Preliminary Study on the Use of Biodiesel Obtained from Waste Vegetable Oils for Blending with Hydrotreated Kerosene Fossil Fuel Using Calcium Oxide (CaO) from Natural Waste Materials as Heterogeneous Catalyst
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Equipment Used
2.3. Preparation of Natural CaO Catalysts
2.4. Waste Cooking Oil Pre-Treatment
2.5. Transesterification Reaction Process
2.6. Biodiesel Purification Process
3. Results and Discussion
3.1. Catalyst Characterization
3.2. Biodiesel Characterization
3.3. Biodiesel Repeatability Test
3.4. Catalytic Stability Test
3.5. Kinetic Tests
3.6. Determination of Physical Properties of Produced Biodiesel
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, W.; Tao, L.; Markham, J.; Zhang, Y.; Tan, E.; Batan, L.; Warner, E.; Biddy, M. Review of Biojet Fuel Conversion Technologies; National Renewable Energy Laboratory: Golden, CO, USA, 2016. [Google Scholar]
- Deane, P.; Shea, R.; Gallachoir, B.; Buchholz, D. Biofuels for Aviation; Rapid Response Energy Brief; Insight-E: Brussels, Belgium, 2015. [Google Scholar]
- Korres, D.M.; Karonis, D.; Lois, E.; Linck, M.B.; Gupta, A.K. Aviation fuel JP-5 and biodiesel on a diesel engine. Fuel 2008, 87, 70–78. [Google Scholar] [CrossRef]
- Hari, T.K.; Yaakob, Z.; Binitha, N.N. Aviation biofuel from renewable resources: Routes, opportunities and challenges. Renew. Sustain. Energy Rev. 2015, 42, 1234–1244. [Google Scholar] [CrossRef]
- Tunca, I. Biyodizel hakkında herşey. Biyodizel Derg. 2013, 8, 41–49. (In Turkish) [Google Scholar]
- Bartan, A. Biyodizel Üretiminde Heterojen Katalizör Geliştirilmesi. Master’s Thesis, Gazi Üniversitesi, Ankara, Turkey, 2009. (In Turkish). [Google Scholar]
- Puna, J.; Gomes, J.; Correia, M.J.N.; Dias, A.S.; Bordado, J.; Puna, J.; Correia, M.J.N.; Dias, A.P.S. Advances on the development of novel heterogeneous catalysts for transesterification of triglycerides in biodiesel. Fuel 2010, 89, 3602–3606. [Google Scholar] [CrossRef]
- Puna, J.F.; Correia, M.J.N.; Dias, A.P.S.; Gomes, J.; Bordado, J. Biodiesel production from waste frying oils over lime catalysts. React. Kinet. Mech. Catal. 2013, 109, 405–415. [Google Scholar] [CrossRef][Green Version]
- Marchetti, J.M.; Miguel, V.; Errazu, A. Possible methods for biodiesel production. Renew. Sustain. Energy Rev. 2007, 11, 1300–1311. [Google Scholar] [CrossRef]
- Demirbaş, A. Progress and recent trends in biodiesel fuels. Energy Convers. Manag. 2009, 50, 14–34. [Google Scholar] [CrossRef]
- Boro, J.; Deka, D.; Thakur, A.J. A review on solid oxide derived from waste shells as catalyst for biodiesel production. Renew. Sustain. Energy Rev. 2012, 16, 904–910. [Google Scholar] [CrossRef]
- BP Statistical Review of World Energy December 2018. Available online: https://www.bp.com/content/dam/bp/business-sites/en/global/corporate/pdfs/energy-economics/statistical-review/bp-stats-review-2019-full-report.pdf (accessed on 25 October 2019).
- Ozcimen, D.; Yucel, S. Novel methods in biodiesel production. Proc. Biofuel Eng. Process Technol. 2011, 8, 353–384. [Google Scholar]
- Shan, R.; Zhao, C.; Lv, P.; Yuan, H.; Yao, J. Catalytic applications of calcium rich waste materials for biodiesel: Current state and perspectives. Energy Convers. Manag. 2016, 127, 273–283. [Google Scholar] [CrossRef]
- Puna, J.F.; Gomes, J.F.; Bordado, J.C.; Correia, M.J.N.; Dias, A.P.S. Biodiesel production over lithium modified lime catalysts: Activity and deactivation. Appl. Catal. A Gen. 2014, 470, 451–457. [Google Scholar] [CrossRef]
- Gomes, J.F.; Puna, J.F.; Gonçalves, L.M.; Bordado, J.C. Study on the use of MgAl hydrotalcites as solid heterogeneous catalysts for biodiesel production. Energy 2011, 36, 6770–6778. [Google Scholar] [CrossRef]
- Granados, M.L.; Poves, M.Z.; Alonso, D.M.; Mariscal, R.; Galisteo, F.C.; Moreno-Tost, R.; Santamaria, J.; Fierro, J. Biodiesel from sunflower oil by using activated calcium oxide. Appl. Catal. B Environ. 2007, 73, 317–326. [Google Scholar] [CrossRef]
- Dias, A.P.S.; Puna, J.; Gomes, J.; Correia, M.J.N.; Bordado, J. Biodiesel production over lime. Catalytic contributions of bulk phases and surface Ca species formed during reaction. Renew. Energy 2016, 99, 622–630. [Google Scholar] [CrossRef]
- Catarino, M.; Ramos, M.; Dias, A.P.S.; Santos, M.T.; Puna, J.F.; Gomes, J.F. Calcium Rich Food Wastes Based Catalysts for Biodiesel Production. Waste Biomass Valorization 2017, 8, 1699–1707. [Google Scholar] [CrossRef]
- Semwal, S.; Arora, A.; Badoni, P.; Tuli, D. Biodiesel production using heterogeneous catalysts. Bioresour. Technol. 2011, 102, 2151–2161. [Google Scholar] [CrossRef] [PubMed]
- Marwaha, A.; Rosha, P.; Mohapatra, S.K.; Mahla, S.K.; Dhir, A. Waste materials as potential catalysts for biodiesel production: Current state and future scope. Fuel Process. Technol. 2018, 181, 175–186. [Google Scholar] [CrossRef]
- Lee, S.L.; Wong, Y.C.; Tan, Y.P.; Yew, S.Y. Transesterification of palm oil to biodiesel by using waste obtuse horn shell-derived CaO catalyst. Energy Convers. Manag. 2015, 93, 282–288. [Google Scholar] [CrossRef]
- Nakatani, N.; Takamori, H.; Takeda, K.; Sakugawa, H. Transesterification of soybean oil using combusted oyster shell waste as a catalyst. Bioresour. Technol. 2009, 100, 1510–1513. [Google Scholar] [CrossRef] [PubMed]
- Boey, P.L.; Maniam, G.P.; Hamid, S.A. Biodiesel production via transesterification of palm oil using waste mud crab (Scylla serrata) shell as a heterogeneous catalyst. Bioresour. Technol. 2009, 100, 6362–6368. [Google Scholar] [CrossRef] [PubMed]
- Viriya-Empikul, N.; Krasae, P.; Nualpaeng, W.; Yoosuk, B.; Faungnawakij, K. Biodiesel production over Ca-based solid catalysts derived from industrial wastes. Fuel 2012, 92, 239–244. [Google Scholar] [CrossRef]
- Viriya-Empikul, N.; Krasae, P.; Puttasawat, B.; Yoosuk, B.; Chollacoop, N.; Faungnawakij, K. Waste shells of mollusk and egg as biodiesel production catalysts. Bioresour. Technol. 2010, 101, 3765–3767. [Google Scholar] [CrossRef] [PubMed]
- Piker, A.; Tabah, B.; Perkas, N.; Gedanken, A. A green and low-cost room temperature biodiesel production method from waste oil using eggshells as catalyst. Fuel 2016, 182, 34–41. [Google Scholar] [CrossRef]
- Kaewdaeng, S.; Sintuya, P.; Nirunsin, J. Biodiesel production using CaO from river snail shell ash as catalyst. Energy Procedia 2017, 138, 937–942. [Google Scholar] [CrossRef]
- Birla, A.; Singh, B.; Upadhyay, S.; Sharma, Y. Kinetics studies of synthesis of biodiesel from waste frying oil using a heterogeneous catalyst derived from snail shell. Bioresour. Technol. 2012, 106, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zheng, Y.; Chen, Y.; Zhu, X. Biodiesel production from waste cooking oil using a heterogeneous catalyst from pyrolyzed rice husk. Bioresour. Technol. 2014, 154, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, S.H.Y.S.; Hanapi, N.H.M.; Azid, A.; Umar, R.; Juahir, H.; Khatoon, H.; Endut, A. A review of biomass-derived heterogeneous catalyst for a sustainable biodiesel production. Renew. Sustain. Energy Rev. 2017, 70, 1040–1051. [Google Scholar] [CrossRef]
- Kulkarni, M.G.; Dalai, A.K. Waste Cooking OilAn Economical Source for Biodiesel: A Review. Ind. Eng. Chem. Res. 2006, 45, 2901–2913. [Google Scholar] [CrossRef]
- Melero, J.A.; Iglesias, J.; Morales, G. Heterogeneous acid catalysts for biodiesel production: Current status and future challenges. Green Chem. 2009, 11, 1285–1308. [Google Scholar] [CrossRef]
- Shan, R.; Lu, L.; Shi, Y.; Yuan, H.; Shi, J. Catalyst from renewable sources for biodiesel production. Energy Convers. Manag. 2018, 178, 277–289. [Google Scholar] [CrossRef]
- Dias, A.P.S.; Puna, J.; Correia, M.J.N.; Nogueira, I.; Gomes, J.; Bordado, J. Effect of the oil acidity on the methanolysis performances of lime catalyst biodiesel from waste frying oils (WFO). Fuel Process. Technol. 2013, 116, 94–100. [Google Scholar] [CrossRef]
- Lam, M.K.; Lee, K.T.; Mohamed, A.R. Homogeneous, heterogeneous and enzymatic catalysis for transesterification of high free fatty acid oil (waste cooking oil) to biodiesel: A review. Biotechnol. Adv. 2010, 28, 500–518. [Google Scholar] [CrossRef] [PubMed]
- Kouzu, M.; Kasuno, T.; Tajika, M.; Sugimoto, Y.; Yamanaka, S.; Hidaka, J. Calcium oxide as a solid base catalyst for transesterification of soybean oil and its application to biodiesel production. Fuel 2008, 87, 2798–2806. [Google Scholar] [CrossRef]
- Vujicic, D.; Comic, D.; Zarubica, A.; Micic, R.; Bošković, G. Kinetics of biodiesel synthesis from sunflower oil over CaO heterogeneous catalyst. Fuel 2010, 89, 2054–2061. [Google Scholar] [CrossRef]
- Khemthong, P.; Luadthong, C.; Nualpaeng, W.; Changsuwan, P.; Tongprem, P.; Viriya-Empikul, N.; Faungnawakij, K. Industrial eggshell wastes as the heterogeneous catalysts for microwave-assisted biodiesel production. Catal. Today 2012, 190, 112–116. [Google Scholar] [CrossRef]
- Miladinović, M.R.; Krstić, J.B.; Tasić, M.B.; Stamenković, O.S.; Veljković, V.B. A kinetic study of quicklime-catalyzed sunflower oil methanolysis. Chem. Eng. Res. Des. 2014, 92, 1740–1752. [Google Scholar] [CrossRef]
- Chouhan, A.S.; Sarma, A. Modern heterogeneous catalysts for biodiesel production: A comprehensive review. Renew. Sustain. Energy Rev. 2011, 15, 4378–4399. [Google Scholar] [CrossRef]

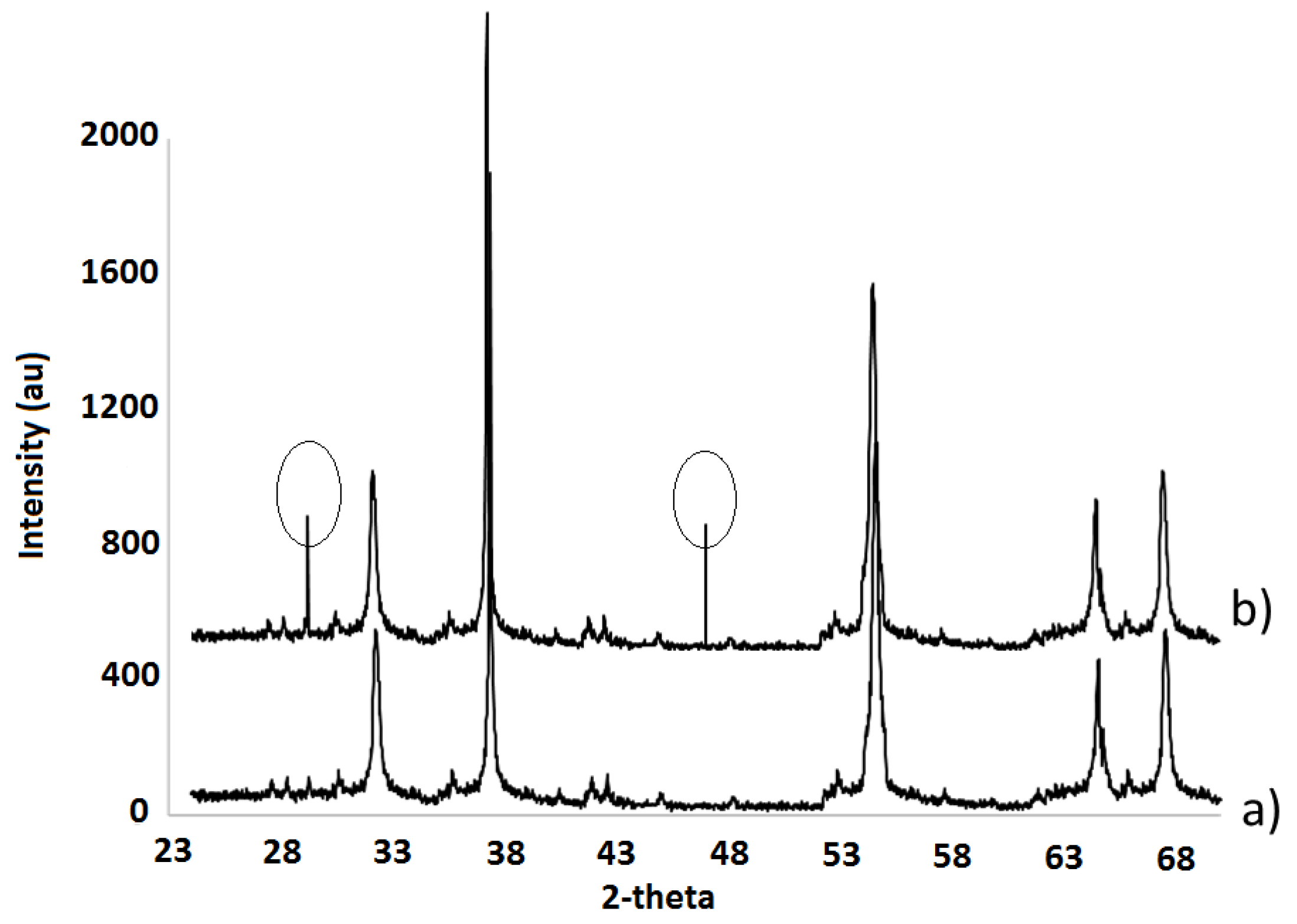
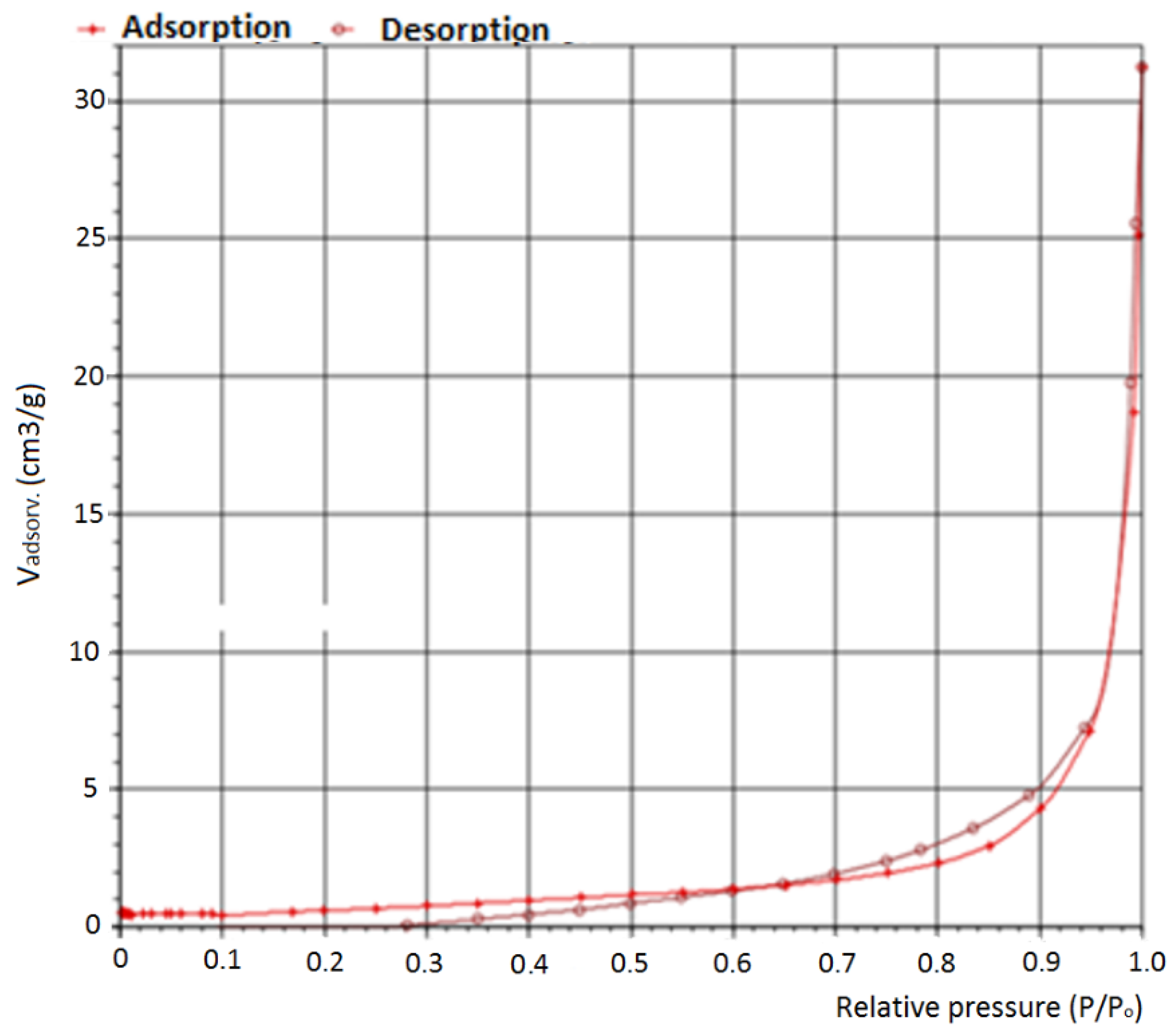
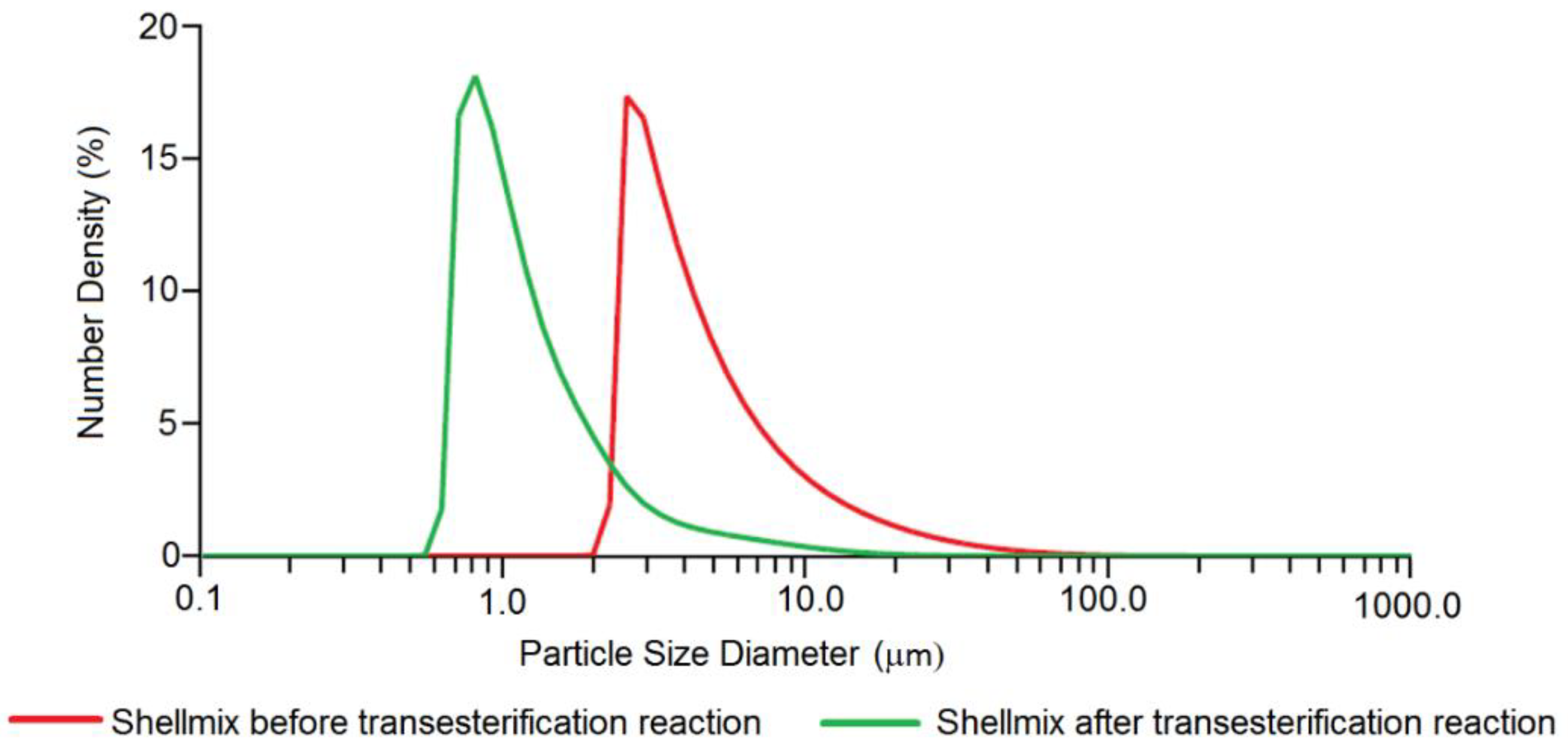
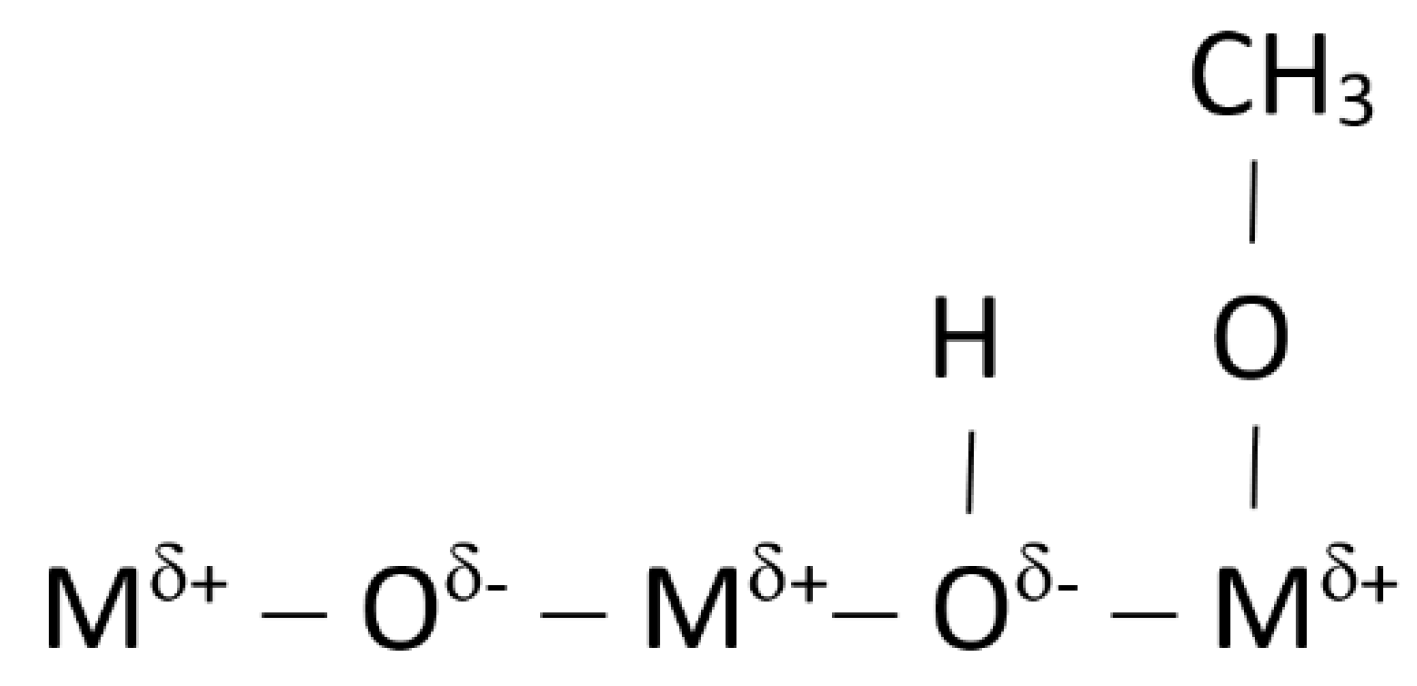
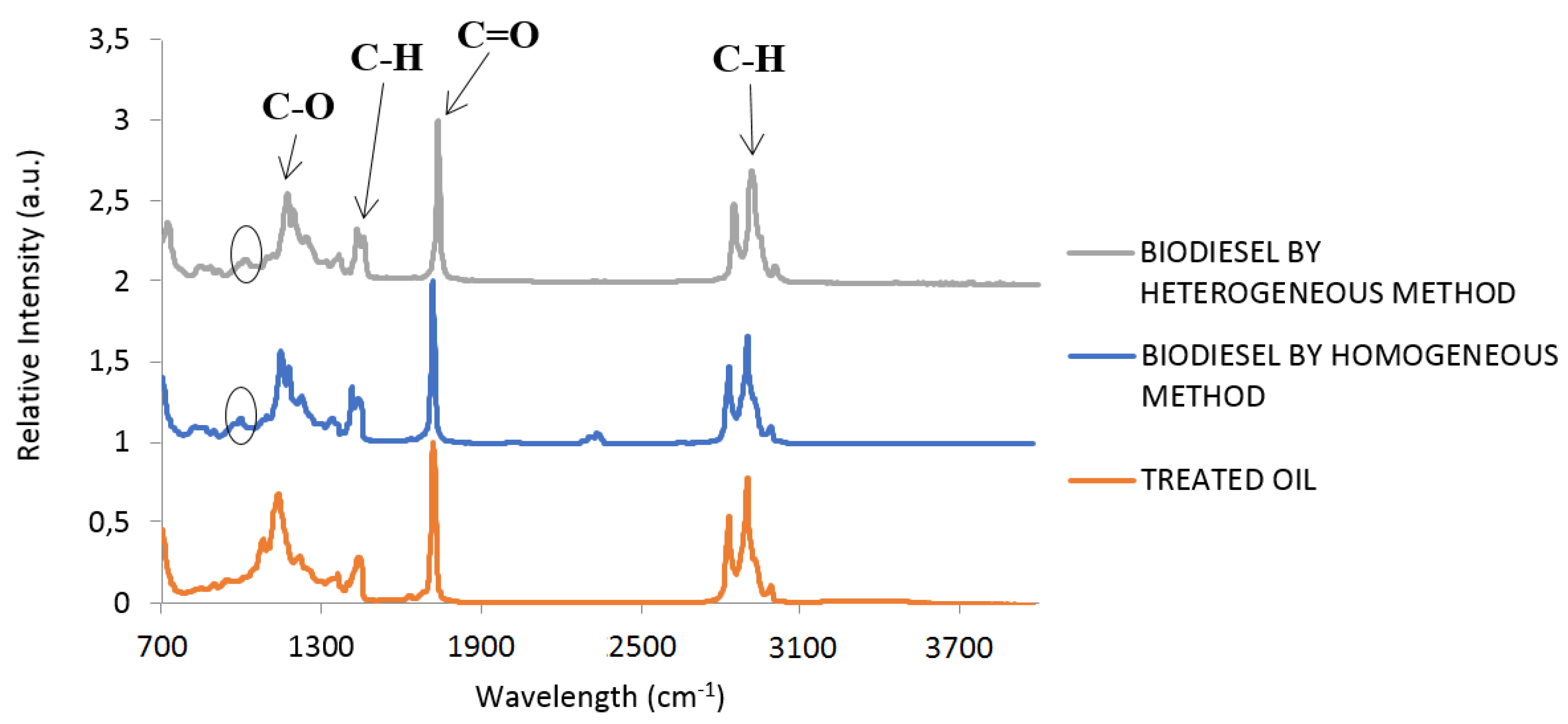
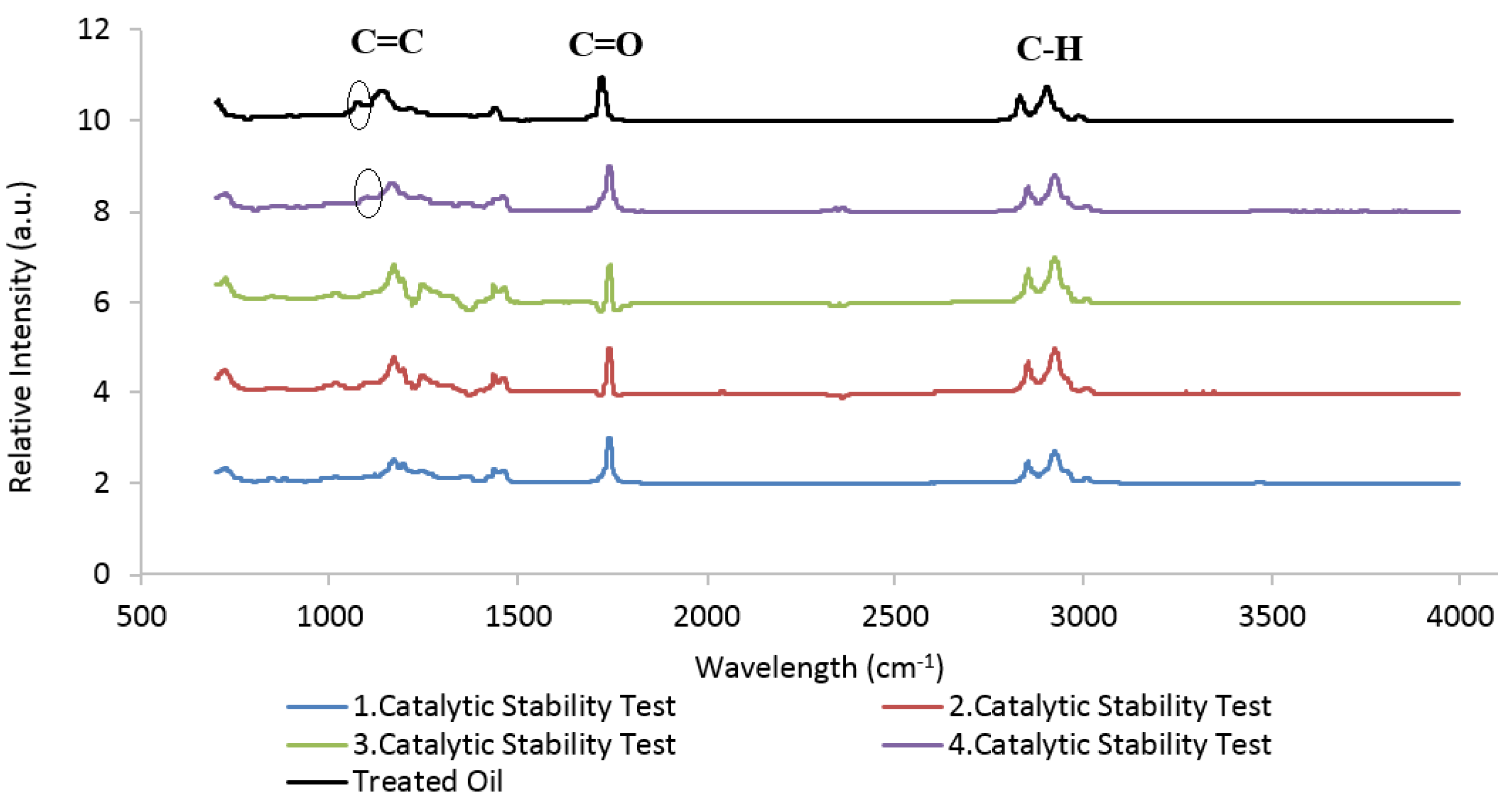

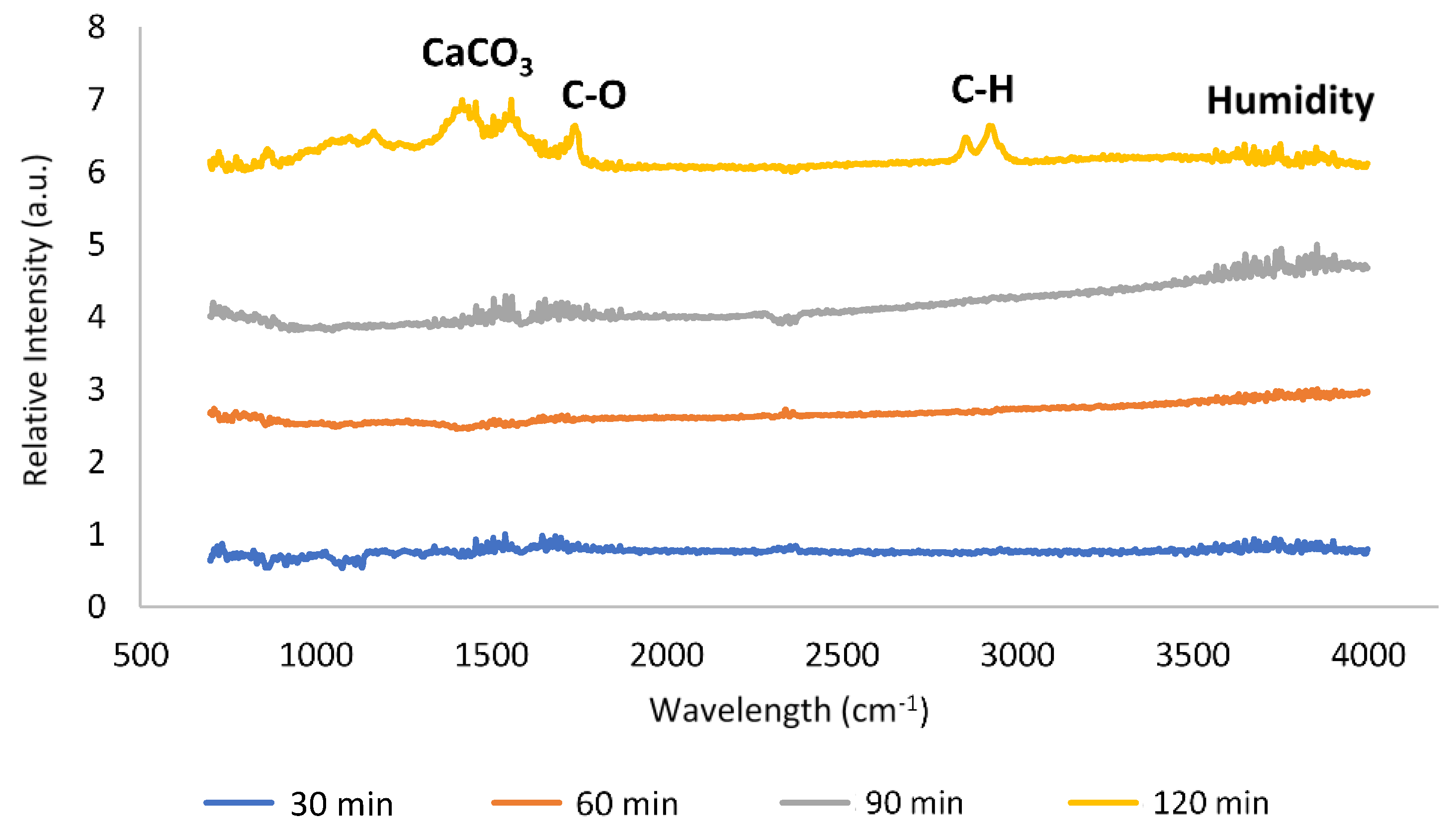
| Element | Heterogeneous Process | Homogeneous Process |
|---|---|---|
| T (°C) | 60 | 60 |
| t (h) | 2 | 2 |
| MeOH/WCO molar ratio | 12 | 6 |
| Catalyst | Shell mix (CaO), c. 850 °C | NaOH with methanol |
| % (Wcat./WWCO) | 5.0% | 0.6% |
| Element | “Shellm.” before Calcination | “Shellm.” after Calcination | “Shellm.” after First Reaction |
|---|---|---|---|
| C | 21.6 ± 1.0% | 4.39 ± 1.1% | 8.1 ± 1.0% |
| O | 59.3 ± 1.3% | 59.9 ± 1.1% | 63.5 ± 1.2% |
| Ca | 19.1 ± 0.9% | 35.7 ± 1.0% | 28.4 ± 1.1% |
| Type of Catalyst | Reaction Time (h) | Refractive Index, 25 °C | FAME Conversion (%) |
|---|---|---|---|
| Shell mix (former composition) | 4 | 1.47735 | 1.10 |
| 5 | 1.47731 | 1.20 | |
| 6 | 1.47729 | 1.30 | |
| 5 (separately calcinated) | 1.46709 | 49.9 | |
| 2 (Methanol + catalyst, 3 h) | 1.47733 | 1.20 | |
| 1 (Methanol + catalyst, 7 h) | 1.45669 | >99.5 | |
| 1 (Methanol + catalyst, 3 h) (a) | 1.47725 | 1.80 | |
| White-colored clam | 6 | 1.46700 | 50.0 |
| Shrimp shell | 6 | 1.45665 | >99.5 |
| “Navalha” shell | 6 | 1.45663 | >99.5 |
| Dark-colored clam | 6 | 1.45662 | >99.5 |
| Ca(OMe)2 | 5 | 1.47730 | 1.20 |
| 7 | 1.46716 | 50.8 | |
| 8 | 1.45661 | >99.5 | |
| Ca(OH)2 | 5 | 1.47727 | 1.40 |
| 7 | 1.47726 | 1.40 | |
| 8 | 1.45661 | >99.5 | |
| Glycerol + Methanol (b) | 2 | 1.45662 | >99.5 |
| Shell mix 5%(w/w) (c) | 6 | 1.45662 | >99.5 |
| Shell mix 3%(w/w) (c) | 6 | 1.46690 | 50.4 |
| Number of Experiment (#) | FAME Yield (% Wbiod./WWCO) |
|---|---|
| #1 | 99.9 |
| #2 | 99.8 |
| #3 | 99.9 |
| #4 | 99.7 |
| #5 | 99.8 |
| Number of Experiment (#) | FAME Yield (% Wbiod./WWCO) |
|---|---|
| #1 | 99.9 ± 1.0% |
| #2 | 99.9 ± 1.2% |
| #3 | 99.8 ± 0.9% |
| #4 | 28.9 ± 2.2% |
| Reaction Time (min.) | FAME Yield (% Wbiod./WWCO) |
|---|---|
| 0 | 0.00 |
| 30 | 1.30 |
| 45 | 1.50 |
| 60 | 1.80 |
| 75 | 24.8 |
| 80 | 25.0 |
| 90 | 99.7 |
| 120 | 99.8 |
| 180 | 99.8 |
| 240 | 99.8 |
| 300 | 99.9 |
| 360 | 99.9 |
| Biodiesel Samples | Acidity Index (AI) Control 1 | Acidity Index (AI) Control 2 | Average Acidity Index (AI) |
|---|---|---|---|
| #1 | 0.57 | 0.56 | 0.57 ± 1.3% |
| #2 | 0.55 | 0.53 | 0.54 ± 2.6% |
| #3 | 0.42 | 0.44 | 0.43 ± 1.6% |
| #4 | 0.56 | 0.55 | 0.56 ± 1.3% |
| #5 | 0.55 | 0.56 | 0.56 ± 1.3% |
| Sample | Density (kg/m3) |
|---|---|
| Biodiesel by homogeneous method | 865.0 (25 °C) |
| Biodiesel by heterogeneous method (first try of catalytic stability test) | 890.5 (25 °C) 842.2 (40 °C) |
| Biodiesel by heterogeneous method (second try of catalytic stability test) | 889.9 (25 °C) 839.5 (40 °C) |
| Biodiesel by heterogeneous method (third try of catalytic stability test) | 892.7 (25 °C) 841.3 (40 °C) |
| Biodiesel by heterogeneous method (four try of catalytic stability test) | 919.8 (25 °C) 868.6 (40 °C) |
| Biodiesel according to EN 14214 | 860–900 (15 °C) |
| Treated oil | 925.0 (25 °C) 877.6 (40 °C) |
| Jet-A1 | 793.8 (25 °C) |
| Blend of HK fuel (1% biodiesel additive) | 793.6 (25 °C) |
| Blend of HK fuel (2% biodiesel additive) | 794.8 (25 °C) |
| Blend of HK fuel (3% biodiesel additive) | 795.0 (25 °C) |
| Blend of HK fuel (5% biodiesel additive) | 796.2 (25 °C) |
| Blend of HK fuel (10% biodiesel additive) | 801.9 (25 °C) |
| Jet-A1 specifications | 775.0–840.0 (15 °C) |
| Sample | Kinematic Viscosity (mm2·s−1), 40 °C |
|---|---|
| Biodiesel by homogeneous method | 3.50 |
| Biodiesel by heterogeneous method (first try out of catalytic stability test) | 3.72 |
| Biodiesel by heterogeneous method (second try out of catalytic stability test) | 3.52 |
| Biodiesel by heterogeneous method (third try out of catalytic stability test) | 3.60 |
| Biodiesel by heterogeneous method (four try out of catalytic stability test) | 12.6 |
| Biodiesel according to EN 14214 | 3.5–5.0 |
| Treated oil | 32.9 |
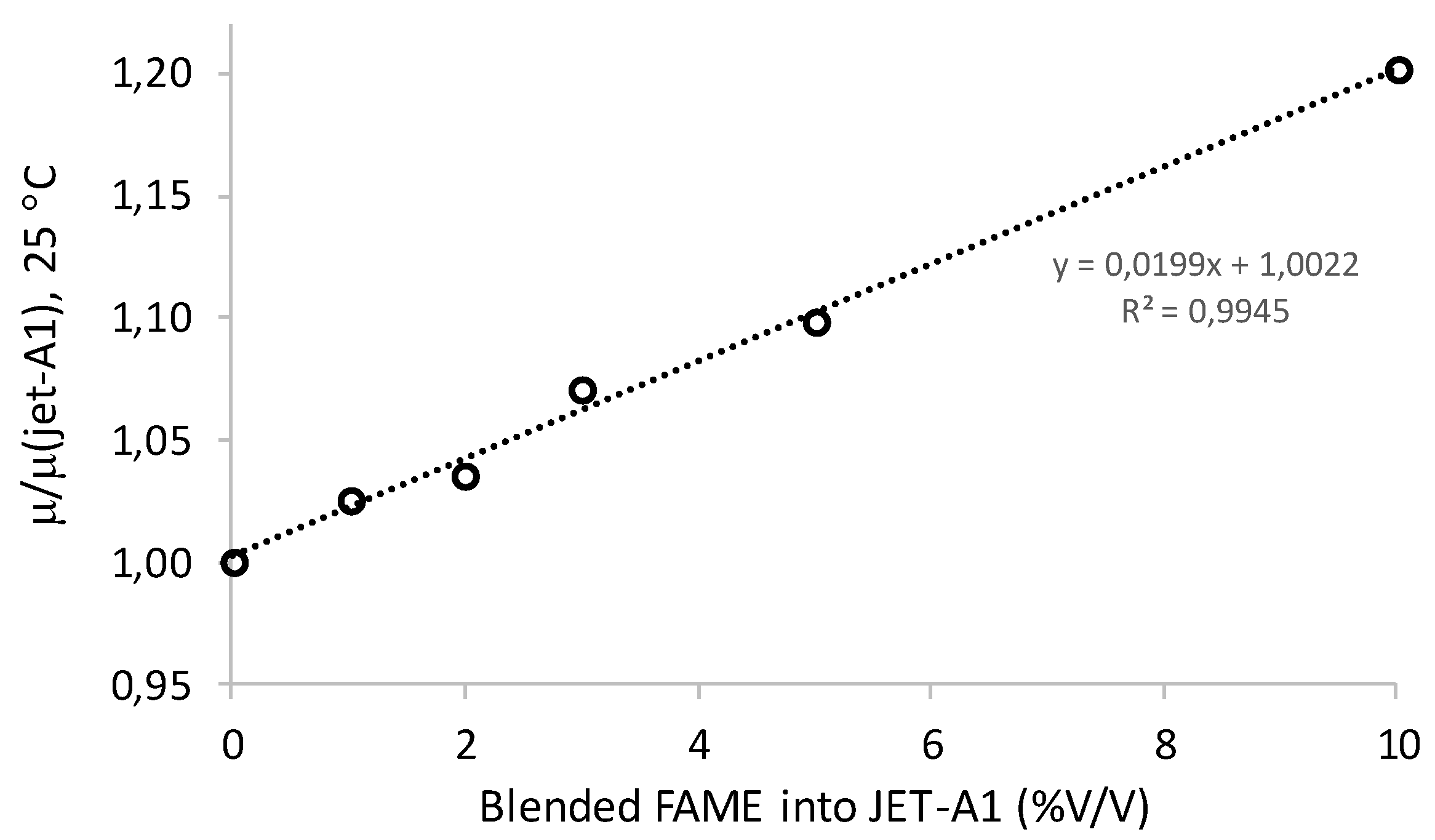
| Jet-A1 | 1.00 (*) |
| Blend of HK fuel (1% biodiesel additive) | 1.03 (*) |
| Blend of HK fuel (2% biodiesel additive) | 1.04 (*) |
| Blend of HK fuel (3% biodiesel additive) | 1.07 (*) |
| Blend of HK fuel (5% biodiesel additive) | 1.10 (*) |
| Blend of HK fuel (10% biodiesel additive) | 1.20 (*) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ozkan, S.; Puna, J.F.; Gomes, J.F.; Cabrita, T.; Palmeira, J.V.; Santos, M.T. Preliminary Study on the Use of Biodiesel Obtained from Waste Vegetable Oils for Blending with Hydrotreated Kerosene Fossil Fuel Using Calcium Oxide (CaO) from Natural Waste Materials as Heterogeneous Catalyst. Energies 2019, 12, 4306. https://doi.org/10.3390/en12224306
Ozkan S, Puna JF, Gomes JF, Cabrita T, Palmeira JV, Santos MT. Preliminary Study on the Use of Biodiesel Obtained from Waste Vegetable Oils for Blending with Hydrotreated Kerosene Fossil Fuel Using Calcium Oxide (CaO) from Natural Waste Materials as Heterogeneous Catalyst. Energies. 2019; 12(22):4306. https://doi.org/10.3390/en12224306
Chicago/Turabian StyleOzkan, S., J. F. Puna, J. F. Gomes, T. Cabrita, J. V. Palmeira, and M. T. Santos. 2019. "Preliminary Study on the Use of Biodiesel Obtained from Waste Vegetable Oils for Blending with Hydrotreated Kerosene Fossil Fuel Using Calcium Oxide (CaO) from Natural Waste Materials as Heterogeneous Catalyst" Energies 12, no. 22: 4306. https://doi.org/10.3390/en12224306
APA StyleOzkan, S., Puna, J. F., Gomes, J. F., Cabrita, T., Palmeira, J. V., & Santos, M. T. (2019). Preliminary Study on the Use of Biodiesel Obtained from Waste Vegetable Oils for Blending with Hydrotreated Kerosene Fossil Fuel Using Calcium Oxide (CaO) from Natural Waste Materials as Heterogeneous Catalyst. Energies, 12(22), 4306. https://doi.org/10.3390/en12224306





