Use of Buckwheat Straw to Produce Ethyl Alcohol Using Ionic Liquids
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Ionic Liquids
2.3. Enzymatic Hydrolysis
2.4. Alcoholic Fermentation
2.5. Analytical Methods
- Ce—ethanol concentration (g/dm3)
- V—sample volume (dm3)
- M—total amount of substrate in the sample (g s.s.)
- C—cellulose concentration in the material (%)
- 1.1—cellulose to glucose conversion factor
- 0.51—glucose to ethanol conversion factor
3. Results and Discussion
3.1. Influence of Ionic Liquids on the Structure of Buckwheat Straw
3.2. Influence of the Use of Ionic Liquids on Enzymatic Hydrolysis and Alcoholic Fermentation
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zarzecka, K.; Gugała, M.; Mystkowska, I. Nutritional value and opportunities of using buckwheat. Postępy Fito. 2014, 1, 28. [Google Scholar]
- Alvira, P.; Tomas-Pejo, E.; Ballesteros, M.; Negro, M.J. Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: A Review. Bioresour. Technol. 2010, 101, 4851. [Google Scholar] [CrossRef]
- Holm, J.; Lassi, U. Ionic liquids in pretreatment of lignocellulosic biomass In Ionic Liquids: Application and Perspectives; Kokorin, A., Ed.; IntechOpen: Rijeka, Croatia, 2011; pp. 546–560. [Google Scholar]
- Pinkert, A.; Marsh, K.N.; Pang, S.; Staiger, M.P. Ionic liquids and their interaction with cellulose. Chem. Rev. 2009, 109, 6712. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S. Use of ionic liquids for the efficient utilization of lignocellulosic materials. J. Chem. Technol. Biotechnol. 2008, 83, 777. [Google Scholar] [CrossRef]
- Samayam, I.P.; Hanson, L.B.; Langan, P.; Schall, C.A. Ionic-liquid induced changes in cellulose structure associated with enhanced biomass hydrolysis. Biomacromolecules 2011, 12, 3091. [Google Scholar] [CrossRef]
- Brandt, A.; Hallett, J.P.; Leak, D.J.; Murphy, R.J.; Welton, T. The effect of the ionic liquid anion in the pretreatment of pine wood chips. Green Chem. 2010, 12, 672–679. [Google Scholar] [CrossRef]
- Liu, K.; Lin, X.; Yue, J.; Li, X.; Fang, X.; Zhu, M.; Lin, J.; Qua, J.; Xiao, L. High concentration ethanol production from corncob residues by fed-batch strategy. Bioresour. Technol. 2010, 101, 4952–4958. [Google Scholar] [CrossRef]
- Smuga-Kogut, M.; Zgórska, K.; Szymanowska-Powałowska, D. Influence of the crystalline structure of cellulose on the production of ethanol from lignocellulose biomass. Int. Agrophys. 2016, 30, 83–88. [Google Scholar] [CrossRef]
- Li, H.Y.; Chen, X.; Wang, C.Z.; Sun, S.N.; Sun, R.C. Evaluation of the two-step treatment with ionic liquids and alkali for enhancing enzymatic hydrolysis of Eucalyptus: Chemical and anatomical changes. Biotechnol. Biofuels 2016, 9, 166. [Google Scholar] [CrossRef] [PubMed]
- Shill, K.; Padmanabhan, S.; Xin, Q.; Prausnitz, J.M.; Clark, D.S.; Blanch, H.W. Ionic liquid pretreatment of cellulosic biomass: Enzymatic hydrolysis and ionic liquid recycle. Biotechnol. Bioeng. 2011, 108, 511. [Google Scholar] [CrossRef]
- Qiu, Z.; Aita, G.M.; Walker, M.S. Effect of ionic liquid pretreatment on the chemical composition, structure and enzymatic hydrolysis of energy cane bagasse. Bioresour. Technol. 2012, 117, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Doherty, T.V.; Mora-Pale, M.; Foley, S.E.; Linhardt, R.J.; Dordick, J.S. Ionic liquid solvent properties as predictors of lignocellulose pretreatment efficacy. Green Chem. 2010, 12, 1967–1975. [Google Scholar] [CrossRef]
- Tuchołka, D. Methods of Absorption and Reflection Spectroscopy in Chemical Analysis. Ph.D. Thesis, University of Agriculture in Poznan, Poznan, Poland, 1996. [Google Scholar]
- Kocjan, R. Analytical Chemistry T. 2 Instrumental Analysis; Medical Publishing PZWL: Warsaw, Poland, 2014; pp. 231–247. [Google Scholar]
- Ji, Z.; Zhang, X.; Ling, Z.; Zhou, X.; Ramaswamy, S.; Xu, F. Visualization of Miscanthus giganteus cell wall deconstruction subjected to dilute acid pretreatment for enhanced enzymatic digestibility. Biotechnol. Biofuels 2015, 8, 1. [Google Scholar] [CrossRef]
- Horn, S.J.; Vaaje-Kolstad, G.; Westereng, B.; Eijsink, V.G.H. Novel enzymes for the degradation of cellulose. Biotechnol. Biofuels 2012, 5, 45. [Google Scholar] [CrossRef]
- Uju, S.Y.; Nakamoto, A.; Goto, M.; Tokuhara, W.; Noritake, Y.; Katahira, S.; Ishida, N.; Nakashima, K.; Ogino, C.; Kamiya, N. Short time ionic liquids pretreatment on lignocellulosic biomass to enhance enzymatic saccharification. Bioresour. Technol. 2012, 103, 446–452. [Google Scholar] [CrossRef]
- Sun, S.; Cao, X.; Sun, S.; Xu, F.; Song, X.; Jones, G. Improving the enzymatic hydrolysis of thermo-mechanical fiber from Eucalyptus urophylla by a combination of hydrothermal pretreatment and alkali fractionation. Biotechnol. Biofuels 2014, 7, 116. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Doherty, T.V.; Linhardt, R.J.; Dordick, J.S. Ionic liquid-mediated selective extraction of lignin from wood leading to enhanced enzymatic cellulose hydrolysis. Biotechnol. Bioeng. 2009, 102, 1368–1376. [Google Scholar] [CrossRef]
- Boeriu, C.G.; Bravo, D.; Gosselink, R.J.; van Dam, J.E. Characterization of structure-dependent functional properties of lignin with infrared spectroscopy. Ind. Crops Prod. 2004, 20, 205–218. [Google Scholar] [CrossRef]
- Hou, X.D.; Li, N.; Zong, M.H. Renewable bio ionic liquids-water mixtures-mediated selective removal of lignin from rice straw: Visualization of changes in composition and cell wall structure. Biotechnol. Bioeng. 2013, 110, 1895–1902. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, S.; Seydibeyoglu, M.O.; Mohanty, A.K.; Misra, M. Characterization of industrial lignins for their utilization in future value added applications. Biomass Bioenergy 2011, 35, 4230–4237. [Google Scholar] [CrossRef]
- Cotana, F.; Cavalaglio, G.; Nicolini, A.; Gelosia, M.; Coccia, V.; Petrozzi, A.; Brinchi, L. Lignin as co-product of second generation bioethanol production from lignocellulosic. Energy Procedia 2014, 45, 52–60. [Google Scholar] [CrossRef]
- Mehmood, N.; Husson, E.; Jacquard, C.; Wewetzer, S.; Büchs, J.; Sarazin, C.I. Impact of two ionic liquids, 1-ethyl-3-methylimidazolium acetate and 1-ethyl-3-methylimidazolium methylphosphonate, on Saccharomyces cerevisiae: Metabolic, physiologic, and morphological investigations. Biotechnol. Biofuels 2015, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Sitepu, I.R.; Shi, S.; Simmons, B.A.; Singer, S.W.; Boundy-Mills, K.; Simmons, C.W. Yeast tolerance to the ionic liquid 1-ethyl-3-methylimidazolium acetate. FEMS Yeast Res. 2014, 14, 1286–1294. [Google Scholar] [CrossRef] [PubMed]
- Qing, Q.; Yang, B.; Wyman, C.E. Xylooligomers are strong inhibitors of cellulose hydrolysis by enzymes. Bioresour. Technol. 2010, 101, 9624–9630. [Google Scholar] [CrossRef]
- Giri, F.M.; Fonseca, C.; Carvalheiro, F.; Duarte, L.C.; Marques, S.; Bogel-Lukasic, R. Hemicelluloses for fuel ethanol: A review. Bioresour. Technol. 2010, 101, 4775–4800. [Google Scholar] [CrossRef]
- Gamage, J.; Howard, L.; Zisheng, Z. Bioethanol production from lignocellulosic biomass. J. Biobased Mater. Bioenerg. 2010, 4, 3–11. [Google Scholar] [CrossRef]
- Garver, M.P.; Liu, S. Development of thermochemical and biochemical technologies for biorefineries. In Bioenergy Research: Advances and Applications, 1st ed.; Gupta, V.K., Tuohy, M.G., Kubicek, C.P., Saddler, J., Xu, F., Eds.; Publisher: Elsevier, Netherlands, 2014; Volume 27, pp. 457–488. [Google Scholar]
- Álvarez, C.; Manuel Reyes-Sosa, F.; Díez, B. Enzymatic hydrolysis of biomass from wood. Microbiol. Biotechnol. 2016, 9, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Pan, X. Correlation between lignin physicochemical properties and inhibition to enzymatic hydrolysis of cellulose. Biotechnol. Bioeng. 2016, 113, 1213–1224. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.G.; Jin, Y.S.; Cha, Y.L.; Seo, J.H. Bioethanol production from cellulosic hydrolysates by engineered industrial Saccharomyces cerevisiae. Bioresour. Technol. 2017, 228, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, Q.; Bottoms, S.; Hinchman, L.; McIlwain, S.; Li, S.; Myers, C.L.; Boone, C.; Coon, J.J.; Hebert, A.; Sato, T.K.; et al. Mechanism of imidazolium ionic liquids toxicity in Saccharomyces cerevisiae and rational engineering of a tolerant, xylose-fermenting strain. Microbial. Cell Fact. 2016, 15, 17. [Google Scholar] [CrossRef] [PubMed]

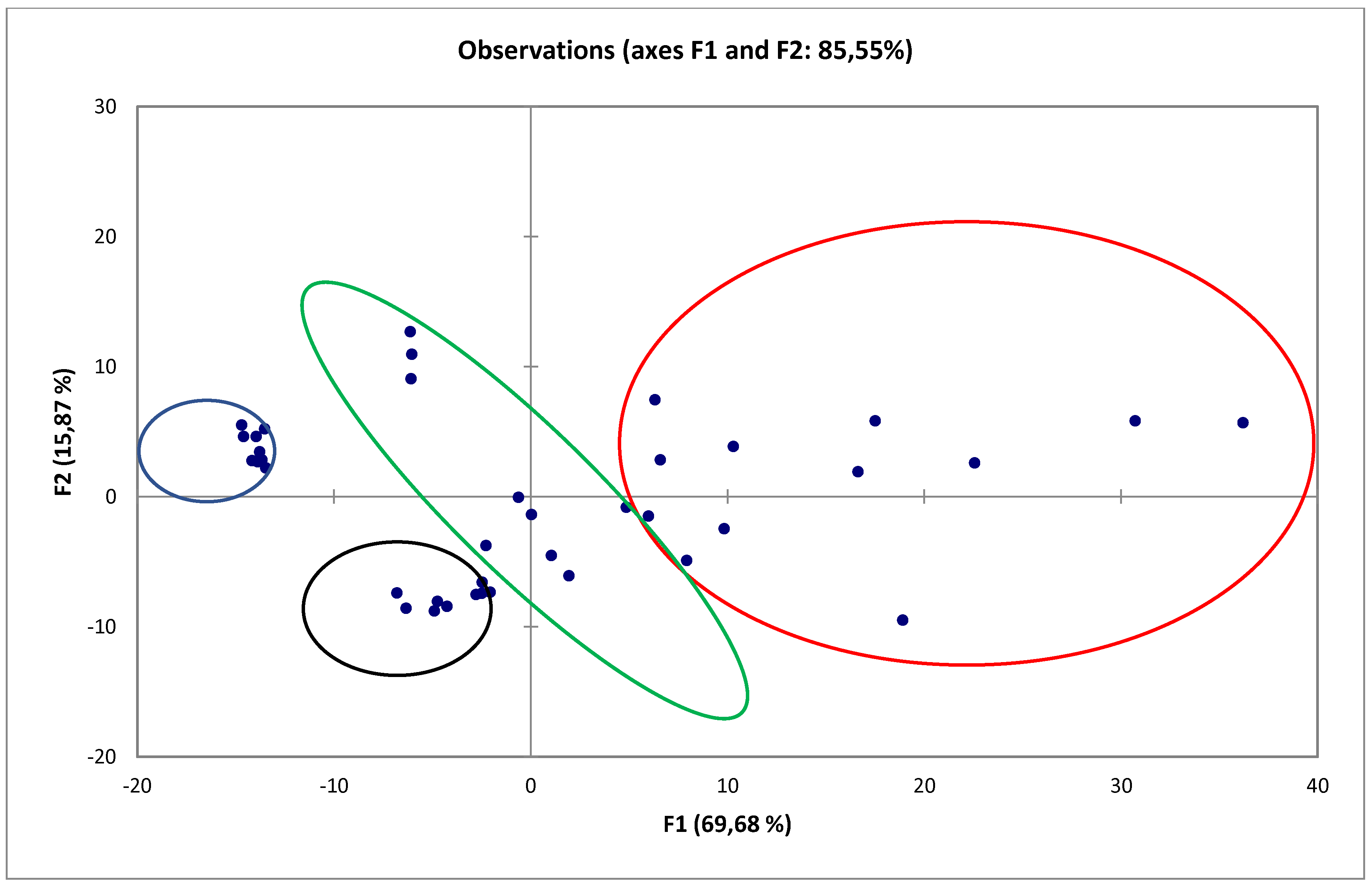
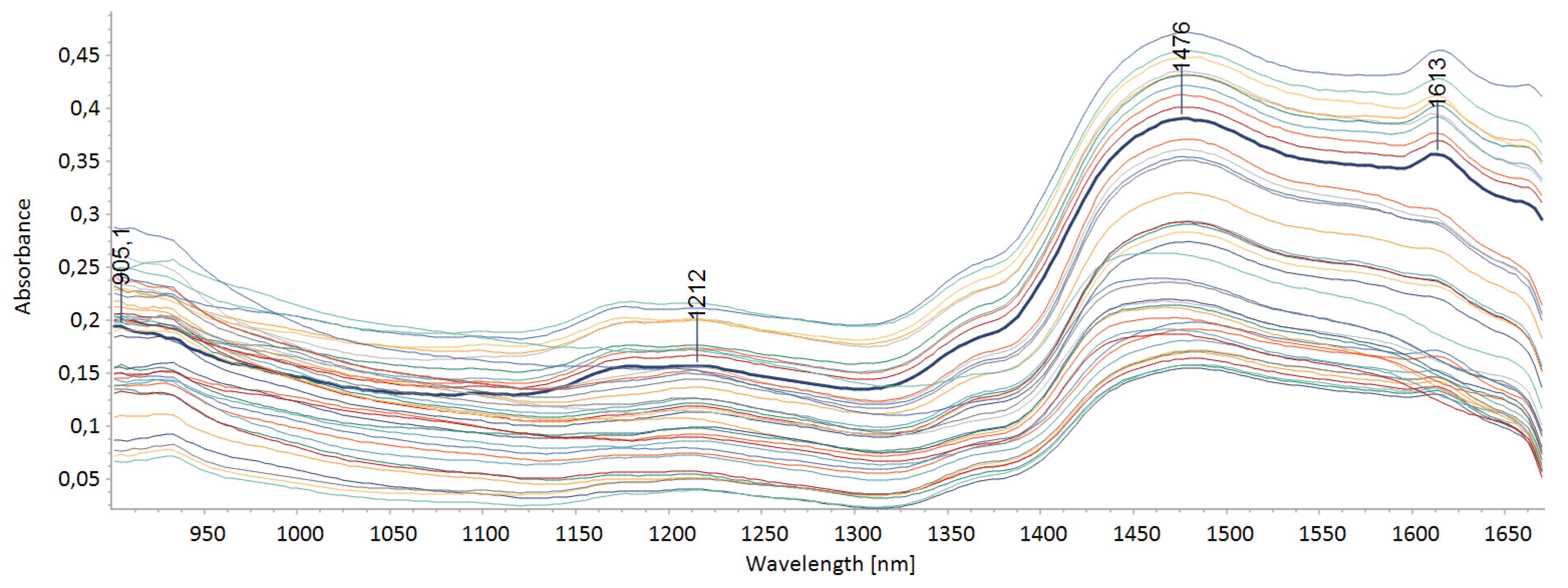
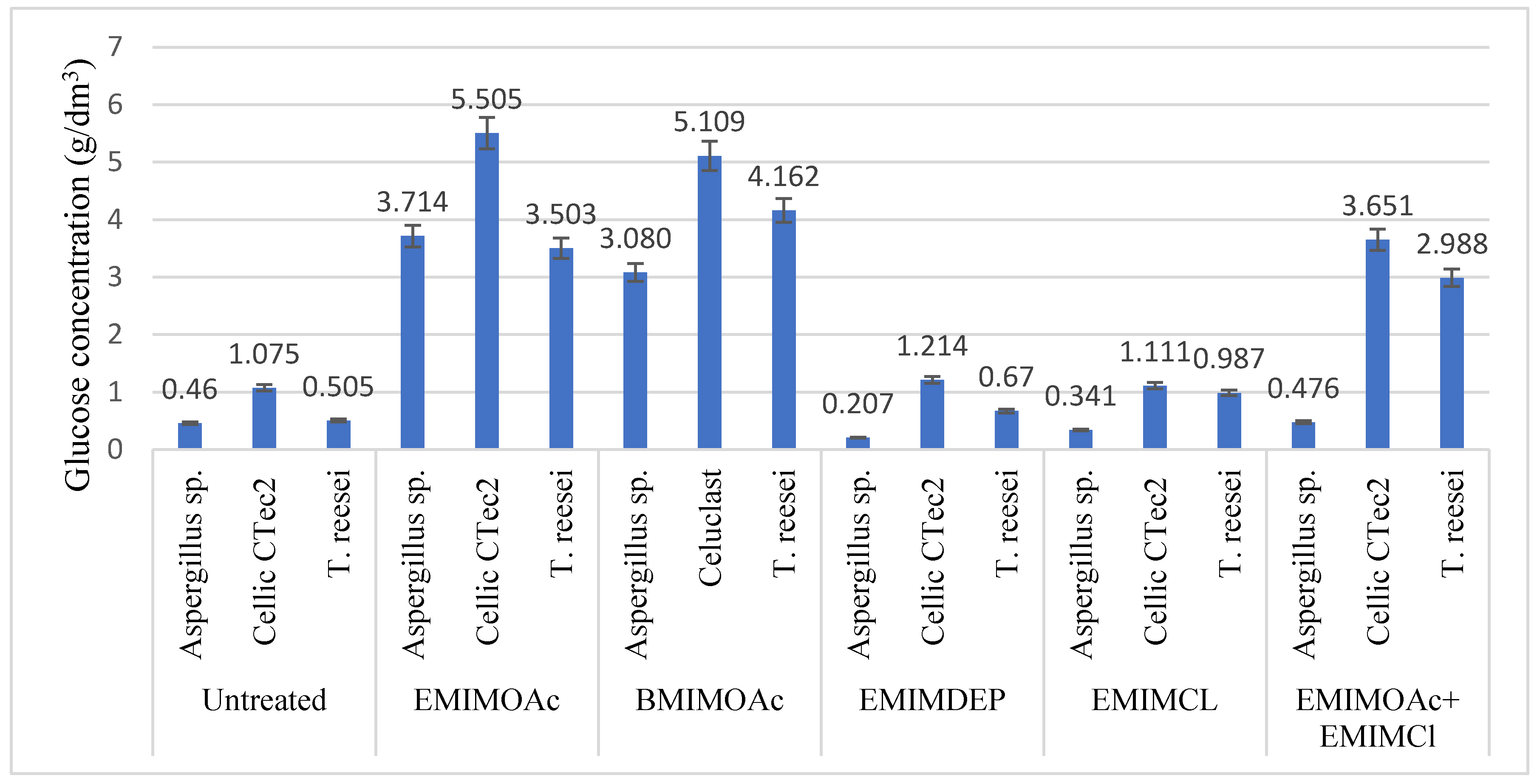
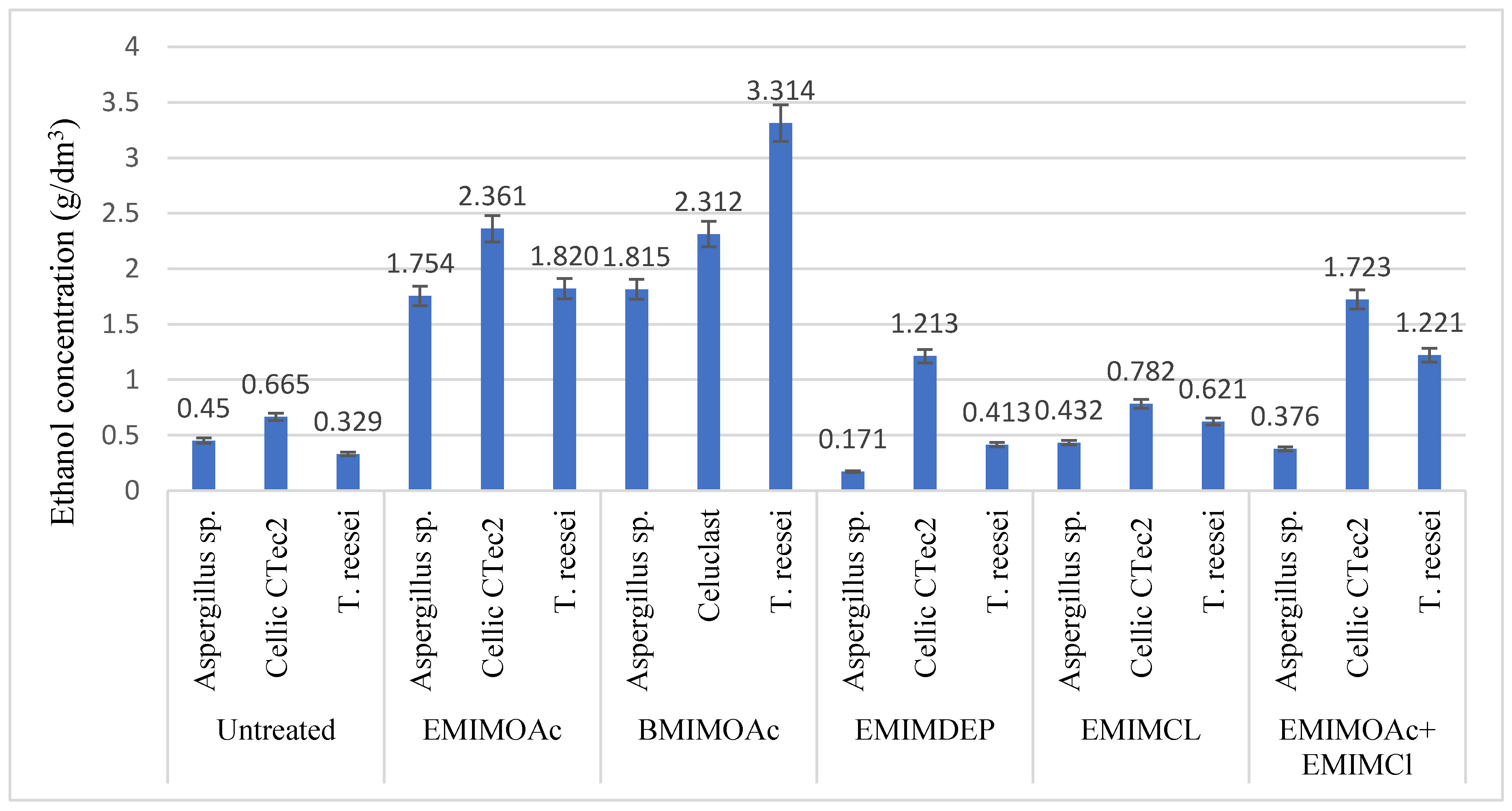
| Type of Pretreatment | Cellulose [%] | Hemicellulose [%] | Lignin [%] |
|---|---|---|---|
| Untreated | 42.08 ± 0.12 | 8.38 ± 0.02 | 16.97 ± 0.13 |
| EMIMCl | 29.05 ± 0.21 | 18.47 ± 0.11 | 11.98 ± 0.23 |
| EMIMDEP | 41.49 ± 0.10 | 22.27 ± 0.03 | 15.26 ± 0.19 |
| EMIMOAc | 39.48 ± 0.07 | 8.81 ± 0.07 | 20.66 ± 0.08 |
| BMIMOAc | 36.91 ± 0.17 | 9.33 ± 0.14 | 14.32 ± 0.11 |
| EMIMOAc + EMIMCl | 33.27 ± 0.08 | 18.56 ± 0.22 | 8.71 ± 0.12 |
| Model | Range (nm) | Number of Spectra | Number of Variables | R2 | MSE | RMSE |
|---|---|---|---|---|---|---|
| PLS | 900–1680 | 50 | 1 | 0.9593 | 1.1311 | 1.0635 |
| Cellulose Content [%] | PLS | |
|---|---|---|
| Predicted Cellulose Content [%] | Deviation [%] | |
| 29.05 | 29.63 | 2.04 |
| 41.49 | 42.83 | 1.65 |
| 39.48 | 38.00 | 1.52 |
| 42.08 | 41.64 | 2.08 |
| Sample | Enzyme Type | Cell Viability (%) | Cell Count (CFU) | Cellulose to Ethanol Conversion (%) |
|---|---|---|---|---|
| Untreated | Aspergillus sp. | 55% ± 1.1% | 4.0 × 106 | 15.25% |
| Cellic CTec2 | 61% ± 0.9% | 3.1 × 105 | 22.54% | |
| T. reesei | 60% ± 1.9% | 2.9 × 105 | 11.15% | |
| EMIMOAc | Aspergillus sp. | 69% ± 0.7% | 9.0 × 106 | 63.23% |
| Cellic CTec2 | 78% ± 0.9% | 8.4 × 105 | 83.10% | |
| T. reesei | 75% ± 2.1% | 7.6 × 105 | 65.76% | |
| BMIMOAc | Aspergillus sp. | 67% ± 2.2% | 6.0 × 106 | 70.14% |
| Cellic CTec2 | 72% ± 1.9% | 8.0 × 105 | 89.35% | |
| T. reesei | 70% ± 1.8% | 5.5 × 105 | 128.07% | |
| EMIMDEP | Aspergillus sp. | 54% ± 1.4% | 3.0 × 106 | 5.88% |
| Cellic CTec2 | 57% ± 2.1% | 3.4 × 105 | 41.71% | |
| T. reesei | 56% ± 1.4% | 2.6 × 105 | 14.20% | |
| EMIMCl | Aspergillus sp. | 51% ± 0.7% | 3.2 × 106 | 21.22% |
| Cellic CTec2 | 55% ± 0.6% | 4.5 × 105 | 38.42% | |
| T. reesei | 43% ± 1.5% | 4.0 × 105 | 30.51% | |
| EMIMOAc+ EMIMCl | Aspergillus sp. | 74% ± 0.5% | 5.0 × 106 | 16.22% |
| Cellic CTec2 | 78% ± 2.1% | 6.6 × 105 | 73.86% | |
| T. reesei | 81% ± 2.9% | 4.5 × 105 | 52.34% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smuga-Kogut, M.; Bychto, L.; Walendzik, B.; Cielecka-Piontek, J.; Marecik, R.; Kobus-Cisowska, J.; Grajek, K.; Szymanowska-Powałowska, D. Use of Buckwheat Straw to Produce Ethyl Alcohol Using Ionic Liquids. Energies 2019, 12, 2014. https://doi.org/10.3390/en12102014
Smuga-Kogut M, Bychto L, Walendzik B, Cielecka-Piontek J, Marecik R, Kobus-Cisowska J, Grajek K, Szymanowska-Powałowska D. Use of Buckwheat Straw to Produce Ethyl Alcohol Using Ionic Liquids. Energies. 2019; 12(10):2014. https://doi.org/10.3390/en12102014
Chicago/Turabian StyleSmuga-Kogut, Małgorzata, Leszek Bychto, Bartosz Walendzik, Judyta Cielecka-Piontek, Roman Marecik, Joanna Kobus-Cisowska, Katarzyna Grajek, and Daria Szymanowska-Powałowska. 2019. "Use of Buckwheat Straw to Produce Ethyl Alcohol Using Ionic Liquids" Energies 12, no. 10: 2014. https://doi.org/10.3390/en12102014
APA StyleSmuga-Kogut, M., Bychto, L., Walendzik, B., Cielecka-Piontek, J., Marecik, R., Kobus-Cisowska, J., Grajek, K., & Szymanowska-Powałowska, D. (2019). Use of Buckwheat Straw to Produce Ethyl Alcohol Using Ionic Liquids. Energies, 12(10), 2014. https://doi.org/10.3390/en12102014






