Investigation of the Relationship between Bacteria Growth and Lipid Production Cultivating of Microalgae Chlorella Vulgaris in Seafood Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microalgae Strain and Wastewater Sample
2.2. Experimental Set-up
2.3. Analysis
2.3.1. Microalgae Growth
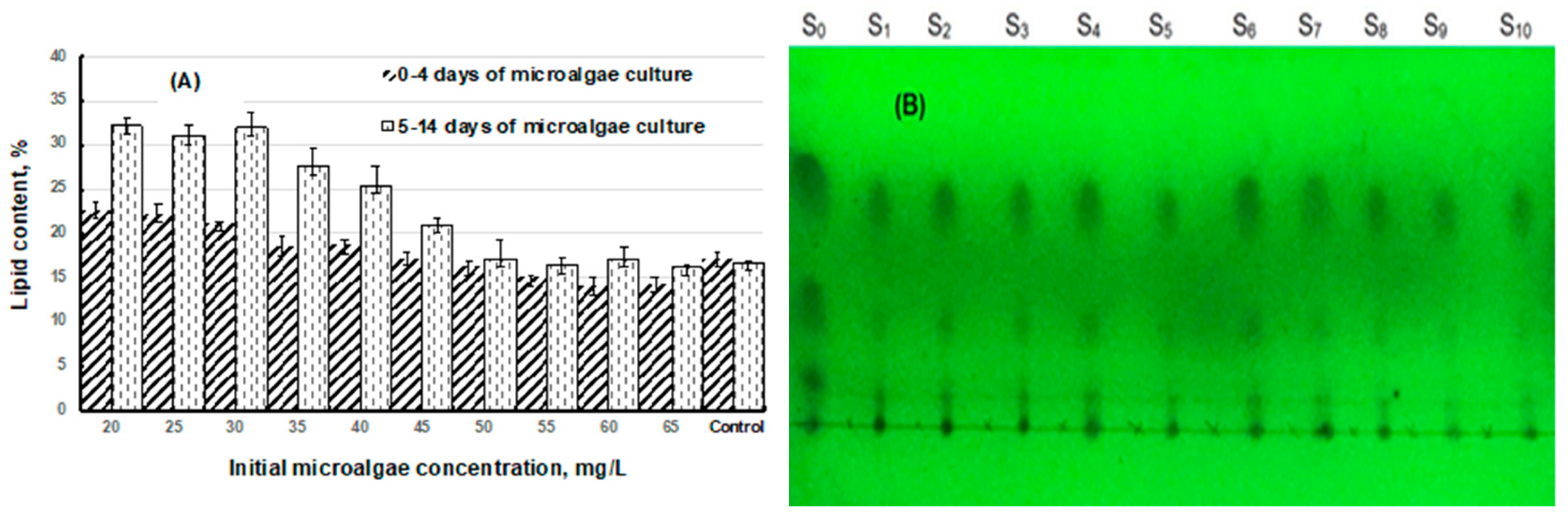
2.3.2. Total Lipid Content
2.3.3. Lipid Analysis
2.3.4. Bacteria Count
2.3.5. Water Quality
3. Results
3.1. Effect of Growth Rate of Initial Microalgae Cell
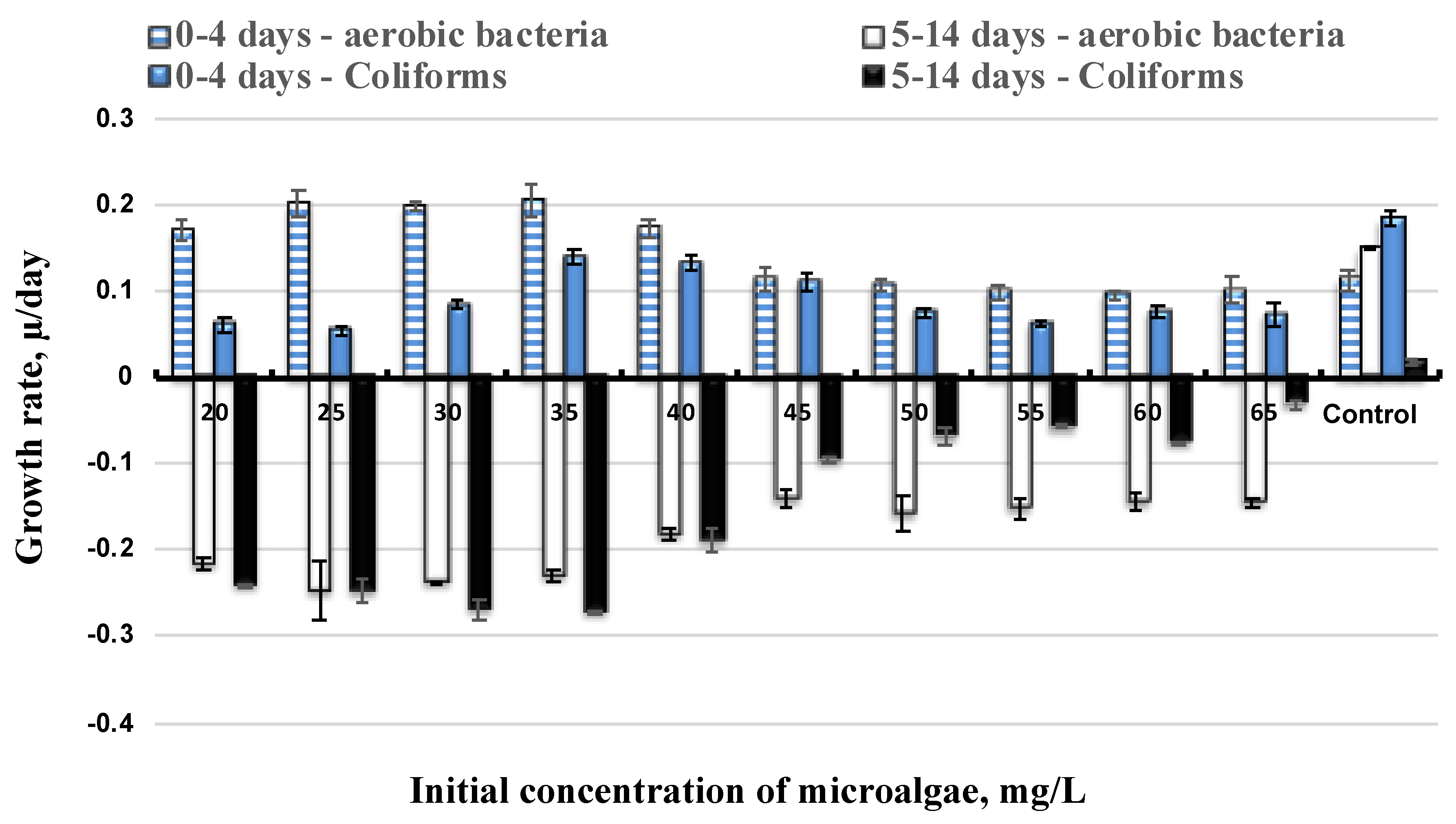
3.2. Effect of Initial Microalgae Cell and Bacteria Growth
3.3. Bacteria Inhibition to Lipid Production
3.4. Effects of Total-N and Total-P to Lipid Production
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef] [Green Version]
- Cheirsilp, B.; Torpee, S. Enhanced growth and lipid production of microalgae under mixotrophic culture condition: Effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour. Technol. 2012, 110, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.S.; Choi, H.I.; Choi, J.W.; Lee, J.S.; Sung, Y.J.; Sim, S.J. Multilateral approach on enhancing economic viability of lipid production from microalgae: A review. Bioresour. Technol. 2018, 258, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Atta, M.; Idris, A.; Bukhari, A.; Wahidin, S. Intensity of blue LED light: A potential stimulus for biomass and lipid content in fresh water microalgae Chlorella vulgaris. Bioresour. Technol. 2013, 148, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Hultberg, M.; Jönsson, H.L.; Bergstrand, K.J.; Carlsson, A.S. Impact of light quality on biomass production and fatty acid content in the microalga Chlorella vulgaris. Bioresour. Technol. 2014, 159, 465–467. [Google Scholar] [CrossRef] [PubMed]
- Han, F.; Huang, J.; Li, Y.; Wang, W.; Wan, M.; Shen, G.; Wang, J. Enhanced lipid productivity of Chlorella pyrenoidosa through the culture strategy of semi-continuous cultivation with nitrogen limitation and pH control by CO2. Bioresour. Technol. 2013, 136, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Mujtaba, G.; Choi, W.; Lee, C.G.; Lee, K. Lipid production by Chlorella vulgaris after a shift from nutrient-rich to nitrogen starvation conditions. Bioresour. Technol. 2012, 123, 279–283. [Google Scholar] [CrossRef]
- Xin, L.; Hong-ying, H.; Yu-ping, Z. Growth and lipid accumulation properties of a freshwater microalga Scenedesmus sp. under different cultivation temperature. Bioresour. Technol. 2011, 102, 3098–3102. [Google Scholar] [CrossRef]
- Yeh, K.L.; Chang, J.S. Effects of cultivation conditions and media composition on cell growth and lipid productivity of indigenous microalga Chlorella vulgaris ESP-31. Bioresour. Technol. 2012, 105, 120–127. [Google Scholar] [CrossRef]
- Widjaja, A.; Chien, C.C.; Ju, Y.H. Study of increasing lipid production from fresh water microalgae Chlorella vulgaris. J. Taiwan Inst. Chem. Eng. 2009, 40, 13–20. [Google Scholar] [CrossRef]
- Feng, D.; Chen, Z.; Xue, S.; Zhang, W. Increased lipid production of the marine oleaginous microalgae Isochrysis zhangjiangensis (Chrysophyta) by nitrogen supplement. Bioresour. Technol. 2011, 102, 6710–6716. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Li, C.; Zhang, D. Lipid production of Chlorella vulgaris cultured in artificial wastewater medium. Bioresour. Technol. 2011, 102, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Illman, A.M.; Scragg, A.H.; Shales, S.W. Increase in Chlorella strains calorific values when grown in low nitrogen medium. Enzyme Microb. Technol. 2000, 27, 631–635. [Google Scholar] [CrossRef]
- Reitan, K.I.; Rainuzzo, J.R.; Olsen, Y. Effect of nutrient limitation on fatty acid and lipid content of marine microalgae. J. Phycol. 1994, 30, 972–979. [Google Scholar] [CrossRef]
- Takagi, M.; Yoshida, T. Effect of salt concentration on intracellular accumulation of lipids and triacylglyceride in marine microalgae Dunaliella cells. J. Biosci. Bioeng. 2006, 101, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Prathima Devi, M.; Venkata Subhash, G.; Venkata Mohan, S. Heterotrophic cultivation of mixed microalgae for lipid accumulation and wastewater treatment during sequential growth and starvation phases: Effect of nutrient supplementation. Renew. Energy 2012, 43, 276–283. [Google Scholar] [CrossRef]
- Shi, X.M.; Chen, F.; Yuan, J.P.; Chen, H. Heterotrophic production of lutein by selected Chlorella strains. J. Appl. Phycol. 1997, 9, 445–450. [Google Scholar] [CrossRef]
- Nguyen, T.D.P.; Frappart, M.; Jaouen, P.; Pruvost, J.; Bourseau, P. Harvesting Chlorella vulgaris by natural increase in pH: Effect of medium composition. Environ. Technol. 2014, 35, 1378–1388. [Google Scholar] [CrossRef]
- Nguyen, T.D.P.; Le, T.V.A.; Show, P.L.; Nguyen, T.T.; Tran, M.H.; Tran, T.N.T.; Lee, S.Y. Bioflocculation formation of microalgae-bacteria in enhancing microalgae harvesting and nutrient removal from wastewater effluent. Bioresour. Technol. 2018, 272, 34–39. [Google Scholar] [CrossRef]
- Nguyen, T.D.P.; Tran, T.N.T.; Le, T.V.A.; Phan, T.X.N.; Show, P.L.; Chia, S.R. Auto-flocculation through cultivation of Chlorella vulgaris in seafood wastewater discharge: Influence of culture conditions on microalgae growth and nutrient removal. J. Biosci. Bioeng. 2019, 127, 492–498. [Google Scholar] [CrossRef]
- Krzemińska, I.; Pawlik-Skowrońska, B.; Trzcińska, M.; Tys, J. Influence of photoperiods on the growth rate and biomass productivity of green microalgae. Bioprocess Biosyst. Eng. 2014, 37, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Pruvost, J.; Van Vooren, G.; Le Gouic, B.; Couzinet-Mossion, A.; Legrand, J. Systematic investigation of biomass and lipid productivity by microalgae in photobioreactors for biodiesel application. Bioresour. Technol. 2011, 102, 150–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hazan, R.; Que, Y.A.; Maura, D.; Rahme, L.G. A method for high throughput determination of viable bacteria cell counts in 96-well plates. BMC Microbiol. 2012, 12, 1. [Google Scholar] [CrossRef] [PubMed]
- APHA/AWWA/WEF. Standard Methods for the Examination of Water and Wastewater. Available online: https://www.google.com.tw/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&cad=rja&uact=8&ved=2ahUKEwjH3bDW05ziAhVjF6YKHQuGDMMQFjAAegQIAhAC&url=https%3A%2F%2Fwww.researchgate.net%2Fprofile%2FArif_Reza%2Fpost%2FCan_you_give_me_the_APHA_method_for_trace_metal_analysis%2Fattachment%2F59d63565c49f478072ea35b5%2FAS%253A273663767580673%25401442257934916%2Fdownload%2Fstandard%2Bmethods%2Bfor%2Bthe%2Bexamination%2Bof%2Bwater%2Band%2Bwastewater%2B%25281000-3000%2529.pdf&usg=AOvVaw1054PsR-j-GFOAdq-aW9sx (accessed on 20 April 2019).
- Van Vooren, G.; Le Grand, F.; Legrand, J.; Cuiné, S.; Peltier, G.; Pruvost, J. Investigation of fatty acids accumulation in Nannochloropsis oculata for biodiesel application. Bioresour. Technol. 2012, 124, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; He, B.; Sun, Z.; Chen, Y.F. Chemically enhanced lipid production from microalgae under low sub-optimal temperature. Algal Res. 2016, 16, 20–27. [Google Scholar] [CrossRef]
- Zhang, Y.; Su, H.; Zhong, Y.; Zhang, C.; Shen, Z.; Sang, W.; Yan, G.; Zhou, X. The effect of bacterial contamination on the heterotrophic cultivation of Chlorella pyrenoidosa in wastewater from the production of soybean products. Water Res. 2012, 46, 5509–5516. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhou, W.; Fu, Z.; Cheng, Y.; Min, M.; Liu, Y.; Zhang, Y.; Chen, P.; Ruan, R. Effect of wastewater-borne bacteria on algal growth and nutrients removal in wastewater-based algae cultivation system. Bioresour. Technol. 2014, 167, 8–13. [Google Scholar] [CrossRef]
- Quijano, G.; Arcila, J.S.; Buitrón, G. Microalgal-bacterial aggregates: Applications and perspectives for wastewater treatment. Biotechnol. Adv. 2017, 35, 772–781. [Google Scholar] [CrossRef]
- Fukami, K.; Nishijima, T.; Ishida, Y. Stimulative and inhibitory effects of bacteria on the growth of microalgae. Hydrobiologia 1997, 358, 185–191. [Google Scholar] [CrossRef]
- Han, J.; Zhang, L.; Wang, S.; Yang, G.; Zhao, L.; Pan, K. Co-culturing bacteria and microalgae in organic carbon containing medium. J. Biol. Res.-Thessalon. 2016, 23, 8. [Google Scholar] [CrossRef] [Green Version]
- Abinandan, S.; Subashchandrabose, S.R.; Venkateswarlu, K.; Megharaj, M. Nutrient removal and biomass production: Advances in microalgal biotechnology for wastewater treatment. Crit. Rev. Biotechnol. 2018, 38, 1244–1260. [Google Scholar] [CrossRef] [PubMed]
- Alcántara, C.; Domínguez, J.M.; García, D.; Blanco, S.; Pérez, R.; García-Encina, P.A.; Muñoz, R. Evaluation of wastewater treatment in a novel anoxic-aerobic algal-bacterial photobioreactor with biomass recycling through carbon and nitrogen mass balances. Bioresour. Technol. 2015, 191, 173–186. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Park, S.Y.; Li, Y. Nutrient recovery from wastewater streams by microalgae: Status and prospects. Renew. Sustain. Energy Rev. 2013, 19, 360–369. [Google Scholar] [CrossRef]
- Li, X.; Li, W.; Zhai, J.; Wei, H.; Wang, Q. Effect of ammonium nitrogen on microalgal growth, biochemical composition and photosynthetic performance in mixotrophic cultivation. Bioresour. Technol. 2019, 273, 368–376. [Google Scholar] [CrossRef] [PubMed]

| Initial Microalgae Density (mg·L−1) | 20 | 25 | 30 | 35 | 40 | 45 | 50 | 55 | 60 | 65 | Micro-Algae in Sueoka (Control) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Maximum biomass concentration (mg·L−1) | 820 ± 12 | 750 ± 5.7 | 560 ± 10.0 | 500 ± 8.5 | 500 ± 17 | 440 ± 12.1 | 270 ± 15.2 | 270 ± 10.2 | 250 ± 10.45 | 250 ± 20.1 | 800 ± 25.5 |
| μ (day5−1)a | 0.6 ± 0.016 | 0.65 ± 0.01 | 0.61 ± 0.012 | 0.54 ± 0.015 | 0.52 ± 0.02 | 0.41 ± 0.011 | 0.32 ± 0.014 | 0.21 ± 0.011 | 0.188 ± 0.016 | 0.18 ± 0.013 | 0.57 ± 0.02 |
| Chlorophyll a (mg·L−1) | 21.14 ± 1.68 | 20.45 ± 2.5 | 17.35 ± 1.25 | 14.5 ± 2.2 | 14.75 ± 1.6 | 13.75 ± 1.5 | 9.25 ± 1.2 | 9.72 ± 2.1 | 8.87 ± 0.75 | 6.8 ± 0.5 | 19.92 ± 2.6 |
| % TSS removal (%) | 88 ± 0.7 | 90 ± 3.7 | 88 ± 1.2 | 85.7 ± 1.5 | 91 ± 2.25 | 91.1 ± 2.5 | 88.9 ± 2.1 | 92.2 ± 3.5 | 93.8 ± 1.5 | 90.5 ± 1.5 | - |
| Lipid content (5–14 days) (%) | 32.15 ± 1.45 | 31 ± 1.28 | 32 ± 1.55 | 27.56 ± 2.15 | 25.5 ± 2.02 | 21.02 ± 0.85 | 17.25 ± 2.25 | 16.41 ± 1.5 | 17.2 ± 1.5 | 17.5 ± 2.5 | 16.8 ± 0.5 |
| Ci (mg·L−1) | 20 | 25 | 30 | 35 | 40 | 45 | 50 | 55 | 60 | 65 | Control |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Total-N removal (%) | 82.5 ± 2.5 | 81.8 ± 5.6 | 80.7 ± 0.75 | 70.17 ± 1.2 | 70.6 ± 3.0 | 66.66 ± 0.5 | 70.1 ± 1.23 | 57.84 ± 2.25 | 41.7 ± 2.05 | 41.8 ± 0.5 | 38.3 ± 0.35 |
| Total-P removal (%) | 77.71 ± 0.5 | 79.02 ± 0.45 | 78.4 ± 1.05 | 75.33 ± 0.5 | 71.47 ± 0.72 | 66.07 ± 0.5 | 62.22 ± 0.32 | 63.76 ± 0.75 | 59.9 ± 0.5 | 57.59 ± 0.24 | 48.34 ± 0.67 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.D.P.; Nguyen, D.H.; Lim, J.W.; Chang, C.-K.; Leong, H.Y.; Tran, T.N.T.; Vu, T.B.H.; Nguyen, T.T.C.; Show, P.L. Investigation of the Relationship between Bacteria Growth and Lipid Production Cultivating of Microalgae Chlorella Vulgaris in Seafood Wastewater. Energies 2019, 12, 2282. https://doi.org/10.3390/en12122282
Nguyen TDP, Nguyen DH, Lim JW, Chang C-K, Leong HY, Tran TNT, Vu TBH, Nguyen TTC, Show PL. Investigation of the Relationship between Bacteria Growth and Lipid Production Cultivating of Microalgae Chlorella Vulgaris in Seafood Wastewater. Energies. 2019; 12(12):2282. https://doi.org/10.3390/en12122282
Chicago/Turabian StyleNguyen, Thi Dong Phuong, Duc Huy Nguyen, Jun Wei Lim, Chih-Kai Chang, Hui Yi Leong, Thi Ngoc Thu Tran, Thi Bich Hau Vu, Thi Trung Chinh Nguyen, and Pau Loke Show. 2019. "Investigation of the Relationship between Bacteria Growth and Lipid Production Cultivating of Microalgae Chlorella Vulgaris in Seafood Wastewater" Energies 12, no. 12: 2282. https://doi.org/10.3390/en12122282







