Hydrothermal Carbonization of Various Paper Mill Sludges: An Observation of Solid Fuel Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Hydrothermal Carbonization (HTC)
2.3. Characterization of Hydrochars
3. Results and Discussion
3.1. Product Distribution and Energy Densification
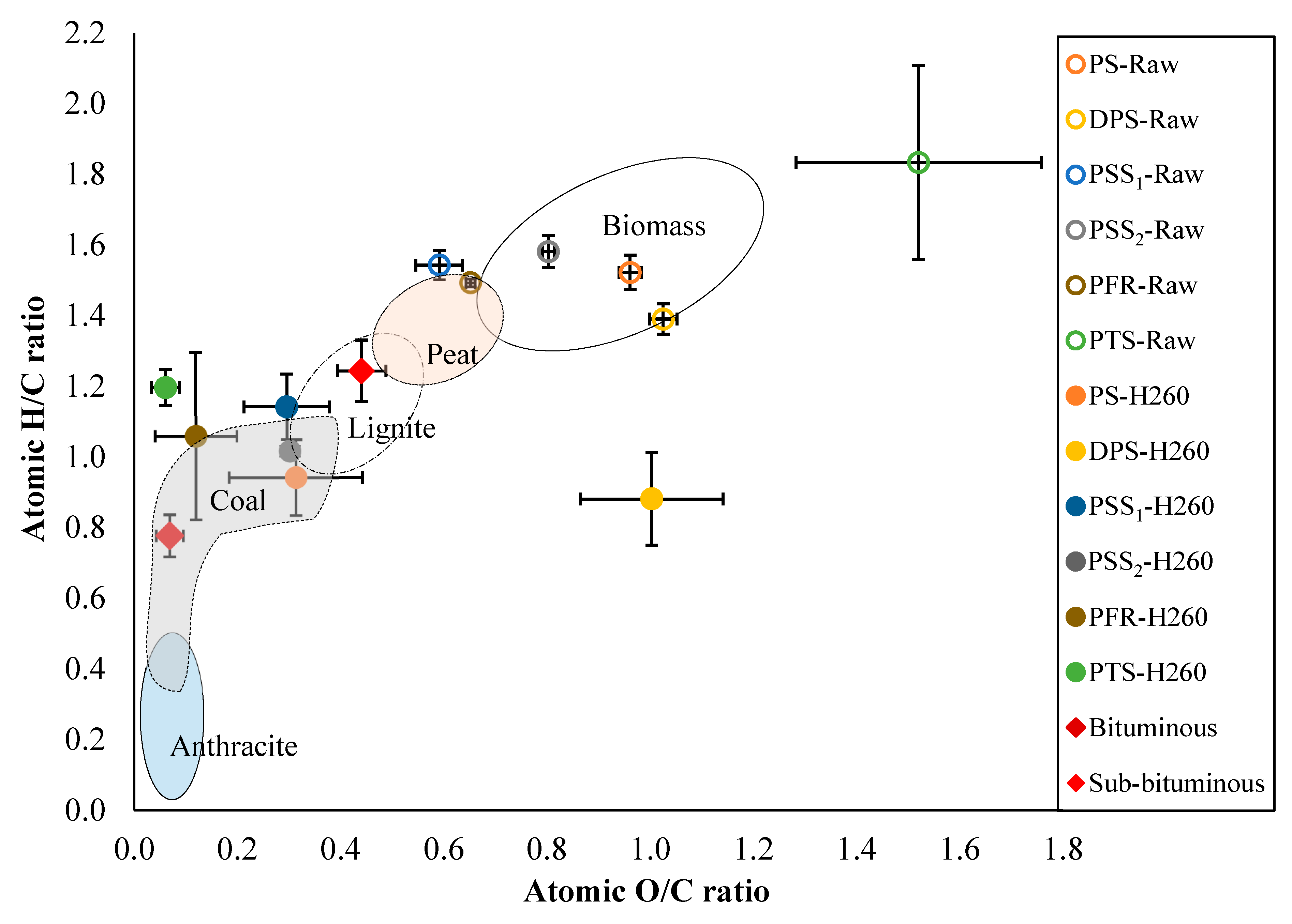
3.2. Chemical Characteristics of PMS Hydrochars Produced at 260 °C
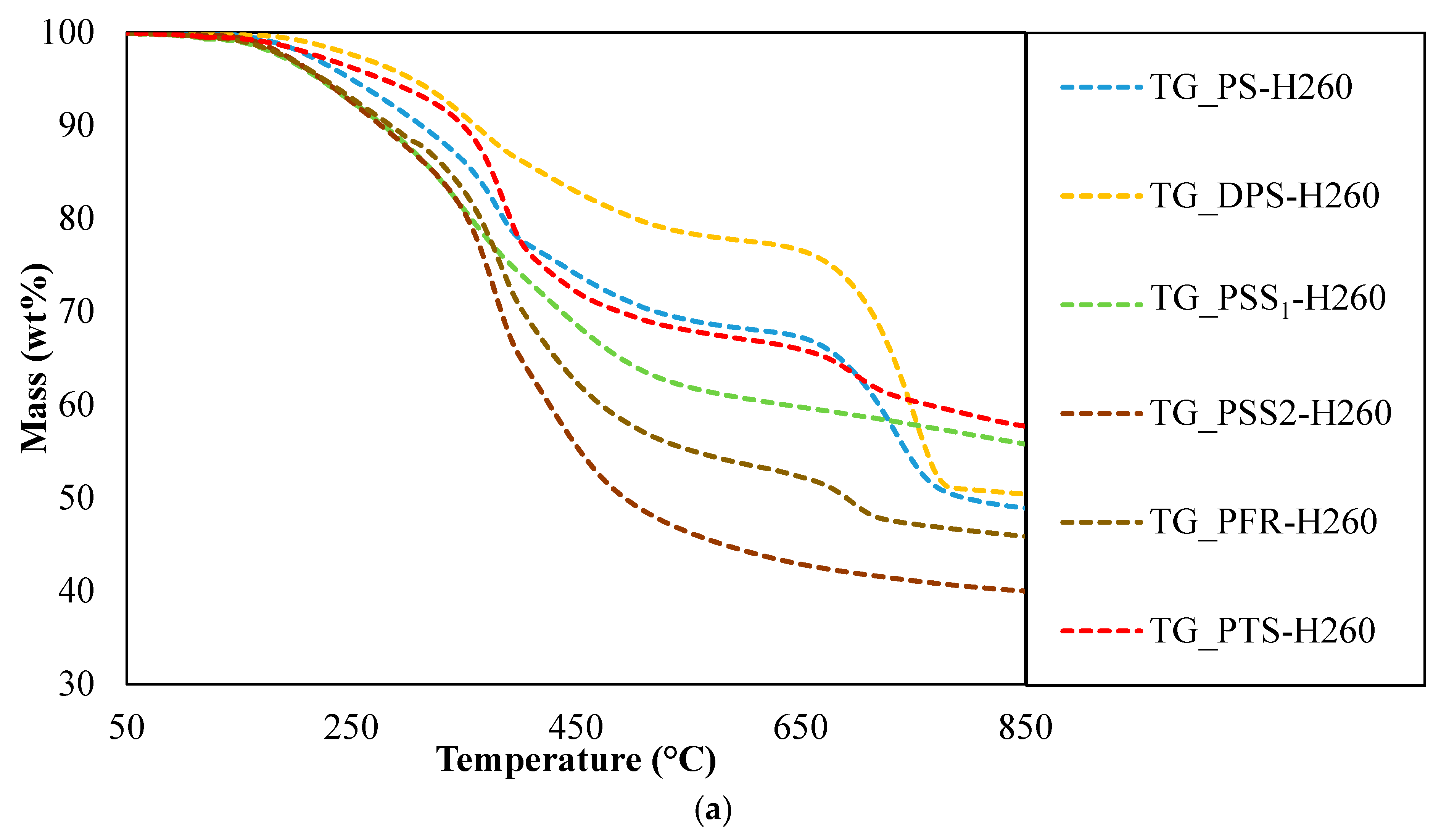
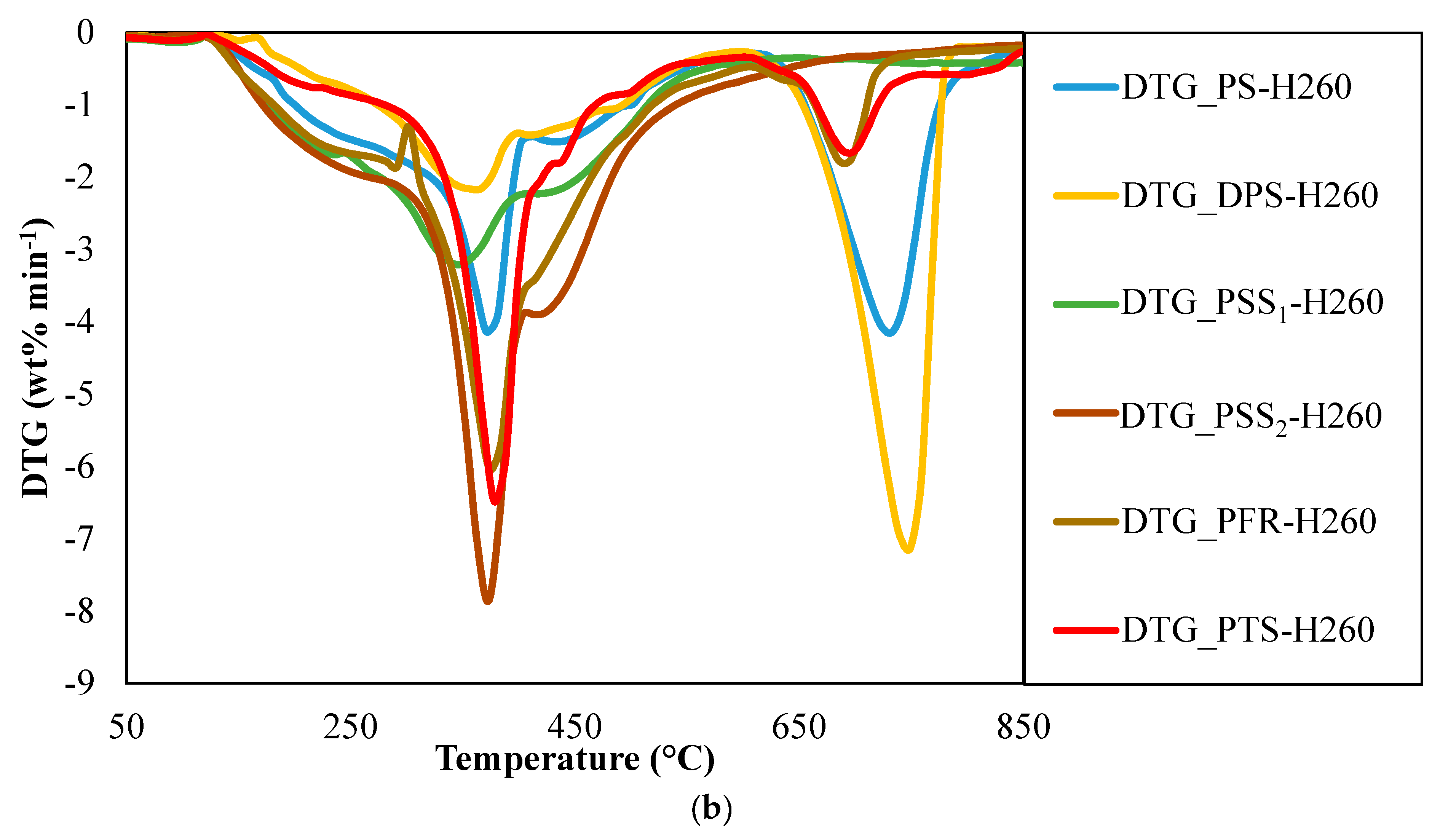
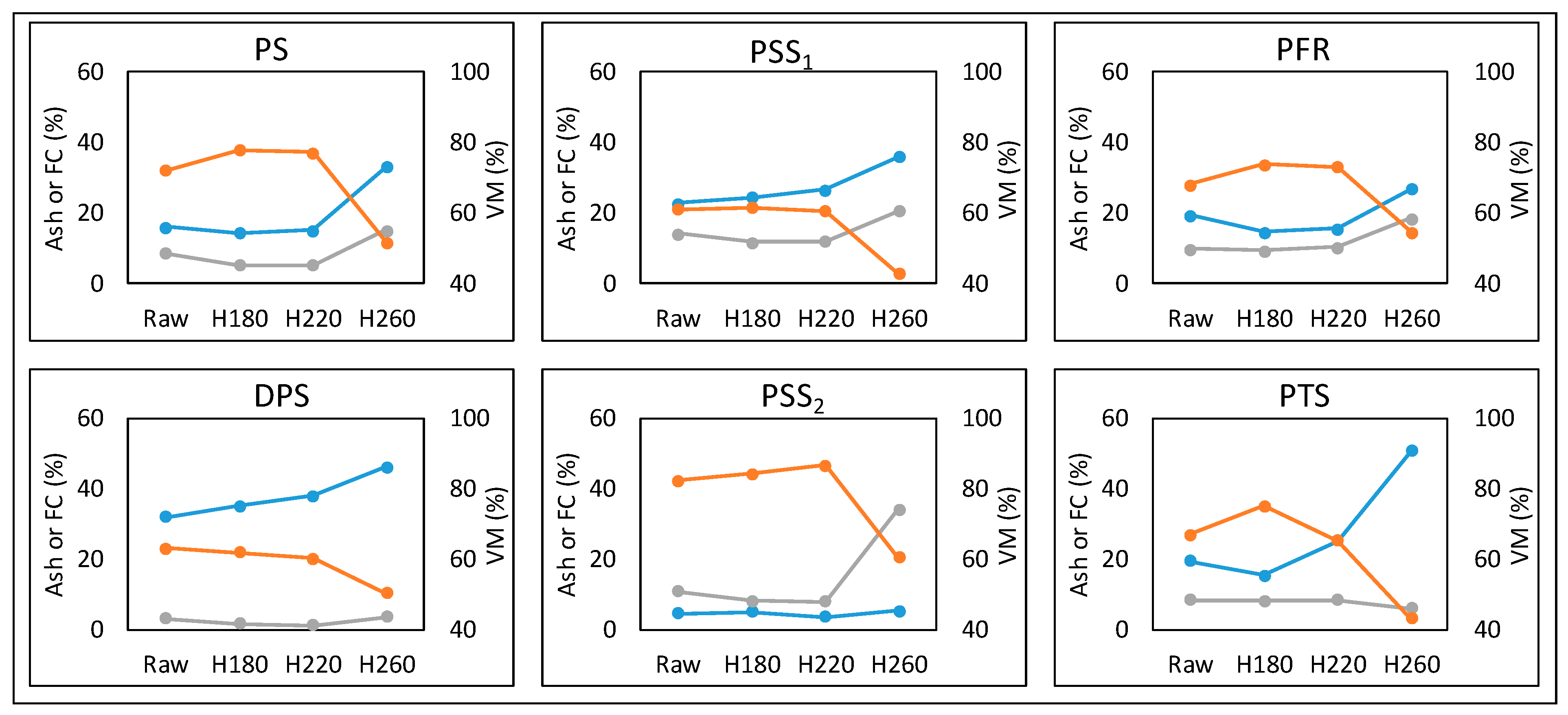
3.3. Fuel Characteristics of Hydrochars
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shaykheeva, D.; Panasyuk, M.; Malganova, I.; Khairullin, I. World Population Estimates and Projections: Data and Methods. J. Econ. Econ. Educ. Res. 2016, 17, 237–247. [Google Scholar]
- Annual Energy Outlook 2016. Available online: http://large.stanford.edu/courses/2016/ph240/martelaro1/docs/0383-2016.pdf (accessed on 5 March 2019).
- United States Department of Energy. Billion Ton Update: Biomass Supply for a Bioenergy and Bioproducts Industry; Oak Ridge National Laboratory: Oak Ridge, TN, USA, 2016.
- Bird, M.; Talberth, J. Waste Stream Reduction and Re-Use in the Pulp and Paper Sector; Washington State Department of Ecology: Santa Fe, NM, USA, 2008. [Google Scholar]
- Aspitarte, T.R.; Rosenfield, A.S.; Smale, B.C.; Amberg, H.R. Methods for Pulp and Paper Mill Sludge Utilization and Disposal; USEPA: Washington, DC, USA, 1973.
- Mun, T.Y.; Tumsa, T.Z.; Lee, U.; Yang, W. Performance evaluation of co-firing various kinds of biomass with low rank coals in a 500 MWe coal-fired power plant. Energy 2016, 115, 954–962. [Google Scholar] [CrossRef]
- Tsai, M.-Y.; Wu, K.-T.; Huang, C.-C.; Lee, H.-T. Co-firing of paper mill sludge and coal in an industrial circulating fluidized bed boiler. Waste Manag. 2002, 22, 439–442. [Google Scholar] [CrossRef]
- Bajpai, P. Generation of Waste in Pulp and Paper Mills. In Management of Pulp and Paper Mill Waste; Springer International Publishing: Basel, Switzerland, 2015. [Google Scholar]
- Monte, M.C.; Fuente, E.; Blanco, A.; Negro, C. Waste management from pulp and paper production in the European Union. Waste Manag. 2009, 29, 293–308. [Google Scholar] [CrossRef] [PubMed]
- IPPC. Reference Document on Best Available Techniques in the Pulp and Paper Industry; European IPPC Bureau: Seville, Spain, 2001. [Google Scholar]
- Soucy, J.; Koubaa, A.; Migneault, S.; Riedl, B. Chemical Composition and Surface Properties of Paper Mill Sludge and their Impact on High Density Polyethylene (HDPE) Composites. J. Wood Chem. Technol. 2016, 36, 77–93. [Google Scholar] [CrossRef]
- Bajpai, P. Recycling and Deinking of Recovered Paper; Elsevier Science: Amsterdam, The Netherlands, 2013. [Google Scholar]
- CANMET. Pulp and Paper Sludge to Energy—Preliminary Assessment of Technologies, Canada; CANMET Energy Technology Centre: Ottawa, ON, Canada, 2005. [Google Scholar]
- Scott, G.M.; Smith, A. Sludge characteristics and disposal alternatives for the pulp and paper industry. In Proceedings of the 1995 International Environmental Conference, Atlanta, GA, USA, 7–10 May 1995; pp. 269–279. [Google Scholar]
- Reza, M.T. Upgrading Biomass by Hydrothermal and Chemical Conditioning; University of Nevada Reno: Reno, NV, USA, 2013. [Google Scholar]
- Reza, M.T.; Andert, J.; Wirth, B.; Busch, D.; Pielert, J.; Lynam, J.G.; Mumme, J. Hydrothermal Carbonization of Biomass for Energy and Crop Production. Appl. Bioenergy 2014, 1, 11–29. [Google Scholar] [CrossRef]
- Reza, M.T.; Coronella, C.; Holtman, K.M.; Franqui-Villanueva, D.; Poulson, S.R. Hydrothermal Carbonization of Autoclaved Municipal Solid Waste Pulp and Anaerobically Treated Pulp Digestate. ACS Sustain Chem. Eng. 2016, 4, 3649–3658. [Google Scholar] [CrossRef]
- Reza, M.T.; Mumme, J.; Ebert, A. Characterization of Hydrochar Obtained from Hydrothermal Carbonization of Wheat Straw Digestate. Biomass Convers. Biorefin. 2015. [Google Scholar] [CrossRef]
- Kruse, A.; Dinjus, E. Hot compressed water as reaction medium and reactant: Properties and synthesis reactions. J. Supercrit. Fluids 2007, 39, 362–380. [Google Scholar] [CrossRef]
- Bandura, A.V.; Lvov, S.N. The ionization constant of water over wide ranges of temperature and density. J. Phys. Chem. Ref. Data 2006, 35, 15–30. [Google Scholar] [CrossRef]
- Reza, M.T.; Uddin, M.H.; Lynam, J.; Hoekman, S.K.; Coronella, C. Hydrothermal carbonization of loblolly pine: Reaction chemistry and water balance. Biomass Convers. Biorefin. 2014, 4, 311–321. [Google Scholar] [CrossRef]
- Reza, M.T.; Rottler, E.; Herklotz, L.; Wirth, B. Hydrothermal carbonization (HTC) of wheat straw: Influence of feedwater pH prepared by acetic acid and potassium hydroxide. Bioresour. Technol. 2015, 182, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Reza, M.T.; Wirth, B.; Luder, U.; Werner, M. Behavior of selected hydrolyzed and dehydrated products during hydrothermal carbonization of biomass. Bioresour. Technol. 2014, 169, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Reza, M.T.; Yan, W.; Uddin, M.H.; Lynam, J.G.; Hoekman, S.K.; Coronella, C.J.; Vasquez, V.R. Reaction kinetics of hydrothermal carbonization of loblolly pine. Bioresour. Technol. 2013, 139, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Lynam, J.G.; Reza, M.T.; Yan, W.; Vásquez, V.R.; Coronella, C.J. Hydrothermal carbonization of various lignocellulosic biomass. Biomass Convers. Biorefin. 2015, 5, 173–181. [Google Scholar] [CrossRef]
- Makela, M.; Benavente, V.; Fullana, A. Hydrothermal carbonization of lignocellulosic biomass: Effect of process conditions on hydrochar properties. Appl. Energy 2015, 155, 576–584. [Google Scholar] [CrossRef]
- Reza, M.T.; Lynam, J.G.; Vasquez, V.R.; Coronella, C.J. Pelletization of Biochar from Hydrothermally Carbonized Wood. Environ. Prog. Sustain. Energy 2012, 31, 225–234. [Google Scholar] [CrossRef]
- Reza, M.T.; Yang, X.; Coronella, C.J.; Lin, H.; Hathwaik, U.; Shintani, D.; Neupane, B.P.; Miller, G.C. Hydrothermal Carbonization (HTC) and Pelletization of Two Arid Land Plants Bagasse for Energy Densification. ACS Sustain. Chem. Eng. 2015. [Google Scholar] [CrossRef]
- Saha, N.; Saba, A.; Reza, M.T. Effect of hydrothermal carbonization temperature on pH, dissociation constants, and acidic functional groups on hydrochar from cellulose and wood. J. Anal. Appl. Pyrolysis 2019, 137, 138–145. [Google Scholar] [CrossRef]
- Naisse, C.; Alexis, M.; Plante, A.F.; Wiedner, K.; Glaser, B.; Pozzi, A.; Carcaillet, C.; Criscuoli, I.; Rumpel, C. Can biochar and hydrochar stability be assessed with chemical methods? Org. Geochem. 2013, 60, 40–44. [Google Scholar] [CrossRef]
- Wiedner, K.; Naisse, C.; Rumpel, C.; Pozzi, A.; Wieczorek, P.; Glaser, B. Chemical modification of biomass residues during hydrothermal carbonization—What makes the difference, temperature or feedstock? Org. Geochem. 2013, 54, 91–100. [Google Scholar] [CrossRef]
- Wiedner, K.; Rumpel, C.; Steiner, C.; Pozzi, A.; Maas, R.; Glaser, B. Chemical evaluation of chars produced by thermochemical conversion (gasification, pyrolysis and hydrothermal carbonization) of agro-industrial biomass on a commercial scale. Biomass Bioenergy 2013, 59, 264–278. [Google Scholar] [CrossRef]
- Ronix, A.; Pezoti, O.; Souza, L.S.; Souza, I.P.A.F.; Bedin, K.C.; Souza, P.S.C.; Silva, T.L.; Melo, S.A.R.; Cazetta, A.L.; Almeida, V.C. Hydrothermal carbonization of coffee husk: Optimization of experimental parameters and adsorption of methylene blue dye. J. Environ. Chem. Eng. 2017, 5, 4841–4849. [Google Scholar] [CrossRef]
- Kannan, S.; Gariepy, Y.; Vijaya Raghavan, G.S. Optimization of the conventional hydrothermal carbonization to produce hydrochar from fish waste. Biomass Convers. Biorefin. 2018, 8, 563–576. [Google Scholar] [CrossRef]
- Kruse, A.; Zevaco, A.T. Properties of Hydrochar as Function of Feedstock, Reaction Conditions and Post-Treatment. Energies 2018, 11, 674. [Google Scholar] [CrossRef]
- Román, S.; Libra, J.; Berge, N.; Sabio, E.; Ro, K.; Li, L.; Ledesma, B.; Álvarez, A.; Bae, S. Hydrothermal Carbonization: Modeling, Final Properties Design and Applications: A Review. Energies 2018, 11, 216. [Google Scholar] [CrossRef]
- Kempegowda, R.S.; Skreiberg, Ø.; Tran, K.-Q.; Selvam, P.V.P. Techno-economic Assessment of Thermal Co-pretreatment and Co-digestion of Food Wastes and Sewage Sludge for Heat, Power and Biochar Production. Energy Procedia 2017, 105, 1737–1742. [Google Scholar] [CrossRef]
- Escala, M.; Zumbuhl, T.; Koller, C.; Junge, R.; Krebs, R. Hydrothermal Carbonization as an Energy-Efficient Alternative to Established Drying Technologies for Sewage Sludge: A Feasibility Study on a Laboratory Scale. Energy Fuel 2013, 27, 454–460. [Google Scholar] [CrossRef]
- Lucian, M.; Fiori, L. Hydrothermal Carbonization of Waste Biomass: Process Design, Modeling, Energy Efficiency and Cost Analysis. Energies 2017, 10, 211. [Google Scholar] [CrossRef]
- Areeprasert, C.; Coppola, A.; Urciuolo, M.; Chirone, R.; Yoshikawa, K.; Scala, F. The effect of hydrothermal treatment on attrition during the fluidized bed combustion of paper sludge. Fuel Process. Technol. 2015, 140, 57–66. [Google Scholar] [CrossRef]
- Areeprasert, C.; Zhao, P.T.; Ma, D.C.; Shen, Y.F.; Yoshikawa, K. Alternative Solid Fuel Production from Paper Sludge Employing Hydrothermal Treatment. Energy Fuel 2014, 28, 1198–1206. [Google Scholar] [CrossRef]
- Zhao, P.T.; Ge, S.F.; Ma, D.C.; Areeprasert, C.; Yoshikawa, K. Effect of Hydrothermal Pretreatment on Convective Drying Characteristics of Paper Sludge. ACS Sustain. Chem. Eng. 2014, 2, 665–671. [Google Scholar] [CrossRef]
- Areeprasert, C.; Ma, D.; Prayoga, P.; Yoshikawa, K. A Review on Pilot-Scale Applications of Hydrothermal Treatment for Upgrading Waste Materials. Int. J. Environ. Sci. Dev. 2016, 7, 425–430. [Google Scholar] [CrossRef]
- Areeprasert, C.; Scala, F.; Coppola, A.; Urciuolo, M.; Chirone, R.; Chanyavanich, P.; Yoshikawa, K. Fluidized bed co-combustion of hydrothermally treated paper sludge with two coals of different rank. Fuel Process Technol. 2016, 144, 230–238. [Google Scholar] [CrossRef]
- Mäkelä, M.; Forsberg, J.; Söderberg, C.; Larsson, S.H.; Dahl, O. Process water properties from hydrothermal carbonization of chemical sludge from a pulp and board mill. Bioresour. Technol. 2018, 263, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Makela, M.; Benavente, V.; Fullana, A. Hydrothermal carbonization of industrial mixed sludge from a pulp and paper mill. Bioresour. Technol. 2016, 200, 444–450. [Google Scholar] [CrossRef] [PubMed]
- TAPPI. Acid-Insoluble Lignin in Wood and Pulp; (Reaffirmation of T 222 om-02); TAPPI: Peachtree Corners, GA, USA, 2006; Available online: https://www.tappi.org/content/SARG/T222.pdf (accessed on 4 March 2019).
- Sequeira, R.M.; Lew, R.B. Carbohydrate composition of almond hulls. J. Agric. Food Chem. 1970, 18, 950–951. [Google Scholar] [PubMed]
- Rong, H.; Wang, T.; Zhou, M.; Wang, H.; Hou, H.; Xue, Y. Combustion Characteristics and Slagging during Co-Combustion of Rice Husk and Sewage Sludge Blends. Energies 2017, 10, 438. [Google Scholar] [CrossRef]
- Vamvuka, D.; El Chatib, N.; Sfakiotakis, S. Measurements of ignition point and combustion characteristics of biomass fuels and their blends with lignite. Combustion 2011, 2015, 95. [Google Scholar]
- Funke, A.; Ziegler, F. Hydrothermal carbonization of biomass: A summary and discussion of chemical mechanisms for process engineering. Biofuels Bioprod. Biorefin. 2010, 4, 160–177. [Google Scholar] [CrossRef]
- Hoekman, S.K.; Broch, A.; Robbins, C. Hydrothermal Carbonization (HTC) of Lignocellulosic Biomass. Energy Fuel 2011, 25, 1802–1810. [Google Scholar] [CrossRef]
- Sheng, C.; Azevedo, J.L.T. Estimating the higher heating value of biomass fuels from basic analysis data. Biomass Bioenergy 2005, 28, 499–507. [Google Scholar] [CrossRef]
- Zhao, C.; Jiang, E.; Chen, A. Volatile production from pyrolysis of cellulose, hemicellulose and lignin. J. Energy Inst. 2017, 90, 902–913. [Google Scholar] [CrossRef]
- Kang, S.; Li, X.; Fan, J.; Chang, J. Characterization of Hydrochars Produced by Hydrothermal Carbonization of Lignin, Cellulose, d-Xylose, and Wood Meal. Ind. Eng. Chem. Res. 2012, 51, 9023–9031. [Google Scholar] [CrossRef]
- Lin, Y.; Ma, X.; Peng, X.; Hu, S.; Yu, Z.; Fang, S. Effect of hydrothermal carbonization temperature on combustion behavior of hydrochar fuel from paper sludge. Appl. Therm. Eng. 2015, 91, 574–582. [Google Scholar] [CrossRef]
- Kim, J.S.; Lee, Y.Y.; Park, S.C. Pretreatment of Wastepaper and Pulp Mill Sludge by Aqueous Ammonia and Hydrogen Peroxide. In Twenty-First Symposium on Biotechnology for Fuels and Chemicals, Proceedings of the Twenty-First Symposium on Biotechnology for Fuels and Chemicals, Fort Collins, CO, USA, 2–6 May 1999; Finkelstein, M., Davison, B.H., Eds.; Humana Press: Totowa, NJ, USA, 2000; pp. 129–139. [Google Scholar] [CrossRef]
- Saba, A.; Saha, P.; Reza, M.T. Co-Hydrothermal Carbonization of coal-biomass blend: Influence of temperature on solid fuel properties. Fuel Process. Technol. 2017, 167, 711–720. [Google Scholar] [CrossRef]
- García, G.; Arauzo, J.; Gonzalo, A.; Sánchez, J.L.; Ábrego, J. Influence of feedstock composition in fluidised bed co-gasification of mixtures of lignite, bituminous coal and sewage sludge. Chem. Eng. J. 2013, 222, 345–352. [Google Scholar] [CrossRef]
- Grénman, H.; Eränen, K.; Krogell, J.; Willför, S.; Salmi, T.; Murzin, D.Y. Kinetics of Aqueous Extraction of Hemicelluloses from Spruce in an Intensified Reactor System. Ind. Eng. Chem. Res. 2011, 50, 3818–3828. [Google Scholar] [CrossRef]
- McGaughy, K.; Reza, M.T. Recovery of Macro and Micro-Nutrients by Hydrothermal Carbonization of Septage. J. Agric. Food Chem. 2018, 66, 1854–1862. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhai, Y.; Zhu, Y.; Li, C.; Zeng, G. A review of the hydrothermal carbonization of biomass waste for hydrochar formation: Process conditions, fundamentals, and physicochemical properties. Renew. Sustain. Energy Rev. 2018, 90, 223–247. [Google Scholar] [CrossRef]
- McKendry, P. Energy production from biomass (part 1): Overview of biomass. Bioresour. Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef]
- Stricker, G.D.; Flores, R.M.; Trippi, M.H.; Ellis, M.S.; Olson, C.M.; Sullivan, J.E.; Takahashi, K.I. Coal Quality and Major, Minor, and Trace Elements in the Powder River, Green River, and Williston basins, Wyoming and North Dakota: U.S. Geological Survey; U.S. Department of the Interior: Reston, VA, USA, 2007.
- El-Sayed, S.A.; Mostafa, M. Pyrolysis characteristics and kinetic parameters determination of biomass fuel powders by differential thermal gravimetric analysis (TGA/DTG). Energy Convers. Manag. 2014, 85, 165–172. [Google Scholar] [CrossRef]
- Yanfen, L.; Xiaoqian, M. Thermogravimetric analysis of the co-combustion of coal and paper mill sludge. Appl. Energy 2010, 87, 3526–3532. [Google Scholar] [CrossRef]
- Volpe, M.; Goldfarb, J.L.; Fiori, L. Hydrothermal carbonization of Opuntia ficus-indica cladodes: Role of process parameters on hydrochar properties. Bioresour. Technol. 2018, 247, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Wang, X.; Wang, J.; Li, X.; Cheng, J.; Yang, H.; Chen, H. Effect of residence time on chemical and structural properties of hydrochar obtained by hydrothermal carbonization of water hyacinth. Energy 2013, 58, 376–383. [Google Scholar] [CrossRef]
- He, C.; Giannis, A.; Wang, J.-Y. Conversion of sewage sludge to clean solid fuel using hydrothermal carbonization: Hydrochar fuel characteristics and combustion behavior. Appl. Energy 2013, 111, 257–266. [Google Scholar] [CrossRef]
- Varol, M.; Atimtay, A.; Bay, B.; Olgun, H. Investigation of co-combustion characteristics of low quality lignite coals and biomass with thermogravimetric analysis. Thermochim. Acta 2010, 510, 195–201. [Google Scholar] [CrossRef]
| Sludge Sample | ID | MC (%) |
|---|---|---|
| Primary Sludge | PS | 60.1 ± 1.5 |
| De-inked Paper Sludge | DPS | 63.5 ± 0.5 |
| Primary and Secondary Sludge 1 | PSS1 | 64.1 ± 1.0 |
| Primary and Secondary Sludge 2 | PSS2 | 76.5 ± 1.0 |
| Primary Sludge and Fiber Rejects | PFR | 57.1 ± 1.2 |
| Pre-thickened Sludge | PTS | 98.7 ± 0.1 |
| Sample | HTC Temperature (°C) | Solid Mass Yield (%) | Liquid Mass Yield (%) | Gas Mass Yield (%) | HHVdaf (MJ kg−1) | ED |
|---|---|---|---|---|---|---|
| PS | Raw | NA a | NA a | NA a | 15.5 ± 0.0 | NA a |
| 180 | 96.2 ± 2.0 | 3.8 ± 2.0 | 0.0 ± 0.0 | 15.1 ± 0.4 | 1.0 ± 0.0 | |
| 220 | 85.4 ± 2.0 | 11.8 ± 2.2 | 2.8 ± 0.2 | 15.3 ± 0.4 | 1.0 ± 0.0 | |
| 260 | 30.3 ± 1.1 | 58.1 ± 2.4 | 11.6 ± 1.4 | 22.8 ± 0.1 | 1.5 ± 0.0 | |
| DPS | Raw | NA a | NA a | NA a | 13.0 ± 0.0 | NA a |
| 180 | 91.7 ± 0.9 | 8.3 ± 0.9 | 0.0 ± 0.0 | 13.0 ± 0.2 | 1.0 ± 0.0 | |
| 220 | 84.4 ± 0.9 | 13.3 ± 1.4 | 2.3 ± 0.5 | 12.9 ± 0.1 | 1.0 ± 0.0 | |
| 260 | 58.4 ± 2.0 | 33.9 ± 1.3 | 7.2 ± 1.3 | 11.4 ± 0.7 | 0.9 ± 0.1 | |
| PSS1 | Raw | NA a | NA a | NA a | 21.4 ± 0.1 | NA a |
| 180 | 81.2 ± 0.3 | 18.3 ± 1.0 | 0.0 ± 0.0 | 21.7 ± 0.1 | 1.0 ± 0.0 | |
| 220 | 74.1 ± 0.0 | 22.2 ± 0.4 | 3.6 ± 0.5 | 21.7 ± 0.1 | 1.0 ± 0.0 | |
| 260 | 54.1 ± 2.0 | 38.8 ± 3.6 | 7.1 ± 1.6 | 27.4 ± 0.1 | 1.3 ± 0.0 | |
| PSS2 | Raw | NA a | NA a | NA a | 19.6 ± 0.2 | NA a |
| 180 | 90.9 ± 0.5 | 9.1 ± 0.5 | 0.0 ± 0.0 | 19.6 ± 0.1 | 1.0 ± 0.0 | |
| 220 | 78.9 ± 1.2 | 18.4 ± 1.2 | 2.7 ± 0.0 | 20.2 ± 0.1 | 1.0 ± 0.0 | |
| 260 | 41.1 ± 1.7 | 49.9 ± 2.1 | 9.0 ± 0.4 | 28.9 ± 0.1 | 1.5 ± 0.0 | |
| PFR | Raw | NA a | NA a | NA a | 19.7 ± 0.2 | NA a |
| 180 | 93.2 ± 2.1 | 6.8 ± 2.1 | 0.0 ± 0.0 | 18.5 ± 0.0 | 0.9 ± 0.0 | |
| 220 | 81.9 ± 1.2 | 15.6 ± 0.5 | 2.5 ± 0.7 | 19.3 ± 0.2 | 1.0 ± 0.0 | |
| 260 | 45.4 ± 1.2 | 42.9 ± 0.8 | 10.7 ± 0.4 | 25.2 ± 0.3 | 1.3 ± 0.0 | |
| PTS | Raw | NA a | NA a | NA a | 17.1 ± 0.7 | NA a |
| 180 | 87.5 ± 2.9 | 8.0 ± 0.8 | 0.0 ± 0.0 | 18.6 ± 0.5 | 1.1 ± 0.0 | |
| 220 | 69.8 ± 2.8 | 25.2 ± 1.4 | 5.0 ± 1.4 | 21.6 ± 0.1 | 1.3 ± 0.0 | |
| 260 | 43.8 ± 1.1 | 52.0 ± 4.6 | 4.1 ± 3.5 | 31.5 ± 3.7 | 1.8 ± 0.3 | |
| Clarion # 5a coal | Raw | NA a | NA a | NA a | 30.5 ± 0.3 | NA a |
| Sample | HTC Temperature (°C) | Carbon (%) | Hydrogen (%) | Nitrogen (%) | Sulfur (%) | Oxygen a (%) | Ash (%) | Lignin (%) | C5 and C6 Sugar (mg/g) |
|---|---|---|---|---|---|---|---|---|---|
| PS | Raw | 34.6 ± 0.4 | 4.7 ± 0.1 | 0.3 ± 0.0 | BD b | 44.4 ± 0.8 | 16.0 ± 0.0 | 5.2 ± 0.4 | 2.7 ± 0.3 |
| 260 | 44.3 ± 3.3 | 3.8 ± 0.3 | 0.2 ± 0.0 | 2.0 ± 0.0 | 18.5 ± 7.5 | 33.1 ± 1.7 | 36.9 ± 1.3 | 3.0 ± 0.3 | |
| DPS | Raw | 27.1 ± 0.2 | 3.4 ± 0.1 | 0.7 ± 0.0 | BD b | 37.0 ± 0.9 | 31.9 ± 0.4 | 10.0 ± 1.2 | 2.0 ± 0.2 |
| 260 | 22.2 ± 1.7 | 1.8 ± 0.2 | 0.4 ± 0.1 | 0.1 ± 0.0 | 29.6 ± 3.4 | 46.0 ± 0.3 | 19.7 ± 0.8 | 1.9 ± 0.3 | |
| PSS1 | Raw | 38.4 ± 0.8 | 5.3 ± 0.1 | 2.3 ± 0.2 | 0.9 ± 0.1 | 30.3 ± 2.2 | 22.8 ± 0.4 | 20.2 ± 0.1 | 4.0 ± 1.1 |
| 260 | 40.5 ± 1.5 | 4.2 ± 0.3 | 2.1 ± 0.0 | 1.3 ± 0.0 | 16.0 ± 4.4 | 36.0 ± 1.3 | 46.8 ± 0.8 | 1.4 ± 0.0 | |
| PSS2 | Raw | 42.8 ± 0.0 | 6.1 ± 0.2 | 0.7 ± 0.1 | 0.2 ± 0.0 | 45.8 ± 0.7 | 4.5 ± 0.1 | 17.6 ± 0.5 | 3.5 ± 1.2 |
| 260 | 62.2 ± 0.7 | 5.7 ± 0.1 | 1.6 ± 0.1 | 0.3 ± 0.0 | 25.0 ± 1.5 | 5.2 ± 0.1 | 87.8 ± 5.9 | 3.5 ± 2.5 | |
| PFR | Raw | 39.8 ± 0.1 | 5.3 ± 0.0 | 0.6 ± 0.1 | 0.3 ± 0.0 | 34.6 ± 0.5 | 19.5 ± 0.1 | 22.6 ± 0.3 | 3.8 ± 2.2 |
| 260 | 57.6 ± 2.6 | 5.5 ± 1.2 | 0.6 ± 0.1 | 0.4 ± 0.1 | 9.2 ± 6.1 | 26.8 ± 0.3 | 57.9 ± 1.2 | 2.8 ± 0.3 | |
| PTS | Raw | 23.9 ± 2.7 | 3.9 ± 0.4 | 0.6 ± 0.0 | 6.2 ± 0.6 | 48.4 ± 5.2 | 17.1 ± 0.1 | 20.4 ± 0.5 | 3.2 ± 2.2 |
| 260 | 40.2 ± 1.4 | 4.3 ± 0.1 | 0.4 ± 0.1 | 1.3 ± 0.2 | 3.3 ± 1.5 | 50.5 ± 1.5 | 44.2 ± 0.6 | 2.4 ± 1.2 | |
| Clarion # 5a coal | Raw | 63.8 ± 1.2 | 4.1 ± 0.1 | 1.5 ± 0.0 | 4.6 ± 0.8 | 14.9 ± | 11.1 ± 0.6 | NA c | NA c |
| Bituminous coal [63] | Raw | 75.1 ± 2.8 | 5.3 ± 0.4 | 1.5 ± 0.1 | 1.1 ± 0.8 | 6.9 ± 2.6 | 10.0 ± 1.4 | NA c | NA c |
| Sub-bituminous coal [64] | Raw | 55.8 ± 3.5 | 6.2 ± 0.2 | 0.7 ± 0.1 | 0.3 ± 0.1 | 32.7 ± 2.8 | 4.3 ± 0.9 | NA c | NA c |
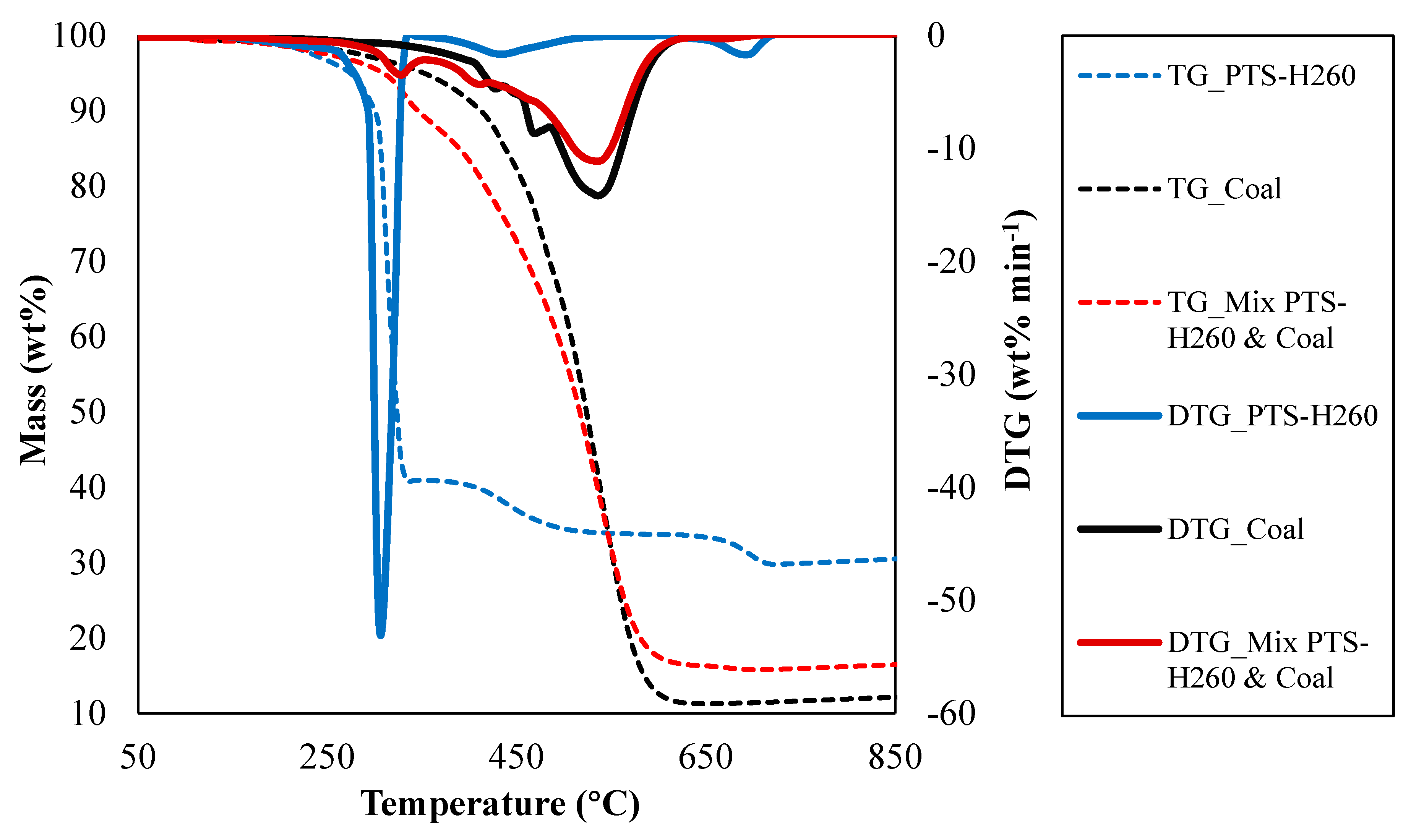
| Sample | Ignition Temperature (°C) | Ignition Time (min) | DTGmax (% min−1) | Burnout Temperature (°C) | Burnout Time (min) | DTGmax Time (min) | Ignition Index, Di | Burnout Index, DB |
|---|---|---|---|---|---|---|---|---|
| PS-H260 | 380.4 | 23.8 | −19.4 | 752.5 | 42.3 | 25.8 | 3.17 × 10−2 | 7.19 × 10−4 |
| DPS-H260 | 628.1 | 36.3 | −6.6 | 788.7 | 44.3 | 41.8 | 4.39 × 10−3 | 9.10 × 10−5 |
| PSS1-H260 | 262.0 | 18.8 | −8.6 | 606.4 | 35.8 | 20.8 | 2.21 × 10−2 | 5.88 × 10−4 |
| PSS2-H260 | 273.0 | 19.3 | −41.7 | 557.1 | 33.3 | 19.8 | 1.10 × 10−1 | 3.26 × 10−3 |
| PFR-H260 | 294.1 | 20.3 | −38.0 | 708.4 | 40.8 | 21.3 | 8.82 × 10−2 | 2.10 × 10−3 |
| PTS-H260 | 294.2 | 20.3 | −52.1 | 718.5 | 41.3 | 20.8 | 1.24 × 10−1 | 2.98 × 10−3 |
| Coal | 466.7 | 28.8 | −14.2 | 627.8 | 36.8 | 32.3 | 1.53 × 10−2 | 4.19 × 10−4 |
| 50% Coal + 50% PS-H260 | 305.3 | 20.8 | −14.5 | 739.4 | 42.3 | 21.8 | 3.22 × 10−2 | 7.49 × 10−4 |
| 50% Coal + 50% PSS1-H260 | 386.3 | 24.8 | −7.0 | 648.0 | 37.8 | 31.8 | 8.97 × 10−3 | 2.10 × 10−4 |
| 50% Coal + 50% PSS2-H260 | 398.7 | 24.8 | −9.9 | 660.3 | 37.8 | 31.3 | 1.28 × 10−2 | 3.16 × 10−4 |
| 50% Coal + 50% PFR-H260 | 410.3 | 25.3 | −7.5 | 701.8 | 39.8 | 31.8 | 9.38 × 10−3 | 2.46 × 10−4 |
| 50% Coal + 50% PTS-H260 | 430.5 | 26.8 | −11.1 | 692.0 | 39.8 | 31.8 | 1.31 × 10−2 | 3.12 × 10−4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saha, N.; Saba, A.; Saha, P.; McGaughy, K.; Franqui-Villanueva, D.; Orts, W.J.; Hart-Cooper, W.M.; Reza, M.T. Hydrothermal Carbonization of Various Paper Mill Sludges: An Observation of Solid Fuel Properties. Energies 2019, 12, 858. https://doi.org/10.3390/en12050858
Saha N, Saba A, Saha P, McGaughy K, Franqui-Villanueva D, Orts WJ, Hart-Cooper WM, Reza MT. Hydrothermal Carbonization of Various Paper Mill Sludges: An Observation of Solid Fuel Properties. Energies. 2019; 12(5):858. https://doi.org/10.3390/en12050858
Chicago/Turabian StyleSaha, Nepu, Akbar Saba, Pretom Saha, Kyle McGaughy, Diana Franqui-Villanueva, William J. Orts, William M. Hart-Cooper, and M. Toufiq Reza. 2019. "Hydrothermal Carbonization of Various Paper Mill Sludges: An Observation of Solid Fuel Properties" Energies 12, no. 5: 858. https://doi.org/10.3390/en12050858
APA StyleSaha, N., Saba, A., Saha, P., McGaughy, K., Franqui-Villanueva, D., Orts, W. J., Hart-Cooper, W. M., & Reza, M. T. (2019). Hydrothermal Carbonization of Various Paper Mill Sludges: An Observation of Solid Fuel Properties. Energies, 12(5), 858. https://doi.org/10.3390/en12050858






