Biological CO2-Methanation: An Approach to Standardization
Abstract
1. Introduction
2. Methodology and Motivation
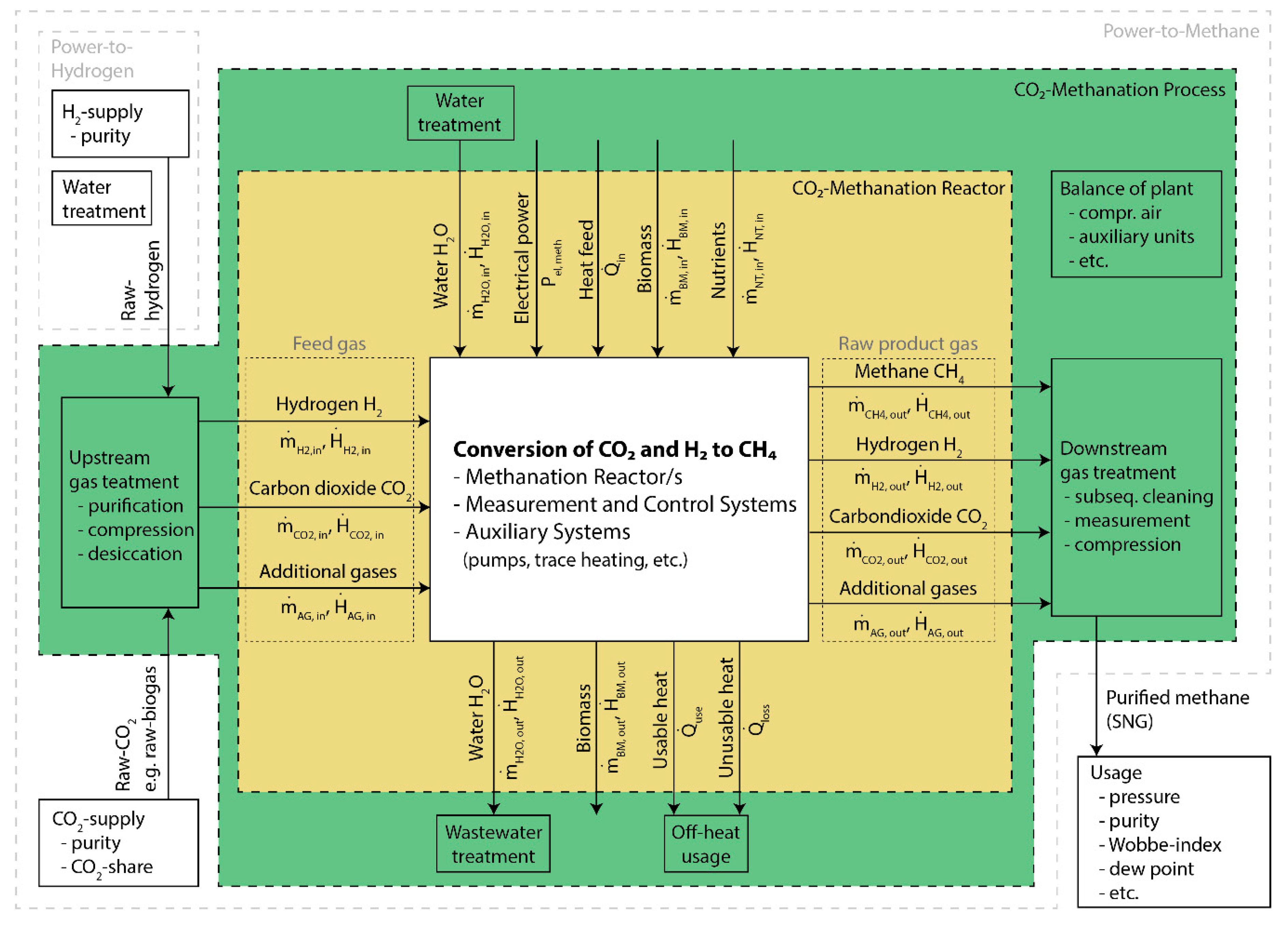
3. Power-to-Gas—Definitions and System Boundaries
3.1. Methanation System
3.1.1. CO2-Methanation Reactor
3.1.2. CO2-Methanation Process
3.2. Power-to-Hydrogen
3.3. Power-to-Methane
4. System Parameters and Characteristic Variables
- (1)
- A-Standard: detailed scientific data and background information
- (2)
- B-Standard: basic information including economic aspects
- (3)
- C-Standard: basic information on most important parameters
4.1. Standard Conditions
4.2. Feed-in Relevant Standards and Regulations
4.3. System Related Parameters
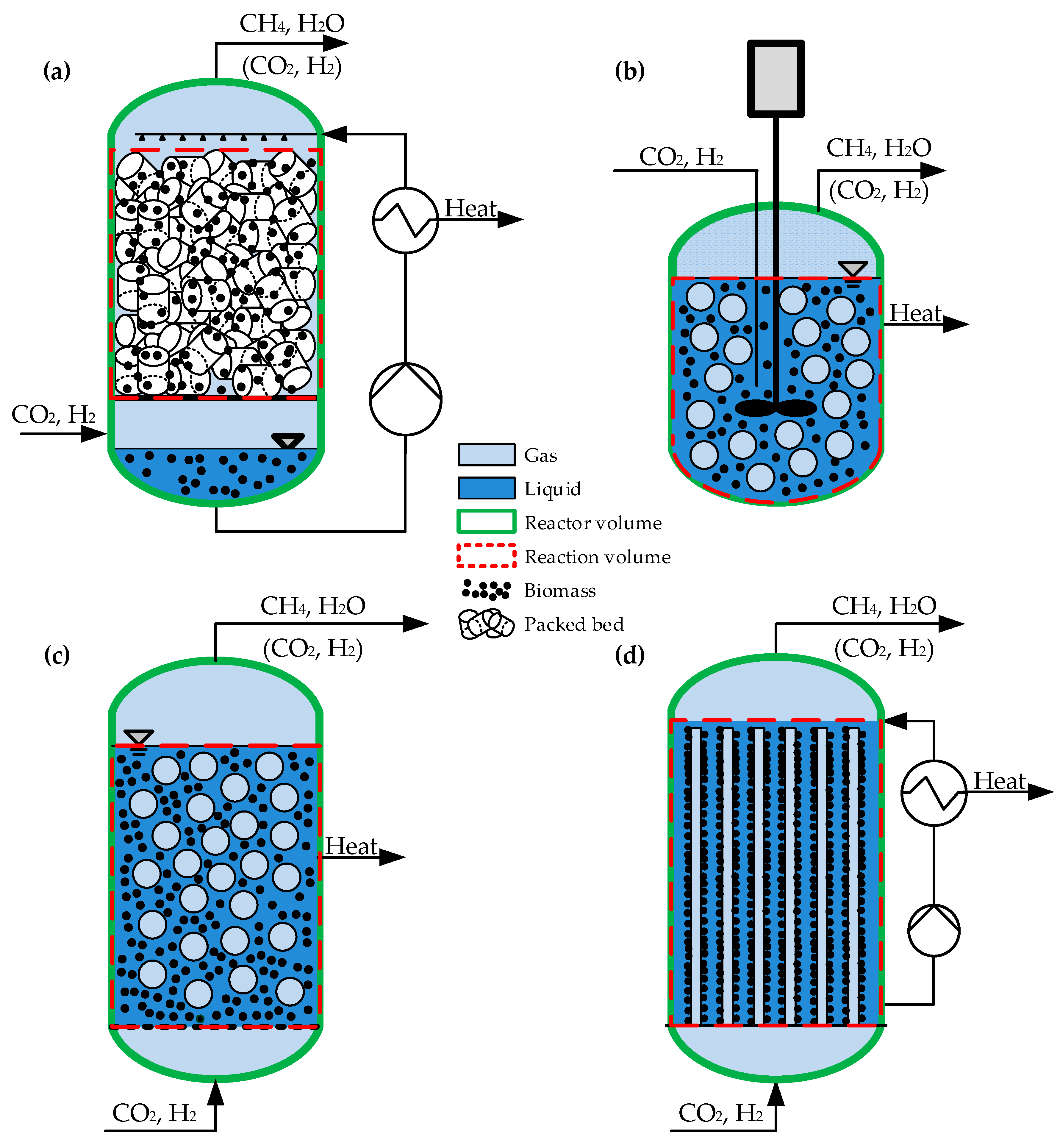
4.3.1. Reactor and Plant Type
- Reactor type (TBR, CSTR, BCR, MB, etc.; explanations are given below)
- Mode of operation (e.g., batch/fed-batch/continuous/semi-continuous) and
- Required plant components, according to the ‘CO2-Methanation process’ boundary definition,
- Potential specific characteristics of the plant/process concept e.g., co-current/counter-current mode, flow chart, etc.
4.3.2. Reactor-, Gas-, Reaction- and Liquid Volume, h/d-Ratio, Packing Volume
4.3.3. Plant Capacity, Size and Footprint
- Reactor volume VR (see Section 4.3.2)
- Nominal capacity of the methanation given by:
- ⚬
- Methane production rate in nominal point (MPR) in m³/h and related to the lower heating value MPRLHV in kW/m³ (see Section 4.4.6)
- ⚬
- The lower heating value (LHV) of the methane produced (in kW or MW). The LHVs of potential further product gas components n are not considered here but can be summed up as .
- The footprint of the methanation plant, both, absolute FPMeth in square meters (m2) and as specific area value fpMeth, where the area is normalized by the nominal capacity of the methanation plant referred to the lower heating value of the methane produced (m2/kW)
- The volume specific power input calculated as in Wh/m³
- Nominal capacity of the entire plant PN (connected electric power of the electrolyzer and methanation including all peripheral systems in kW or MW)
4.3.4. Plant Operating States
- In the ‘nominal operating state’, the plant is operating at its nominal capacity (PN,Meth).
- When the plant is operated in a ‘partial load operation state’, the load should be provided as percent of the nominal operating capacity.
- In ‘intermittent operation state’ (i.e., load following), the plant is operated in accordance to the availability of (renewable) electric power for hydrogen generation which is the essential idea behind the Power-to-Gas concepts. For this operating mode there is no specific parameter definition, but the conditions of operation should be provided, i.e., frequency and order of magnitude of load changes.
- ‘Hot standby’ (HSB) is defined as the operation state that is chosen during short breaks of operation with the possibility to change quickly back to one of the aforementioned operation states. Therefore, temperature and pressure settings in warm standby are expected to be as at nominal operating state, but the reactant gas feed is stopped.
- The plant is put in ‘cold standby’ (CSB) during longer shut down of operation. In this mode, most components are not in operation to save energy. Accordingly, the temperature is allowed to decrease to a critical value where heating is only switched on to avoid damage to the process e.g., due to freezing of liquids. System pressure and gas composition can be different from operating conditions.
- ‘Shut down’ (SD) describes the state of no operation. Only components to ensure plant safety like gas detection or fire detection are in operation.
4.3.5. Reactant and Product Gas Specification
4.4. Performance Parameters
4.4.1. Gas Hourly Space Velocity
4.4.2. Gas Retention Time
4.4.3. Hydraulic Retention Time and Liquid Recirculation
4.4.4. Gas Conversion Rate
4.4.5. Methane Yield
4.4.6. Methane Production Rate
4.4.7. Methane Production Dynamics or Load Change Rates
- Start-up time from any operating state to partial and full load (partial loads should be given as percent of the nominal operating capacity, see Section 4.3.4).
- In case of load following operation, no specific states as for the nominal operating are specified. But the reaction times should be specified together with a description of the expected load variation dynamics.
4.4.8. Methanation Efficiency
4.4.9. Specific Power Demand
4.4.10. System Availability
4.4.11. Gas and Liquid Hold-up, Effective Surface Area
4.5. Microbiology-Based Parameters
4.5.1. Methanogenic Archaea
4.5.2. Type and Origin of the Inoculum
4.5.3. Nomenclature of Species/Genera of Methanogenic Archaea
4.5.4. Biocatalyst Concentration
4.5.5. Nutrients and other Supplements
4.5.6. Reactor-related Parameters
4.6. Cost Parameters
- Building works on site or construction site preparation (e.g., foundations, fences Specification of required connections and weight of the methanation unit by provider)
- Water supply and sewage hook-up on site (definition of water quality, delivery pressure, amounts and connection size specified by supplier)
- Gas connections for methane available on site (definition of gas amounts, pressure and size of the injection station by provider)
- Connection to hydrogen and carbon dioxide source available on site
- Heating management: heating and cooling water circuit available on site
- Pressurized air available on site
- Flare available
- All project development costs (e.g., environmental impact assessments, noise insulation/protection certificates, architectural surveys, landscape planning, permits, fire protection, feasability studies, property/land costs, etc.)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| Acronym | Meaning | Units |
| A | System availability | % |
| AFA | Antifoam agents | - |
| AG | Additional gases | - |
| Al2O3 | Aluminium oxide | - |
| Cross-sectional area of the reactor | m3 | |
| Specific surface area | m3/m3 | |
| BCR | Bubble column reactor | - |
| BM | Biomass | - |
| CAPEX | Capital expenditures | €/kW |
| CH4 | Methane | - |
| Concentration of i in gas phase | mol/m3 | |
| Concentration of i in liquid phase | mol/m3 | |
| CNG | Compressed natural gas | - |
| CO2 | Carbon dioxide | - |
| Co | Cobalt | - |
| COS | Carbonyl sulfide | y |
| CSB | Cold standby | - |
| CSTR | Continuously stirred tank reactor | - |
| Reactor diameter | m | |
| Packing diameter | m | |
| DVGW | German Technical and Scientific Association for Gas and Water | - |
| Absolute energy demand of biological CO2-Methanation | kWh/d | |
| Specific power demand biol. CO2-Methanation | ||
| Specific power demand biol. CO2-Meth. during cold standby | ||
| Specific power demand biol. CO2-Meth. during hot standby | ||
| Specific power demand biol. CO2-Meth. during shut-down | ||
| Gas hold-up | - | |
| Liquid hold-up | - | |
| ηtotal | Methanation efficiency | - |
| Fe | Iron | - |
| Absolute footprint of methanation plant | m3 | |
| Footprint of methanation plant (specific area value) | m3/kW | |
| Gas hourly space velocity related to hydrogen supplied | h−1 | |
| Gas hourly space velocity related to entire reactant gas mixture | h−1 | |
| Dimensionless Henry-coefficient | - | |
| Reactor height | m | |
| Packing height | m | |
| H2 | Hydrogen | - |
| H2O | Water | - |
| H2S | Hydrogen sulfide | - |
| HSB | Hot stand-by | - |
| kL,i | Liquid mass transfer coefficient | m/s |
| Load change rate from shut down to nominal operating point | kW/s | |
| Load change rate from hot standby to nominal operating point | kW/s | |
| Load change rate from cold standby to nominal operating point | kW/s | |
| Load change rate from partial load x to nominal operating point | kW/s | |
| LHV | Lower heating value | kW or MW |
| LPSN | List of Prokaryotic Names with Standing in Nomenclature | - |
| Mass flow rate | kg s−1 | |
| MPR | Methane production rate (referred to volume) | |
| Methane production rate normalized to reactor volume | h−1 | |
| Methane production rate normalized to liquid volume | h−1 | |
| Methane production rate (referred to energy/LHV) | kW/m3 | |
| MR | Membrane reactor | - |
| N | Nitrogen | - |
| Material/molar flow rate | mol s−1 | |
| Ni | Nickel | - |
| NT | Nutrients | - |
| O2 | Oxygen | - |
| OPEX | Operational expenditure | €/kWh |
| P | Phosphor | - |
| Total nominal capacity | kW or MW | |
| Nominal capacity of methanation (gas capacity rel. to LHV) | ||
| p | (Process) Pressure | bar(a) |
| Reactor pressure at nominal point | bar(a) | |
| Volume specific power input | Wh/m3 | |
| pH | pH-Value | - |
| PtG | Power-to-Gas | - |
| PtG-CH4 | Power-to-Methane, Power-to-Gas-Methane | - |
| PtG-H2 | Power-to-Hydrogen, Power-to-Gas-Hydrogen | - |
| Liquid recirculation rate | m3/h | |
| Ratio of H2/CO2 used in the methanation process | %/% | |
| S | Sulphur | - |
| SD | Shut-down | - |
| Se | Selenium | - |
| SNG | Synthetic natural gas | - |
| STP | Standard Temperature and Pressure | - |
| - | System availability | h/a |
| tmaint | Maintenance time | h/a |
| Operation time | h/a | |
| T | (Process) Temperature | °C |
| TBR | Trickle-bed reactor | - |
| Reactor temperature at nominal point | °C | |
| Gas retention time | s | |
| Gas retention time referred to average gas flows | s | |
| Hydraulic retention time | s | |
| UHV | Upper heating value | kW or MW |
| Volumetric flow rate | m3/h | |
| Total gas inflow rate | m3/h | |
| Gas velocity | m/s | |
| Gas volume | m3 | |
| Liquid volume | m3 | |
| Packing volume | m3 | |
| Reactor volume | m3 | |
| Reaction volume | m3 | |
| Carbon dioxide conversion rate | - | |
| Hydrogen conversion rate | - | |
| Absolute methane yield related to hydrogen | - | |
| Absolute methane yield related to carbon dioxide | - | |
| Relative methane yield | - | |
| Maximum methane mole fraction | % | |
| Nominal methane mole fraction | % | |
| Hydrogen mole fraction | % | |
| Carbon dioxide mole fraction | % |
Appendix A
| Parameters | Indications from Literature | Units from Literature |
|---|---|---|
| Process temperature | K/°C [35,40] | |
| °C [33,34,36,38,42,43,44,45,46] | ||
| Process pressure | ambient [43,44,46]; atm [35,40] | |
| bar/mbar [33,36,38,45] | ||
| Methane concentration | [36,37,43,44] CH4 at outlet [33] CH4 concentrations [33] End concentrationT [38] Gas components in outlet gas [34] Methane concentration(T) [36,43,44] Methane content of the exit gas [42]; (in/of the product gasT) [37,38] | % [33,34,37,38,42,44,45,46] vol% [43] Vol.-% [36] vol.% [47]2 vol-% [54]2 |
| Reactant gas composition | Feeding gas mixture [48] Gas (inlet) composition [38,40] H2/CO2 ratio [33,38]; H2/CO2 ratio [33]; RatioT H2:CO2 [37] H2-CO2-relationship [44] H2/CO2 (gas) mixture [41,43] Loading rateT C/H [45] Mixed gas feed rate [42] v/v CO2/H2 volumetric shareT [37] | [43]; [38,42]; [41] [33] mole percent CO2, mole percent H2 [35] Nl/h [45] v/v [37,42]; %v/v [41] % vol. [39] |
| Loading rate | bR [36]; H2 [43] | [33] |
| Gas feedT [37]; Hydrogen feed rate [34] | [36] | |
| G (gas flow rate) [35] | (T) [43,44] | |
| H2 loading rate [33,43] | [37] | |
| LRH2 [33,44]; Loading rateT C/H [45] | [34] | |
| OLR (Organic loading rate) [43] | mL/h [35]; Nl/h [45] | |
| Injection rate [50]1 | [54]2 | |
| Volumetric gas loading rate [50]1 | ml/min [50]2 | |
| Hydrogen conversion rate | H2 consumption [35]; H2 conversion [33,34] H2-degradation rate [43] Removal efficiency [39] Utilisation efficiency of H2 [48] Conversion rate [50]1 | % [33,34,36,39,42,43,48] mmol/h [35]; mmol/hr [40] |
| Carbon dioxide conversion rate | analogous to Hydrogen conversion rate | analogous to Hydrogen conversion rate |
| Flow Rate (Gas) | Flow rate [43] G [40]; qin [43] Gas flow rate [35,40]; Inflow rate [41] Hydrogen feed rate [34]; Gas feedT [37] Loading rateT C/H [45] | l/h [42]; L/h [35]; Nl/h [45] ml/min [34,41] mL/hr [40]; mL/h [40] [43] |
| (Specific) Recirculation rate of the trickle medium | Liquid recycle rate [35] Medium feed rate [42] Nutrient solution circulation [100] Qfl [36]; QRV [44] Recirculation (rate) [34,43] SprinklingT [36] | m3/s [33] mL/min [35,100]; l/min [38]; ml/h [42]; ml/72 h [34] m3/(m3·d) [36] (T) [44] [43] |
| Methane yield | CH4 yield on H2 [40]; Methane yield(T) [36,43]; CH4 yield [54]1 Y [36,43]; YCH4 [42]; Y(x/CH4) [47]1 Yield [35] Growth product yield [47]1 | % [40,42] mol/mol [35] [36,43] [47]2 [54]2 |
| Gas retention time | (Average) retention time [33,43,44] [39,43,44] Gas retention time (GRT) [39,48,50]1 | h [33,34,39,44] |
| Hydraulic retention time | (Calcul. average) retention time [34,39] τ [39] | h [34,39,44] |
| Reactor volume3 | Fixed-bed volume [43]; Packed volume [100]; Volume of packed support [42]; VSV [44]; Trickle-bedT [37] Volume [33,44] Working volumeT [37] | mL [40]; l [34,36,38,41,43,44,45]; L [37,46] cm3 [35,42]; m3 [33,100] |
| Parameter | Unit | Standard | |||
|---|---|---|---|---|---|
| A | B | C | |||
| System-related Parameters | |||||
| Reactor and Plant Type | |||||
| Reactor type | - | x | x | x | |
| Required plant components (according to ‘CO2-Methanation process’ boundary) | - | x | |||
| Mode of operation | - | x | |||
| Plant/process characteristics | - | x | |||
| Reactor and Liquid Volume, h/d-Ratio, Packing Volume and Surface, Hold-up | |||||
| Reactor volume () | m3 | x | x | x | |
| Reactor height () | m | x | |||
| Reactor diameter () | m | x | |||
| h/d ratio (h/d) | - | x | |||
| Liquid volume () | m3 | x | |||
| Packing volume () | m3 | x | |||
| Packing height () | m | x | |||
| Packing diameter () | m | x | |||
| Gas volume ( | m3 | x | |||
| Reaction volume ( | m3 | x | |||
| Liquid hold-up ( | - | x | |||
| Gas hold-up () | - | x | |||
| Effective surface for gas-liquid mass transfer () | m−1 | x | |||
| Plant Size | |||||
| Nominal capacity of methanation () | kW or MW | x | x | ||
| Methanation plant footprint () | m3 | x | |||
| Normalized methanation plant footprint ( | m3/kW | x | |||
| Volume specific power input ( | Wh/m3 | x | |||
| Nominal plant capacity () | kW or MW | x | x | x | |
| Operating States (indicate at least T, p, MPR for each state) | |||||
| Nominal operating state (TN; pN; MPRN) | °C; bar(a); h−1 | x | |||
| Partial load operating state (TPL; pPL; MPRPL) | °C; bar(a); h−1 | x | |||
| Intermittent operating state (TIM; pIM; MPRIM) | °C; bar(a); h−1 | x | |||
| Hot standby (HSB) | °C; bar(a); h−1 | x | |||
| Cold standby (CSB) | °C; bar(a); h−1 | x | |||
| Shut down (SD) | °C; bar(a); h−1 | x | |||
| Reactant and Product Gas Specification | |||||
| Reactant gas mole fraction (, , ) | % | x | |||
| Maximum methane purity () | % | x | x | x | |
| Nominal methane purity () | % | x | x | x | |
| Product gas mole fraction (, ) | % | x | |||
| Contamination levels of oxygen, hydrogen sulfide, ammonia tolerated by the system | ppm or ppb | x | |||
| Thresholds of components tolerated by the methanation process (only feed gas) | - | x | |||
| Moisture content/humidity (only product gas) | x | ||||
| Performance Parameters | |||||
| Gas hourly space velocity related to entire reactant gas mixture () | h−1 | x | x | ||
| Gas hourly space velocity related to hydrogen supplied () | h−1 | x | x | ||
| Gas retention time () | s | x | x | ||
| Gas retention time () | s | x | |||
| Hydraulic retention time () | s | x | |||
| Liquid recirculation () | m3/h | x | |||
| Carbon dioxide gas conversion rate () | - | x | |||
| Hydrogen gas conversion rate () | - | x | |||
| Absolute methane yield related to hydrogen () | x | ||||
| Absolute methane yield rel. to carbon dioxide () | x | ||||
| Relative methane yield () | - | x | |||
| Methane production rate normalized to reactor volume () | h−1 | x | x | x | |
| Methane production rate relating to lower heating value of methane produced () | x | x | x | ||
| Absolute Methane production rate (MPR) | x | ||||
| Methane production rate normalized to liquid volume (MPRL) | h−1 | x | |||
| Load change rate from shut down to nominal operating point () | kW/s | x | x | ||
| Load change rate from hot standby to nominal operating point () | kW/s | x | |||
| Load change rate from cold standby to nominal operating point () | kW/s | x | |||
| Load change rate from partial load x to nominal operating point () | kW/s | x | |||
| Methanation efficiency () | % | x | x | ||
| Specific power demand () | x | ||||
| Specific power demand during hot standby () | x | ||||
| Specific power demand during cold standby () | x | ||||
| Specific power demand during shut-down () | x | ||||
| Absolute energy demand biological CO2-Methanation (EBM) | kWh/d | x | |||
| System availability top and A | h/a and % | x | |||
| Maintenance time | h/a | x | |||
| Microbiology-based Parameters | |||||
| Type and Origin of the Inoculum | |||||
| Information, whether pure/defined mixed cultures or organic material (like slurry, etc.) is used* | pc, dmc, om* | x | x | x | |
| For pure/defined mixed cultures (pc/dmc) | |||||
| Name(s) of species | - | x | x | ||
| Strain designation (incl. type strain or not) | - | x | |||
| Culture origin (culture collection no. or providing lab) | - | x | x | ||
| If own strain(s): origin of sample and enrichment procedure | - | x | |||
| For organic material (om) like sludge or slurry | |||||
| Sampling site (name and location of plant incl. date of collection) | - | x | x | ||
| Inoculation ratio | - | x | |||
| Number of methanogens in inoculum | % of all organisms | x | x | ||
| Most abundant genera/species in inoculum | gen./spec. name | x | |||
| Biocatalyst Concentration | |||||
| Cell density | cells/ml or gDW/ml | x | x | ||
| Amount of adherent cells (if biofilm) | - | x | x | ||
| Number of methanogens during operation (if sludge or slurry as inoculum) | % of all organisms | x | x | ||
| Most abundant genera/species in established reactor system (if sludge or slurry as inoculum) | - | x | |||
| Nutrients and Supplements | |||||
| Media composition | - | x | x | ||
| Additives at operation start (like reducing agent, buffers, etc.) | - | x | x | ||
| Additives during operation (like reducing agents, AFA, etc.) | - | x | |||
| Reactor-related Parameters | |||||
| Reactor/process temperature nominal () | °C | x | x | x | |
| pH-value | - | x | x | ||
| Reactor/process pressure () | bar(a) | x | x | x | |
| Rotation speed (ωST) and energy input (EST) of the stirrer (for CSTR) | rpm and kWh/d | x | |||
| Cost Parameters | |||||
| Capital expenditures (CAPEX) | €/kW | x | x | ||
| Operational expenditures (OPEX) | €/kWh | x | x | ||
| Contact Information | |||||
| Contact person biology (name, affiliation, email, phone) | x | x | x | ||
| Contact person engineering (name, affiliation, email, phone) | x | x | x | ||
References
- Masson-Delmotte, V.; Zhai, P.; Pörtner, H.O.; Roberts, D.; Skea, J.; Shukla, P.R. Summary for Policymakers. In Global Warming of 1.5 °C; IPCC: Geneva, Switzerland, 2018. [Google Scholar]
- Conference of the Parties. Paris Agreement. 2015. Available online: https://unfccc.int/resource/docs/2015/cop21/eng/l09r01.pdf (accessed on 16 April 2019).
- United Nations Framework Convention on Climate Change (UNFCCC). 24th Conference of the Parties: COP24. Available online: https://cop24.gov.pl/ (accessed on 18 December 2018).
- IRENA. Global Energy Transformation. A Roadmap to 2050. 2018. Available online: https://www.irena.org//media/Files/IRENA/Agency/Publication/2018/Apr/IRENA_Report_GET_2018.pdf (accessed on 16 April 2019).
- WRI, (n.d.). Höhe der Weltweiten Treibhausgasemissionen Nach Quellgruppe im Jahr 2014 (in Millionen Tonnen CO2-Äquivalent). Available online: https://de.statista.com/statistik/daten/studie/311844/umfrage/globale-treibhausgasemissionen-nach-quellgruppe/ (accessed on 6 September 2018).
- IEA, (n.d.). Verteilung der Energiebedingten CO2-Emissionen Weltweit Nach Sektor im Jahr 2015. Available online: https://de.statista.com/statistik/daten/studie/167957/umfrage/verteilung-der-co-emissionen-weltweit-nach-bereich/ (accessed on 16 April 2019).
- Blanco, H.; Faaij, A. A review at the role of storage in energy systems with a focus on Power to Gas and long-term storage. Renew. Sustain. Energy Rev. 2018, 81, 1049–1086. [Google Scholar] [CrossRef]
- Götz, P.; Huneke, F.; Lenck, T.; Linkenheil, C.P. Minimaler Bedarf an Langfristiger Flexibilität im Stromsystem bis 2050: Studienerweiterung; Greenpeace Energy: Berlin, Germany, 2016. [Google Scholar]
- Energiespeicher. Bedarf, Technologien, Integration, 2nd ed.; Sterner, M., Stadler, I., Eds.; Springer: Berlin, Germany, 2017; ISBN 978-3-662-48892-8. [Google Scholar]
- Thema, M.; Sterner, M.; Lenck, T.; Götz, P. Necessity and Impact of Power-to-gas on Energy Transition in Germany. Energy Procedia 2016, 99, 392–400. [Google Scholar] [CrossRef]
- Sedlacek. Untertage-Erdgasspeicherung in Deutschland: Underground Gas Storage in Germany. Erdgasspeicherung. Erdöl Erdgas Kohle 2016, 132, 409–417. [Google Scholar]
- Angenent, L.T.; Usack, J.G.; Xu, J.; Hafenbradl, D.; Posmanik, R.; Tester, J.W. Integrating electrochemical, biological, physical, and thermochemical process units to expand the applicability of anaerobic digestion. Bioresour. Technol. 2018, 247, 1085–1094. [Google Scholar] [CrossRef]
- Vereinigung der Fernleitungsnetzbetreiber Gas e.V. Netzentwicklungsplan Gas 2012–2014. FNBGas. Available online: https://www.fnb-gas.de/files/2015-01-28_nep_gas_2014.pdf (accessed on 10 April 2019).
- Newton, J. Power-to-Gas and Methanation-Pathways to a ‘Hydrogen Economy’. In Proceedings of the 14th Annual Apgtf Workshop, London, UK, 12–13 March 2014. [Google Scholar]
- Wiede, T.; Land, A. Sektorenkopplung: Amprion und Open Grid Europe Geben Power-to-Gas in Deutschland einen Schub; Amprion GmbH, Open Grid Europe: Berlin, Germany, 2018. [Google Scholar]
- Northseawindpowerhub.eu. Power to Gas: North Sea Wind Power Hub. Available online: https://northseawindpowerhub.eu/ (accessed on 16 April 2019).
- Jürgensen, L.; Ehimen, E.A.; Born, J.; Holm-Nielsen, J.B. Utilization of surplus electricity from wind power for dynamic biogas upgrading: Northern Germany case study. Biomass Bioenergy 2014, 66, 126–132. [Google Scholar] [CrossRef]
- Gas for Climate-A path to 2050. Role of Renewable Gas and the Existing Gas Infrastructure in 2050. Available online: https://gasforclimate2050.eu/news (accessed on 16 April 2019).
- Bär, K.; Mörs, F.; Götz, M.; Graf, F. Vergleich der biologischen und katalytischen Methanisierung für den Einsatz bei PtG-Konzepten. Gwf-Gas 2015, 156, 466–473. [Google Scholar]
- Götz, M.; Lefebvre, J.; Mörs, F.; McDaniel Koch, A.; Graf, F.; Bajohr, S.; Reimert, R.; Kolb, T. Renewable Power-to-Gas: A technological and economic review. Renew. Energy 2016, 85, 1371–1390. [Google Scholar] [CrossRef]
- Ghaib, K.; Ben-Fares, F.-Z. Power-to-Methane: A state-of-the-art review. Renew. Sustain. Energy Rev. 2018, 81, 433–446. [Google Scholar] [CrossRef]
- Neubert, M.; Treiber, P.; Krier, C.; Hackel, M.; Hellriegel, T.; Dillig, M.; Karl, J. Influence of hydrocarbons and thiophene on catalytic fixed bed methanation. Fuel 2017, 207, 253–261. [Google Scholar] [CrossRef]
- Neubert, M.; Hauser, A.; Pourhossein, B.; Dillig, M.; Karl, J. Experimental evaluation of a heat pipe cooled structured reactor as part of a two-stage catalytic methanation process in power-to-gas applications. Appl. Energy 2018, 229, 289–298. [Google Scholar] [CrossRef]
- Rönsch, S.; Schneider, J.; Matthischke, S.; Schlüter, M.; Götz, M.; Lefebvre, J.; Prabhakaran, P.; Bajohr, S. Review on methanation-From fundamentals to current projects. Fuel 2016, 166, 276–296. [Google Scholar] [CrossRef]
- Götz, M.; McDaniel Koch, A.; Graf, F. State of the Art and Perspectives of CO2 Methanation Process Concepts for Power-to-Gas Applications. Conference Paper. 2014. Available online: https://www.researchgate.net/publication/273139805_State_of_the_Art_and_Perspectives_of_CO2_Methanation_Process_Concepts_for_Power-to-Gas_Applications (accessed on 16 April 2019).
- Frontera, P.; Macario, A.; Ferraro, M.; Antonucci, P. Supported Catalysts for CO2 Methanation: A Review. Catalysts 2017, 7, 59. [Google Scholar] [CrossRef]
- Bailera, M.; Lisbona, P.; Romeo, L.M.; Espatolero, S. Power to Gas projects review: Lab, pilot and demo plants for storing renewable energy and CO2. Renew. Sustain. Energy Rev. 2017, 69, 292–312. [Google Scholar] [CrossRef]
- Martin, M.R.; Fornero, J.J.; Stark, R.; Mets, L.; Angenent, L.T. A Single-Culture Bioprocess of Methanothermobacter thermautotrophicus to upgrade Digester Biogas by CO2-to-CH4 Conversion with H2. Archaea 2013, 2013, 157529. [Google Scholar] [CrossRef]
- Enzmann, F.; Mayer, F.; Rother, M.; Holtmann, D. Methanogens: Biochemical background and biotechnological applications: Mini-Review. AMB Express 2018, 8, 1. [Google Scholar] [CrossRef]
- Lecker, B.; Illi, L.; Lemmer, A.; Oechsner, H. Biological hydrogen methanation-A Review. Bioresour. Technol. 2017, 245, 1220–1228. [Google Scholar] [CrossRef]
- Geppert, F.; Liu, D.; van Eerten-Jansen, M.; Weidner, E.; Buisman, C.; ter Heijne, A. Bioelectrochemical Power-to-Gas: State of the Art and Future Perspectives: Review. Focus on Bioelectrochemistry. Trends Biotechnol. 2016, 34, 879–894. [Google Scholar] [CrossRef]
- Graf, F.; Krajete, A.; Schmack, U. Techno-Ökonomische Studie zur Biologischen Methanisierung bei Power-to-Gas-Konzepten. Abschlussbericht. 2014. Available online: http://www.dvgw-innovation.web33.dvgw-sc.de/fileadmin/dvgw/angebote/forschung/innovation/pdf/g3_01_13.pdf (accessed on 16 April 2019).
- Rachbauer, L.; Voitl, G.; Bochmann, G.; Fuchs, W. Biological biogas upgrading capacity of a hydrogenotrophic community in a trickle-bed reactor. Appl. Energy 2016, 180, 483–490. [Google Scholar] [CrossRef]
- Alitalo, A.; Niskanen, M.; Aura, E. Biocatalytic methanation of hydrogen and carbon dioxide in a fixed bed bioreactor. Bioresour. Technol. 2015, 196, 600–605. [Google Scholar] [CrossRef]
- Kimmel, D.E.; Klasson, K.T.; Clausen, E.C.; Gaddy, J.L. Performance of trickle-bed bioreactors for converting synthesis gas to methane. Appl. Biochem. Biotechnol. 1991, 28–29, 457–469. [Google Scholar] [CrossRef]
- Burkhardt, M.; Tietze, M.; Jordan, I.; Behrens, J. Biologische Methanisierung im Rieselbettverfahren: Untersuchung im Technikumsmaßstab und scale-up. In Biologische Methanisierung. 2. Fachforum Biologische Methanisierung, Regensburg, Germany, 25 October 2016; Ostbayerisches Technologie-Transfer-Institut e.V., Ed.; OTTI: Regensburg, Germany, 2016; pp. 31–41. [Google Scholar]
- Strübing, D.; Huber, B.; Lebuhn, M.; Drewes, J.E.; Koch, K. Biologische Methanisierung in Rieselbettreaktoren durch Mischbiozönosen unter Thermophilen Bedingungen. In Biologische Methanisierung. 2. Fachforum Biologische Methanisierung, Regensburg, Germany, 25 October 2016; Ostbayerisches Technologie-Transfer-Institut e.V., Ed.; OTTI: Regensburg, Germany, 2016; pp. 43–53. [Google Scholar]
- Dröge, S.; Kreubel, J.; König, H. Biologische Methanisierung im Rieselstrom-Reaktor unter thermophilen Bedingungen. In Biologische Methanisierung. 1. Fachforum Biologische Methanisierung, Regensburg, Germany, 11 November 2015; Ostbayerisches Technologie-Transfer-Institut e.V., Ed.; OTTI: Regensburg, Germany, 2015; pp. 47–57. [Google Scholar]
- Dupnock, T.L.; Deshusses, M.A. High-Performance Biogas Upgrading Using a Biotrickling Filter and Hydrogenotrophic Methanogens. Appl. Biochem. Biotechnol. 2017, 183, 488–502. [Google Scholar] [CrossRef]
- Klasson, K.T.; Cowger, J.P.; Ko, C.W.; Vega, J.L.; Clausen, E.C.; Gaddy, J.L. Methane production from synthesis gas using a mixed culture of R. rubrum, M. barkeri, and M. formicicum. Appl. Biochem. Biotechnol. 1990, 24–25, 317–328. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, Z.; Lu, J.; Maekawa, T. Continuous methane fermentation and the production of vitamin B12 in a fixed-bed reactor packed with loofah. Bioresour. Technol. 2004, 92, 285–290. [Google Scholar] [CrossRef]
- Jee, H.S.; Nishio, N.; Nagai, S. Continuous CH4 Production from H2 and CO2 by Methanobacterium thermoautotrophicum in a fixed-bed reactor. J. Ferment. Technol. 1988, 66, 235–238. [Google Scholar] [CrossRef]
- Burkhardt, M.; Busch, G. Methanation of hydrogen and carbon dioxide. Appl. Energy 2013, 111, 74–79. [Google Scholar] [CrossRef]
- Burkhardt, M.; Koschack, T.; Busch, G. Biocatalytic methanation of hydrogen and carbon dioxide in an anaerobic three-phase system. Bioresour. Technol. 2015, 178, 330–333. [Google Scholar] [CrossRef]
- Ullrich, T. Einsatz der Biologischen Methanisierung für Power-to-Gas-Konzepte: Hochdruckmethanisierung von H2; Universität Hohenheim, Ostbayerisches Technologie-Transfer-Institut e.V. (OTTI): Regensburg, Germany, 2016. [Google Scholar]
- Strübing, D.; Huber, B.; Lebuhn, M.; Drewes, J.E.; Koch, K. High performance biological methanation in a thermophilic anaerobic trickle bed reactor. Bioresour. Technol. 2017, 245, 1176–1183. [Google Scholar] [CrossRef]
- Bernacchi, S.; Krajete, A.; Herwig, C. Experimental workflow for developing a feed forward strategy to control biomass growth and exploit maximum specific methane productivity of Methanothermobacter marburgensis in a biological methane production process (BMPP). AIMS Microbiol. 2016, 2, 262–277. [Google Scholar] [CrossRef]
- Porté, H.; Kougias, P.G.; Alfaro, N.; Treu, L.; Campanaro, S.; Angelidaki, I. Process performance and microbial community structure in thermophilic trickling biofilter reactors for biogas upgrading. Sci. Total Environ. 2019, 655, 529–538. [Google Scholar] [CrossRef]
- Szuhaj, M.; Acs, N.; Tengolics, R.; Bodor, A.; Rakhely, G.; Kovacs, K.L.; Bagi, Z. Conversion of H2 and CO2 to CH4 and acetate in fed-batch biogas reactors by mixed biogas community: A novel route for the power-to-gas concept. Biotechnol. Biofuels 2016, 9, 102. [Google Scholar] [CrossRef]
- Lee, J.C.; Kim, J.H.; Chang, W.S.; Pak, D. Biological conversion of CO2 to CH4 using hydrogenotrophic methanogen in a fixed bed reactor. J. Chem. Technol. Biotechnol. 2012, 87, 844–847. [Google Scholar] [CrossRef]
- Savvas, S.; Donnelly, J.; Patterson, T.; Chong, Z.S.; Esteves, S.R. Biological methanation of CO2 in a novel biofilm plug-flow reactor: A high rate and low parasitic energy process. Appl. Energy 2017, 202, 238–247. [Google Scholar] [CrossRef]
- Biologische Methanisierung; Ostbayerisches Technologie-Transfer-Institut e.V. (OTTI), Ed.; 1. Fachforum Biologische Methanisierung, Regensburg, Sorat Hotel, 11 November 2015; OTTI: Regensburg, Germany, 2015. [Google Scholar]
- Seifert, A.H.; Rittmann, S.; Herwig, C. Analysis of process related factors to increase volumetric productivity and quality of biomethane with Methanothermobacter marburgensis. Appl. Energy 2014, 132, 155–162. [Google Scholar] [CrossRef]
- Laborde, H.P. Comparison of Different Reactor Configurations for Ex-Situ Biological Biogas Upgrading. Masterarbeit; Linköping University: Linköping, Sweden, 2016. [Google Scholar]
- Friedl, M.; Meier, B.; Ruoss, F.; Schmidlin, L. Thermodynamik von Power-to-Gas; Hochschule für Technik Rapperswil: Rapperswil, Switzerland, 2016. [Google Scholar]
- Sterner, M. Bioenergy and Renewable Power Methane in Integrated 100% Renewable Energy Systems. Limiting Global Warming by Transforming Energy Systems. Ph.D. Thesis, University of Kassel, Kassel, Germany, 2009. [Google Scholar]
- Hou, P.; Enevoldsen, P.; Eichman, J.; Hu, W.; Jacobson, M.Z.; Chen, Z. Optimizing investments in coupled offshore wind -electrolytic hydrogen storage systems in Denmark. J. Power Sources 2017, 359, 186–197. [Google Scholar] [CrossRef]
- Kopp, M.; Coleman, D.; Stiller, C.; Scheffer, K.; Aichinger, J.; Scheppat, B. Energiepark Mainz: Technical and economic analysis of the worldwide largest Power-to-Gas plant with PEM electrolysis. Int. J. Hydrogen Energy 2017, 42, 13311–13320. [Google Scholar] [CrossRef]
- Muñoz, R.; Meier, L.; Diaz, I.; Jeison, D. A review on the state-of-the-art of physical/chemical and biological technologies for biogas upgrading. Rev. Environ. Sci. Biotechnol. 2015, 14, 727–759. [Google Scholar] [CrossRef]
- Lemmer, A.; Ullrich, T. Effect of Different Operating Temperatures on the Biological Hydrogen Methanation in Trickle Bed Reactors. Energies 2018, 11, 1344. [Google Scholar] [CrossRef]
- Buttler, A.; Spliethoff, H. Current status of water electrolysis for energy storage, grid balancing and sector coupling via power-to-gas and power-to-liquids: A review. Renew. Sustain. Energy Rev. 2018, 82, 2440–2454. [Google Scholar] [CrossRef]
- Glenk, G.; Reichelstein, S. Economics of converting renewable power to hydrogen. Nat. Energy 2019, 4, 216–222. [Google Scholar] [CrossRef]
- DVGW Deutscher Verein des Gas- und Wasserfaches e. V. DVGW G 260 (A) Gasbeschaffenheit; Wirtschafts- und Verlagsgesellschaft Gas und Wasser mbH: Bonn, Germany, 2013. [Google Scholar]
- Swiss Gas and Water Industry Association (SVGW). Richtlinie für die Einspeisung von Erneuerbaren Gasen. SVGW G 13, 2016 (G13). Available online: https://epaper.svgw.ch/Epaper/Render/Download/?editionId=279ae750-a781-e711-80d8-001dd8b729e1 (accessed on 16 April 2019).
- Österreichische Vereinigung für das Gas- und Wasserfach (ÖVGW). National Gas in Austria-Gas Quality; Österreichische Vereinigung für das Gas- und Wasserfach (ÖVGW): Wien, Austria, 2001; (ÖVGW G 31). [Google Scholar]
- Deutsches Institut für Normung e.V. Referenzzustand, Normzustand, Normvolumen; Begriffe und Werte. Reference conditions, Normal Conditions, Normal Volume; Concepts and Values; Beuth Verlag GmbH: Berlin, Germany, 1990. [Google Scholar]
- Deutsches Institut für Normung e.V. DIN EN ISO 13443 Natural Gas-Standard Reference Conditions. Erdgas-Standardbezugsbedingungen; Beuth Verlag GmbH: Berlin, Germany, 2007. [Google Scholar]
- International Organization for Standardization. Petroleum Liquids and Liquefied Petroleum Gases-Measurement-Standard Reference Conditions, 75.180.30; ISO 5024; International Organization for Standardization: Geneva, Switzerland, 1999. [Google Scholar]
- DIN Deutsches Institut für Normung e.V. Natural Gas-Calculation of Calorific Values, Density, Relative Density and Wobbe Indices from Composition; Beuth Verlag GmbH: Berlin, Germany, 2016; 75.060 (DIN EN ISO 6976). [Google Scholar]
- Deutsches Institut für Normung e.V. Standard Atmosphere. Normatmosphäre; Beuth Verlag GmbH: Berlin, Germany, 1979; 551.51 (DIN ISO 2533). [Google Scholar]
- Altfeld, K.; Pinchbeck, D. Admissible hydrogen concentrations in natural gas systems: There are proposals to inject hydrogen (H2) from renewable sources in the natural gas network. This measure would allow the very large transport and storage capacities of the existing infrastructure, particularly high-pres- sure pipelines, to be used for indirect electricity transport and storage. Gas Energy 2013, 2013, 1–12, Reprint. [Google Scholar]
- European Association for the Streamlining of Energy Exchange-gas (EASEE-gas). Harmonization of Natural Gas Quality. Common Business Practice. 2005-001/02. 2008. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=2ahUKEwjuoo311O3fAhVRKFAKHdNaDRQQFjAAegQICRAC&url=https%3A%2F%2Feasee-gas.eu%2Fdownload_file%2FDownloadFile%2F4%2Fcbp-2005-001-02-harmonisation-of-natural-gas-quality&usg=AOvVaw0najV_hNvzYHY6HmfdWbbh (accessed on 15 January 2019).
- European Commission. Gas Quality Harmonisation. 2019. Available online: https://ec.europa.eu/energy/en/topics/markets-and-consumers/wholesale-market/gas-quality-harmonisation (accessed on 15 January 2019).
- Gikopoulos, C. Einspeisung von Biogas in ein Erdgasnetz. Qualitätserfordernisse aus der Sicht eines Gasnetzbetreibers. Gas&Wärme Energie Steiermark. Available online: http://www.lev.at/Download/Int_Biogas_Ex_StT1/Biogaseinspeisung_Gikopoulos.pdf (accessed on 16 April 2019).
- Bryant, N.; Paxton, A. Gas Quality Harmonisation Cost Benefit Analysis. Final report. 2012. Available online: https://ec.europa.eu/energy/sites/ener/files/documents/2012_gas_quality_harmonisation_cost_benefit_analysis_.pdf (accessed on 15 January 2019).
- Van Renssen, S.; Edwardes-Evans, H. Power in Europe: French prompt surges into 2017. S&P Glob. Platts 2017, 5–8. Available online: http://www.itm-power.com/wp-content/uploads/2017/01/platts-itm-article-Januart-2016.pdf (accessed on 26 April 2019).
- Dansk Gasteknisk Center a/s. Kontrolmanual til Måling af Biogas og Bionaturgas. Naturgasselskabernes Kontrolmanual til Måling af Biogas og Bionaturgas. Kontrolmanual 3. Ugdave. 2015. Available online: https://www.dgc.dk/sites/default/files/filer/publikationer/M02_kontrolmanual_bionaturgas_0.pdf (accessed on 29 January 2019).
- Bekendtgørelse om gasreglementets afsnit C-12, bestemmelser om gaskvaliteter. BEK nr 1264 af 14/12/2012. 2012. Available online: https://www.retsinformation.dk/Forms/R0710.aspx?id=144715 (accessed on 16 April 2019).
- DVGW Deutscher Verein des Gas- und Wasserfaches e. V. DVGW G 262 (A) Nutzung von Gasen aus regenerativen Quellen in der öffentlichen Gasversorgung; Wirtschafts- und Verlagsgesellschaft Gas und Wasser mbH: Bonn, Germany, 2011; G 262 (A). [Google Scholar]
- Berger, R. Development of Business Cases for Fuel Cells and Hydrogen Applications for Regions and Cities. Hydrogen Injection into the Natural Gas Grid; Roland Berger: Munich, Germany, 2017. [Google Scholar]
- Maćkowiak, J. Modellierung des flüssigkeitsseitigen Stoffüberganges in Kolonnen mit klassischen und gitterförmigen Füllkörpern. Chemie Ingenieur Technik 2008, 80, 57–77. [Google Scholar] [CrossRef]
- Chemical Reactors for Gas-Liquid Systems, 1st ed.; Kaštánek, F., Sharp, D.H., Eds.; Ellis Horwood: New York, NY, USA, 1993. [Google Scholar]
- Baerns, M.; Hofmann, H.; Renken, A. Chemische Reaktionstechnik. Lehrbuch der Technischen Chemie-Band 1. Mit 41 Tabellen, 3rd ed.; Thieme: Stuttgart, Germany, 1999; ISBN 313687501X. [Google Scholar]
- Wanner, O.; Debus, O.; Reichert, P. Modelling the spatial distribution and dynamics of a xylene-degrading microbial population in a membrane-bound biofilm. Water Sci. Technol. 1994, 29, 243–251. [Google Scholar] [CrossRef]
- Shen, Y.; Brown, R.; Wen, Z. Syngas fermentation of Clostridium carboxidivoran P7 in a hollow fiber membrane biofilm reactor: Evaluating the mass transfer coefficient and ethanol production performance. Biochem. Eng. J. 2014, 85, 21–29. [Google Scholar] [CrossRef]
- Lee, K.-C.; Rittmann, B.E. A novel hollow-fibre membrane biofilm reactor for autohydrogenotrophic denitrification of drinking water. Water Sci. Technol. 2000, 41, 219–226. [Google Scholar] [CrossRef]
- Biegger, P.; Medved, A.R.; Lehner, M.; Ebner, H.M.; Friedacher, A. Methanisierung im Umfeld von “Power to Gas”. In Kurzfassungsband zum 14. Symposium Energieinnovation, Graz; Verlag der Technischen Universität: Graz, Austria, 2016. [Google Scholar]
- Hertwig, K.; Martens, L.; Hamel, C. Chemische Verfahrenstechnik. Berechnung, Auslegung und Betrieb Chemischer Reaktoren, 3rd ed.; De Gruyter Inc.: Berlin, Germany; Boston, MA, USA, 2018; ISBN 9783110500998. [Google Scholar]
- Electrochaea.dk ApS. Power-to-Gas via Biological Catalysis (P2G-BioCat). Available online: http://biocat-project.com (accessed on 14 December 2018).
- Burkhardt, M.; Buschmann-Kosel, J.; Tietze, M.; Hilse, H. Biologische Methanisierung im Rieselbettverfahren: Leistungs- und Flexibilitätsnachweis im Technikumsmaßstab. Energie Wasser Praxis 2018, 8, 42–45. [Google Scholar]
- Woese, C.R.; Kandler, O.; Wheelis, M.L. Towards a natural system of organisms: Proposal for the domains Archaea, Bacteria, and Eucarya. Proc. Natl. Acad. Sci. USA 1990, 87, 4576–4579. [Google Scholar] [CrossRef]
- Hedderich, R.; Whitman, W.B. Physiology and Biochemistry of the Methane-Producing Archaea. In The Prokaryotes, 4th ed.; Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., Thompson, F.L., Eds.; Springer: Berlin, Germany, 2013–2014; pp. 635–662. ISBN 978-3-642-30140-7.
- Euzéby, J.P. List of Bacterial Names with Standing in Nomenclature: A folder available on the Internet. Int. J. Syst. Bacteriol. 1997, 47, 590–592. [Google Scholar] [CrossRef]
- Parte, A.C. LPSN-list of prokaryotic names with standing in nomenclature. Nucleic Acids Res. 2014, 42, D613–D616. [Google Scholar] [CrossRef]
- Parte, A.C. LPSN-List of Prokaryotic names with Standing in Nomenclature (bacterio.net), 20 years on. Int. J. Syst. Evol. Microbiol. 2018, 68, 1825–1829. [Google Scholar] [CrossRef]
- Bacterio.net. List of Procariotic Names with Standing in Nomenclature: LPSN. Available online: http://www.bacterio.net/ (accessed on 14 December 2018).
- Wasserfallen, A.; Nölling, J.; Pfister, P.; Reeve, J.; Conway de Macario, E. Phylogenetic analysis of 18 thermophilic Methanobacterium isolates supports the proposals to create a new genus, Methanothermobacter gen. nov., and to reclassify several isolates in three species, Methanothermobacter thermautotrophicus comb. nov., Methanothermobacter wolfeii comb. nov., and Methanothermobacter marburgensis sp. nov. Int. J. Syst. Evol. Microbiol. 2000, 50 Pt 1, 43–53. [Google Scholar] [CrossRef]
- Validation of publication of new names and new combinations previously effectively published outside the IJSEM. Int. J. Syst. Evol. Microbiol. 2002, 52, 685–690. [CrossRef]
- Vardar-Sukan, F. Foaming: Consequences, prevention and destruction. Biotechnol. Adv. 1998, 16, 913–948. [Google Scholar] [CrossRef]
- Rachbauer, L.; Beyer, R.; Bochmann, G.; Fuchs, W. Characteristics of adapted hydrogenotrophic community during biomethanation. Sci. Total Environ. 2017, 595, 912–919. [Google Scholar] [CrossRef]
| Parameters | Indications from Literature | Units from Literature |
|---|---|---|
| Methane production rate | CH4 production rate [39] CH4 production [35,40] MBR [36] Methane formation [41] Methane evolution rate MER [47] Methane formation rateT [36,37,38] Methane productivity [33,34,35,48] Methane production rate [41,42,48] P [43] [33,44,48] Specific methane production [43,44] | l/(l*d) [45]; [34] l/l-reactor/h [41]; [49]2 [42] [39]; [46]; [33]; [44]; [50]2; v/v/d [51]2 ml/h [42]; ml/min [52]2; mmol/h [35]; mmol/hr [40] [35,40]; [53]2; [47]2; [37] Nl/h per m3 fixed bed vol.T [38] [36]; (T) [43,44] [48,54]2 |
| System Boundaries and Components | CO2-Methanation Reactor | CO2-Methanation Process | Power-to-Hydrogen | Power-to-Methane |
|---|---|---|---|---|
| Upstream gas treatment | - | x | x | x |
| H2-supply (e.g., electrolysis) | - | - | x | x |
| Water treatment | - | x | x | x |
| Conversion of CO2 and H2 to CH4 | x | x | - | x |
| Off-heat usage | - | x | - | x |
| Wastewater treatment | - | x | - | x |
| Downstream gas treatment | - | x | - | x |
| Standard, Title | Pressure | Temperature | |
|---|---|---|---|
| DIN 1343 | Physikalischer Normzustand; STP | pn = 101,325 Pa (=1.01325 bar) | Tn = 273.15 K (tn = 0 °C) |
| DIN EN ISO 13443 | Natural gas—Standard reference conditions | 1.013250 bar | 0 °C and 288.15 K (15 °C) |
| ISO 5024 | Petroleum liquids and gases; Measurement; Standard reference conditions | 101.325 kPa | 288.15 K (15 °C) |
| DIN ISO 2533 | Standard Atmosphere | 1.013250 bar | 288.15 K1 (15 °C) |
| Country | In SNG | In grid (d, t) | References | |||||
|---|---|---|---|---|---|---|---|---|
| CH4 % | CO2 % | CO2 % | H2 % | H2S mg/m3 | Wobbe-Index WI | |||
| Lower WI MJ/ms3 | Upper WI MJ/ms3 | |||||||
| Austria | - | 2 | 2 | 4 | 5 | 45.42 | 53.62 | [65,74,75] |
| Belgium | - | 2 | 2 | 0 | 5** | 46.61 | 53.90 | [14,75,76] |
| Bulgaria | - | 1 | 1 | - | 2 | - | - | [75] |
| Canada | - | - | - | 4 | - | - | - | [76] |
| Czech Republic | - | 3 | 3 | 2 | 2 | 45.70 | 52.20 | [75,76] |
| Denmark | - | 3.0 | 3.0d/2.5t | - | 5** | 48.19 | 52.93 | [75,77,78] |
| France | - | 2.5 | 2.5 | 6 | 5** | 46.47 | 53.48 | [14,75,76] |
| EASEE-gas | - | - | 2.5 | - | 5** | 46.45 | 53.99 | [75] |
| Estonia | - | - | 1.5* | - | - | 46.65 | 47.31 | [75] |
| Germany | > 95 | 6 | - | 2/101 | 5 | 43.62 | 53.46 | [63,75,76,79] |
| Greece | - | 3 | 3 | - | 5.4 | 44.29 | 55.32 | [75] |
| Hungary | - | - | - | - | 20 | 43.71 | 53.57 | [75] |
| Ireland | - | 2 | 2 | - | 5 | 47.20 | 51.41 | [75] |
| Italy | - | 3 | 3 | 0.5 | 6.6 | 47.31 | 52.33 | [75,76] |
| Latvia | - | - | - | - | 20 | 39.06 | 51.67 | [75] |
| Luxembourg | - | - | - | - | - | 46.45 | 53.99 | [75] |
| Netherlands | - | - | - | 12 | 5 | 41.23 | 42.13 | [75,76] |
| New Zealand | - | - | - | 0 | - | - | - | [76] |
| Poland | - | 3 | - | - | 7 | - | - | [75] |
| Portugal | - | - | - | - | 5 | 45.70 | 54.70 | [75] |
| Romania | - | 8 | 8 | - | 6.8 | - | - | [75] |
| Slovakia | - | 3 | 3 | - | 2 | - | - | [75] |
| Slovenia | - | 1.575 | 1.575 | - | 6.3 | - | - | [75] |
| Spain | - | 2.5 | 2.5 | 5 | 15** | 45.65 | 54.70 | [75,76] |
| Sweden | - | - | - | 0.5 | 5 | 43.73 | 53.60 | [14,75,76,80] |
| Switzerland | > 96 | - | - | 4 | - | - | - | [64,76] |
| United Kingdom | - | - | - | 0.1 | 5 | 47.20 | 51.41 | [75,76] |
| Parameter | Unit | Trickle-Bed Reactor (TBR) | Continuous Stirred Tank Reactor (CSTR) | Bubble Column Reactor (BCR) | Membrane Reactor (MR) |
|---|---|---|---|---|---|
| Gas hold-up | - | 0.75–0.98 | 0.05–0.3 | 0.02–0.4 | |
| Liquid hold-up1 | - | 0.5–0.2 | 0.7–0.95 | 0.7–0.95 | |
| Effective surface area | m−1 | 60–640 | 100–1500 | 100–1000 | 70–180 |
| Mass transfer coefficient | m/s | 0.4–210−4 | 0.3–410−4 | 1–410−4 | 1–1010−4 |
| Volume specific power input | Wh/m3 | 4.3 | 50 | 12.5–15.6 |
| Feed/Reactant Gas | Product Gas |
|---|---|
| Thresholds of components tolerated by the methanation process | Moisture content/humidity (mg/m3) |
| Mole fraction , , in % | Maximum methane conc. in % Nominal methane conc. in % Mole fraction , in % |
| Contamination levels of oxygen, hydrogen sulfide, ammonia | |
| (Process) Pressure p in bar(a) | |
| (Process) Temperature T in °C1 | |
| CAPEXLHV in €/kW | OPEXLHV in €/kWh | |
|---|---|---|
| Fixed Operating Costs | Variable Operating Costs | |
| Development and construction costs Material costs Production costs Transport costs Test and commissioning costs Costs for authorization plans Power- and water connection (transformer, etc.) Gas conditioning (if needed) | Personnel costs Catalyst costs First fill Commissioning Media and nutrients | Costs for hydrogen and carbon dioxide (or e.g., biogas) Electricity costs for methanation Costs for heating/cooling of methanation and gas processing Costs for other gases (e.g., nitrogen, calibration gases) Water supply and wastewater management Further operating resources Costs for repair, maintenance and spare parts |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thema, M.; Weidlich, T.; Hörl, M.; Bellack, A.; Mörs, F.; Hackl, F.; Kohlmayer, M.; Gleich, J.; Stabenau, C.; Trabold, T.; et al. Biological CO2-Methanation: An Approach to Standardization. Energies 2019, 12, 1670. https://doi.org/10.3390/en12091670
Thema M, Weidlich T, Hörl M, Bellack A, Mörs F, Hackl F, Kohlmayer M, Gleich J, Stabenau C, Trabold T, et al. Biological CO2-Methanation: An Approach to Standardization. Energies. 2019; 12(9):1670. https://doi.org/10.3390/en12091670
Chicago/Turabian StyleThema, Martin, Tobias Weidlich, Manuel Hörl, Annett Bellack, Friedemann Mörs, Florian Hackl, Matthias Kohlmayer, Jasmin Gleich, Carsten Stabenau, Thomas Trabold, and et al. 2019. "Biological CO2-Methanation: An Approach to Standardization" Energies 12, no. 9: 1670. https://doi.org/10.3390/en12091670
APA StyleThema, M., Weidlich, T., Hörl, M., Bellack, A., Mörs, F., Hackl, F., Kohlmayer, M., Gleich, J., Stabenau, C., Trabold, T., Neubert, M., Ortloff, F., Brotsack, R., Schmack, D., Huber, H., Hafenbradl, D., Karl, J., & Sterner, M. (2019). Biological CO2-Methanation: An Approach to Standardization. Energies, 12(9), 1670. https://doi.org/10.3390/en12091670






