Combustion and Emission Reduction Characteristics of GTL-Biodiesel Fuel in a Single-Cylinder Diesel Engine
Abstract
1. Introduction
2. Experimental Section and Procedure
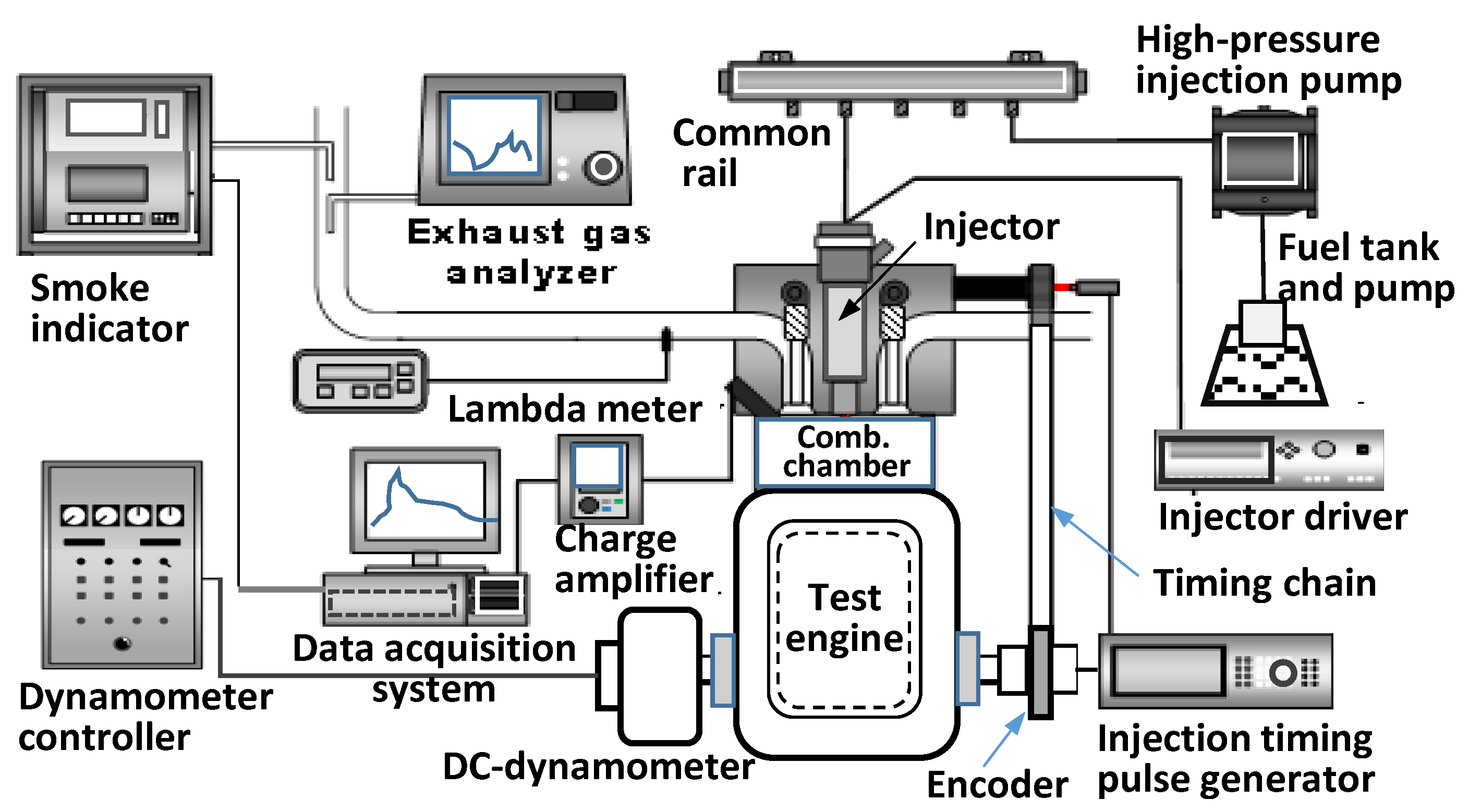
2.1. Test Engine with Various Measurement System
2.2. Test Fuels and Experimental Procedure
3. Results and Discussions
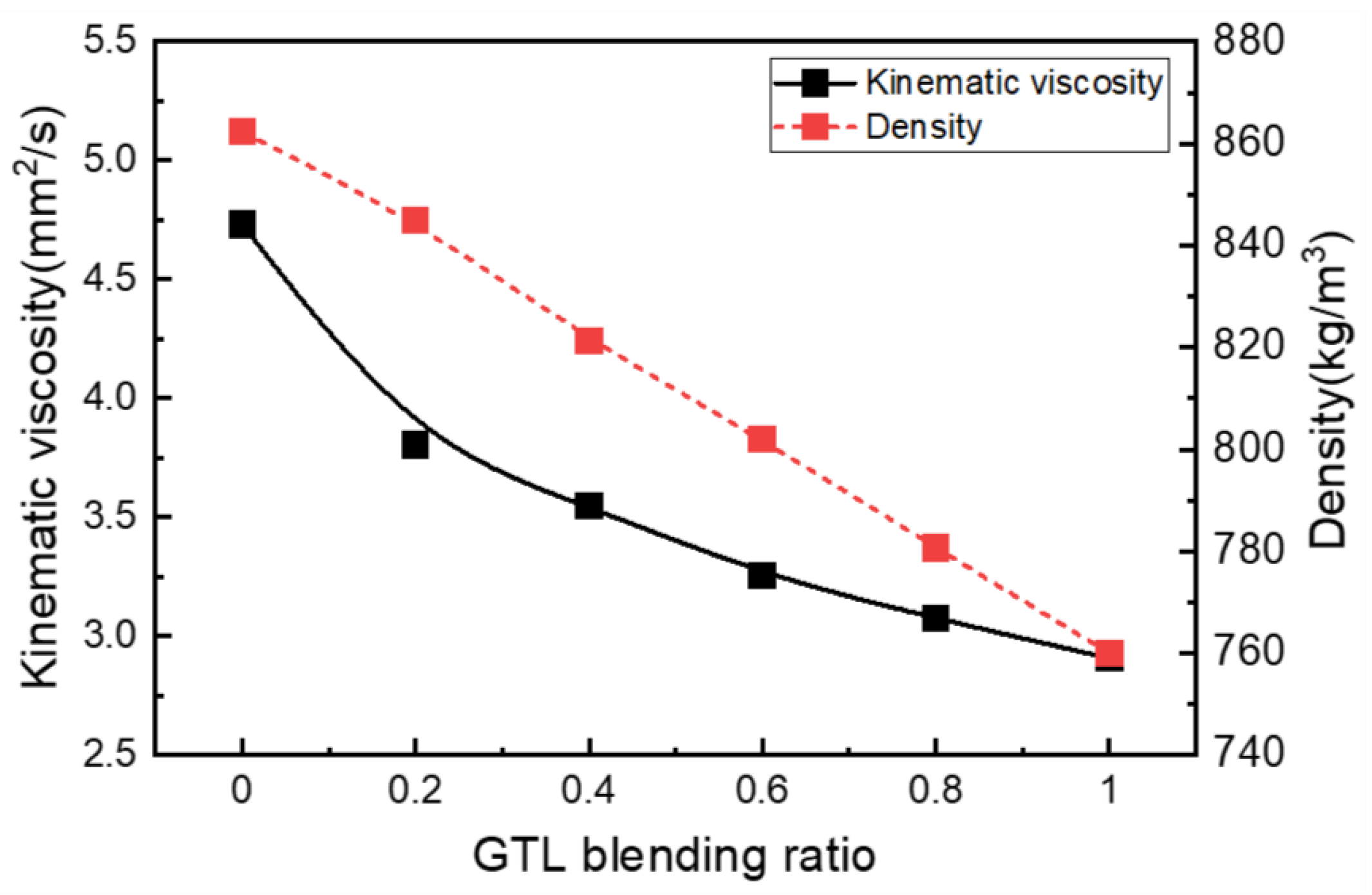
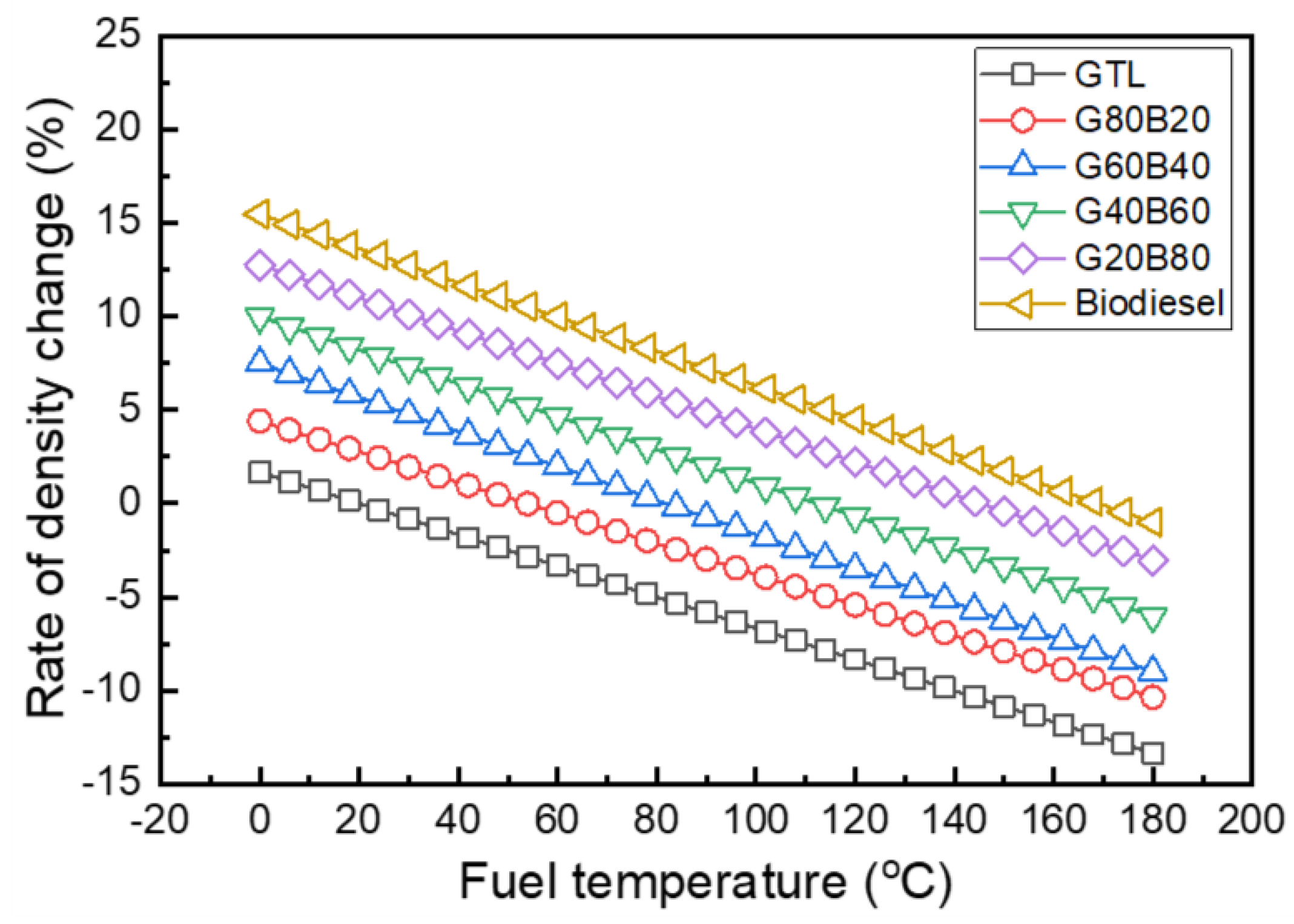
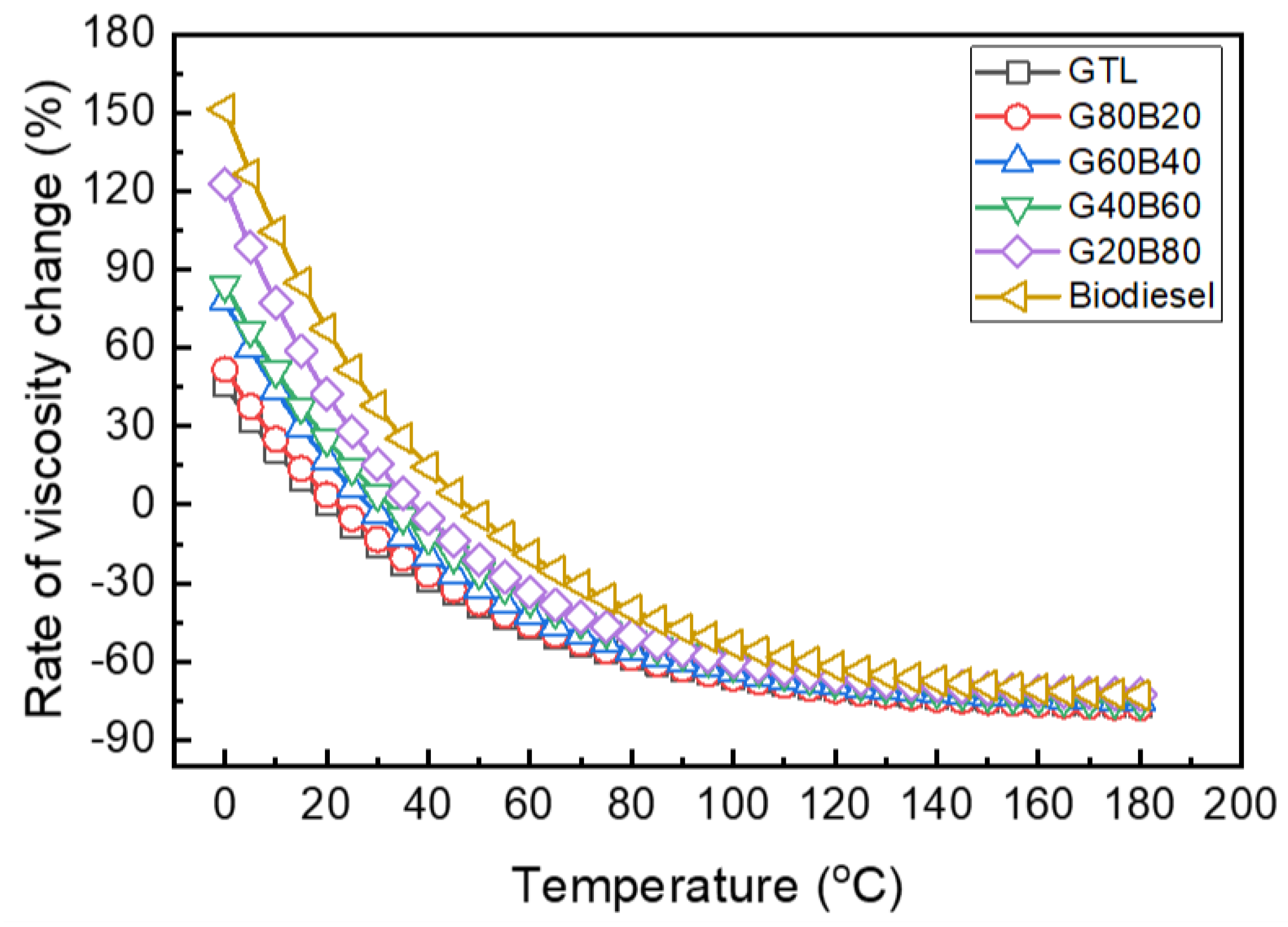
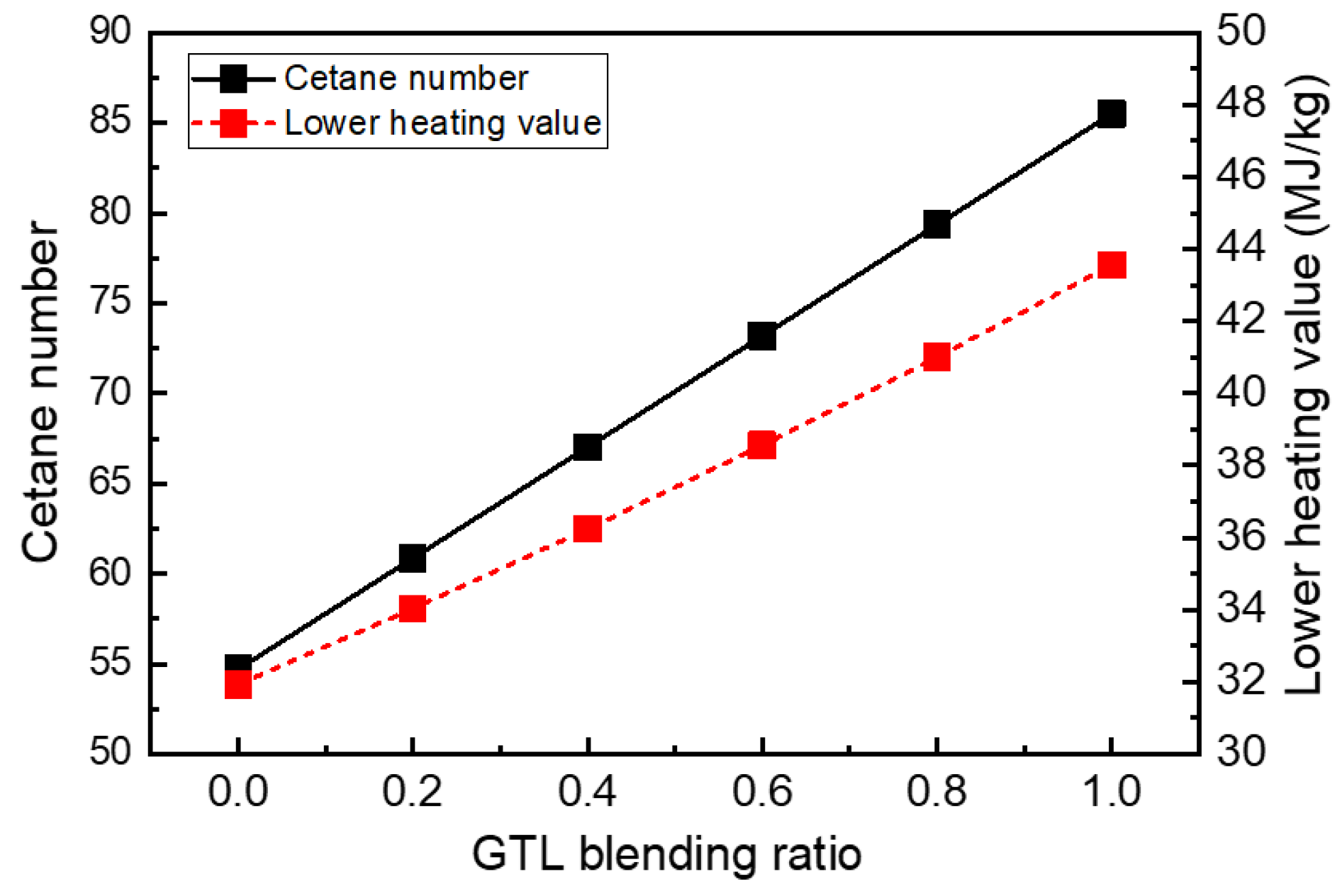
3.1. Fuel Properties of GTL-Biodiesel Blends
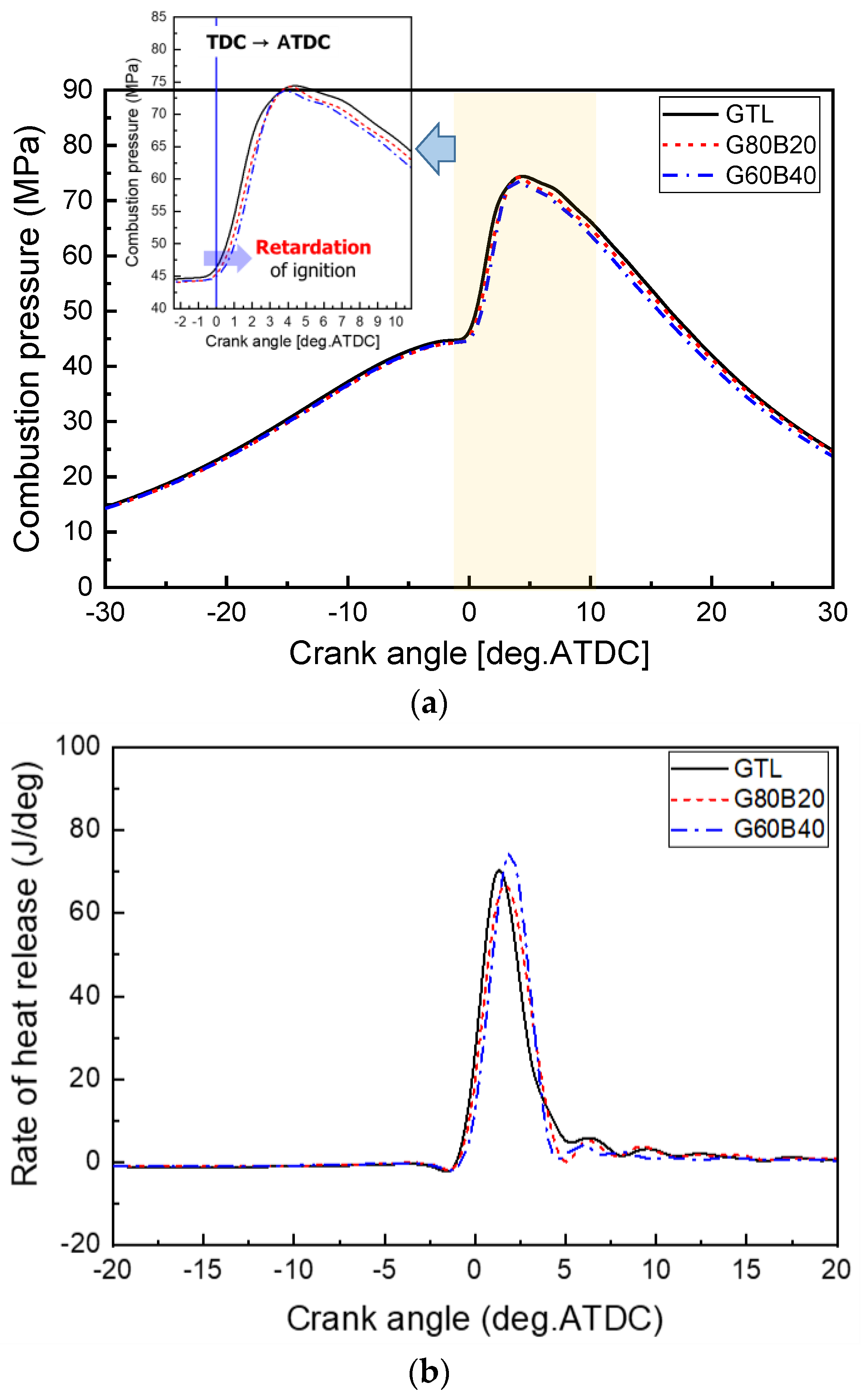
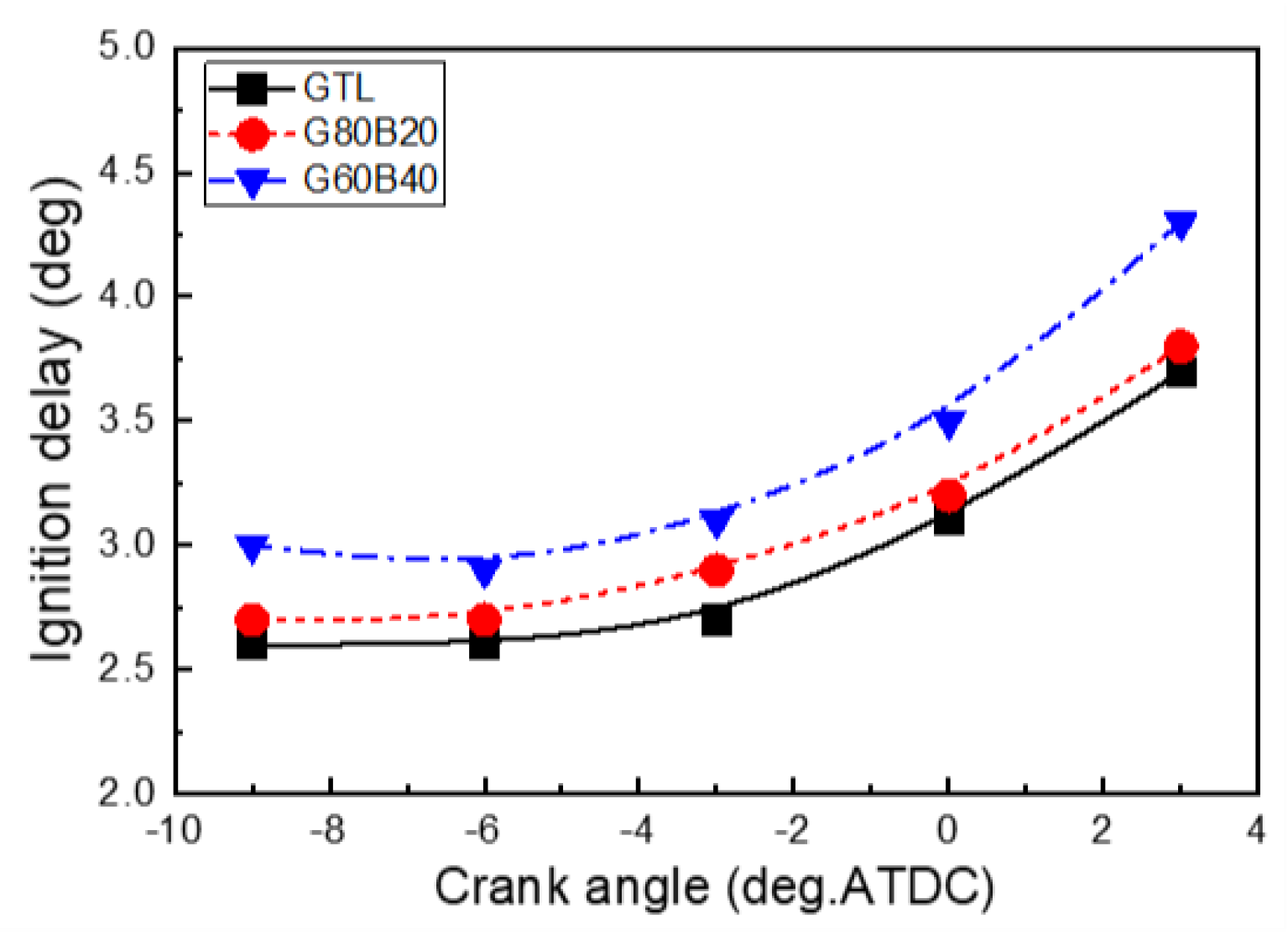
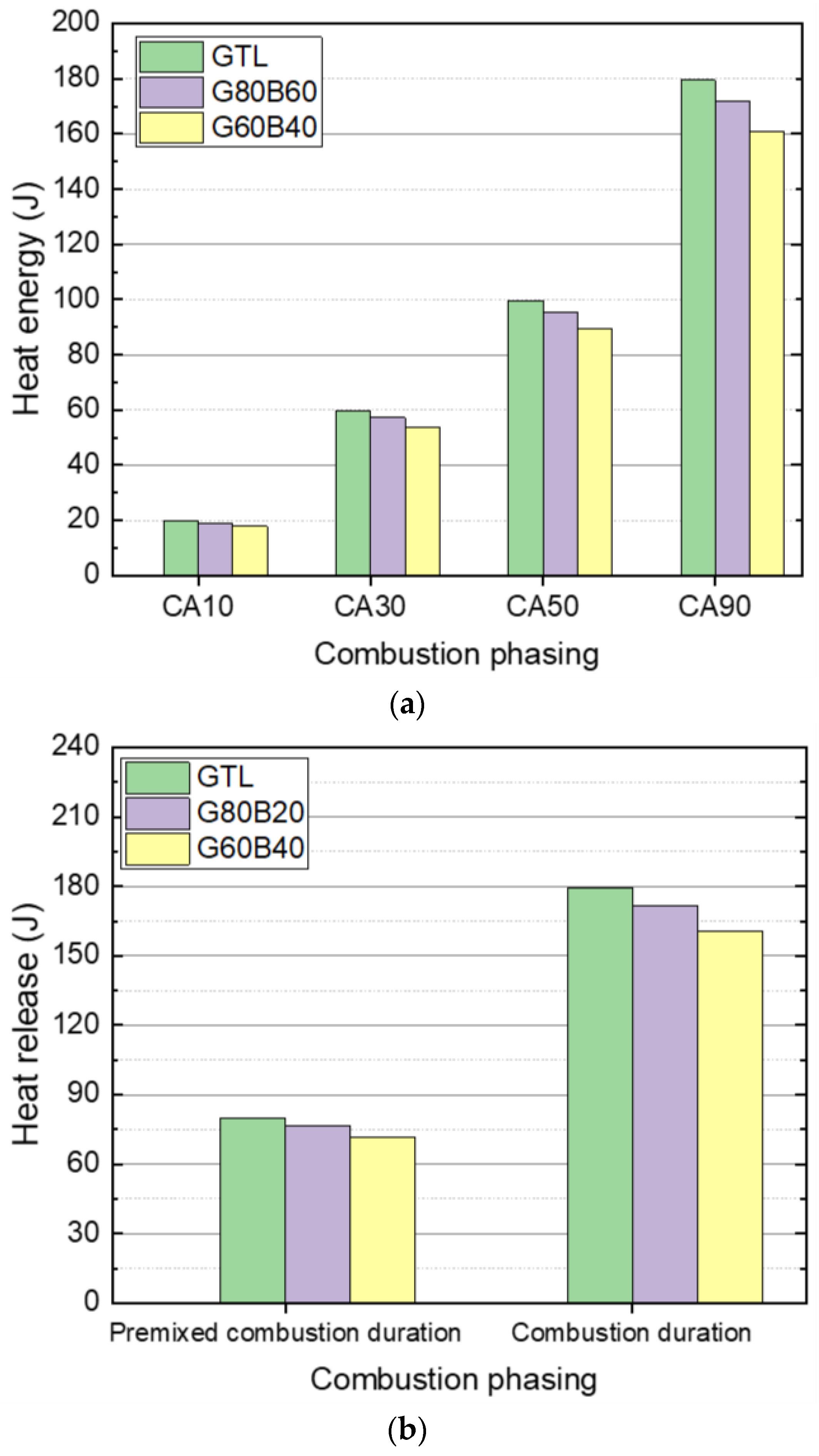
3.2. Combustion and Emission Characteristics
4. Conclusions
- The increasing rate of the density and kinematic viscosity of GTL-biodiesel blends at various temperature conditions was increased with increasing biodiesel volumetric fractions. Based on the investigation results of the GTL-biodiesel blending ratio, the increase in the GTL fraction in the GTL-biodiesel blends led to increases in the CN and LHV of blends.
- The use of biodiesel-blended GTL fuels resulted in the extension of the ignition delay due to the increased biodiesel fraction in the GTL-biodiesel fuel. The increase in the GTL blending ratio led to a decrease in the delay period because of the higher vaporization property.
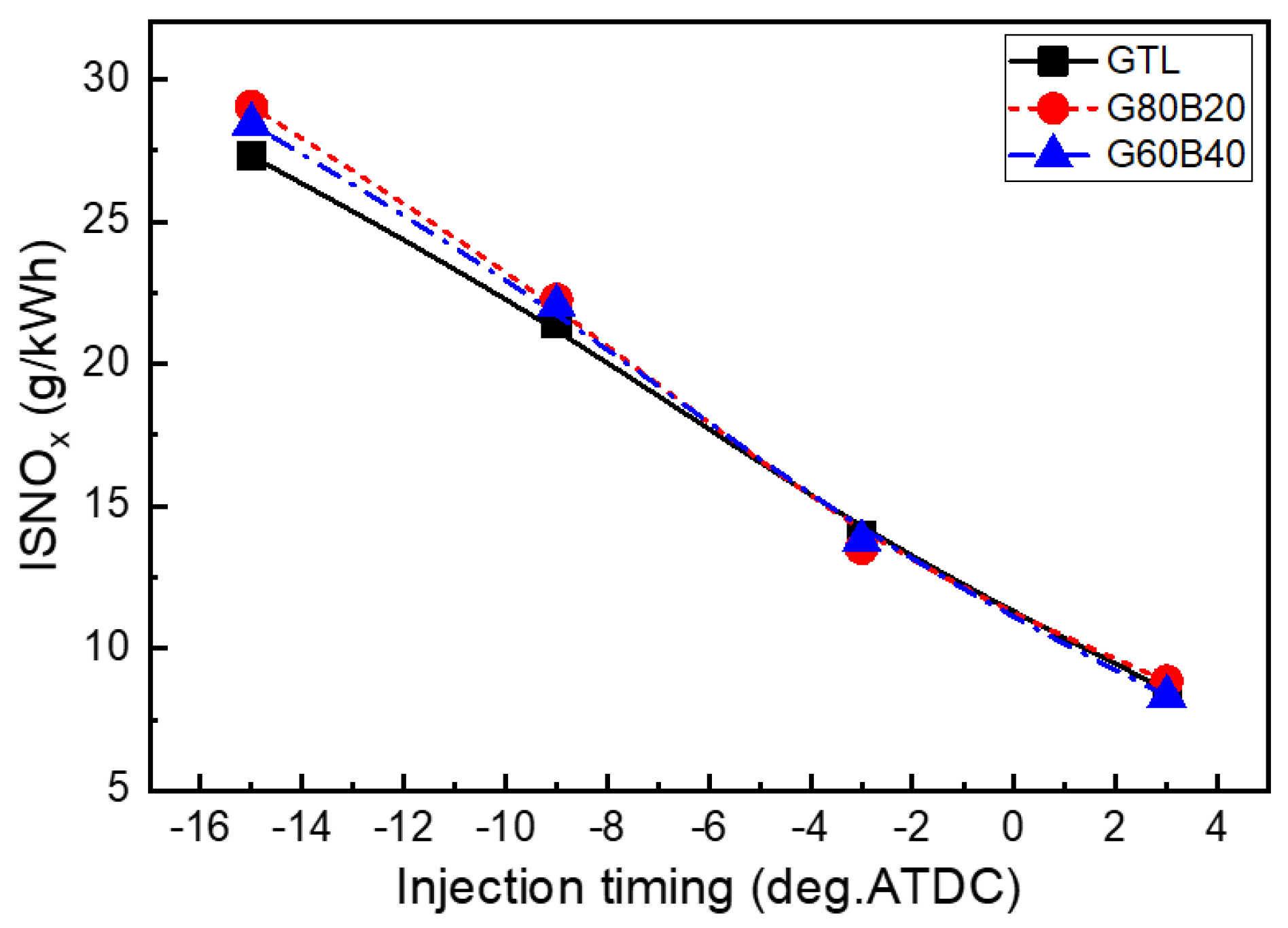
- The GTL fuel exhibits lower NOx emissions than conventional diesel and biodiesel blended GTL fuel throughout the entire range of fuel injection timings. The NOx emissions increase with increasing biodiesel fractions in the blends.
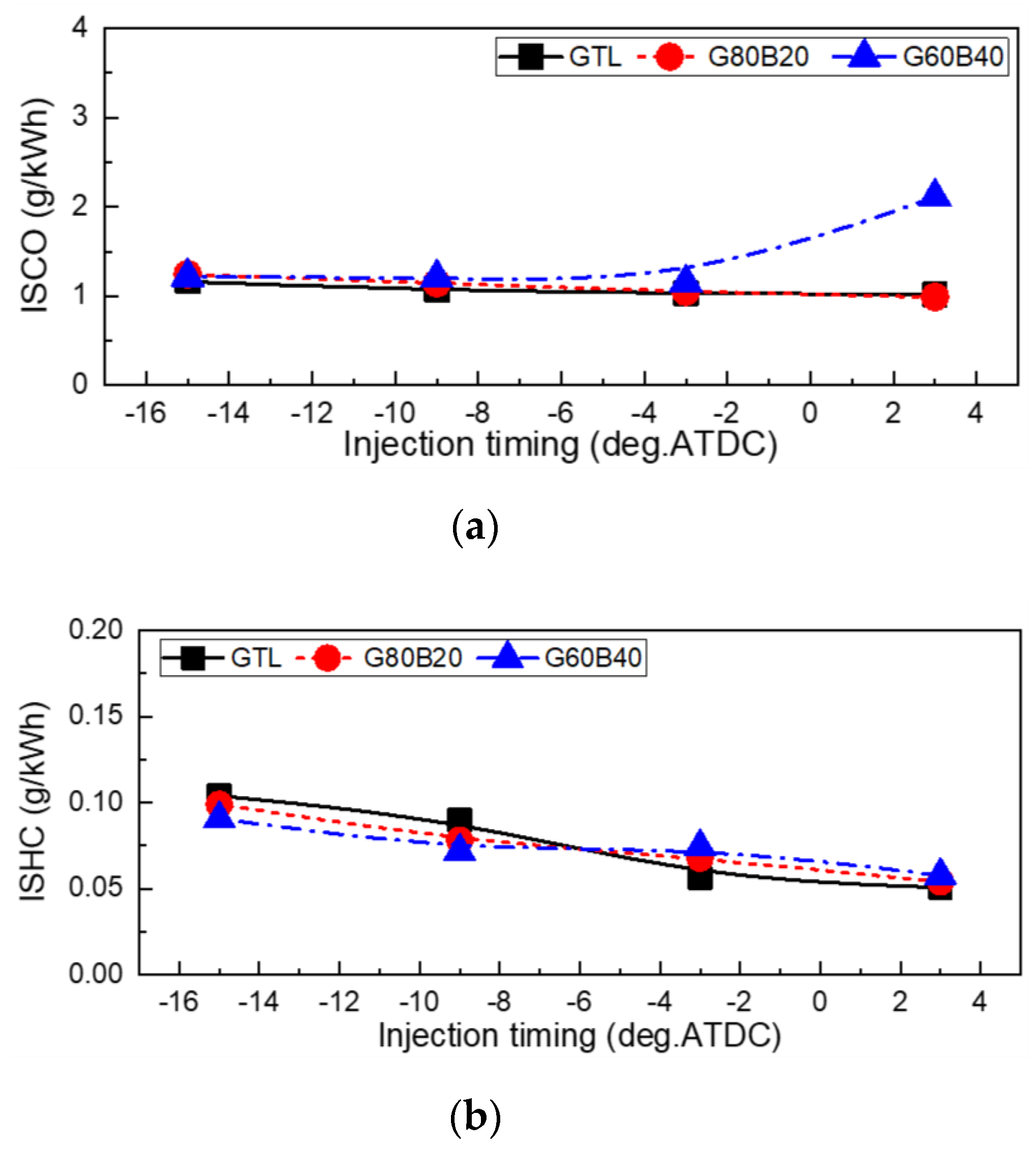
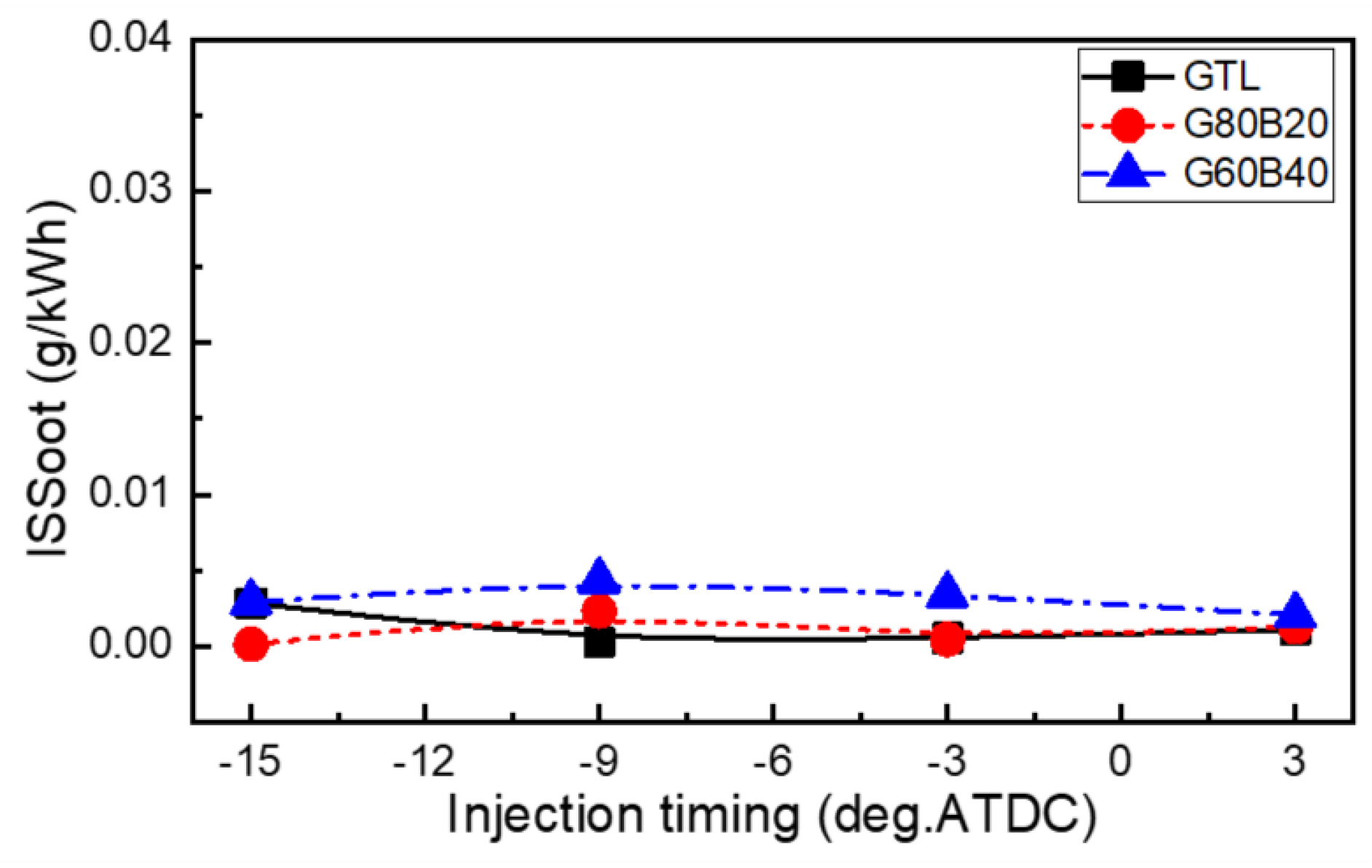
- The effects of GTL-biodiesel blends on the soot emission are small, but the GTL fuel showed a lower value than the other fuels. In the range between 15°BTDC and 3°BTDC of the injection timing, the CO emissions showed almost similarly low values for GTL-biodiesel blends, while the retarded range showed slightly lower and slightly increased HC emissions than those of the GTL fuel at advanced and retarded injection timings, respectively.
- Unlike other studies that reveal combustion characteristics, the results of this study are thought to be of great benefit to alternative fuel research, such as GTL, biodiesel, and GTL-biodiesel blended fuels, for compression ignition diesel engines, by comprehensively studying the properties, as well as the combustion and exhaust emission characteristics of GTL-biodiesel blended fuels.
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| ATDC | after top dead center |
| BTDC | before top dead center |
| CAxx | crank angle at xx% of the cumulative heat release |
| CN | cetane number (-) |
| GTL | gas-to-liquid |
| GxxByy | xx% GTL + yy% biodiesel blended fuel |
| ISNOx | indicated specific nitrogen oxides (g/kWh) |
| ISCO | indicated specific carbon monoxide (g/kWh) |
| ISHC | indicated specific hydrocarbon (g/kWh) |
| ISSoot | indicated specific soot (g/kWh) |
| LHV | lower heating value (MJ/kg) |
| Pinj | injection pressure (Mpa) |
| ROHR | rate of heat release (J/deg) |
| T | temperature (oC) |
| T90 | 90% distillation point |
| TDC | top dead center |
| θinj | energizing duration (degree) |
| ρblend | the density of the blend (kg/m3) |
| rDensity | the rate of the density change (%) |
| ρGTL,20 °C | the GTL density at 20oC and 1atm (kg/m3) |
| νblend | the blend viscosity (mm2/s) |
| rviscosity | the rate of the kinematic viscosity change (%) |
| τid | ignition delay |
References
- Swain, P.K.; Das, L.M.; Naik, S.N. Biomass to liquid: A prospective challenge to research and development in 21st century. Renew. Sustain. Energy Rev. 2011, 15, 4917–4933. [Google Scholar] [CrossRef]
- Bezergianni, S.; Dimitriadis, A. Comparison between different types of renewable diesel. Renew. Sustain. Energy Rev. 2013, 21, 110–116. [Google Scholar] [CrossRef]
- Armas, O.; Yehliu, K.; Boehman, A.L. Effect of alternative fuels on exhaust emissions during diesel engine operation with matched combustion phasing. Fuel 2010, 89, 438–456. [Google Scholar] [CrossRef]
- Oguma, M.; Goto, S.; Oyama, K.; Suguyama, K.; Mori, M. The possibility of gas to liquid (GTL) as a fuel of direct injection diesel engine. SAE Technol. Pap. 2002, 2002, 1706. [Google Scholar]
- Sajjad, H.; Masjuki, H.H.; Varman, M.; Kalam, M.A.; Arbab, M.I.; Imtenan, S.; Ashraful, A.M. Influence of gas-to-liquid (GTL) fuel in the blends of Calophyllum inophyllum biodiesel and diesel: An analysis of combustion–performance–emission characteristics. Energy Convers. Manag. 2015, 97, 42–52. [Google Scholar] [CrossRef]
- Sajjad, H.; Masjuki, H.H.; Varman, M.; Kalam, M.A.; Arbab, M.I.; Imtenan, S.; Ashrafur Rahman, S.M. Engine combustion, performance and emission characteristics of gas to liquid (GTL) fuels and its blends with diesel and bio-diesel. Renew. Sustain. Energy Rev. 2014, 30, 961–986. [Google Scholar] [CrossRef]
- Armas, O.; Gómez, A.; Ramos, A. Comparative study of pollutant emissions from engine starting with animal fat biodiesel and GTL fuels. Fuel 2013, 113, 560–570. [Google Scholar] [CrossRef]
- Abdul-Manan, A.F.N.; Bakor, R.Y.; Zubail, A.H. Analyzing the effects of Gas-to-Liquid (GTL) diesel blending on the efficiency and emissions of petroleum refineries and transport fuels in the U.S. and Europe. Transp. Res.-D 2018, 59, 259–267. [Google Scholar] [CrossRef]
- Du, J.; Sun, W.; Wang, X.; Li, G.; Fan, L. Experimental study on combustion and particle size distribution of a common rail diesel engine fueled with GTL/diesel blends. Appl. Therm. Eng. 2014, 70, 430–440. [Google Scholar] [CrossRef]
- Ezzitouni, S.; Soriano, J.A.; Gómez, A.; Armas, O. Impact of injection strategy and GTL fuels on combustion process and performance under diesel engine start. Atmos. Environ. 2018, 177, 214–221. [Google Scholar] [CrossRef]
- Wang, H.; Hao, H.; Li, X.; Zhang, K.; Ouyang, M. Performance of Euro III common rail heavy duty diesel engine fueled with Gas to Liquid. Appl. Energy 2009, 86, 2257–2261. [Google Scholar] [CrossRef]
- Lapuerta, M.; Armas, O.; Hernadez, J.J.; Tsolake, A. Potential for reducing emissions in a diesel engine by fueling with conventional biodiesel and Fischer-Tropsch diesel. Fuel 2010, 89, 3106–3113. [Google Scholar] [CrossRef]
- Soltic, P.; Edenhauser, D.; Thurnheer, T.; Schreiber, D.; Sankowski, A. Experimental investigation of mineral diesel fuel, GTL fuel, RME and neat soybean and rapeseed oil combustion in a heavy duty on-road engine with exhaust gas after-treatment. Fuel 2009, 88, 1–8. [Google Scholar] [CrossRef]
- Hassaneen, A.; Munack, A.; Ruschel, Y.; Schroeder, O.; Krahl, J. Fuel economy and emission characteristics of Gas-to-Liquid (GTL) and Rapeseed Methyl Ester (RME) as alternative fuels for diesel engines. Fuel 2012, 97, 125–130. [Google Scholar] [CrossRef]
- Li, X.; Huang, C. Emission reduction potential of using gas-to liquid and dimethyl ether fuels on a turbocharged diesel engine. Sci. Total Environ. 2009, 407, 2234–2244. [Google Scholar]
- Huang, Y.; Zhou, L.; Wang, S.; Liu, S. Study on the performance and emissions of a compression ignition engine fuelled with Fischer–Tropsch diesel fuel. J. Automob. Eng. 2006, 220, 827–835. [Google Scholar]
- Abián, M.; Martín, C.; Nogueras, P.; Sánchez-Valdepeñas, J.; Alzueta, M.U. Interaction of diesel engine soot with NO2 and O2 at diesel exhaust conditions. Effect of fuel and engine operation mode. Fuel 2018, 212, 455–461. [Google Scholar] [CrossRef]
- Park, S.H.; Lee, D.; Lee, C.S. Influence of gas-to-liquid fuel on the combustion and pollutant emission characteristics. J. Automob. Eng. 2014, 228, 85–93. [Google Scholar] [CrossRef]
- Abu-Jrai, A.; Tsolakis, A.; Theinnoi, K.; Cracknell, R.; Megaritis, A.; Wyszynski, M.L.; Golunski, S.E. Effect of Gas-to-Liquid Diesel Fuels on Combustion Characteristics, Engine Emissions, and Exhaust Gas Fuel Reforming Comparative Study. Energy Fuel 2006, 20, 2377–2384. [Google Scholar] [CrossRef]
- Li, X.; Huang, Z.; Wang, J.; Zhang, W. Particle size distribution from a GTL engine. Sci. Total Environ. 2007, 382, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Choi, K.; Kim, M.Y.; Lee, C.S. Experimental investigation and prediction of density and viscosity of GTL, GTL-biodiesel, and GTL-diesel blends as a function of temperature. Eenergy Fuel 2013, 27, 56–65. [Google Scholar] [CrossRef]
- Yehliu, K.; Boehman Andre, L.; Armas, O. Emission from different alternative fuels operating with single and split fuel injection. Fuel 2010, 89, 423–437. [Google Scholar] [CrossRef]
| [Ref.#] First Author (Year) | Engine Type (Disp.) | Test Fuels | Featured Results |
|---|---|---|---|
| [5] Sajjad et al. (2015) | 4-cylinder Water-cooled Turbocharged Diesel engine (2477 cc) | Diesel |
|
| CI20 (20% CIBD + 80% Diesel) | |||
| G20 (20% GTL + 80% Diesel) | |||
| DCIG20 (30% CIBD + 20% GTL + 50 Diesel) | |||
| [7] Octavio et al. (2013) | 4-cylinder Intercooled Turbocharged Light-duty diesel engine (2000 cc) | Diesel |
|
| GTL | |||
| Biodiesel | |||
| [9] Jiakun et al. (2014) | 4-cylinder Intercooled Turbocharged Light-duty diesel engine (3168 cc) | Diesel GTL 10, 20, 30, 60, 100 [Vol %] |
|
| [10] Samir et al. (2017) | 4-cylinder VGT Intercooled, Light-duty diesel engine (1994 cc) | Diesel |
|
| Summer GTL | |||
| Winter GTL | |||
| [11] Wang et al. (2009) | 6-cylinder Intercooled Turbocharged diesel engine (5900 cc) | Diesel |
|
| GTL | |||
| [12] Magin et al. (2010) | 4-cylinder Intercooled Turbocharged diesel engine (2200 cc) | Diesel |
|
| Biodiesel | |||
| GTL | |||
| G30B70 | |||
| (30 GTL + 70% Biodiesel) |
| Item | Specification | |
|---|---|---|
| Engine type | Direct injection diesel engine | |
| Number of cylinders | 1 | |
| Bore × stroke | 75 mm × 84.5 mm | |
| Displacement volume | 0.3733 L | |
| Compression ratio | 17.3 | |
| Valve mechanism | DOHC 4 valves | |
| Fuel injection system | Bosch common rail | |
| Injector | Hole diameter | 0.131 mm |
| Number of holes | 8 | |
| Spray angle | 153° | |
| Valve timing | Intake valve | Open BTDC 8° |
| Close ABDC 52° | ||
| Exhaust valve | Open BBDC 8° | |
| Close ATDC 38° | ||
| Exhaust Emission Analyzer for NOx, HC, CO | |
|---|---|
| Item | Specification |
| Model | MEXA-554JK |
| Principle of measurement | CO, HC: Non-dispersive infrared rays |
| NOx: Chemical method (ECS sensor) | |
| Range | HC: 0 ~ 10,000 ppm vol. |
| CO: 0 ~ 10% vol. | |
| NOx: 0 ~ 4000 ppm | |
| Repeatability | HC: ±12 ppm vol. |
| CO: ±0.06% vol. | |
| NOx: Less than ±1.0% | |
| Response | HC and CO: 90% response within 10 s |
| NOx: within 30 s | |
| Exhaust Emission Analyzer for Soot | |
| Item | Specification |
| Model | AVL-415S |
| Principle of measurement | Filter paper method |
| Measuring range | 0 ~ 10 FSN (0 ~ 32,000 mg/m3) |
| Repeatability | 0.005 FSN +3% |
| Response | 0.001 FSN/0.01 mg/m3 |
| Test Items | Test Method | Diesel | GTL | Biodiesel |
|---|---|---|---|---|
| Density (kg/m3at 15 °C) | EN 12185 | 826.2 | 778.4 | 882.2 |
| Viscosity (mm2/s, at 40 °C) | EN ISO 3104 | 2.359 | 2.704 | 4.2 |
| Total sulfur (mg/kg) | EN ISO 20846 | 7.4 | 7.4 | 1 |
| Cetane index | EN ISO 4264 | 53.2 | 85.5 | 54.7 |
| Flash point (°C) | EN ISO 2719 | 61.0 | 85.0 | 180 |
| Pour point (°C) | ASTM D 97 | −39 | −24 | −5 |
| Cloud point (°C) | ASTM D 2500 | −10 | −19 | 4 |
| Ash content (wt%) | EN ISO 6245 | 0.001 | 0.001 | 0.002 |
| Polycyclic aromatic hydrocarbons (wt%) | EN 12916 | 1.6 | 0 | - |
| Lower heating value, MJ/kg | ASTM D 240 | 43.038 | 43.561 | 31.926 |
| CFPP (°C) * | ASTM D 6371 | −27 | −20 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, K.; Park, S.; Roh, H.G.; Lee, C.S. Combustion and Emission Reduction Characteristics of GTL-Biodiesel Fuel in a Single-Cylinder Diesel Engine. Energies 2019, 12, 2201. https://doi.org/10.3390/en12112201
Choi K, Park S, Roh HG, Lee CS. Combustion and Emission Reduction Characteristics of GTL-Biodiesel Fuel in a Single-Cylinder Diesel Engine. Energies. 2019; 12(11):2201. https://doi.org/10.3390/en12112201
Chicago/Turabian StyleChoi, Kibong, Suhan Park, Hyun Gu Roh, and Chang Sik Lee. 2019. "Combustion and Emission Reduction Characteristics of GTL-Biodiesel Fuel in a Single-Cylinder Diesel Engine" Energies 12, no. 11: 2201. https://doi.org/10.3390/en12112201
APA StyleChoi, K., Park, S., Roh, H. G., & Lee, C. S. (2019). Combustion and Emission Reduction Characteristics of GTL-Biodiesel Fuel in a Single-Cylinder Diesel Engine. Energies, 12(11), 2201. https://doi.org/10.3390/en12112201






