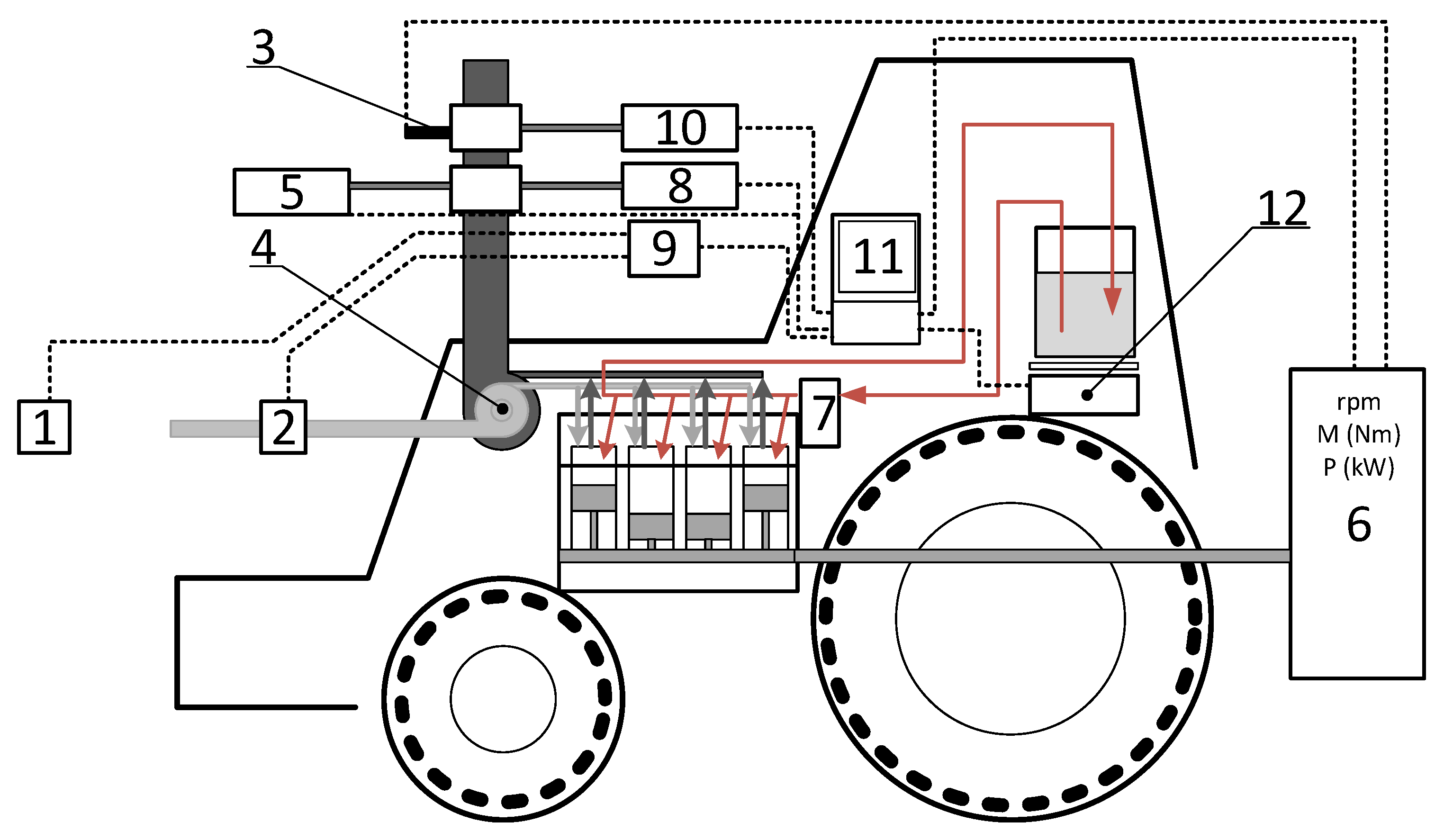
1. Introduction
Considering the globally rising energy consumption and greenhouse gasses (GHG) emissions in the agricultural sector [
1,
2], the utilization of renewable energy sources seems a good alternative to fossil fuels. One of the most common energy sources in the agricultural sector is the diesel engine, in which products or side-products of agricultural production can be used as alternative fuels. For a compression ignition (CI) engine, fuels based on a variety of edible or non-edible vegetable oils were tested as an alternative to diesel fuel [
3,
4,
5,
6,
7,
8].
A variety of vegetable oils were tested as an alternative fuel for CI engines. Vegetable oils, from the viewpoint of a CI engine, differ mainly in physical and chemical parameters, such as cetane number, viscosity, density, calorific value, compositions of fatty acids, miscibility with fossil fuels and many more. Therefore, the effect of different kinds of vegetable oil on the operational parameters of the combustion engine is also different. In general, utilization of vegetable oils in a compression ignition engine requires modification of the fuel properties, especially the viscosity. This can be done by chemical modification, such as transesterification [
9,
10,
11] or hydrotreatment [
12,
13,
14,
15], which require a non-negligible amount of additional energy, preheating of the fuel before entering the engine [
5] or blending the oil with fuel with lower viscosity, such as diesel fuel or alcohols [
3,
16,
17,
18,
19].
Coconut oil is extracted from the kernel of coconut or copra (flesh from a coconut) and it is an edible vegetable oil. Its energy potential lies in the utilization outright in the location of its origin, such as the Pacific Islands or Indonesia, where it is used for transport and electricity generation due to its relatively low local price [
20,
21]. Further, its economic benefits could decrease the transport costs during coconut flesh production, however, it would not dramatically increase the income [
22]. From the viewpoint of storability, coconut oil has a high content of saturated fat, which slows down its oxidation process and makes it resistant to acidification for up to two years [
23]. The main advantage of coconut oil is its relatively high cetane number in comparison with other vegetable oils, such as rapeseed oil (one of the most cultivated energy crops in Europe [
24,
25]) or oil from Jatropha curcas, as can be seen in
Table 1.
Preheated neat coconut oil in comparison with diesel fuel decreases engine performance, engine efficiency, emissions of carbon monoxide (CO) and hydrocarbons (HC), and increases brake specific fuel consumption (BSFC) and engine smoke, especially at higher engine loads [
28,
29].
An increasing concentration of coconut oil in a blend with diesel fuel decreases engine smoke, emissions of nitrogen oxides (NO
X), CO, HC, polycyclic aromatic hydrocarbons (PAH), brake power and exhaust gas temperature, and increases BSFC and emissions of carbon dioxide (CO
2) in comparison with diesel fuel [
21,
23,
30,
31,
32]. However, Machacon et al. [
32] found increased HC and CO with an increasing proportion of coconut oil in comparison with diesel fuel. How et al. [
21] found an increasing trend of smoke and CO emissions at high engine load and 50% coconut oil concentration in diesel fuel in comparison with lower concentrations.
For blending vegetable oils with alcohols, lower alcohols such as methanol or ethanol were tested [
33,
34]. However, fuel properties of higher alcohols, such as butanol, are closer to diesel fuel, especially regarding the higher C/H ratio and cetane number, lower affinity to water, higher calorific value and better miscibility with fossil fuels [
3,
16,
35,
36,
37,
38,
39,
40,
41]. According to Lujaji et al. [
26], the cetane number, density, kinematic viscosity and thermal characteristics of a fuel blend containing 10% vegetable oil, 10% butanol and 80% diesel fuel are more comparable with diesel fuel than methyl esters.
The ternary blends of vegetable oil, diesel fuel and butanol increase emissions of CO and BSFC, and decrease emissions of CO
2, brake power, engine efficiency and engine smoke in comparison with diesel fuel [
16,
38,
42,
43,
44,
45,
46]. However, Atmanli et al. [
47] found lower emissions of CO in comparison with diesel using a diesel fuel-butanol-cotton oil fuel blend. Emissions of NO
X were found increased in some studies [
42,
43,
44,
45] and decreased in other studies [
38,
46,
47]. Emissions of HC were also found increased in a few studies [
38,
46] and decreased in other studies [
42,
43,
45,
47] in comparison with diesel fuel.
Coconut oil in a blend with ethanol and butanol in comparison with diesel fuel increases BSFC and emissions of CO and decreases emissions of NO
X and SO
2. Engine efficiency is increased in lower loads and decreased in higher engine loads. Emissions of CO
2 increased with the coconut oil proportion and decreased with the alcohol proportion in a fuel blend [
48]. In comparison with diesel fuel, coconut oil in a blend with methanol and its biodiesel increases the brake thermal efficiency, especially in higher engine loads, and BSFC, and decreases emissions of NO
X. Higher concentrations of methanol and biodiesel increase emissions of CO and unburned hydrocarbons (UHC). Higher proportions of coconut oil decrease emissions of CO, UHC and CO
2 and increase engine smoke in comparison with diesel fuel [
49].
The aim of the paper was to experimentally determine the influence of coconut oil and n-butanol in ternary blends with diesel fuel on the emissions of CO2, NOX, CO and HC, engine smoke, performance parameters, BSFC and production of solid particles. Butanol in fuel blends is used to improve the fuel properties, especially the viscosity, and to increase the bio-content in the fuel.
3. Results and Discussion
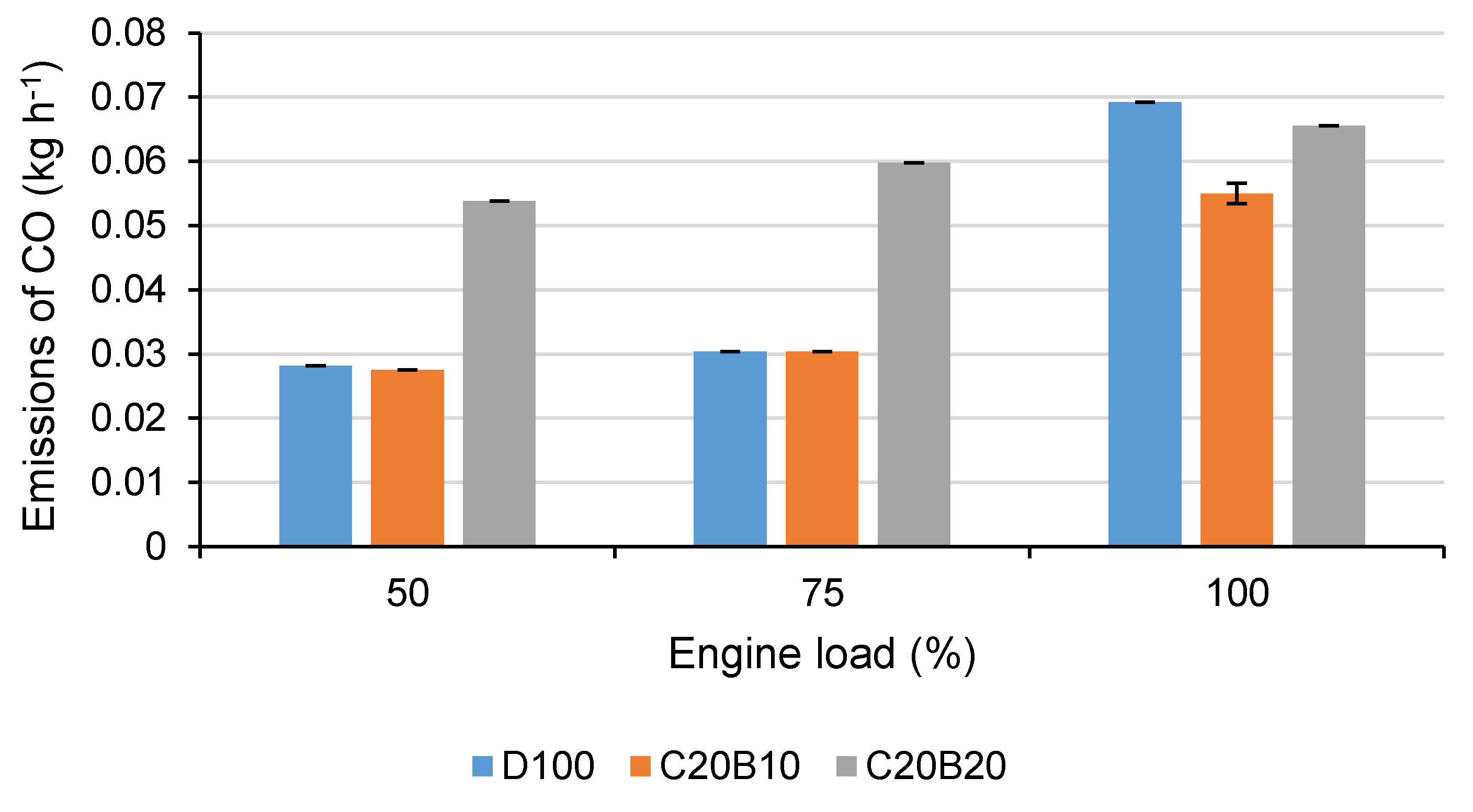
In
Figure 2, the emissions of CO for all tested fuels at all engine loads can be seen. It is evident that both of the tested fuel blends decreased the production of CO at full engine load (fuel blend C20B10 by 20.6% and C20B20 by 5.3%) in comparison with D100. At 50% and 75% engine loads, the fuel blend C20B10 reached approximately the same production of CO as D100 (differences are under the measurement accuracy). On the contrary, compared with D100, the fuel blend C20B20 reached, at the engine loads of 50% and 75%, a higher production of CO by 90.9% and 97%, respectively.
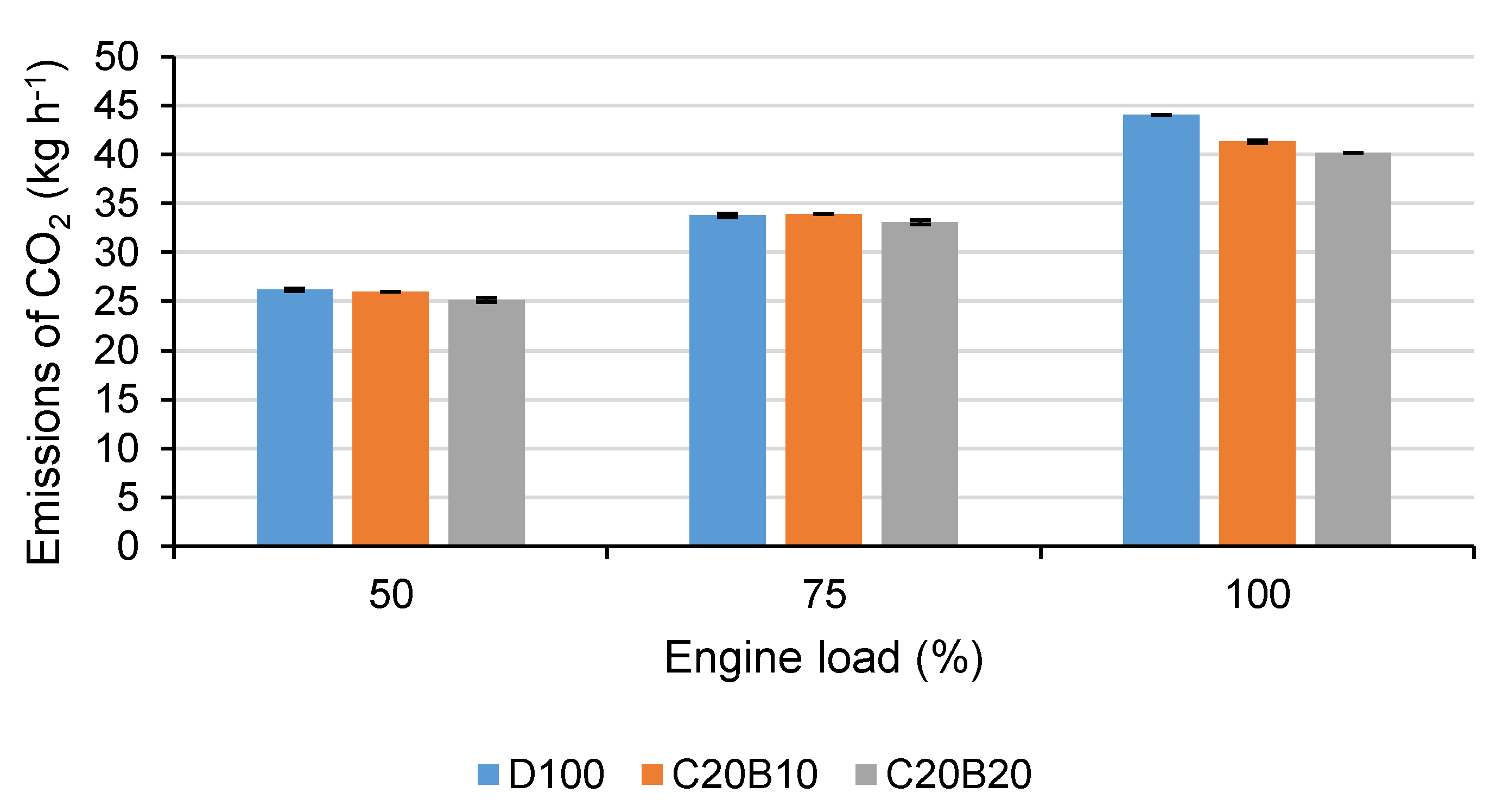
In
Figure 3, the production of CO
2 at all engine loads using all tested fuels is shown. As can be seen from the figure, at engine loads of 50% and 75%, the differences in CO
2 production are relatively small (for C20B10 under 1% and for C20B20 under 4%). At full engine load, the differences are more significant. When using the fuel blend C20B10, the production of CO
2 decreased by approximately 6.2% and C20B20 by approximately 8.8% in comparison with D100. The higher differences at full engine load may be caused by the lower carbon content of fuels containing coconut oil and n-butanol and therefore their lower calorific value as the volume of fuel injected into the cylinder is the same at 100% engine load for all tested fuels. Further, it is evident that with an increasing proportion of n-butanol, the emissions of CO
2 decrease. This is caused by the low cetane number of n-butanol, causing the later start of combustion and therefore ineffective oxidation of CO to CO
2. This is also evident from
Figure 2, where a significant increase in CO for the fuel blend C20B20 at loads 50% and 75% can be seen. At full engine load, the production of CO when using C20B20 is still higher than with C20B10, which confirms the ineffective oxidation caused by n-butanol. Similar results concerning emissions of CO
2 and CO were reached also in other studies dealing with ternary blends of vegetable oil, diesel fuel and butanol in comparison with diesel fuel [
38,
42,
44,
45,
46]. The increasing amount of produced emissions of CO was also found in studies focusing on coconut oil-diesel fuel blends [
32], in which it was explained by the worse atomization of the fuel, due to the higher viscosity, and coconut oil-ethanol-butanol blends [
48].
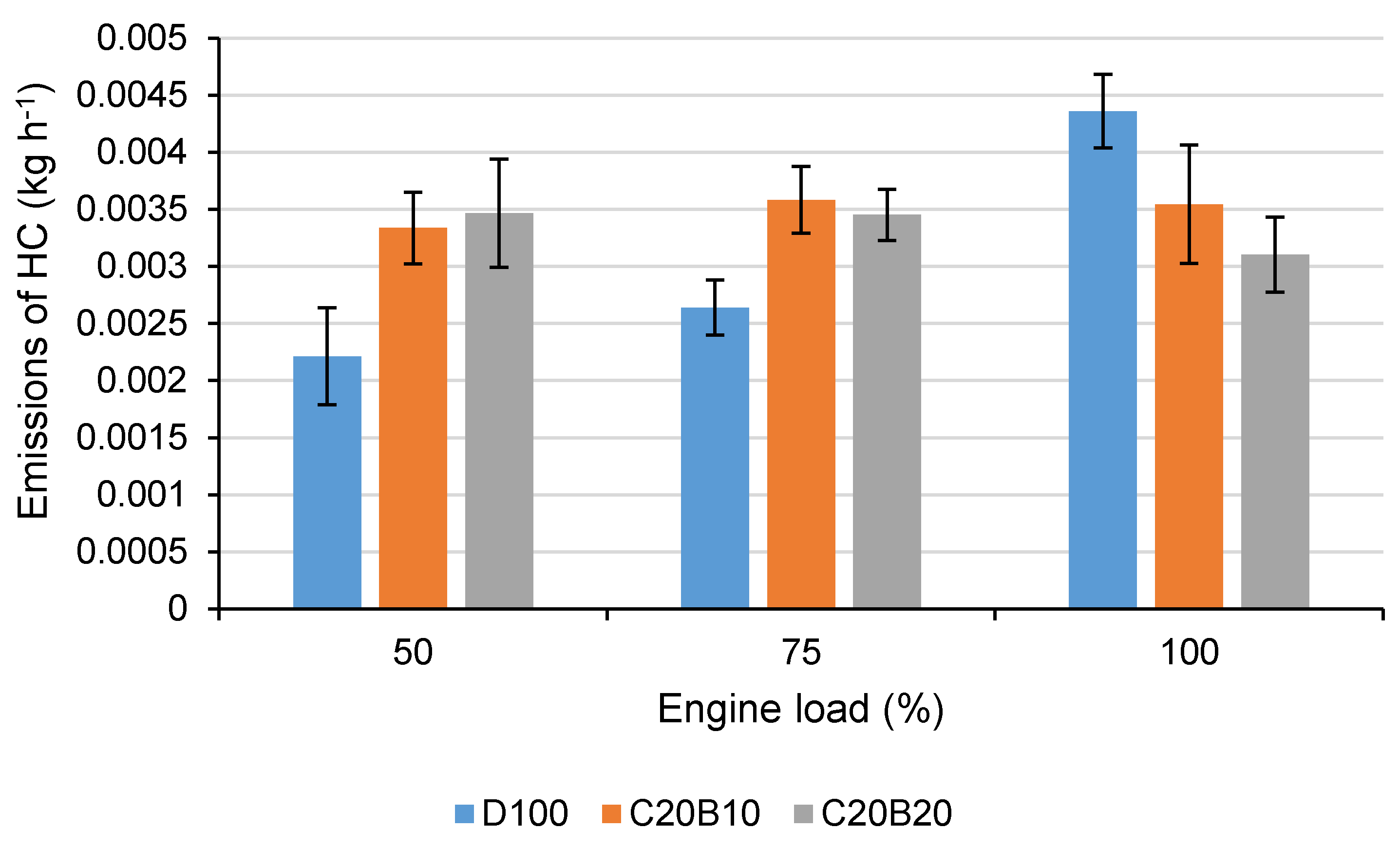
In
Figure 4, the production of emissions of HC using all tested fuels at all tested engine loads is shown. All the differences and even the absolute measured values of volumetric concentrations are under the measurement accuracy. However, from the figure, an increasing trend of HC production at engine loads of 50% and 75% with the blended fuels in comparison with diesel fuel can be observed, which also confirms worsened oxidation since the HC emissions are products of the incomplete oxidation process. At 100% engine load, the trend is decreasing which might be also expected considering the decreased CO
2 and CO emissions due to the lower carbon content of the fuel blends. A similar trend in the emissions of HC was also found in other studies focused on vegetable oil-diesel fuel-butanol blends [
38,
44,
46]. Sharon et al. [
44,
46] explained higher HC emissions, especially in lower and moderate engine loads, by higher hydrogen content and higher viscosity resulting in poor spray characteristics in combination with the presence of butanol, causing poor combustion characteristics. Machacon et al. [
32] also found increasing emissions of HC with an increasing proportion of coconut oil in a diesel fuel–coconut oil blend.
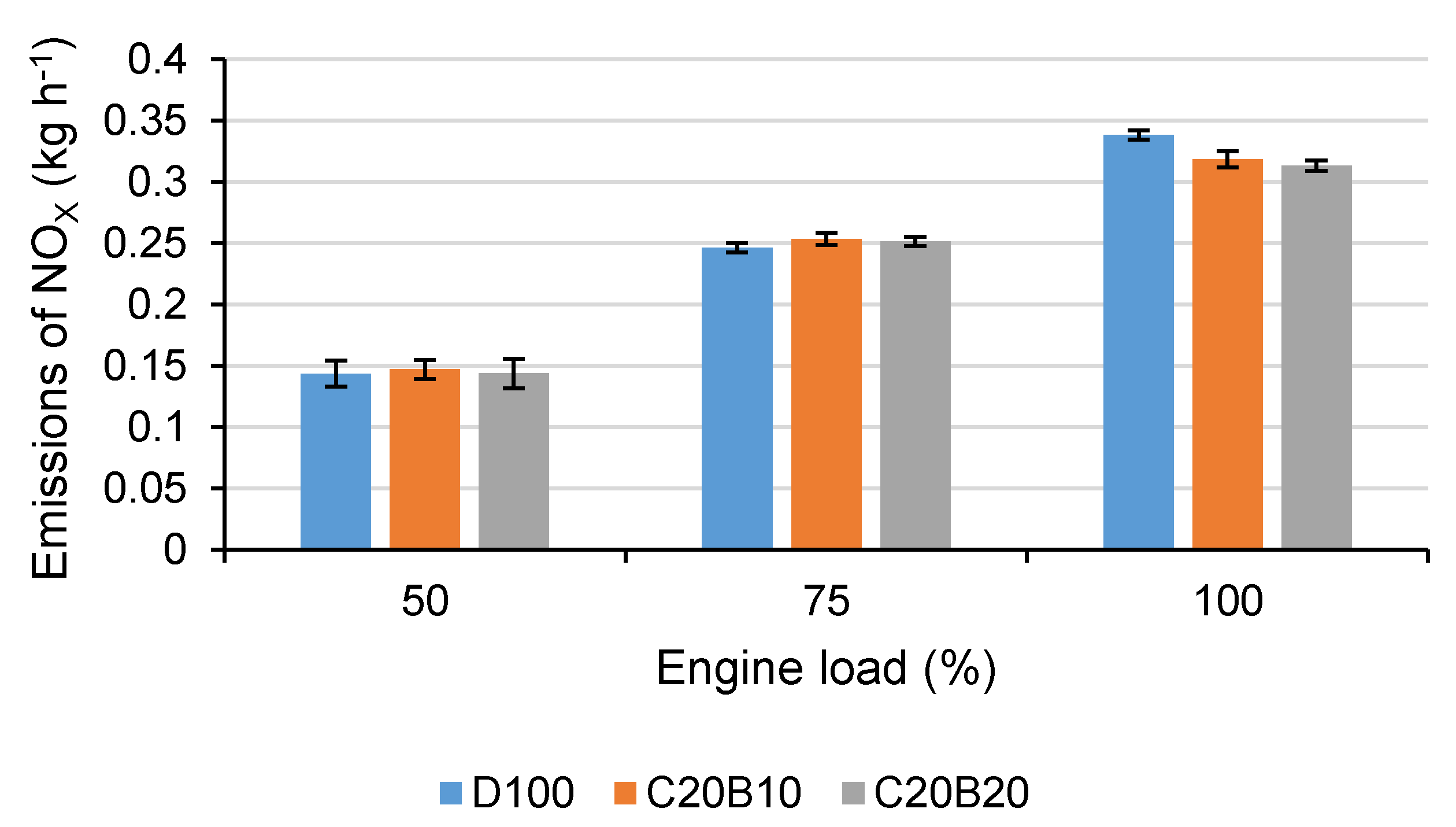
In
Figure 5, the production of emissions of NO
X for all tested fuels at all measured engine loads is shown. At 50% and 75% engine load, the differences of blended fuels in comparison with D100 are relatively small. Using fuel blend C20B10, the emissions of NO
X increased by 2.4% at 50% load and by 2.9% at 75% engine load and using fuel blend C2B20 by 0.9% at 50% engine load and by 2.1% at 75% engine load in comparison with D100. At full engine load, the more significant difference between blended fuels and D100 was observed. Using fuel blend C20B10, the NO
X emissions decreased by 5.9% and C20B20 by 7.4% in comparison with D100. It can be observed that the NO
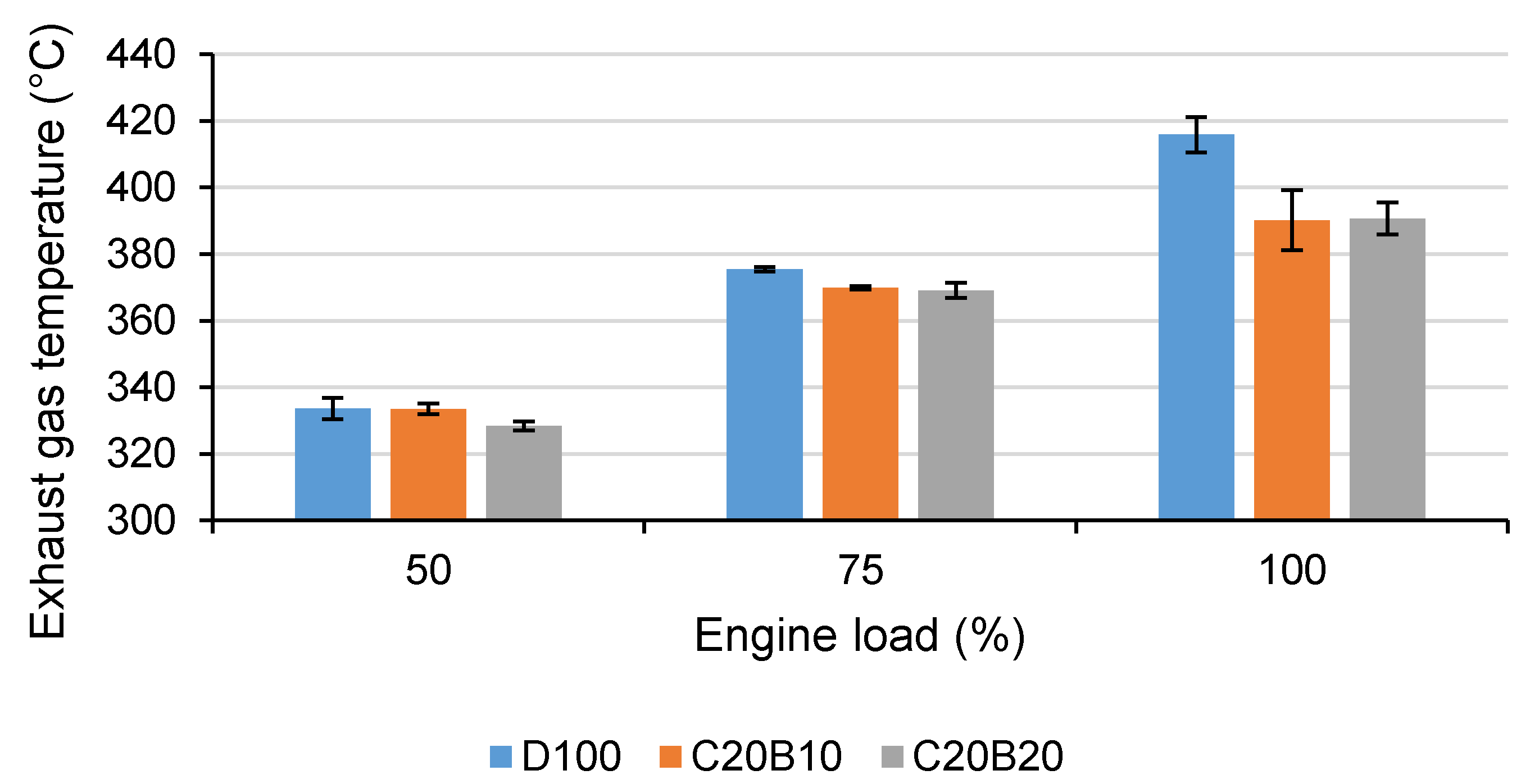
X emissions decreased with the increasing content of n-butanol in the blend despite the increasing oxygen content. This can be explained by the higher latent heat of evaporation of n-butanol, contributing to the lower combustion temperature, in combination with a lower combustion efficiency. This can be also verified by the exhaust gas temperature in
Figure 6, which is decreasing with the increasing proportion of n-butanol at all tested engine loads. Sharon et al. [
38] found similar results concerning lower emissions of NO
X and exhaust gas temperature when using used palm oil–diesel fuel–butanol blends. Lujaji et al. [
44,
46] reached lower emissions of NO
X at low and moderate engine loads. Authors in the studies [
30,
31,
32] dealing with diesel fuel–coconut oil blends also found lower emissions of NO
X with increasing concentration of coconut oil up to 100%. A lower emission of NO
X in comparison with diesel fuel was also reached using coconut oil–ethanol–butanol fuel blends [
48].
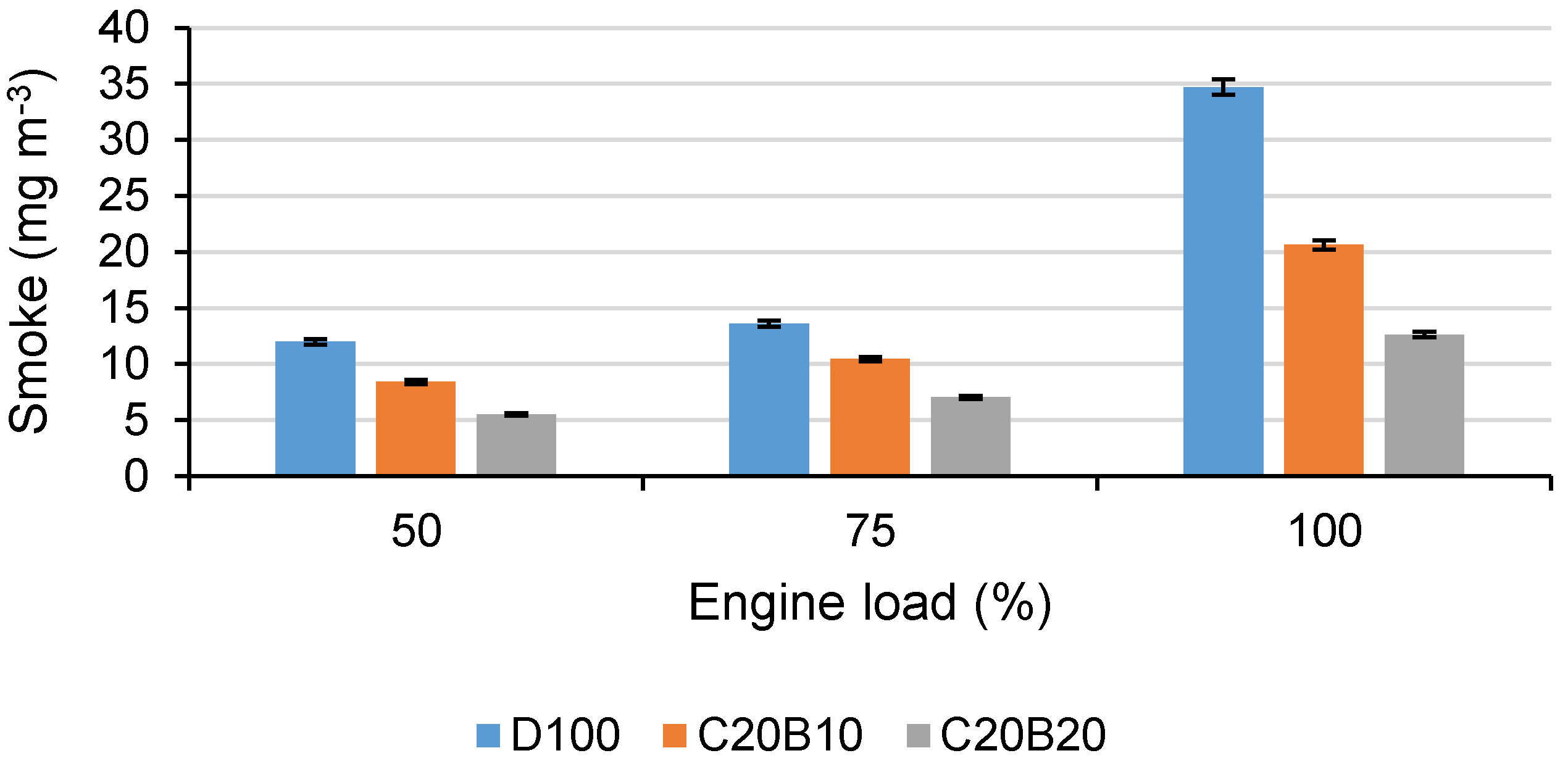
In
Figure 7, the amount of produced smoke by the engine using all tested fuels at all measured engine loads can be seen. It is evident that engine smoke is significantly lower when using both of the tested fuel blends in comparison with D100. Compared with D100, the fuel blend C20B10 showed lower engine smoke by approximately 29.9% at 50% load, 23.2% at 75% engine load and 40.5% at full load. Fuel blend C20B20 showed that engine smoke decreased by approximately 53.9% at 50% load, 48.2% at 75% load and 63.6% at full engine load. It is evident that engine smoke decreases with increasing n-butanol in the fuel blend. This can be explained by a higher oxygen content and higher amount of light fractions in the fuel blends in comparison with D100. Light fractions, such as n-butanol, are more volatile and oxidize with less intermediates in comparison with heavier fractions contained in diesel fuel. Moreover, the oxygen content in coconut oil may contribute to the improved fuel oxidation even in locally rich fuel combustion zones. The increased oxygen concentration in the soot-forming region and the reduced residence time of the fuel element may have improved the smoke reduction. Since the production of soot precursor species strongly depends on the amount of available oxygen in the region, its increased availability in the rich premixed reaction zones results in lower production of soot precursor species and therefore in reduced rates of the reactions producing soot, resulting in lower engine smoke. Further, the lower viscosity of fuel blend C20B20 in comparison with C20B10 contributes to better atomization and therefore lower smoke. The decrease in smoke in comparison with diesel fuel occurs often when using fuels with higher amounts of oxygen. Researchers found lower smoke when using vegetable oil–diesel fuel–butanol blends [
38,
44,
46] and coconut oil-diesel fuel blends [
30,
31,
32] in comparison with diesel fuel. A decreased amount of engine smoke was found also when using other blends containing vegetable oils and/or butanol [
52,
53].
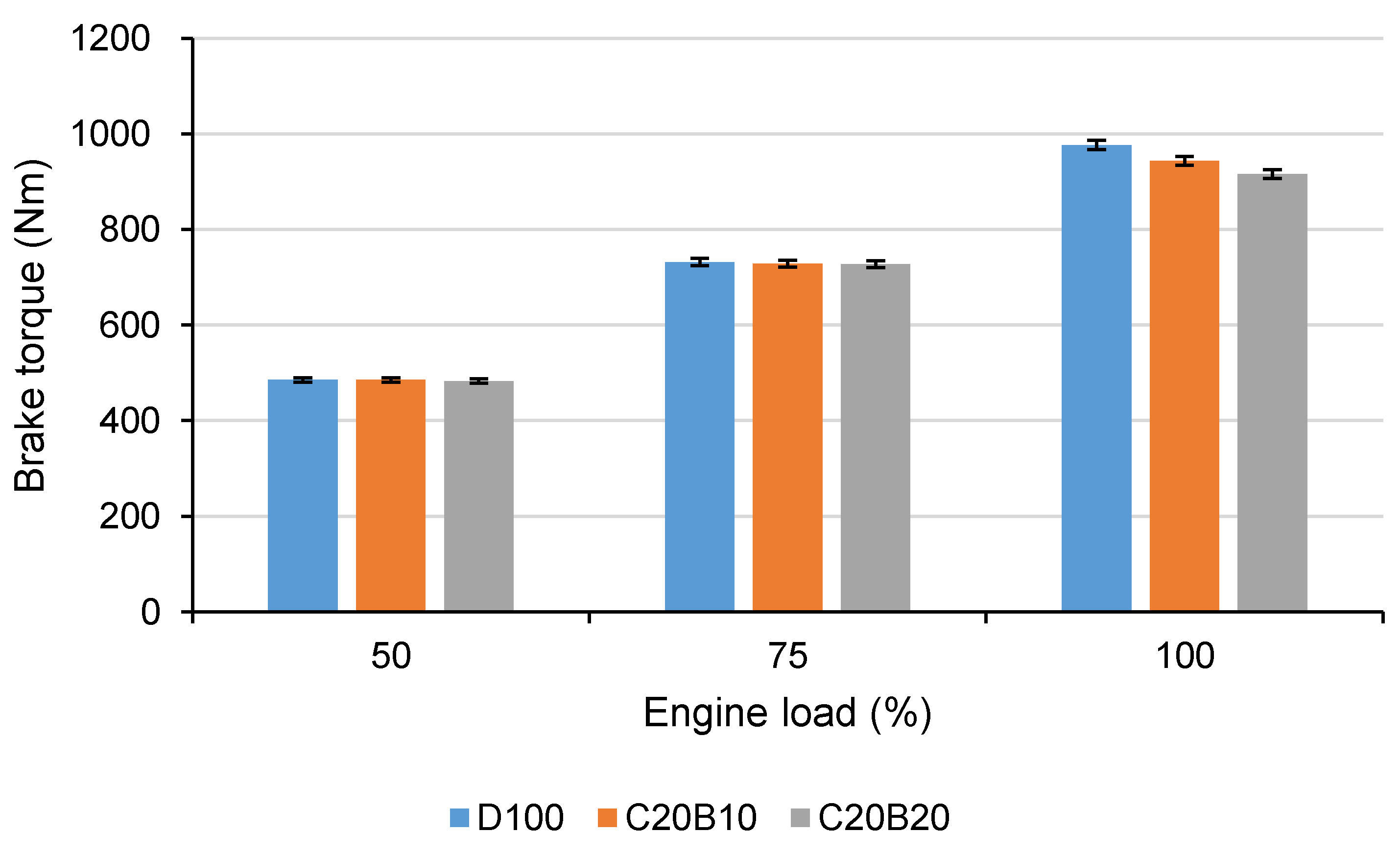
In
Figure 8, the brake torque reached at all measurement points can be seen. The displayed values were reached on the dynamometer using the PTO shaft (PTO gear ratio = 3.543). At 50% and 75% engine loads, the differences between both of the tested fuel blends and D100 are under 0.6%. At full engine load, the effect on maximum engine torque at given speeds can be seen. Fuel blend C20B10 caused a decrease in maximum torque by approximately 3.36% compared with D100 and fuel blend C20B20 by approximately 6.19%. This can be explained by the lower calorific value of blended fuels and worsened efficiency of oxidation. A decrease in engine performance was found when using various diesel fuel–vegetable oil–butanol blends [
42,
43,
45]. Machacon et al. [
30] reported decreased brake mean effective pressure when using coconut oil–diesel fuel blends, where it is explained only by the lower calorific value of the fuel blends in comparison with diesel fuel.
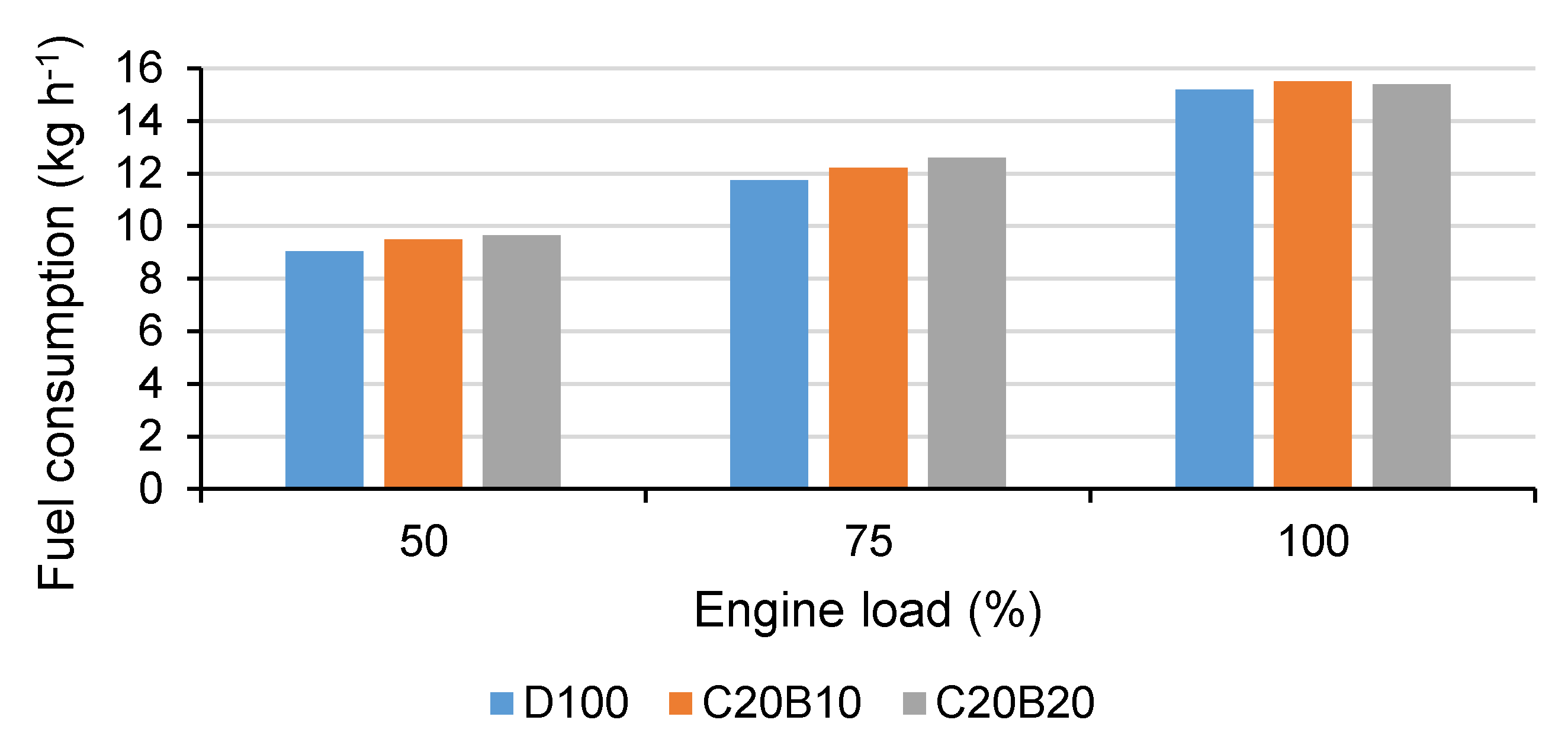
In
Figure 9, the mass fuel consumption (FC) for all tested fuels and all measured engine loads can be seen. From the figure, it is evident that both of the blended fuels caused an increase in FC at all measured engine loads. In comparison with D100, the fuel blend C20B10 caused an increase in FC of approximately 4.72%, 3.81% and 1.93% at 50%, 75% and 100% engine loads, respectively. When using fuel blend C20B20, the increase in FC was approximately 6.17%, 6.79% and 1.29% at 50%, 75% and 100% engine loads, respectively. The increase in FC can be explained above all by the lower calorific value of the blended fuels and their higher density. At 100% engine load, the main reason for the FC increase is the higher density of the blended fuels in comparison with D100, as the volumetric amount of fuel injected into the cylinder is the same at 100% engine load for all tested fuel.
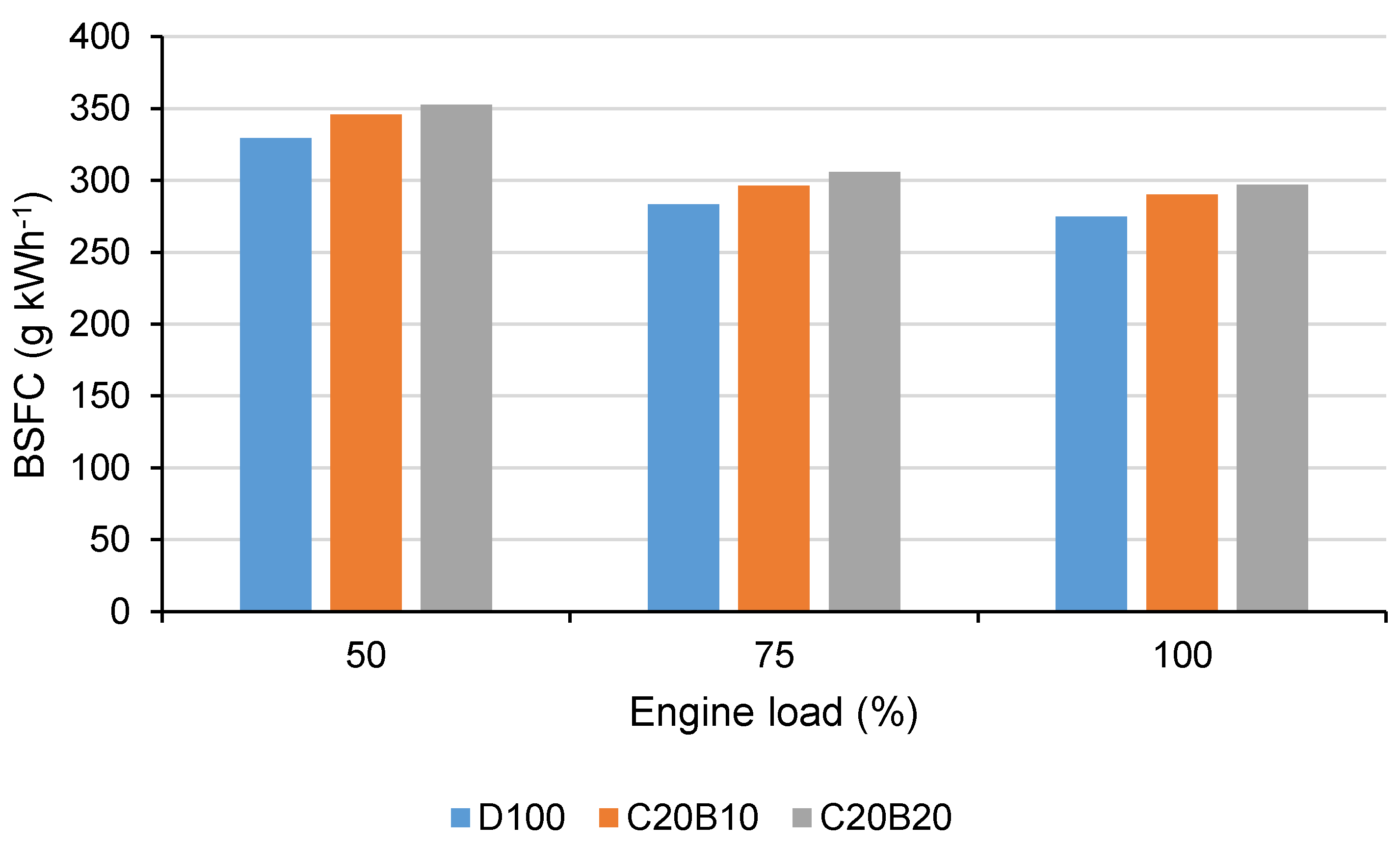
In
Figure 10, the BSFC for all tested engine loads using all tested fuels can be seen. Concerning the lower calorific value of the fuel blends in comparison with D100 and worsened efficiency of oxidation, which is evident from the emissions results, the increased BSFC can be expected. The increase in BSFC when using fuel blend C20B10 was 5% at 50% load, 4.48% at 75% engine load and 5.54% at full engine load. When using fuel blend C20B20, the BSFC increased by approximately 7% at 50% engine load, 7.92% at 75% load and 7.97% at full engine load. The increase in BSFC in comparison with diesel fuel was found using vegetable oil–diesel fuel–butanol blends [
38,
42,
43,
45,
47], coconut oil-diesel fuel blends [
30,
31,
32] and coconut oil-butanol-ethanol blends [
48]. Authors also reported worsened brake thermal efficiency when using vegetable oil-diesel fuel-butanol blends [
38,
42,
43,
45] or increased brake specific energy consumption in the case of coconut oil-diesel fuel blends [
30,
32].
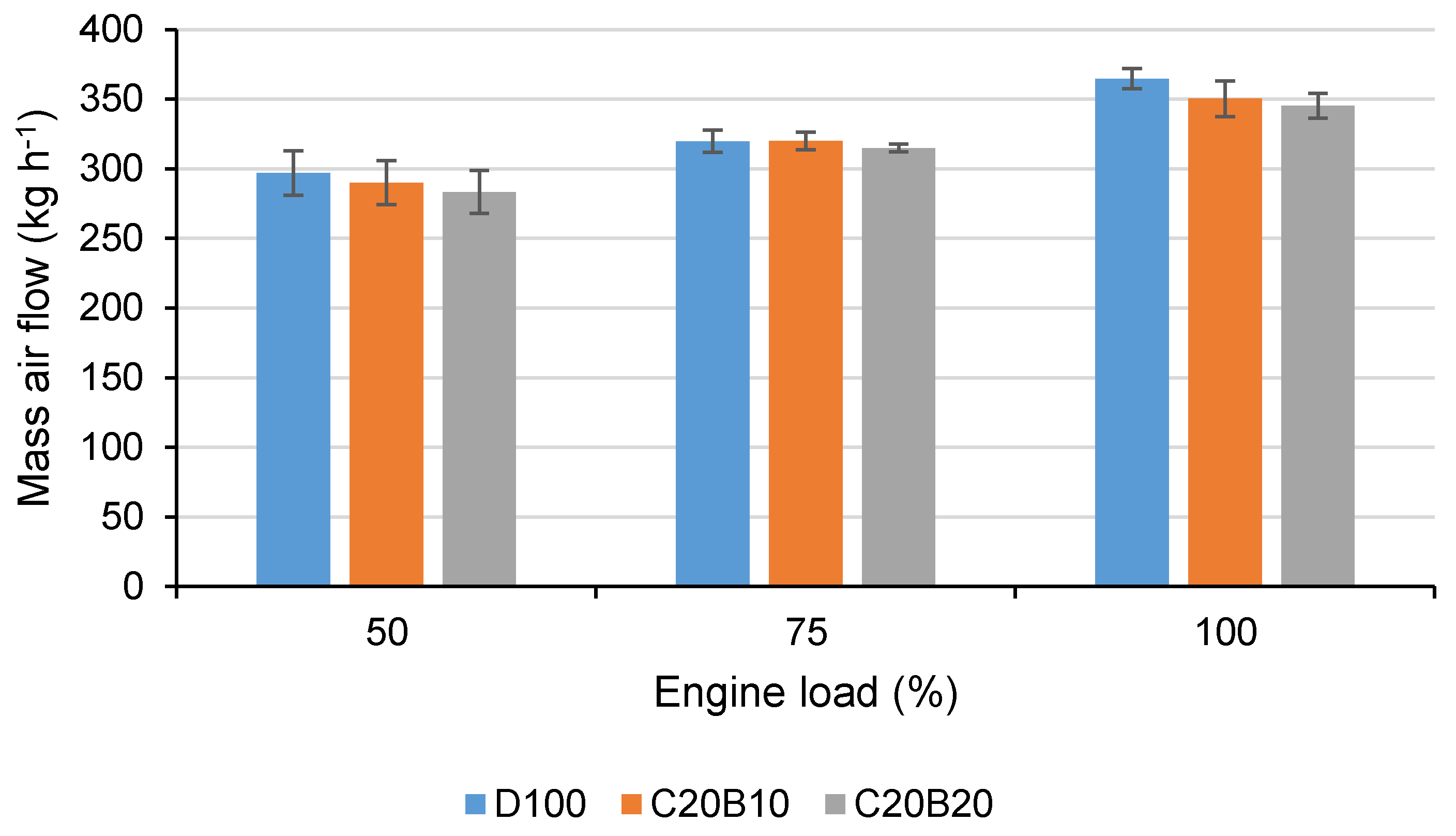
In
Figure 11, the mass air flow (MAF) through the engine for all tested fuels at all measured engine loads is shown. It can be seen that the MAF tends to decrease with increasing amounts of n-butanol in the fuel blend. The differences are relatively small, however, the highest decrease in comparison with D100 (3.93% for C20B10 and 5.3% for C20B20) can be seen at 100% engine load, where it is connected to the lower calorific value of the blended fuels.
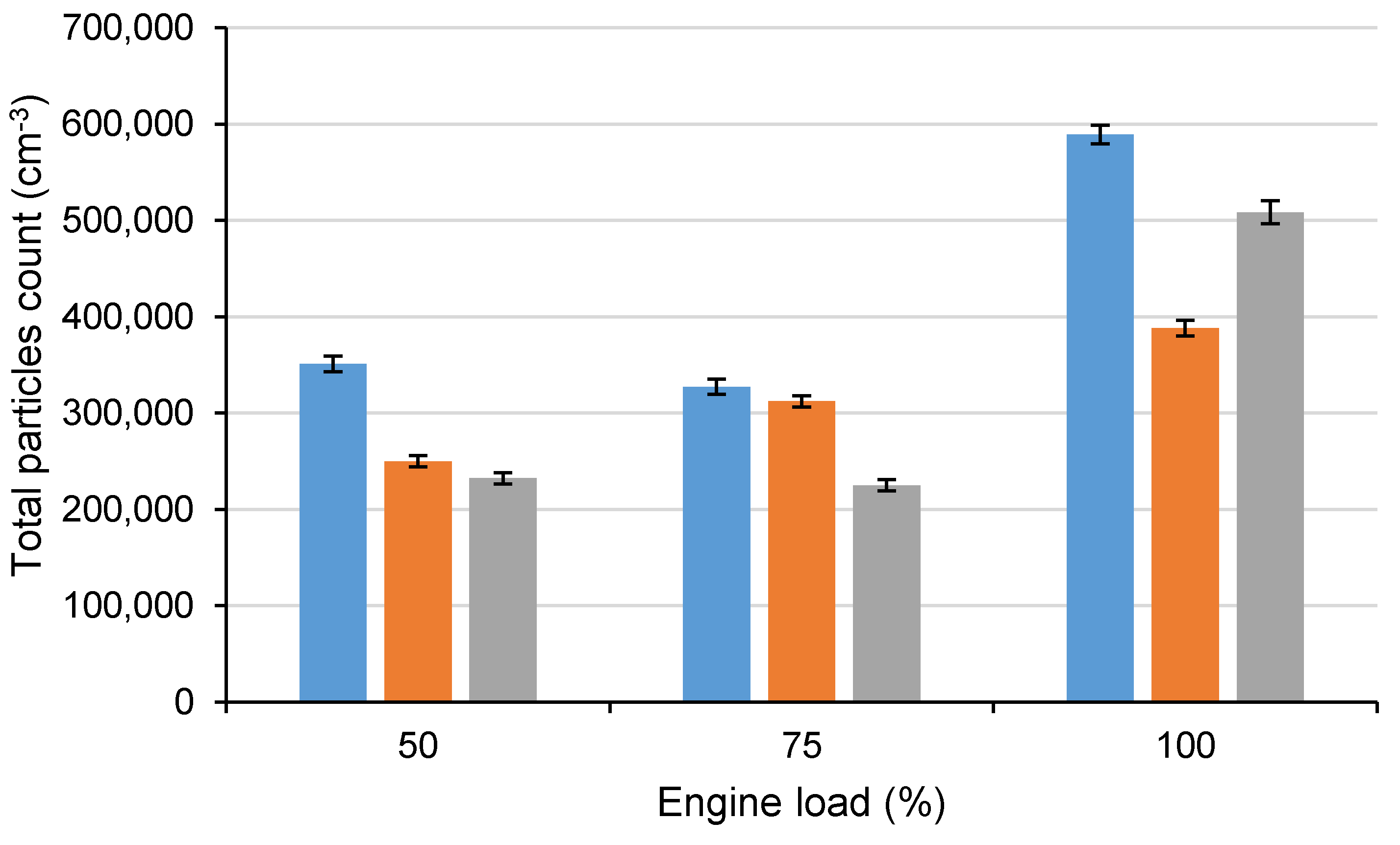
In
Figure 12, the total count of solid particles in the size range of 5.6–560 nm for all tested fuels at all measured engine loads can be seen. In comparison with D100, the total count of solid particles decreased at 50% engine load by approximately 28.81% using C20B10 and by 33.87% using blend C20B20, and at 75% engine load by 4.65% using C20B10 and by 31.29% using C20B20. At full engine load, the decrease was approximately 34.12% using the fuel blend C20B10 and 13.7% when using fuel blend C20B20. The decrease in solid particles was caused, similarly as in the case of engine smoke, mainly by the higher oxygen content in the fuel blends and the higher proportion of light fractions in the fuel blends, causing higher volatility and a faster oxidation process.
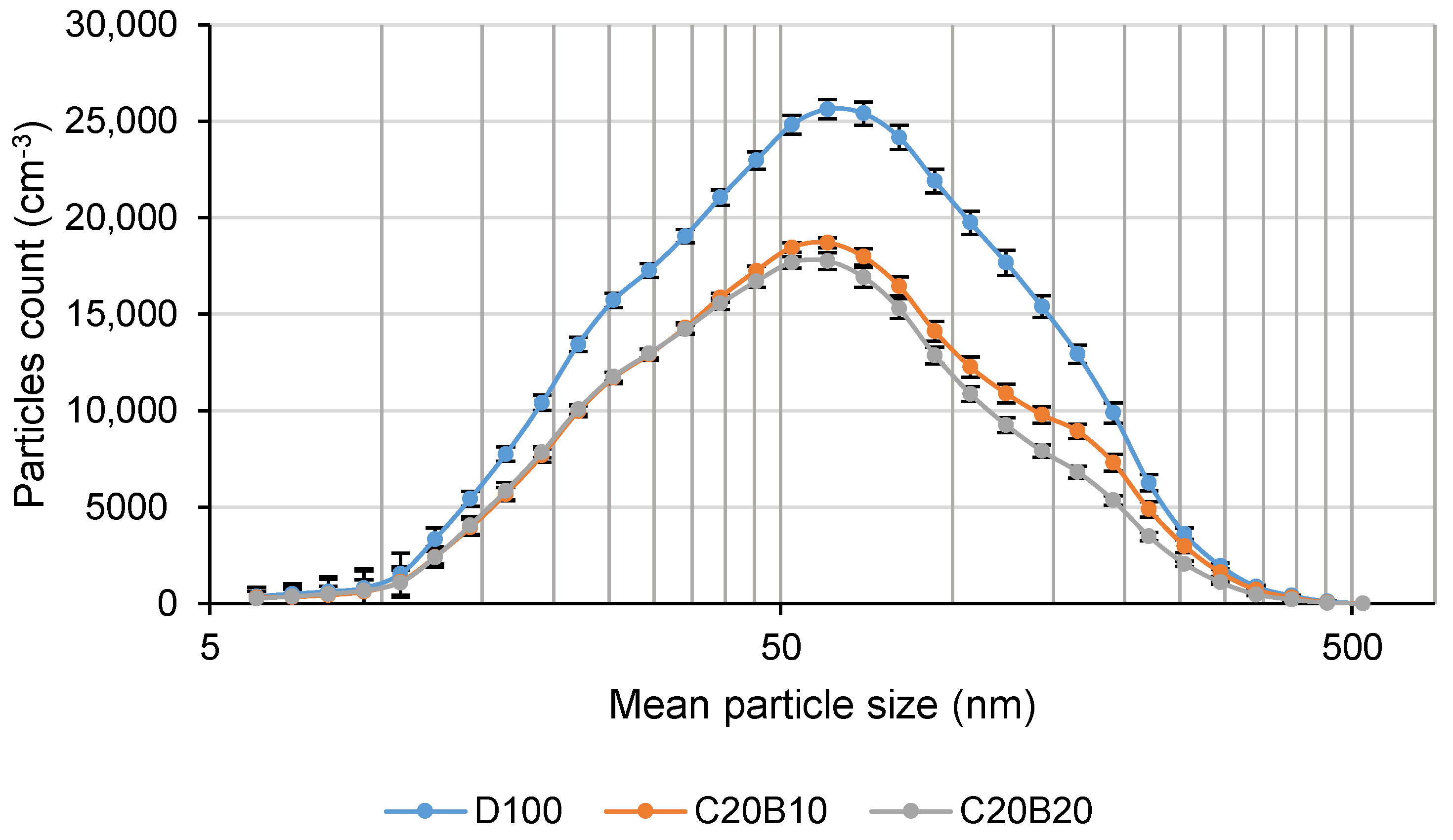
The mean size of the solid particles was calculated as the weighted average and it is shown in
Table 8. As can be seen, both of the tested fuel blends tends to decrease the size of the solid particles (except C20B10 at full engine load). The example of the size distributions of the solid particles for all tested fuels at 50% engine load is shown in
Figure 13. It is evident that the maximum concentration is reached approximately in the size range of 52–70 nm, however, from the size distribution curves, it can be seen that the differences are higher at the bigger sizes of particles (from 52 nm and higher). Geng et al. [
54] reported a lower amount of particles and their smaller mean diameter in comparison with diesel fuel with increasing proportions of n-butanol using waste cooking oil biodiesel-n-butanol blends. A lower concentration of solid particles in the range of 5.6–560 nm, except in idle engine mode, was also found using sunflower oil-diesel fuel blends and rapeseed oil–diesel fuel blends [
55].