An Innovative Procedure to Evaluate the Hydrogen Diffusion Coefficient in Metals from Absorption Measurements
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Hydrogen Absorption Kinetics Measurements
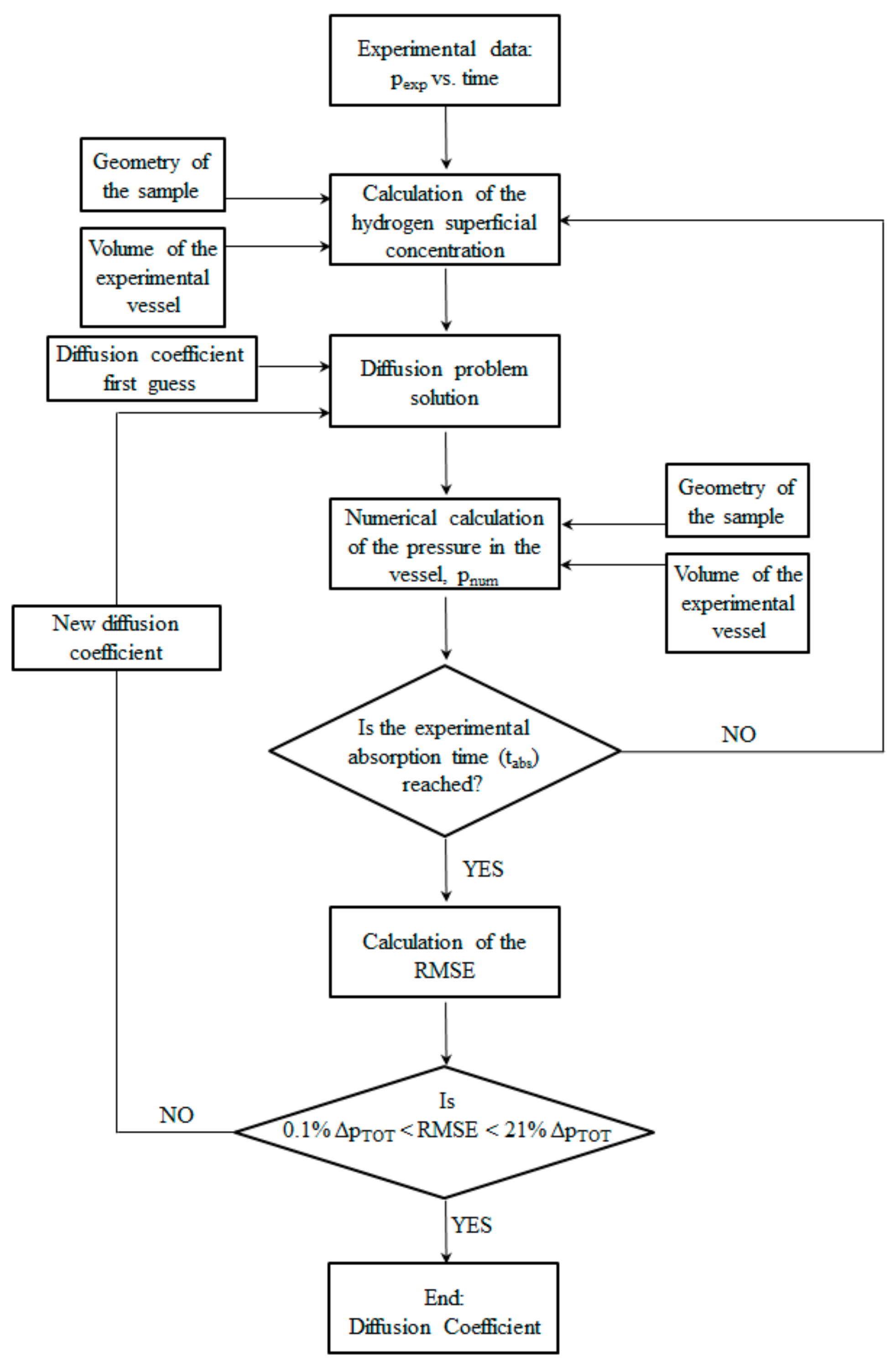
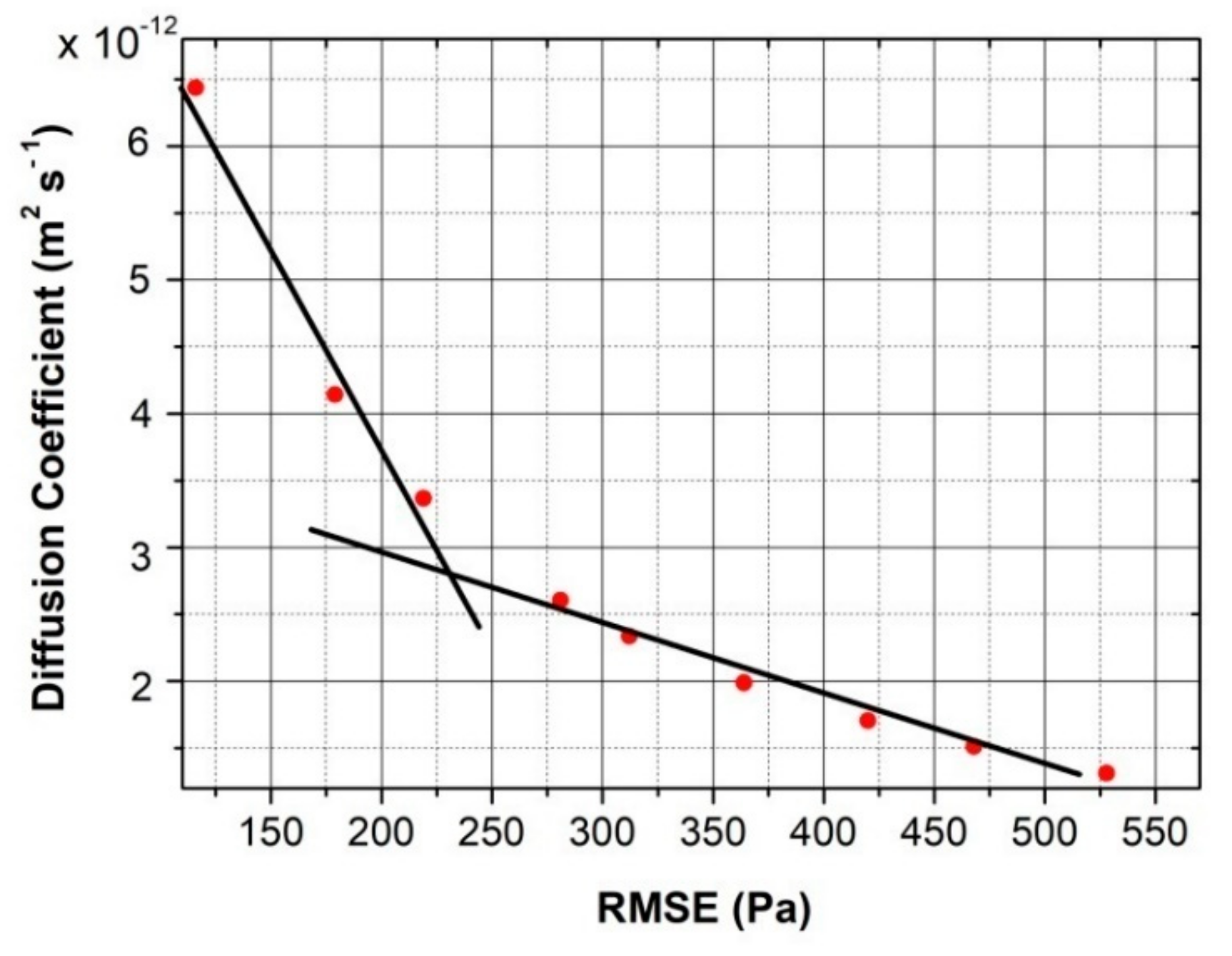
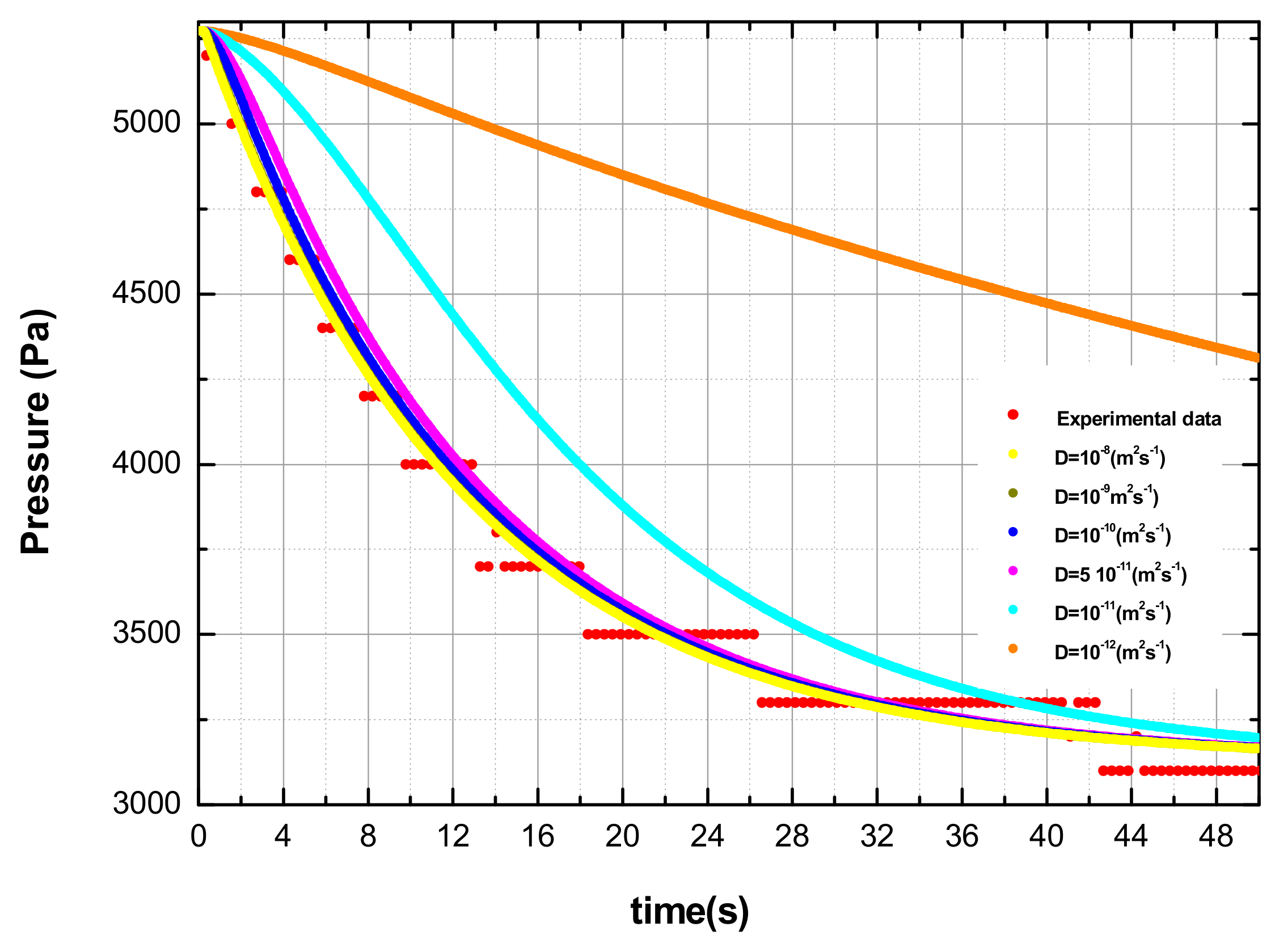
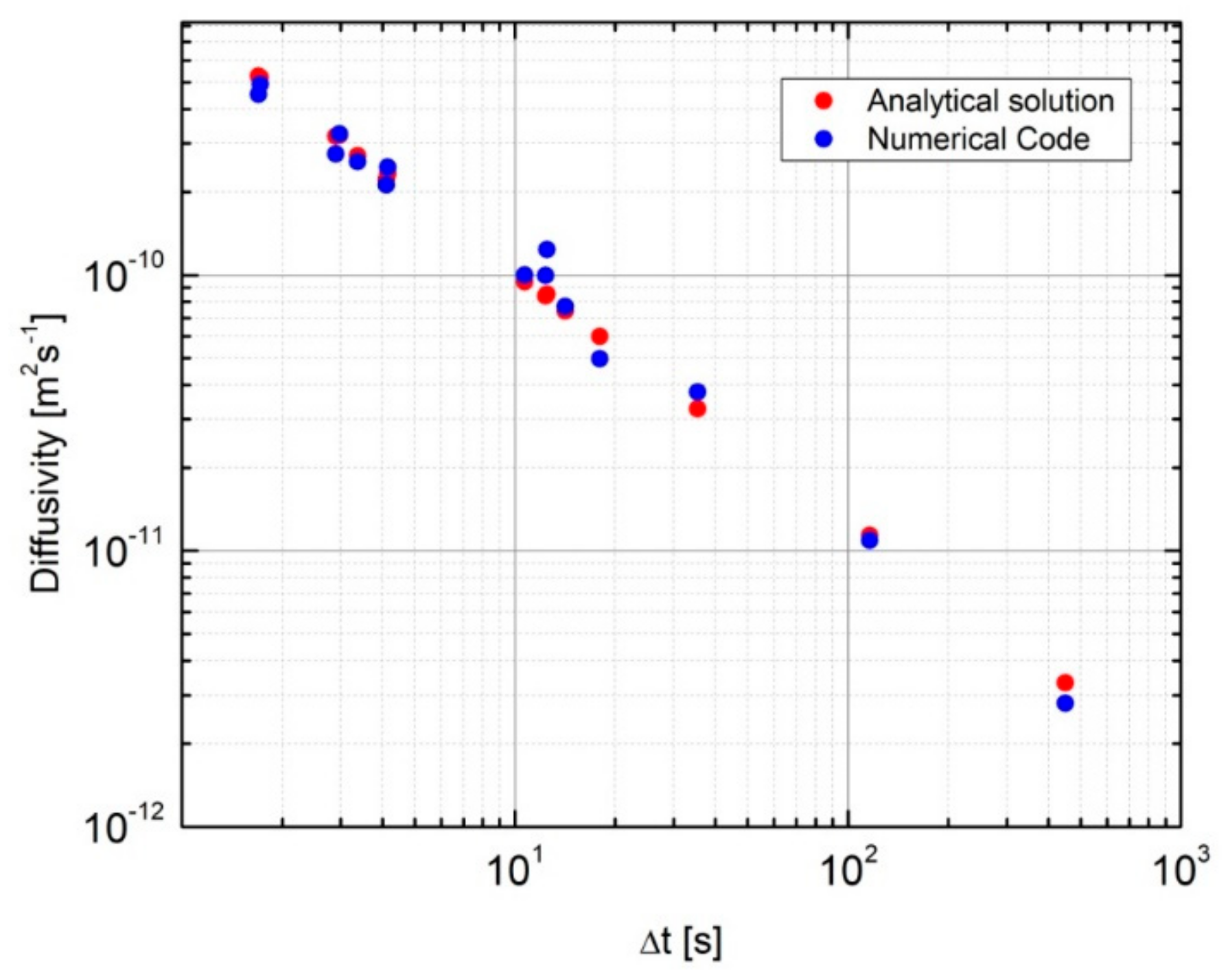
3.2. Numerical Code to Evaluate the Lower Limit of the Hydrogen Diffusion Coefficient
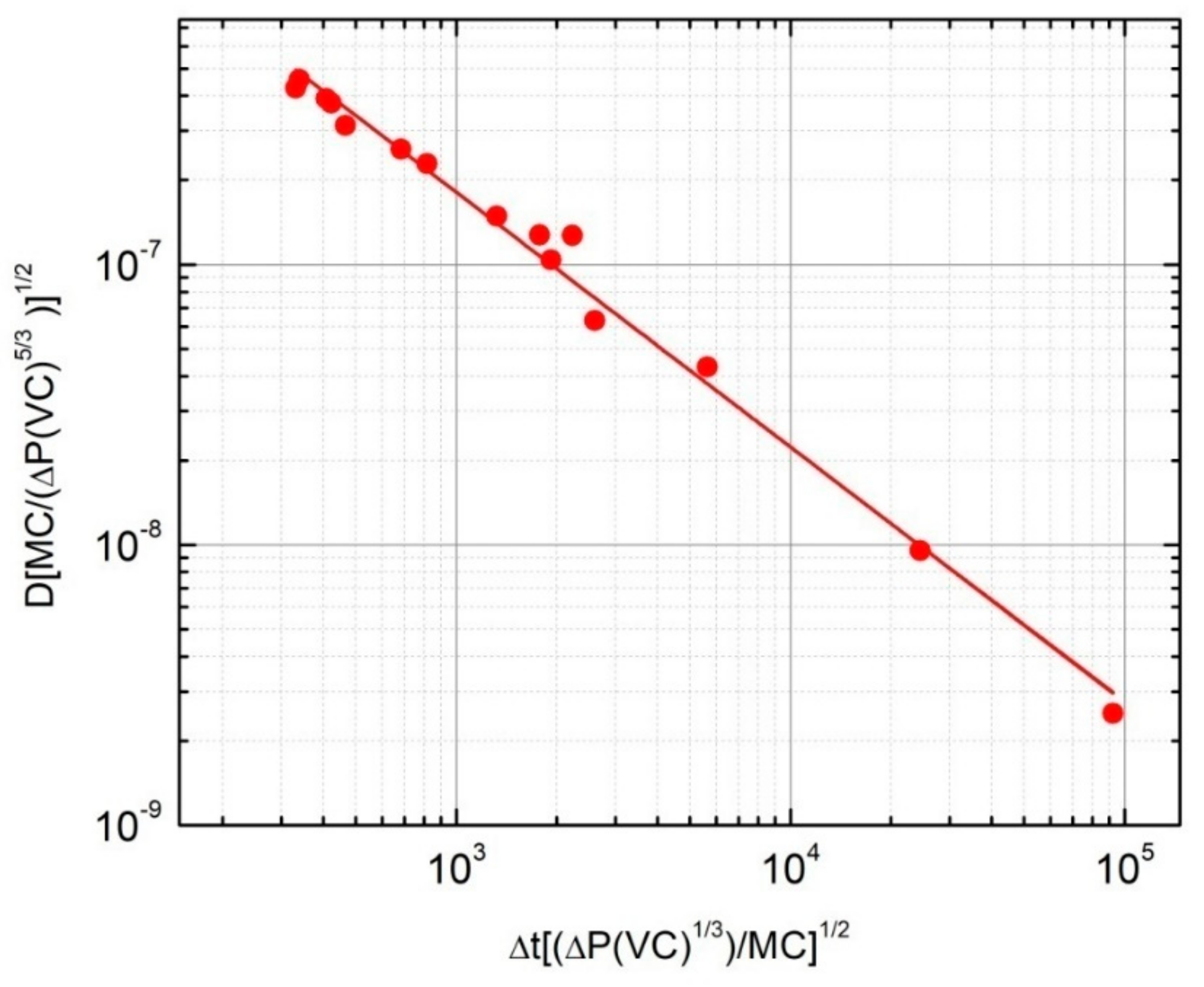
3.3. Dimensional Analysis to Evaluate the Hydrogen Diffusion Coefficient
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ockwig, N.W.; Nenoff, T.M. Membranes for hydrogen separation. Chem. Rev. 2007, 107, 4078–4110. [Google Scholar] [CrossRef]
- Gallucci, F.; Medrano, J.A.; Fernandez, E.; Melendez, J.; van SintAnnaland, M.; Pacheco-Tanaka, D.A. Advances on high temperature Pd-based membranes and membrane reactors for hydrogen purification and production. J. Membr. Sci. Res. 2017, 3, 142–156. [Google Scholar]
- Steward, S.A. Review of hydrogen isotope permeability through metals. In US National Laboratory Report; UCRL-53441; Lawrence Livermore National Lab: Livermore, CA, USA, 1983. [Google Scholar]
- Alefeld, G.; Volkl, J. Hydrogen in Metals; Springer-Verlag: Berlin, Germany, 1978; Volume II. [Google Scholar]
- Santucci, A.; Borgognoni, F.; Vadrucci, M.; Tosti, S. Testing of dense Pd-Ag tubes: Effect of pressure and membrane thickness on the hydrogen permeability. J. Membr. Sci. 2013, 444, 378–383. [Google Scholar] [CrossRef]
- Chapelle, D.; Feng, L.; Rauch, J-Y.; Nardin, P. Assessment of the gas permeation through thin coated polymeric membranes; Improvement of the gas barrier ability for hydrogen storage. In Defect and Diffusion Forum; Trans Tech Publications: Zurich, Switzerland, 2012; pp. 393–399. [Google Scholar] [CrossRef]
- Hara, S.; Caravella, A.; Ishitsuka, M.; Suda, H.; Mukaida, M.; Haraya, K.; Shimano, E.; Tsuji, T. Hydrogen diffusion coefficient and mobility in palladium as a function of equilibrium pressure evaluated by permeation measurement. J. Membr. Sci. 2012, 421, 355–360. [Google Scholar] [CrossRef]
- Flanagan, T.B.; Wang, D. Exponents for the pressure dependence of hydrogen permeation through Pd and Pd−Ag alloy membranes. J. Phys. Chem. C 2010, 114, 14482–14488. [Google Scholar] [CrossRef]
- Palumbo, O.; Brutti, S.; Trequattrini, F.; Sarker, S.; Dolan, M.; Chandra, D.; Paolone, A. Temperature dependence of the elastic modulus of (Ni0.6Nb0.4)1−xZrx membranes: Effects of thermal treatments and hydrogenation. Energies 2015, 8, 3944–3954. [Google Scholar] [CrossRef]
- Palumbo, O.; Trequattrini, F.; Vitucci, F.M.; Bianchin, A.; Paolone, A. Study of the hydrogenation/dehydrogenation process in the Mg-Ni-C-Al system. J. Alloys Compd. 2015, 645, S239–S241. [Google Scholar] [CrossRef]
- Kirchheim, R. Solid solutions of hydrogen in complex materials. Solid State Physics 2004, 59, 203–305. [Google Scholar] [CrossRef]
- Jebaroy, J.M.J.; Morrison, D.; Suni, I.I. Hydrogen diffusion coefficients through Inconel 718 in different metallurgical conditions. Corros. Sci. 2014, 80, 517–522. [Google Scholar] [CrossRef]
- Boes, N.; Zuchner, H. Electrochemical methods for studying diffusion, permeation and solubility of hydrogen in metals. J. Less Comm. Metals 1976, 49, 223–240. [Google Scholar] [CrossRef]
- Yoshinari, O.; Matsuda, H.; Fukuhara, K.; Tanaka, K. Hydrogen diffusivity and solubility in Pd–Y alloys. Mater. Trans. JIM 508–513. [CrossRef]
- Palsson, G.K.; Bliersbach, A.; Wolff, M.; Zamani, A.; Hjorvarsson, B. Using light transmission to watch hydrogen diffuse. Nat. Commun. 2012, 3, 892. [Google Scholar] [CrossRef]
- Huang, W.; Pálsson, G.K.; Brischetto, M.; Palonen, H.; Droulias, S.A.; Hartmann, O.; Wolff, M.; Hjörvarsson, B. Finite size effects: Deuterium diffusion in nm thick vanadium layers. New J. Physics. 2017, 19, 123004. [Google Scholar] [CrossRef]
- Flanagan, T.B.; Wang, D.; Shanahan, K.L. Diffusion of H through Pd membranes: Effects of non-ideality. J. Membr. Sci. 2007, 306, 66–74. [Google Scholar] [CrossRef]
- Tyurin, Y.I.; Larionov, V.V.; Nikitenkov, N.N. A laboratory device for measuring the diffusion coefficient of hydrogen in metals during their simultaneous hydrogenation and electron irradiation. Instrum. Exp. Tech. 2016, 59, 772–774. [Google Scholar] [CrossRef]
- Hara, S.; Caravella, A.; Ishitsuka, M.; Suda, H.; Mukaida, M.; Haraya, K.; Simano, E.; Tsuji, T. Hydrogen diffusion coefficient and mobility in palladium as a function of equilibrium pressure evaluated by permeation measurements. J. Membr. Sci. 2012, 421, 355–360. [Google Scholar] [CrossRef]
- Raina, A.; Sime, N. Effects of anisotropy and regime of diffusion on the measurement of lattice diffusion coefficient of hydrogen inmetals. Proc. R. Soc. A 2018, 474, 20170677. [Google Scholar] [CrossRef]
- Tosti, S.; Fabbricino, M.; Moriani, A.; Agatiello, G.; Scudieri, C.; Borgognoni, F.; Santucci, A. Pressure effect in ethanolsteamreforming via dense Pd-basedmembranes. J. Membr. Sci. 2011, 377, 65–74. [Google Scholar] [CrossRef]
- Hirata, K.; IIkubo, S.; Koyama, K.; Tsuzaki, K.; Ohtani, H. First-principles study on hydrogen diffusivity in BCC, FCC, and HCP iron. Metall. Mat. Mat. Trans. A 2018, 49A, 5015–5022. [Google Scholar] [CrossRef]
- Galindo-Nava, E.I.; Basha, B.I.Y.; Rivera-Díaz-del-Castillo, P.E.J. Hydrogen transport in metals: Integration of permeation, thermal desorption and degassing. J. Mat. Sci. Technol. 2017, 33, 1433–1447. [Google Scholar] [CrossRef]
- Volodin, A.A.; Denys, R.V.; Tsirlina, G.A.; Tarasov, B.P.; Fichtner, M.; Yartys, V.A. Hydrogen diffusion in La1.5Nd0.5MgNi9 alloy electrodes of the Ni/MH battery. J. Alloy. Compd. 2015, 645, S288–S291. [Google Scholar] [CrossRef]
- Dolan, M.D. Non-Pd BCC alloy membranes for industrial hydrogen separation. J. Membr. Sci. 2010, 362, 12–28. [Google Scholar] [CrossRef]
- Sarker, S.; Chandra, D.; Hirscher, M.; Dolan, M.; Isheim, D.; Wermer, J.; Viano, D.; Baricco, M.; Udovic, T.J.; Grant, D.; et al. Developments in the Ni-Nb-Zr amorphous alloy membranes. Appl. Phys. A 2016, 122, 168. [Google Scholar] [CrossRef]
- Paolone, A.; Tosti, S.; Santucci, A.; Palumbo, O.; Trequattrini, F. Hydrogen and deuterium solubility in commercial Pd–Ag alloys for hydrogen purification. ChemEngineering 2018, 1, 14. [Google Scholar] [CrossRef]
- Palumbo, O.; Trequattrini, F.; Sarker, S.; Hulyakar, M.; Pal, N.; Chandra, D.; Dolan, M.; Paolone, S. New studies of the physical properties of metallic amorphous membranes for hydrogen purification. Challenges 2017, 8, 4. [Google Scholar] [CrossRef]
- Kreith, F.; Black, W.B. Basic Heat Transfer; Harper & Row: New York, NY, USA, 1980. [Google Scholar]
- Shu, J.; Grandjean, B.P.A.; Van Neste, A.; Kaliaguine, S. Catalytic palladium-based membrane reactors: A review. Can. J. Chem. Eng. 1991, 69, 1036–1060. [Google Scholar] [CrossRef]
- Nagamoto, H.; Inoue, H. Sorption and desorption of hydrogen on palladiumsheet. NipponKagaku Kaishi 1977, 9, 1264–1267. [Google Scholar] [CrossRef]
- Barlag, H.; Opara, L.; Züchner, H. Hydrogen diffusion in palladium based f.c.c. alloys. J. Alloys Compd. 2002, 330, 434–437. [Google Scholar] [CrossRef]
- Opara, L.; Klein, B.; Züchner, H. Hydrogen-diffusion in Pd1− xAgx (0< x< 1). J. Alloys Compd. 1997, 253, 378–380. [Google Scholar] [CrossRef]
- Holleck Gerhard, L. Diffusion and solubility of hydrogen in palladium and palladium-silver alloys. J. Phys. Chem. 1970, 74, 503–511. [Google Scholar] [CrossRef]
- Sakamoto, Y.; Hirata, S.; Nishikawa, H. Diffusivity and solubiltiy of hydrogen in Pd-Ag and Pd-Au alloys. J. Less Comm. Metals 1982, 88, 387–395. [Google Scholar] [CrossRef]


| Name | Initial Pressure × 105 (Pa) | Sample Temperature (°C) | Final Hydrogen Content (H/M) | Diffusion Coefficient (m2s−1) ×10−10 |
|---|---|---|---|---|
| Test 9 | 0.058 | 150 | 0.02308 | 0.979 |
| Test 7 | 0.104 | 150 | 0.04863 | 0.02806 |
| Test 10 | 0.104 | 200 | 0.03108 | 0.3763 |
| Test 11 | 0.225 | 200 | 0.05084 | 0.1093 |
| Test 12 | 0.110 | 250 | 0.02048 | 0.7703 |
| Test 15 | 0.118 | 250 | 0.02116 | 0.9975 |
| Test 14 | 0.243 | 250 | 0.03609 | 1.237 |
| Test 13 | 0.364 | 250 | 0.04451 | 2.46 |
| Test 16 | 0.102 | 300 | 0.01774 | 1.001 |
| Test 17 | 0.368 | 300 | 0.03264 | 2.749 |
| Test 18 | 0.661 | 300 | 0.04151 | 4.526 |
| Test 19 | 0.936 | 300 | 0.05908 | 3.243 |
| Test 20 | 0.107 | 350 | 0.01006 | 2.117 |
| Test 22 | 0.100 | 350 | 0.04354 | 4.917 |
| Test 24 | 0.360 | 400 | 0.01726 | 2.575 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moriani, A.; Palumbo, O.; Tosti, S.; Santucci, A.; Pozio, A.; Trequattrini, F.; Paolone, A. An Innovative Procedure to Evaluate the Hydrogen Diffusion Coefficient in Metals from Absorption Measurements. Energies 2019, 12, 1652. https://doi.org/10.3390/en12091652
Moriani A, Palumbo O, Tosti S, Santucci A, Pozio A, Trequattrini F, Paolone A. An Innovative Procedure to Evaluate the Hydrogen Diffusion Coefficient in Metals from Absorption Measurements. Energies. 2019; 12(9):1652. https://doi.org/10.3390/en12091652
Chicago/Turabian StyleMoriani, Andrea, Oriele Palumbo, Silvano Tosti, Alessia Santucci, Alfonso Pozio, Francesco Trequattrini, and Annalisa Paolone. 2019. "An Innovative Procedure to Evaluate the Hydrogen Diffusion Coefficient in Metals from Absorption Measurements" Energies 12, no. 9: 1652. https://doi.org/10.3390/en12091652
APA StyleMoriani, A., Palumbo, O., Tosti, S., Santucci, A., Pozio, A., Trequattrini, F., & Paolone, A. (2019). An Innovative Procedure to Evaluate the Hydrogen Diffusion Coefficient in Metals from Absorption Measurements. Energies, 12(9), 1652. https://doi.org/10.3390/en12091652









