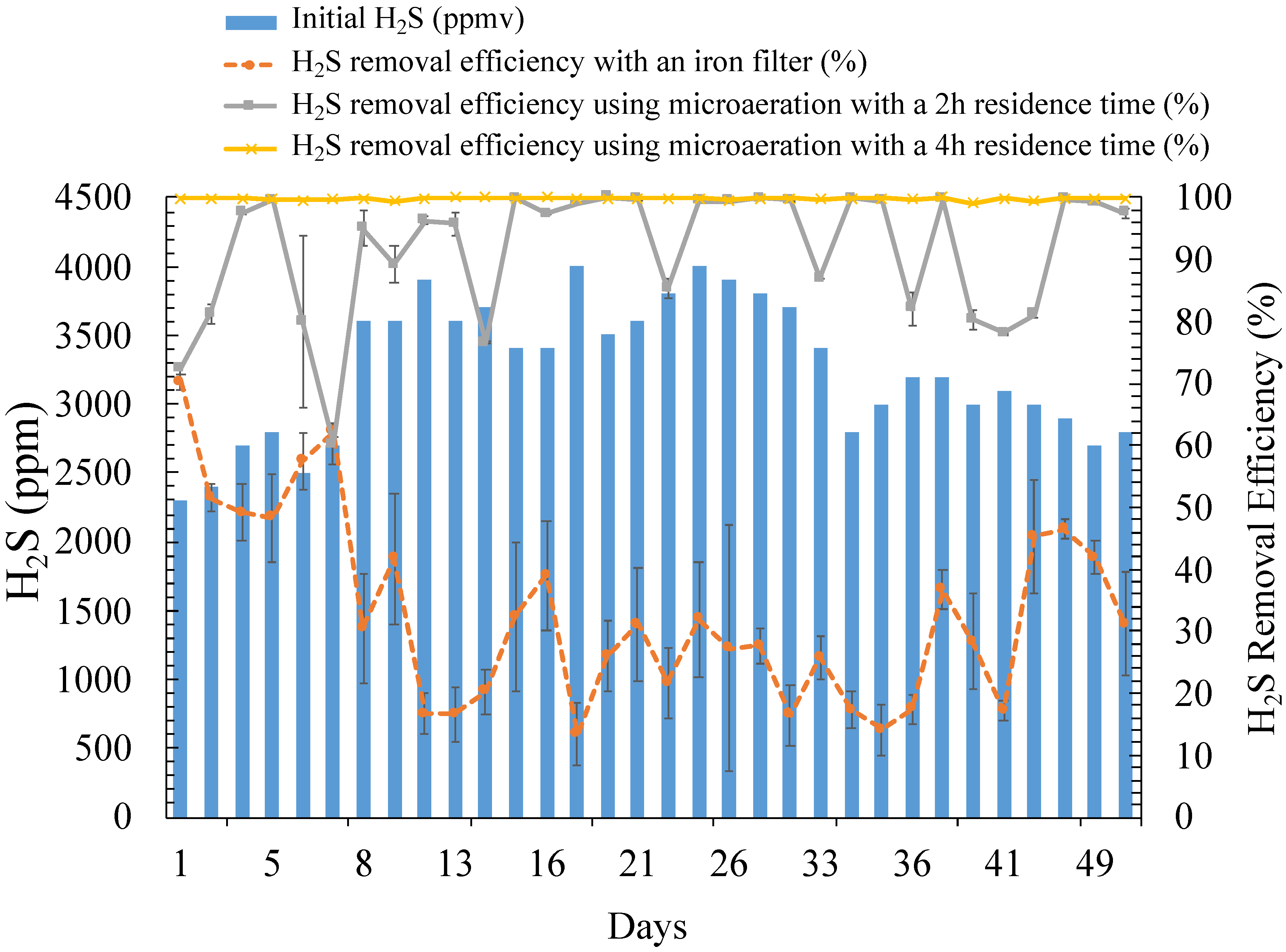
3.1. Digester pH and Temperature and Initial H2S Concentration
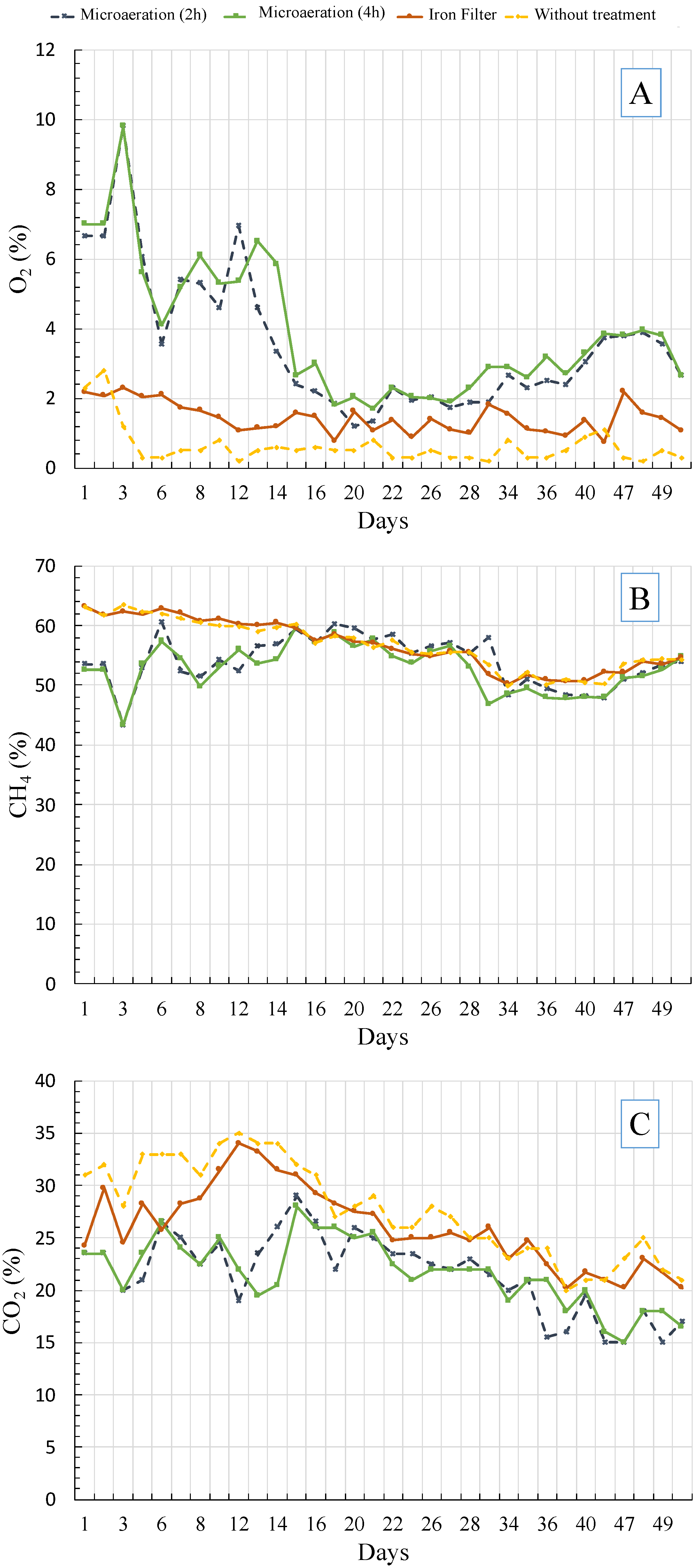
3.2. Biological Desulfurization Using Microaeration
Sulfide Oxidation during Microaeration
3.3. Desulfurization Using the Iron Filter System
3.4. Economic Analysis of Low-Cost H2S Removal Technologies
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Arif, S.; Liaquat, R.; Adil, M. Applications of materials as additives in anaerobic digestion technology. Renew. Sustain. Energy Rev. 2018, 97, 354–366. [Google Scholar] [CrossRef]
- Schieder, D.; Quicker, P.; Schneider, R.; Winter, H.; Prechtl, S.; Faulstich, M. Microbiological removal of hydrogen sulfide from biogas by means of a separate biofilter system: Experience with technical operation. Water Sci. Technol. 2003, 48, 209–212. [Google Scholar] [CrossRef]
- Nägele, H.J.; Steinbrenner, J.; Hermanns, G.; Holstein, V.; Haag, N.L.; Oechsner, H. Innovative additives for chemical desulphurisation in biogas processes: A comparative study on iron compound products. Biochem. Eng. J. 2017. [Google Scholar] [CrossRef]
- Wu, J.; Liu, D.; Zhou, W.; Liu, Q.; Huang, Y. High-Temperature H2S Removal from IGCC Coarse Gas; Springer: Berlin/Heidelberg, Germany, 2018; ISBN 9811068178. [Google Scholar]
- Shelford, T.; Gooch, C.; Choudhury, A.; Lansing, S. A Technical Reference Guide for Dairy-Derived Biogas Production, Treatment, and Utilization. 2019. Available online: https://go.umd.edu/FarmerBiogasHandbook (accessed on 13 September 2020).
- Choudhury, A.; Shelford, T.; Felton, G.; Gooch, C.; Lansing, S. Evaluation of hydrogen sulfide scrubbing systems for anaerobic digesters on two U.S. dairy farms. Energies 2019, 12, 4605. [Google Scholar] [CrossRef]
- Choudhury, A.; Lansing, S. Methane and hydrogen sulfide production from Co-digestion of gummy waste with a food waste, grease waste, and dairy manure mixture. Energies 2019, 12, 4464. [Google Scholar] [CrossRef]
- de Arespacochaga, N.; Valderrama, C.; Mesa, C.; Bouchy, L.; Cortina, J.L. Biogas biological desulphurisation under extremely acidic conditions for energetic valorisation in Solid Oxide Fuel Cells. Chem. Eng. J. 2014. [Google Scholar] [CrossRef]
- Awe, O.W.; Zhao, Y.; Nzihou, A.; Minh, D.P.; Lyczko, N. A review of biogas utilisation, purification and upgrading technologies. Waste Biomass Valorization 2017. [Google Scholar] [CrossRef]
- Allegue, L.B.; Hinge, J. Biogas upgrading evaluation of methods for H2S removal. Danish Technol. Inst. 2014. [Google Scholar] [CrossRef]
- Hassanein, A.; Lansing, S.; Tikekar, R. Impact of metal nanoparticles on biogas production from poultry litter. Bioresour. Technol. 2019. [Google Scholar] [CrossRef]
- Speight, J.G. Natural Gas: A Basic Handbook; Gulf Professional Publishing: Houston, TX, USA, 2018; ISBN 9780128095706. [Google Scholar]
- Sifuentes, G.; de Dios, J. Empleo y Purificación del Biogás Producido en Unitrar. Use and Purification of the Biogas Produced in UNITRAR. 2003. Available online: https://alicia.concytec.gob.pe/vufind/Record/UUNI_69c27d0927f487a5461d296441b87ed2/Description#tabnav (accessed on 13 September 2020).
- Dupnock, T.L.; Deshusses, M.A. Biological Co-treatment of H2S and reduction of CO2 to methane in an anoxic biological trickling filter upgrading biogas. Chemosphere 2020, 256, 127078. [Google Scholar] [CrossRef]
- Kulkarni, M.B.; Ghanegaonkar, P.M. Hydrogen sulfide removal from biogas using chemical absorption technique in packed column reactors. Glob. J. Environ. Sci. Manag. 2019. [Google Scholar] [CrossRef]
- Zhang, Y.; Oshita, K.; Kusakabe, T.; Takaoka, M.; Kawasaki, Y.; Minami, D.; Tanaka, T. Simultaneous removal of siloxanes and H2S from biogas using an aerobic biotrickling filter. J. Hazard. Mater. 2020. [Google Scholar] [CrossRef] [PubMed]
- Raabe, T.; Mehne, M.; Rasser, H.; Krause, H.; Kureti, S. Study on iron-based adsorbents for alternating removal of H2S and O2 from natural gas and biogas. Chem. Eng. J. 2019. [Google Scholar] [CrossRef]
- Schiavon Maia, D.C.; Niklevicz, R.R.; Arioli, R.; Frare, L.M.; Arroyo, P.A.; Gimenes, M.L.; Pereira, N.C. Removal of H2S and CO2 from biogas in bench scale and the pilot scale using a regenerable Fe-EDTA solution. Renew. Energy 2017. [Google Scholar] [CrossRef]
- Montalvo, S.; Huiliñir, C.; Borja, R.; Castillo, A.; Pereda, I. Anaerobic digestion of wastewater rich in sulfate and sulfide: Effects of metallic waste addition and micro-aeration on process performance and methane production. J. Environ. Sci. Heal. Part A Toxic/Hazard. Subst. Environ. Eng. 2019. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, A.; Lansing, S. Biochar addition with Fe impregnation to reduce H2S production from anaerobic digestion. Bioresour. Technol. 2020. [Google Scholar] [CrossRef]
- Verbeeck, K.; De Vrieze, J.; Biesemans, M.; Rabaey, K. Membrane electrolysis-assisted CO2 and H2S extraction as innovative pretreatment method for biological biogas upgrading. Chem. Eng. J. 2019. [Google Scholar] [CrossRef]
- Nishimura, S.; Yoda, M. Removal of hydrogen sulfide from an anaerobic biogas using a big-scrubber. Water Sci. Technol. 1997, 36, 349–356. [Google Scholar] [CrossRef]
- Krayzelova, L.; Bartacek, J.; Díaz, I.; Jeison, D.; Volcke, E.I.P.; Jenicek, P. Microaeration for hydrogen sulfide removal during anaerobic treatment: A review. Rev. Environ. Sci. Biotechnol. 2015. [Google Scholar] [CrossRef]
- Cano, P.I.; Brito, J.; Almenglo, F.; Ramírez, M.; Gómez, J.M.; Cantero, D. Influence of trickling liquid velocity, low molar ratio of nitrogen/sulfur and gas-liquid flow pattern in anoxic biotrickling filters for biogas desulfurization. Biochem. Eng. J. 2019. [Google Scholar] [CrossRef]
- Wu, J.; Jiang, X.; Jin, Z.; Yang, S.; Zhang, J. The performance and microbial community in a slightly alkaline biotrickling filter for the removal of high concentration H2S from biogas. Chemosphere 2020. [Google Scholar] [CrossRef]
- Ángeles Torres, R.; Marín, D.; Rodero, M.d.R.; Pascual, C.; González-Sanchez, A.; de Godos Crespo, I.; Lebrero, R.; Torre, R.M. Biogas treatment for H2S, CO2, and other contaminants removal. In From Biofiltration to Promising Options in Gaseous Fluxes Biotreatment; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar] [CrossRef]
- Vikrant, K.; Kailasa, S.K.; Tsang, D.C.W.; Lee, S.S.; Kumar, P.; Giri, B.S.; Singh, R.S.; Kim, K.H. Biofiltration of hydrogen sulfide: Trends and challenges. J. Clean. Prod. 2018. [Google Scholar] [CrossRef]
- Khanongnuch, R.; Di Capua, F.; Lakaniemi, A.M.; Rene, E.R.; Lens, P.N.L. Transient–state operation of an anoxic biotrickling filter for H2S removal. J. Hazard. Mater. 2019. [Google Scholar] [CrossRef]
- Khanongnuch, R.; Di Capua, F.; Lakaniemi, A.M.; Rene, E.R.; Lens, P.N.L. H2S removal and microbial community composition in an anoxic biotrickling filter under autotrophic and mixotrophic conditions. J. Hazard. Mater. 2019. [Google Scholar] [CrossRef] [PubMed]
- Meier, L.; Stará, D.; Bartacek, J.; Jeison, D. Removal of H2S by a continuous microalgae-based photosynthetic biogas upgrading process. Process. Saf. Environ. Prot. 2018. [Google Scholar] [CrossRef]
- Haosagul, S.; Prommeenate, P.; Hobbs, G.; Pisutpaisal, N. Sulfide-oxidizing bacteria community in full-scale bioscrubber treating H2S in biogas from swine anaerobic digester. Renew. Energy 2020. [Google Scholar] [CrossRef]
- Cheng, Y.; Yuan, T.; Deng, Y.; Lin, C.; Zhou, J.; Lei, Z.; Shimizu, K.; Zhang, Z. Use of sulfur-oxidizing bacteria enriched from sewage sludge to biologically remove H2S from biogas at an industrial-scale biogas plant. Bioresour. Technol. Reports 2018. [Google Scholar] [CrossRef]
- Mulbry, W.; Lansing, S.; Coker, A.C. Microaeration reduces hydrogen sulfi de in biogas. Biocycle 2017, 58, 57–59. [Google Scholar]
- Khoshnevisan, B.; Tsapekos, P.; Alfaro, N.; Díaz, I.; Fdz-Polanco, M.; Rafiee, S.; Angelidaki, I. A review on prospects and challenges of biological H2S removal from biogas with focus on biotrickling filtration and microaerobic desulfurization. Biofuel Res. J. 2017. [Google Scholar] [CrossRef]
- Zhu, H.L.; Papurello, D.; Gandiglio, M.; Lanzini, A.; Akpinar, I.; Shearing, P.R.; Manos, G.; Brett, D.J.L.; Zhang, Y.S. Study of H2S removal capability from simulated biogas by using waste-derived adsorbent materials. Processes 2020, 8, 1030. [Google Scholar] [CrossRef]
- Scarlat, N.; Dallemand, J.F.; Fahl, F. Biogas: Developments and perspectives in Europe. Renew. Energy 2018. [Google Scholar] [CrossRef]
- Hjort-Gregersen, K. Market Overview Micro Scale Digesters; AgroTEch A/S: Aarhus, Denmark, 2015. [Google Scholar]
- Matus, F.J.; Escudey, M.; Förster, J.E.; Gutiérrez, M.; Chang, A.C. Is the Walkley–Black method suitable for organic carbon determination in Chilean volcanic soils? Commun. Soil Sci. Plant Anal. 2009, 40, 1862–1872. [Google Scholar] [CrossRef]
- Kjeldahl, C. A new method for the determination of nitrogen in organic matter. Z. Anal. Chem. 1883, 22, 366–382. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 21st ed.; APHA: Washington, DC, USA, 2005; ISBN 0875532357. [Google Scholar]
- Association, A.P.H.; Association, A.W.W.; Federation, W.P.C.; Federation, W.E. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington DC, USA, 1915; Volume 2, ISBN 8755-3546. [Google Scholar]
- Hurtado, A.; Salamanca, J. Búsqueda de bacterias oxidadoras de ácido sulfhídrico para su potencial uso en la producción de biogás de alta pureza. Rev. Investig. Agrar. Ambient. 2017, 9, 295–304. [Google Scholar] [CrossRef]
- Kohl, A.L.; Nielsen, R. Gas Purification; Elsevier: Amsterdam, The Netherlands, 1997; ISBN 0080507204. [Google Scholar]
- Zicari, S.M. Removal of Hydrogen Sulfide from Biogas Using Cow-Manure Compost. Ph.D. Thesis, Cornell University, New York, NY, USA, 2003. [Google Scholar]
- van der Zee, F.P.; Villaverde, S.; García, P.A.; Fdz.-Polanco, F. Sulfide removal by moderate oxygenation of anaerobic sludge environments. Bioresour. Technol. 2007. [Google Scholar] [CrossRef]
- Ramos, I.; Pérez, R.; Fdz-Polanco, M. Microaerobic desulphurisation unit: A new biological system for the removal of H2S from biogas. Bioresour. Technol. 2013. [Google Scholar] [CrossRef]
- Schneider, R.; Quicker, P.; Anzer, T.; Prechtl, S.; Faulstich, M. Grundlegende Untersuchungen zur effektiven, Kostengünstigen Entfernung von Schwefelwasserstoff aus Biogas–Biogasanlagen: Anforderungen zur Luftreinhaltung; Bayerisches Landesamt für Umweltschutz: Augsburg, Germany, 2002; pp. 25–41. Available online: http://www.link-infos.de/Natur-Links-Energie-Biogas-Biogasanlagen-Anforderungen-Luftreinhaltung.pdf (accessed on 13 September 2020).
- Ramos, I.; Peña, M.; Fdz-Polanco, M. Where does the removal of H2S from biogas occur in microaerobic reactors? Bioresour. Technol. 2014. [Google Scholar] [CrossRef]
- Köchermann, J.; Schneider, J.; Matthischke, S.; Rönsch, S. Sorptive H2S removal by impregnated activated carbons for the production of SNG. Fuel Process. Technol. 2015. [Google Scholar] [CrossRef]
- Díaz, I.; Lopes, A.C.; Pérez, S.I.; Fdz-Polanco, M. Determination of the optimal rate for the microaerobic treatment of several H2S concentrations in biogas from sludge digesters. Water Sci. Technol. 2011. [Google Scholar] [CrossRef]
- Klok, J.B.M.; de Graaff, M.; van den Bosch, P.L.F.; Boelee, N.C.; Keesman, K.J.; Janssen, A.J.H. A physiologically based kinetic model for bacterial sulfide oxidation. Water Res. 2013. [Google Scholar] [CrossRef]
- Giordano, A.; Di Capua, F.; Esposito, G.; Pirozzi, F. Long-term biogas desulfurization under different microaerobic conditions in full-scale thermophilic digesters co-digesting high-solid sewage sludge. Int. Biodeterior. Biodegrad. 2019. [Google Scholar] [CrossRef]
- Brune, D.C. Isolation and characterization of sulfur globule proteins from chromatium vinosum and Thiocapsa roseopersicina. Arch. Microbiol. 1995. [Google Scholar] [CrossRef] [PubMed]
- Steudel, R. Mechanism for the formation of elemental sulfur from aqueous sulfide in chemical and microbiological desulfurization processes. Ind. Eng. Chem. Res. 1996. [Google Scholar] [CrossRef]
- Schlegel, H.G. General Microbiology, 7th ed.; Cambridge University Press: Cambridge, UK, 1993. [Google Scholar]
- Mahdy, A.; Song, Y.; Salama, A.; Qiao, W.; Dong, R. Simultaneous H2S mitigation and methanization enhancement of chicken manure through the introduction of the micro-aeration approach. Chemosphere 2020. [Google Scholar] [CrossRef]
- Fernández, M. Desulfuración de Biogás en Condiciones Anóxicas Mediante Biofiltración. Ph.D. Thesis, Universidad de Cádiz, Cádiz, Spain, 2011. [Google Scholar]
- McCollam, S.W. Viability for Oxidation of H2S Gas Using Low Concentration Solutions of H2O2 Peroxide in Applications for Biogas Purification. Master’s Thesis, University of Louisville, Kentucky, KY, USA, 2009. Available online: https://ir.library.louisville.edu/etd/937/ (accessed on 13 September 2020).
- Pokorna-Krayzelova, L.; Bartacek, J.; Vejmelkova, D.; Alvarez, A.A.; Slukova, P.; Prochazka, J.; Volcke, E.I.P.; Jenicek, P. The use of a silicone-based biomembrane for microaerobic H2S removal from biogas. Sep. Purif. Technol. 2017. [Google Scholar] [CrossRef]
- Tomàs, M.; Fortuny, M.; Lao, C.; Gabriel, D.; Lafuente, J.; Gamisans, X. Technical and economical study of a full-scale biotrickling filter for H2S removal from biogas. Water Pract. Technol. 2009. [Google Scholar] [CrossRef]
- Thompson, K.A.; Shimabuku, K.K.; Kearns, J.P.; Knappe, D.R.U.; Summers, R.S.; Cook, S.M. Environmental comparison of biochar and activated carbon for tertiary wastewater treatment. Environ. Sci. Technol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Surra, E.; Costa Nogueira, M.; Bernardo, M.; Lapa, N.; Esteves, I.; Fonseca, I. New adsorbents from maize cob wastes and anaerobic digestate for H2S removal from biogas. Waste Manag. 2019. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huertas, J.K.; Quipuzco, L.; Hassanein, A.; Lansing, S. Comparing Hydrogen Sulfide Removal Efficiency in a Field-Scale Digester Using Microaeration and Iron Filters. Energies 2020, 13, 4793. https://doi.org/10.3390/en13184793
Huertas JK, Quipuzco L, Hassanein A, Lansing S. Comparing Hydrogen Sulfide Removal Efficiency in a Field-Scale Digester Using Microaeration and Iron Filters. Energies. 2020; 13(18):4793. https://doi.org/10.3390/en13184793
Chicago/Turabian StyleHuertas, Joanna K., Lawrence Quipuzco, Amro Hassanein, and Stephanie Lansing. 2020. "Comparing Hydrogen Sulfide Removal Efficiency in a Field-Scale Digester Using Microaeration and Iron Filters" Energies 13, no. 18: 4793. https://doi.org/10.3390/en13184793
APA StyleHuertas, J. K., Quipuzco, L., Hassanein, A., & Lansing, S. (2020). Comparing Hydrogen Sulfide Removal Efficiency in a Field-Scale Digester Using Microaeration and Iron Filters. Energies, 13(18), 4793. https://doi.org/10.3390/en13184793