Experimental Study of Electromagnetic-Assisted Rare-Earth Doped Yttrium Iron Garnet (YIG) Nanofluids on Wettability and Interfacial Tension Alteration
Abstract
1. Introduction
2. Methodology
2.1. Materials and Methods
2.2. Characterization of RE-YIG Nanoparticles
2.3. Evaluation of RE-YIG Nanofluids
3. Results and Discussions
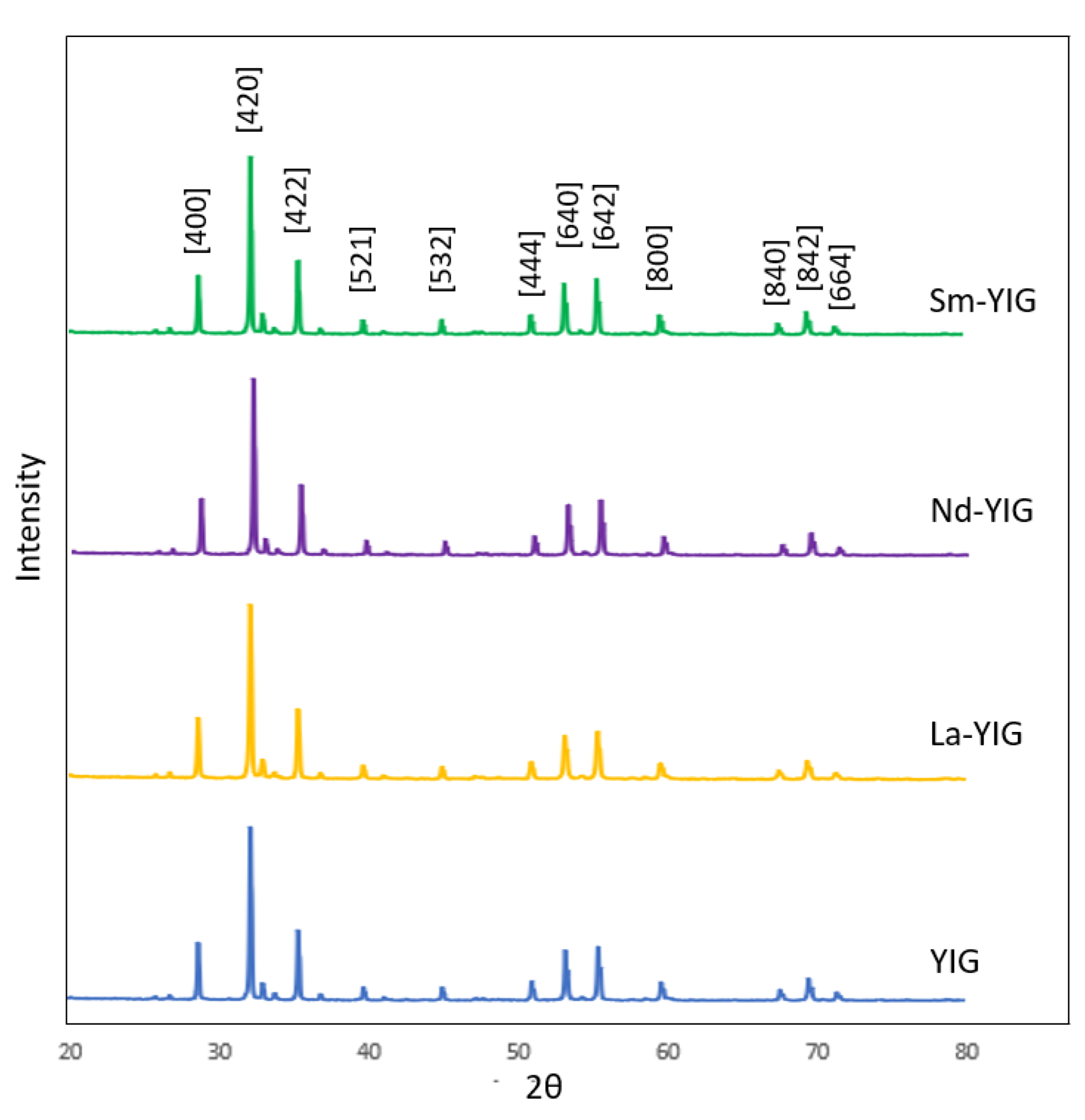
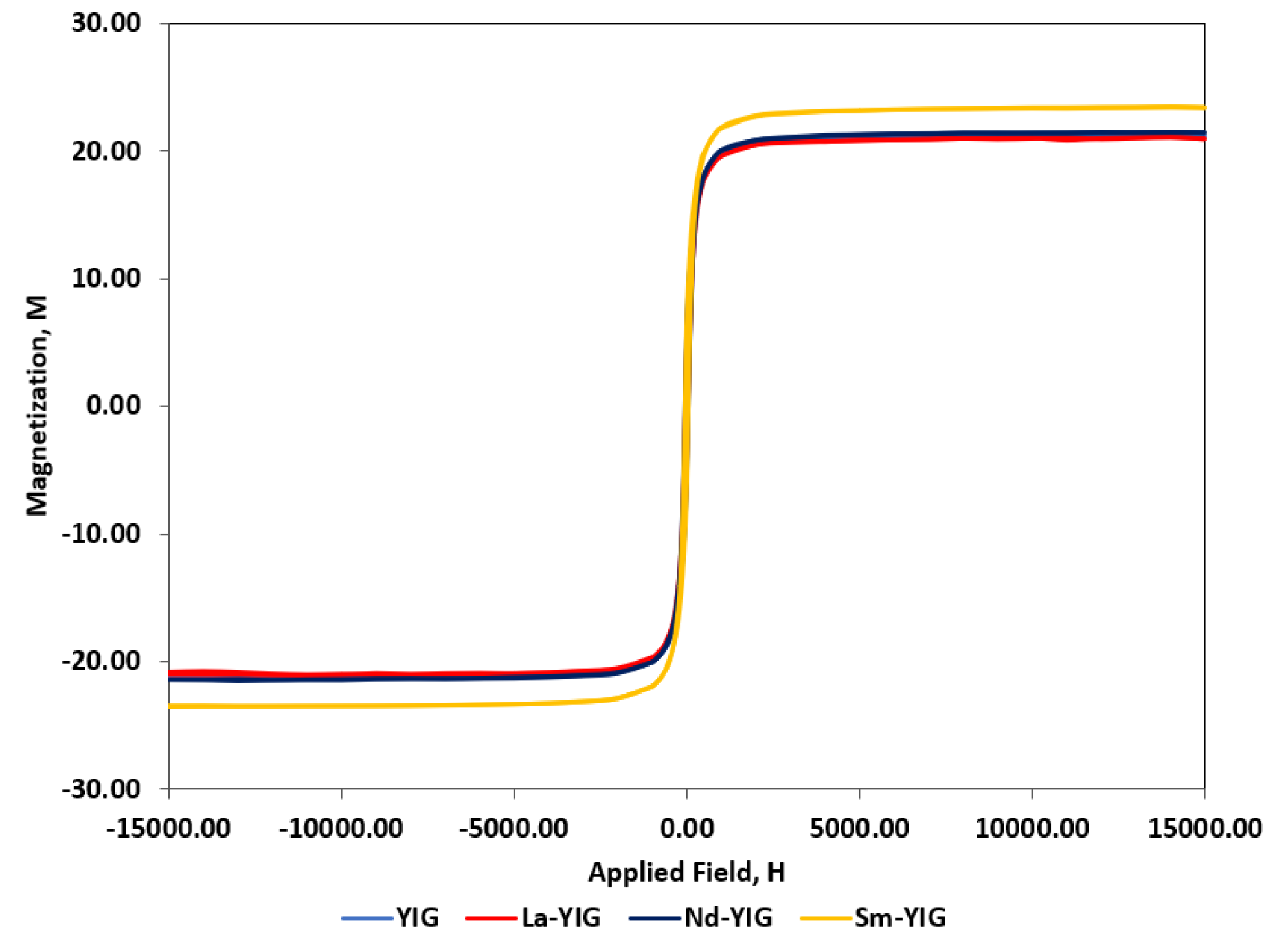
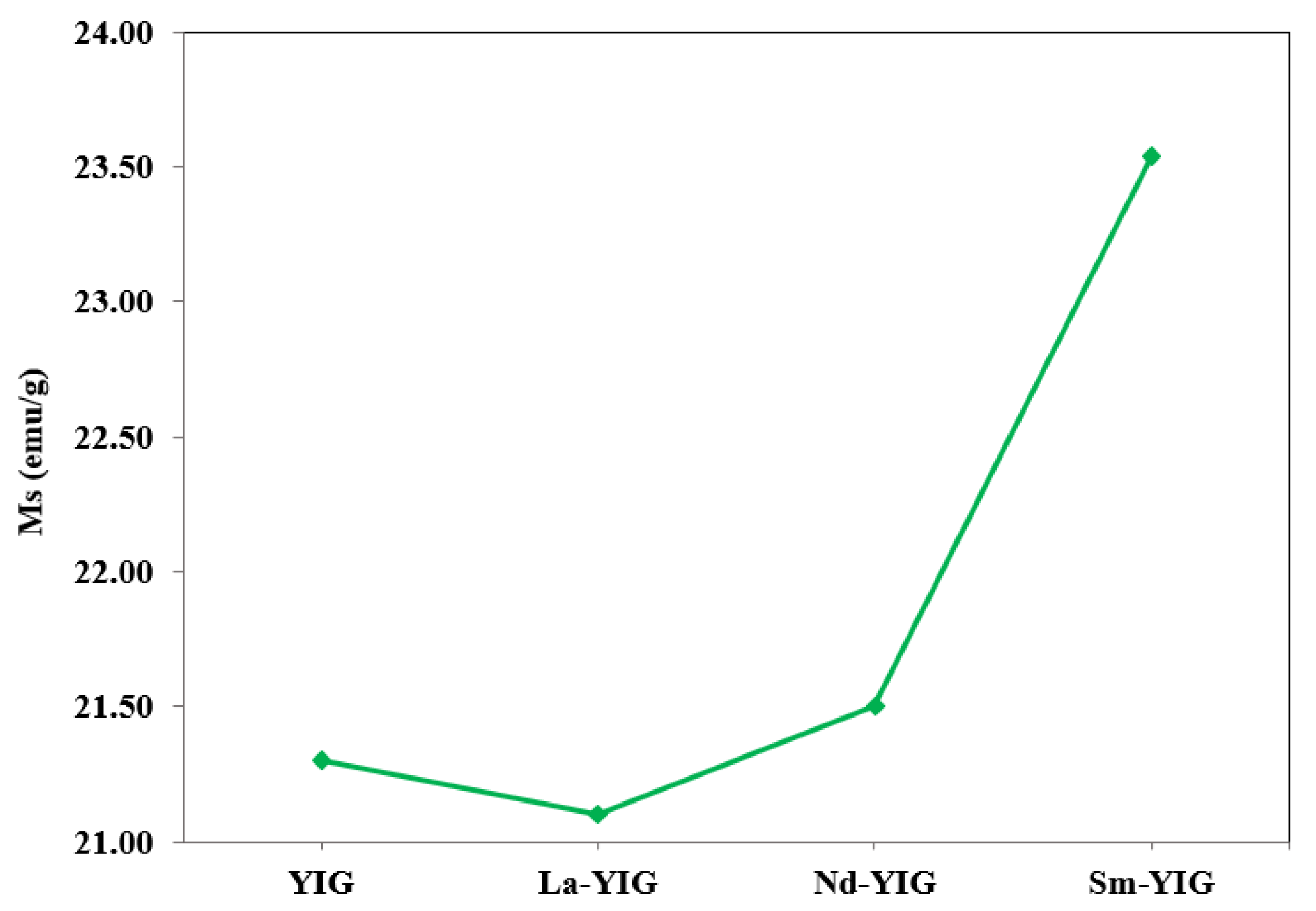
3.1. Characterization of RE-YIG Nanoparticles
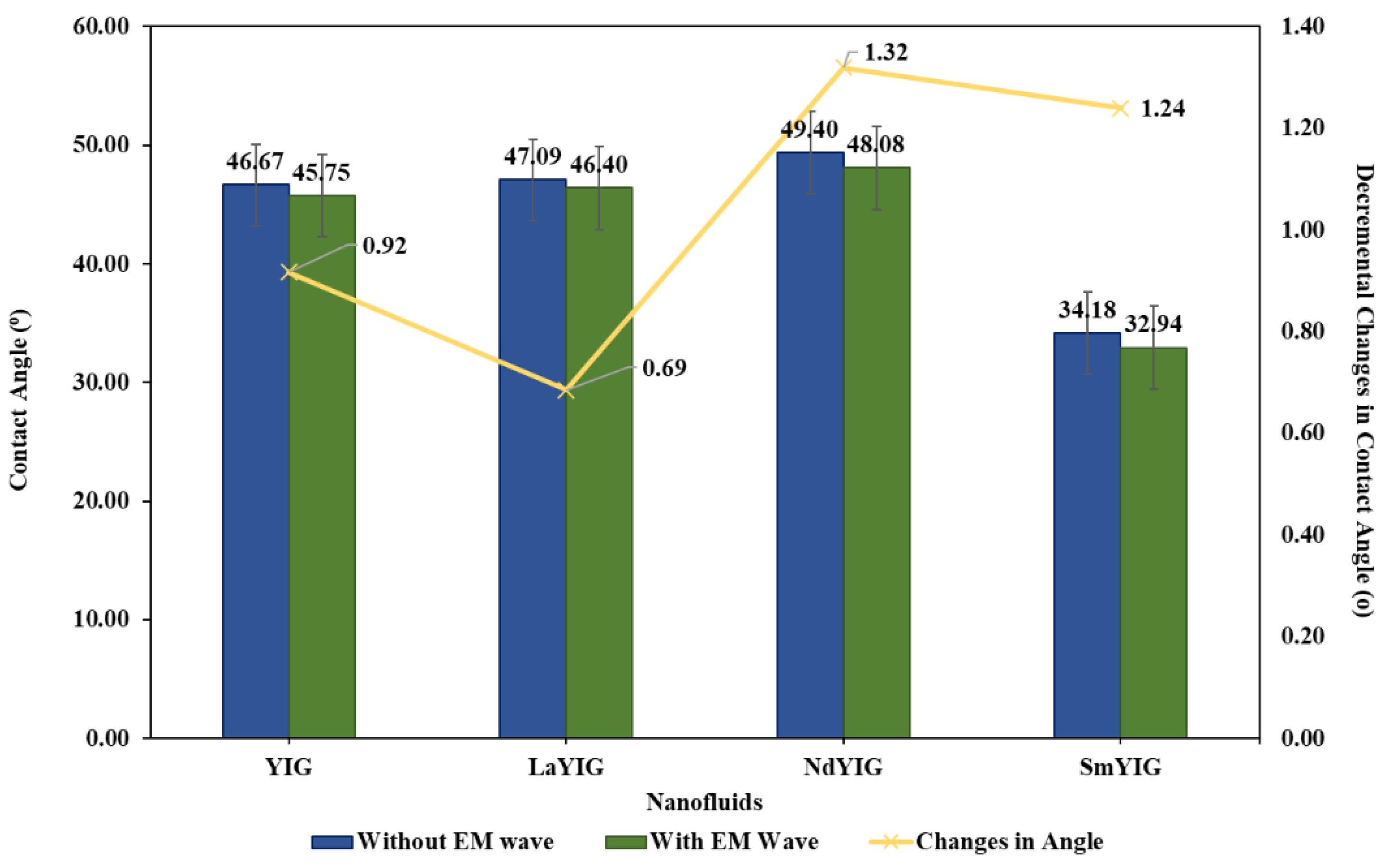
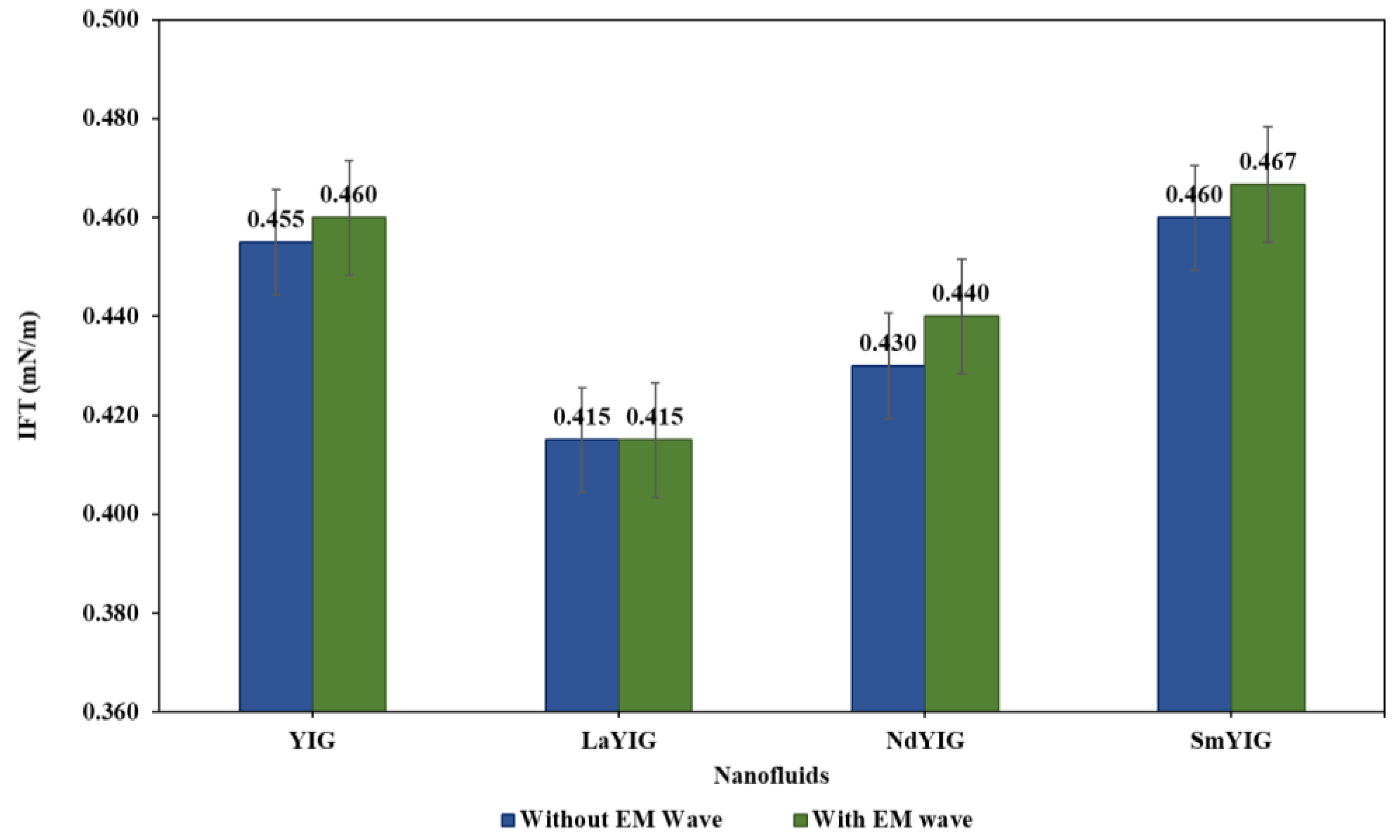
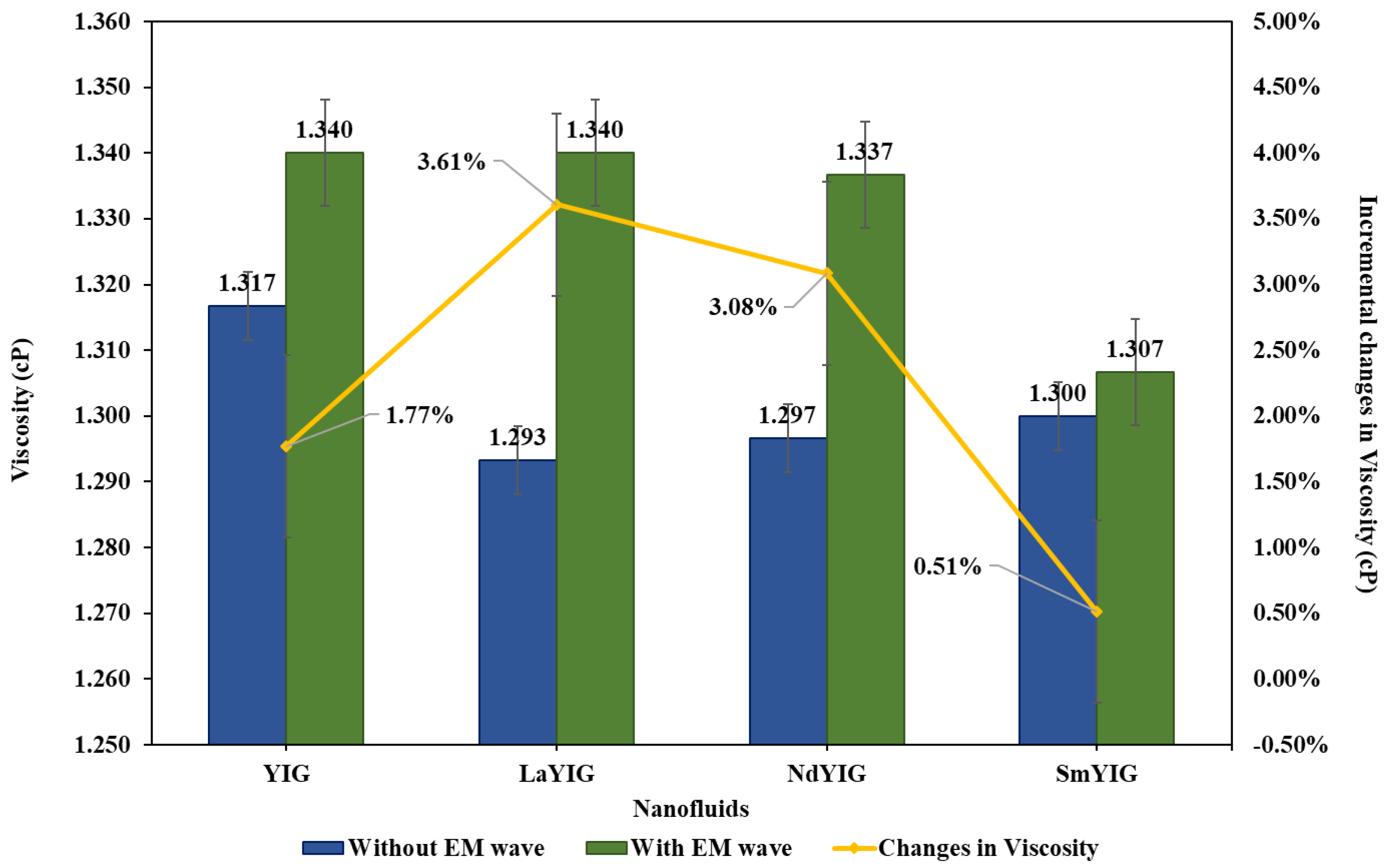
3.2. Evaluation of RE-YIG Nanofluids.
4. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- Bennetzen, M.V.; Mogensen, K. Novel Applications of Nanoparticles for Future Enhanced Oil Recovery. In Proceedings of the International Petroleum Technology Conference, Kuala Lumpur, Malaysia, 10–12 December 2014. [Google Scholar]
- Peng, B.; Tang, J.; Luo, J.; Wang, P.; Ding, B.; Tam, K.C. Applications of Nanotechnology in Oil and Gas Industry: Progress and Perspective. Can. J. Chem. Eng. 2018, 96, 91–100. [Google Scholar] [CrossRef]
- Bhatia, K.H.; Chacko, L.P. Ni-Fe Nanoparticles: A Innovative Approach for Recovery of Hydrates. In Brasil Offshore; Society of Petroleum Engineers: Richardson, TX, USA, 2011. [Google Scholar]
- Pilkington, G.A.; Briscoe, W.H. Nanofluids mediating surface forces. Adv. Colloid Interface Sci. 2012, 179, 68–84. [Google Scholar] [CrossRef]
- Soleimani, H.; Baig, M.K.; Yahya, N.; Khodapanah, L.; Sabet, M.; Demiral, B.M.; Burda, M. Synthesis of ZnO nanoparticles for oil-water interfacial tension reduction in enhanced oil recovery. Appl. Phys. A 2018, 124, 128. [Google Scholar] [CrossRef]
- Ryoo, S.; Rahmani, A.R.; Yoon, K.Y.; Prodanović, M.; Kotsmar, C.; Milner, T.E.; Huh, C.; Johnston, K.P.; Bryant, S.L. Theoretical and experimental investigation of the motion of multiphase fluids containing paramagnetic nanoparticles in porous media. J. Pet. Sci. Eng. 2012, 81, 129–144. [Google Scholar] [CrossRef]
- Yahya, N.; Kashif, M.; Shafie, A.; Soleimani, H.; Zaid, H.M.; Latiff, N.R.A. Improved Oil Recovery by High Magnetic Flux Density Subjected to Iron Oxide Nanofluids. J. Nano Res. 2014, 26, 89–99. [Google Scholar] [CrossRef]
- Zitha, P.; Wessel, F. Fluid Flow Control Using Magnetorheological Fluids; Society of Petroleum Engineers Inc.: Richardson, TX, USA, 2002. [Google Scholar]
- Zaid, H.M.; Latiff, N.R.A.; Yahya, N.; Soleimani, H.; Shafie, A. Application of electromagnetic waves and dielectric nanoparticles in enhanced oil recovery. J. Nano Res. 2014, 26, 135–142. [Google Scholar] [CrossRef]
- Soares, F.S.M.D.A.; Prodanovic, M.; Huh, C. Excitable Nanoparticles for Trapped Oil Mobilization. In SPE Improved Oil Recovery Symposium; Society of Petroleum Engineers: Richardson, TX, USA, 2014. [Google Scholar]
- Soleimani, H.; Yahya, N.; Latiff, N.R.A.; Zaid, H.M.; Demiral, B.; Amighian, J. Novel Enhanced Oil Recovery Method using Co2+xFe2+(1-x)Fe3+2O4 as Magnetic Nanoparticles Activated by Electromagnetic Waves. J. Nano Res. 2014, 26, 111–116. [Google Scholar] [CrossRef]
- Soleimani, H.; Latiff, N.R.A.; Yahya, N.; Sabet, M.; Khodapanah, L.; Kozlowski, G.; Guan, B.H.; Chuan, L.K. Synthesis and Characterization of Yttrium Iron Garnet (YIG) Nanoparticles activated by Electromagnetic wave in Enhanced Oil Recovery. J. Nano Res. 2016, 38, 40–46. [Google Scholar] [CrossRef]
- Akhtar, M.N.; Yousaf, M.; Khan, S.N.; Nazir, M.S.; Ahmad, M.; Khan, M.A. Structural and electromagnetic evaluations of YIG rare earth doped (Gd, Pr, Ho, Yb) nanoferrites for high frequency applications. Ceram. Int. 2017, 43, 17032–17040. [Google Scholar] [CrossRef]
- Wolf, W. Ferromagnetic Alignment by Antiferromagnetic Exchange Interaction. Note on the Magnetic Behaviour of Neodymium Garnet. J. Appl. Phys. 1961, 32, 742. [Google Scholar] [CrossRef]
- Sánchez, R.D.; Rivas, J.; Vaqueiro, P.; Lopez-Quintela, M.A.; Caeiro, D. Particle size effects on magnetic properties of yttrium iron garnets prepared by a sol-gel method. J. Magn. Magn. Mater. 2002, 247, 92–98. [Google Scholar] [CrossRef]
- Vajargah, S.H.; Hosseini, H.M.; Nemati, Z.A. Preparation and characterization of yttrium iron garnet (YIG) nanocrystalline powders by auto-combustion of nitrate-citrate gel. J. Alloys Compd. 2007, 430, 339–343. [Google Scholar] [CrossRef]
- Vajargah, S.H.; Hosseini, H.M.; Nemati, Z.A. Synthesis of nanocrystalline yttrium iron garnets by sol-gel combustion process: The influence of pH of precursor solution. Mater. Sci. Eng. B 2006, 129, 211–215. [Google Scholar] [CrossRef]
- Vaqueiro, P.; Lopez-Quintela, M.A.; Rivas, J.; Greneche, J.M. Annealing dependence of magnetic properties in nanostructured particles of yttrium iron garnet prepared by citrate gel process. J. Magn. Magn. Mater. 1997, 169, 56–68. [Google Scholar] [CrossRef]
- Akhtar, M.N.; Khan, M.A.; Ahmad, M.; Murtaza, G.; Raza, R.; Shaukat, S.F.; Raza, M.R.; Asif, M.H.; Nasir, N.; Abbas, G.; et al. Y3Fe5O12 nanoparticulate garnet ferrites: Comprehensive study on the syntesis and characterization fabricated by various routes. J. Magn. Magn. Mater. 2014, 368, 393–400. [Google Scholar] [CrossRef]
- Janifer, M.A.; Anand, S.; Senthuuran, M.; Pauline, S. Effect of synthesis conditions on yttrium iron garnet (YIG) nanocrystalline powder via sol-gel method. Int. Res. J. Eng. Technol. (IRJET) 2017, 4, 78–81. [Google Scholar]
- Ogolo, N.A.; Olafuyi, O.A.; Onyekonwu, M.O. Enhanced Oil Recovery Using Nanoparticles. In SPE Saudi Arabia Section Technical Symposium and Exhibition; Society of Petroleum Engineers: Richardson, TX, USA, 2012. [Google Scholar]
- Yuan, Y.; Lee, T.R. Chapter 1: Contact angle and wetting properties. In Surface Science Techniques; Springer: Berlin/Heidelberg, Germany, 2013; pp. 3–34. [Google Scholar]
- Herzer, G. Grain size dependence of coercivity and permeability in nanocrystaline ferromagnets. IEEE Trans. Magn. 1990, 26, 1397–1402. [Google Scholar] [CrossRef]
- Kneller, E.F.; Luborsky, F.E. Particles Size Dependence of Coercivity and Remanence of Single Domain Particles. J. Appl. Phys. 1963, 34, 656–658. [Google Scholar] [CrossRef]
- Lu, A.H.; Salabas, E.E.; Schüth, F. Magnetic Nanoparticles: Synthesis, Protection, Functionalization, and Application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar] [CrossRef]
- Uhm, Y.R.; Lim, J.C.; Choi, S.M.; Kim, C.S. Magnetic Properties of R-YIG (R = La, Nd, and Gd) Derived by a Sol-gel Method. J. Magn. 2016, 21, 303–307. [Google Scholar] [CrossRef]
- Mohajeri, M.; Hemmati, M.; Shekarabi, A.S. An experimental study on using a nanosurfactant in an EOR process of heavy oil in fractured micromodel. J. Pet. Sci. Eng. 2015, 126, 162–173. [Google Scholar] [CrossRef]
- Mittal, N.; Deva, D.; Kumar, R.; Sharma, A. Exceptionally robust and conductive superhydrophobic free-standing films of mesoporous carbon nanocapsule/polymer composite for multifunctional applications. Carbon 2015, 93, 492–501. [Google Scholar] [CrossRef]
- Mittal, N.; Kumar, R.; Mishra, G.; Deva, D.; Sharma, A. Mesoporous carbon nanocapsules based coatings with multifunctionalities. Adv. Mater. Interfaces 2016, 3, 1500708. [Google Scholar] [CrossRef]
- Brett, C.J.; Mittal, N.; Ohm, W.; Gensch, M.; Kreuzer, L.P.; Körstgens, V.; Roth, S.V. Water Induced Structural Rearrangements on the Nanoscale in Ultrathin Nanocellulose Films. Macromolecules 2019, 52, 4721–4728. [Google Scholar] [CrossRef]
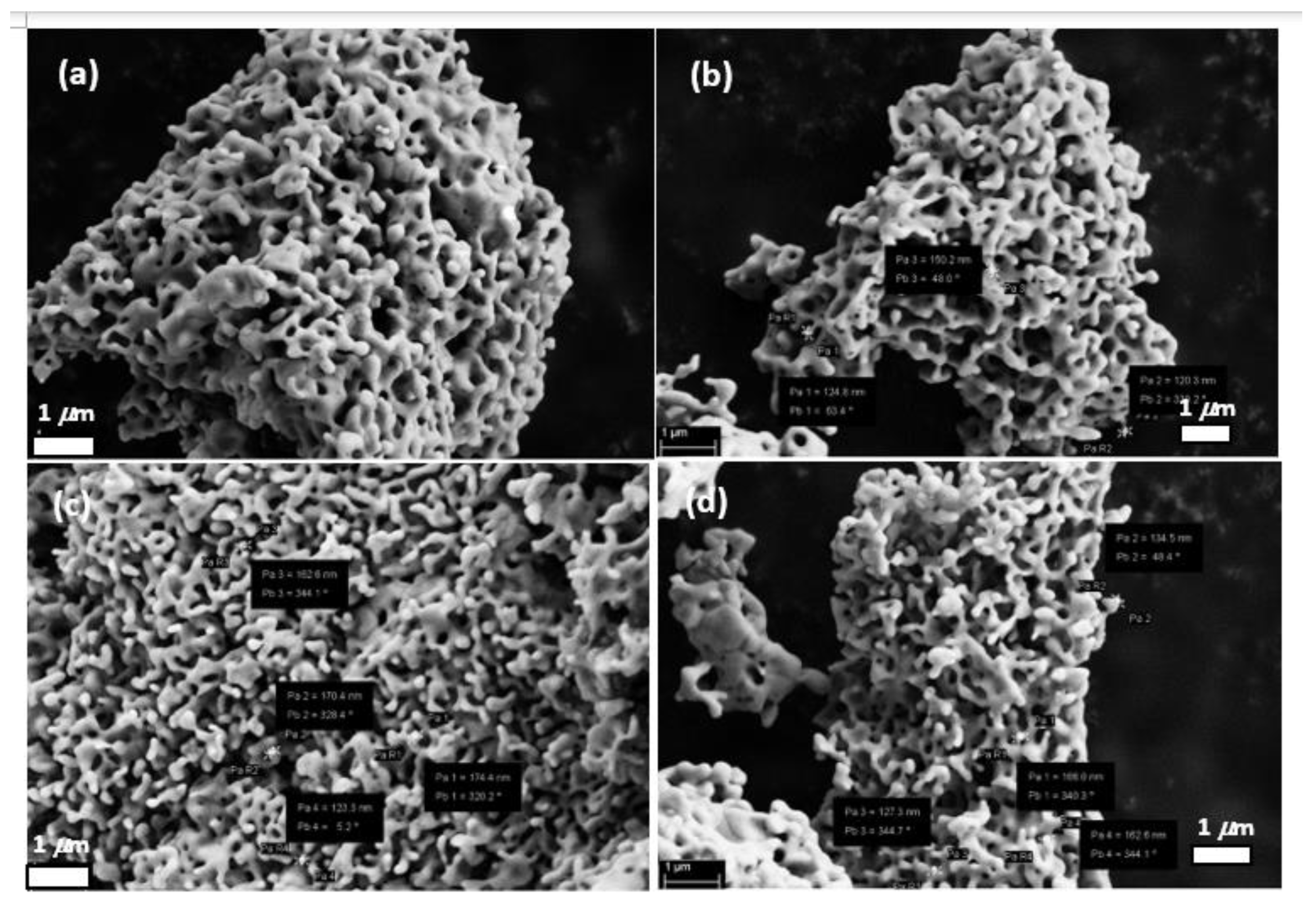
| Element | Samples (Weight %) | |||
|---|---|---|---|---|
| YIG | La-YIG | Nd-YIG | Sm-YIG | |
| Y | 34.37 | 31.44 | 33.19 | 33.38 |
| Fe | 29.89 | 30.95 | 26.05 | 30.18 |
| O | 35.73 | 34.11 | 38.15 | 32.64 |
| RE | 0 | 3.49 | 2.61 | 3.8 |
| Samples | 2θ | FWHM (o) | Crystalline Size, D (nm) | D-Spacing (Å) | |
|---|---|---|---|---|---|
| YIG | 32.324 | 0.163 | 50.058 | 2.767 | 12.374 |
| La-YIG | 32.272 | 0.182 | 44.879 | 2.771 | 12.393 |
| Nd-YIG | 32.298 | 0.158 | 51.870 | 2.769 | 12.383 |
| Sm-YIG | 32.324 | 0.154 | 53.062 | 2.768 | 12.377 |
| Samples | Magnetic Parameters | |||
|---|---|---|---|---|
| Ms (emu/g) | Mr (emu/g) | Hc (Oe) | Hs (Oe) | |
| YIG | 21.30 | 4.45 | 37.09 | 1395.97 |
| La-YIG | 21.10 | 4.41 | 39.68 | 1429.42 |
| Nd-YIG | 21.50 | 5.25 | 46.05 | 1464.67 |
| Sm-YIG | 23.54 | 6.02 | 47.03 | 1503.88 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lau, Z.Y.; Lee, K.C.; Soleimani, H.; Beh, H.G. Experimental Study of Electromagnetic-Assisted Rare-Earth Doped Yttrium Iron Garnet (YIG) Nanofluids on Wettability and Interfacial Tension Alteration. Energies 2019, 12, 3806. https://doi.org/10.3390/en12203806
Lau ZY, Lee KC, Soleimani H, Beh HG. Experimental Study of Electromagnetic-Assisted Rare-Earth Doped Yttrium Iron Garnet (YIG) Nanofluids on Wettability and Interfacial Tension Alteration. Energies. 2019; 12(20):3806. https://doi.org/10.3390/en12203806
Chicago/Turabian StyleLau, Zhen Yin, Kean Chuan Lee, Hassan Soleimani, and Hoe Guan Beh. 2019. "Experimental Study of Electromagnetic-Assisted Rare-Earth Doped Yttrium Iron Garnet (YIG) Nanofluids on Wettability and Interfacial Tension Alteration" Energies 12, no. 20: 3806. https://doi.org/10.3390/en12203806
APA StyleLau, Z. Y., Lee, K. C., Soleimani, H., & Beh, H. G. (2019). Experimental Study of Electromagnetic-Assisted Rare-Earth Doped Yttrium Iron Garnet (YIG) Nanofluids on Wettability and Interfacial Tension Alteration. Energies, 12(20), 3806. https://doi.org/10.3390/en12203806





