Application of Hydrodynamic Cavitation for Improving Methane Fermentation of Sida hermaphrodita Silage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Substrate
2.2. Equipment
2.3. Cavitation
2.4. Determination of Cellulose, Hemicellulose, and Lignin
2.5. Analytical Methods
3. Results and Discussion
3.1. Pretreatment Efficiency
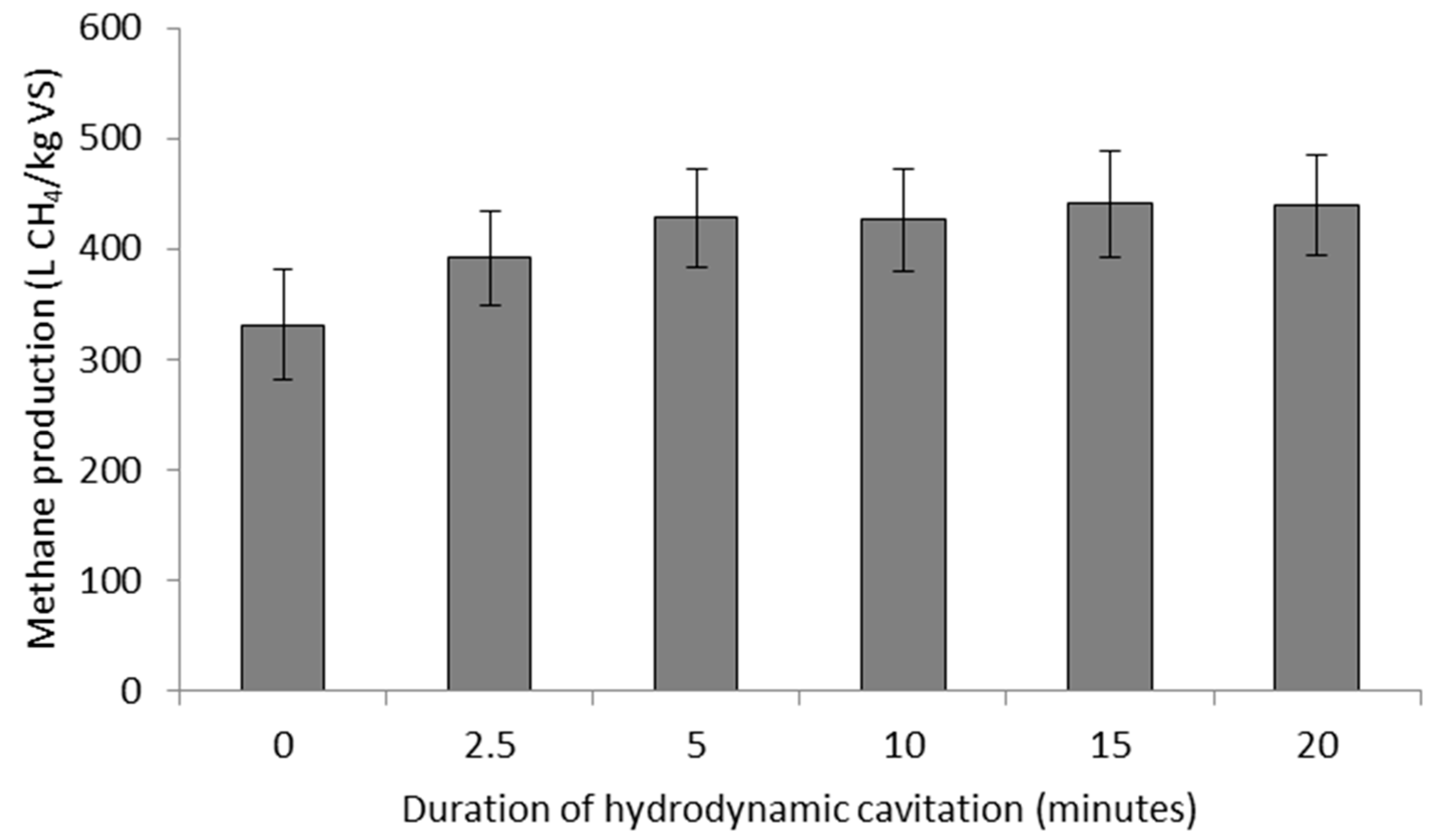
3.2. Methane Production
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bhatia, S.K.; Kim, S.-H.; Yoon, J.-J.; Yang, Y.-H. Current status and strategies for second generation biofuel production using microbial systems. Energy Convers. Manag. 2017, 148, 1142–1156. [Google Scholar] [CrossRef]
- Khanal, S.K.; Rasmussen, M.; Shrestha, P.; Van Leeuwen, H.; Visvanathan, C.; Liu, H. Bioenergy and biofuel production from wastes/residues of emerging biofuel industries. Water Environ. Res. 2008, 80, 1625–1647. [Google Scholar] [CrossRef]
- Paudel, S.R.; Banjara, S.P.; Choi, O.K.; Park, K.Y.; Kim, Y.M.; Lee, J.W. Pretreatment of agricultural biomass for anaerobic digestion: Current state and challenges. Bioresour. Technol. 2017, 245, 1194–1205. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Wang, C.C.J.; Blankschtein, D.; Langer, R. An investigation of the role of cavitation in low-frequency ultrasound-mediated transdermal drug transport. Pharm. Res. 2002, 19, 1160–1169. [Google Scholar] [CrossRef] [PubMed]
- Vanhille, C.; Campos-Pozuelo, C. Nonlinear ultrasonic standing waves: Two-dimensional simulations in bubbly liquids. Ultrason. Sonochem. 2011, 18, 679–682. [Google Scholar] [CrossRef] [PubMed]
- Terán Hilares, R.; Ramos, L.; Da Silva, S.S.; Dragone, G.; Mussatto, S.I.; Dos Santos, J.C. Hydrodynamic cavitation as a strategy to enhance the efficiency of lignocellulosic biomass pretreatment. Crit. Rev. Biotechnol. 2018, 38, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Lee, I.; Jeon, S.H.; Hwang, T.; Han, J.I. Hydrodynamic cavitation as a novel pretreatment approach for bioethanol production from reed. Bioresour. Technol. 2015, 192, 335–339. [Google Scholar] [CrossRef]
- Terán Hilares, R.; Dos Santos, J.C.; Ahmed, M.A.; Jeon, S.H.; Silva, S.S.; Han, J.I. Hydrodynamic cavitation-assisted alkaline pretreatment as a new approach for sugarcane bagasse biorefineries. Bioresour. Technol. 2016, 214, 609–614. [Google Scholar] [CrossRef]
- Nakashima, K.; Ebi, Y.; Shibasaki-Kitakawa, N.; Soyama, H.; Yonemoto, T. Hydrodynamic cavitation reactor for efficient pretreatment of lignocellulosic biomass. Ind. Eng. Chem. Res. 2016, 55, 1866–1871. [Google Scholar] [CrossRef]
- Madison, M.J.; Coward-Kelly, G.; Liang, C.; Karim, M.N.; Falls, M.; Holtzapple, M.T. Mechanical pretreatment of biomass—Part I: Acoustic and hydrodynamic cavitation. Biomass Bioenergy 2016, 98, 500–508. [Google Scholar] [CrossRef]
- Borkowska, H.; Wardzinska, K. Some effects of Sida hermaphrodita R. Cultivation on sewage sludge. Pol. J. Environ. Stud. 2003, 12, 111–122. [Google Scholar]
- Borkowska, H.; Molas, R. Two extremely different crops, Salix and Sida, as sources of renewable bioenergy. Biomass Bioenergy 2012, 36, 234–240. [Google Scholar] [CrossRef]
- Barbosa, D.B.P.; Nabel, M.; Jablonowski, N.D. Biogas-digestate as nutrient source for biomass production of Sida hermaphrodita, Zea mays, L. and Medicago sativa L. Energy Procedia 2014, 59, 120–126. [Google Scholar] [CrossRef]
- Nabel, M.; Bueno, D.; Barbosa, P.; Horsch, D.; Jablonowski, N.D. Energy crop (Sida hermaphrodita) fertilization using digestate under marginal soil conditions: A dose-response experiment. Energy Procedia 2014, 16, 127–133. [Google Scholar] [CrossRef]
- Slepetys, J.; Kadziuliene, Z.; Sarunaite, L.; Tilvikiene, V.; Kryzeviciene, A. Biomass potential of plants grown for bioenergy production. Int. Sci. Conf. Renew. Energy Energy Effic. 2012, 12, 66–72. [Google Scholar]
- Borkowska, H.; Molas, R. Yield comparison of four lignocellulosic perennial energy crop species. Biomass Bioenergy 2013, 51, 145–153. [Google Scholar] [CrossRef]
- Damm, T.; Pattathil, S.; Günl, M.; Jablonowski, N.D.; O’neill, M.; Grün, K.S.; Grande, P.M.; Leitner, W.; Schurr, U.; Usadel, B.; et al. Insights into cell wall structure of Sida hermaphrodita and its influence on recalcitrance. Carbohydr. Polym. 2017, 168, 94–102. [Google Scholar] [CrossRef]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Patil, P.N.; Gogate, P.R. Degradation of methyl parathion using hydrodynamic cavitation: Effect of operating parameters and intensification using additives. Sep. Purif. Technol. 2012, 95, 172–179. [Google Scholar] [CrossRef]
- Barik, A.J.; Gogate, P.R. Degradation of 4-chloro 2-aminophenol using a novel combined process based on hydrodynamic cavitation, UV photolysis and ozone. Ultrason. Sonochem. 2016, 30, 70–78. [Google Scholar] [CrossRef]
- Michalska, K.; Miazek, K.; Krzystek, L.; Ledakowicz, S. Influence of pretreatment with Fenton’s reagent on biogas production and methane yield from lignocellulosic biomass. Bioresour. Technol. 2012, 119, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Krička, T.; Matin, A.; Bilandžija, N.; Jurišić, V.; Antonović, A.; Voća, N.; Grubor, M. Biomass valorisation of Arundo donax L.; Miscanthus × giganteus and Sida hermaphrodita for biofuel production. Int. Agrophys. 2017, 31, 575–581. [Google Scholar] [CrossRef]
- Chen, S.; Wei, L.; Liu, C.; Wen, Z.; Kincaid, R.L.; Harrison, J.H.; Elliot, D.C.; Brown, M.D.; Solana, A.E.; Stevens, D.J. Value-Added Chemicals from Animal Manure; Pacific Northwest National Laboratory: Richlan, WA, USA, 2003; p. 99352. [Google Scholar]
- Habashi, N.; Mehrdadi, N.; Mennerich, A.; Alighardashi, A.; Torabian, A. Hydrodynamic cavitation as a novel approach for pretreatment of oily wastewater for anaerobic co-digestion with waste activated sludge. Ultrason. Sonochem. 2016, 31, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, G.; Langone, M.; Laezza, M.; Andreottola, G. Decolourization of Rhodamine B: A swirling jet-induced cavitation combined with NaOCl. Ultrason. Sonochem. 2016, 32, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Jablonowski, N.D.; Kollmann, T.; Nabel, M.; Damm, T.; Kloose, H.; Müller, M.; Bläsing, M.; Seebold, S.; Krafft, S.; Kuperjans, I.; et al. Valorization of Sida (Sida hermaphrodita) biomass for multiple energy proposes. GCB Bioenergy 2016. [Google Scholar] [CrossRef]
- Patil, P.P.; Gogate, P.R.; Csoka, L.; Dregelyi-Kiss, A.; Horvath, M. Intensification of biogas production using pretreatment based on hydrodynamic cavitation. Ultrason. Sonochem. 2016, 30, 79–86. [Google Scholar] [CrossRef]
- Rusanowska, P.; Zieliński, M.; Dudek, M.R.; Dębowski, M. Mechanical pretreatment of lignocellulosic biomass for methane fermentation in innovative reactor with cage mixing system. J. Ecol. Eng. 2018, 19, 219–224. [Google Scholar] [CrossRef]
- Zieliński, M.; Dębowski, M.; Kisielewska, M. Skuteczność wytwarzania biogazu z wybranych gatunków roślin energetycznych w procesie fermentacji metanowej wspomaganej promieniowaniem mikrofalowym. Ochrona Środowiska 2018, 40, 43–48. [Google Scholar]

| Duration of Hydrodynamic Cavitation (min) | Cellulose (% TS) | Hemicellulose (% TS) | Lignin (% TS) | Glucose Yield (mg/g TS) | Carbohydrate Yield (mg/g TS) |
|---|---|---|---|---|---|
| 0 (control) | 32.8 ± 3.5 | 29.6 ± 3.4 | 19.7 ± 1.3 | 90.2 ± 4.5 | 310.3 ± 9.7 |
| 2.5 | 31.9 ± 2.9 | 28.7 ± 1.4 | 19.5 ± 1.2 | 90.9 ± 2.2 | 324.9 ± 11.8 |
| 5 | 31.6 ± 3.4 | 26.4 ± 2.7 | 19.3 ± 1.5 | 92.1 ± 1.1 | 346.8 ± 19.1 |
| 10 | 30.5 ± 2.1 | 26.2 ± 2.0 | 19.4 ± 2.1 | 93.1 ± 6.3 | 365.5 ± 10.1 |
| 15 | 30.1 ± 2.2 | 26.0 ± 3.1 | 18.5 ± 1.5 | 92.2 ± 3.8 | 366.8 ± 12.3 |
| 20 | 29.5 ± 3.9 | 25.7 ± 1.1 | 18.0 ± 0.9 | 94.3 ± 2.3 | 368.9 ± 14.5 |
| Experiment (min) | TOC Removed (%) | COD Removed (%) | Carbohydrate Removed (%) |
|---|---|---|---|
| 0 (control) | 60.2 ± 5.4 | 53.5 ± 3.2 | 49.1 ± 7.2 |
| 2.5 | 68.1 ± 2.3 | 61.9 ± 5.4 | 53.2 ± 8.1 |
| 5 | 79.3 ± 3.7 | 69.4 ± 4.2 | 59.2 ± 7.9 |
| 10 | 79.4 ± 2.1 | 71.3 ± 2.9 | 59.4 ± 6.3 |
| 15 | 80.4 ± 2.5 | 74.4 ± 3.1 | 60.1 ± 9.2 |
| 20 | 84.1 ± 3.2 | 75.5 ± 6.1 | 63.2 ± 8.8 |
| Experiment (min) | Energy Input (Wh/g TS) | Calorific Value of Produced Biogas (Wh/g TS) | Increase in Energy Due to Cavitation | Energy Profit (Wh/g TS) |
|---|---|---|---|---|
| 0 (control) | 0 | 1.52 | ||
| 2.5 | 0.14 | 1.80 | 0.28 | 0.14 |
| 5 | 0.28 | 1.96 | 0.45 | 0.17 |
| 10 | 0.56 | 1.96 | 0.44 | −0.12 |
| 15 | 0.83 | 2.02 | 0.51 | −0.33 |
| 20 | 1.11 | 2.01 | 0.50 | −0.62 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zieliński, M.; Rusanowska, P.; Krzywik, A.; Dudek, M.; Nowicka, A.; Dębowski, M. Application of Hydrodynamic Cavitation for Improving Methane Fermentation of Sida hermaphrodita Silage. Energies 2019, 12, 526. https://doi.org/10.3390/en12030526
Zieliński M, Rusanowska P, Krzywik A, Dudek M, Nowicka A, Dębowski M. Application of Hydrodynamic Cavitation for Improving Methane Fermentation of Sida hermaphrodita Silage. Energies. 2019; 12(3):526. https://doi.org/10.3390/en12030526
Chicago/Turabian StyleZieliński, Marcin, Paulina Rusanowska, Aleksandra Krzywik, Magda Dudek, Anna Nowicka, and Marcin Dębowski. 2019. "Application of Hydrodynamic Cavitation for Improving Methane Fermentation of Sida hermaphrodita Silage" Energies 12, no. 3: 526. https://doi.org/10.3390/en12030526
APA StyleZieliński, M., Rusanowska, P., Krzywik, A., Dudek, M., Nowicka, A., & Dębowski, M. (2019). Application of Hydrodynamic Cavitation for Improving Methane Fermentation of Sida hermaphrodita Silage. Energies, 12(3), 526. https://doi.org/10.3390/en12030526







