Torrefaction as a Valorization Method Used Prior to the Gasification of Sewage Sludge
Abstract
1. Introduction
1.1. Introduction
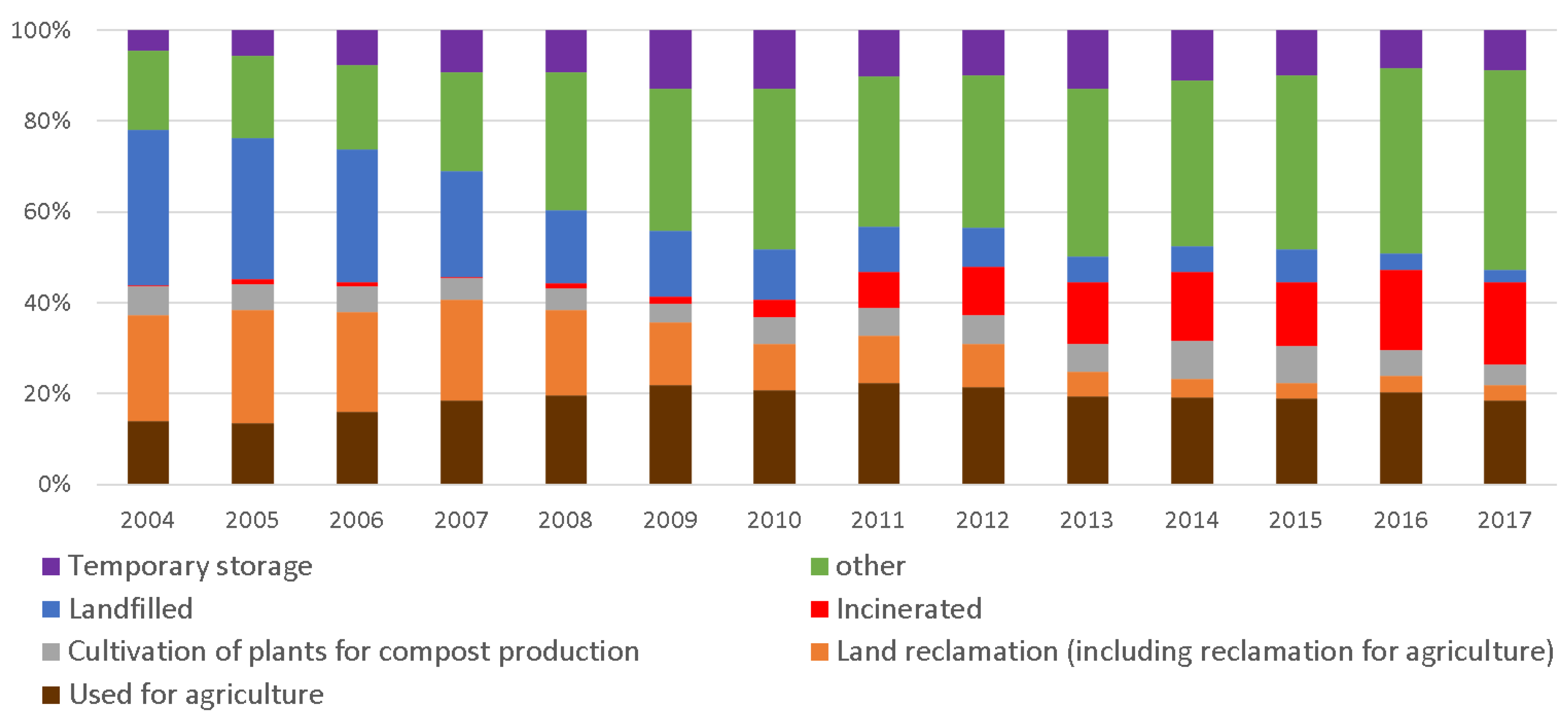
1.2. Torrefaction of Sewage Sludge
1.3. Gasification of Raw and Torrefied Sewage Sludge
2. Novelty, Relevance, Goals and Scope of Work
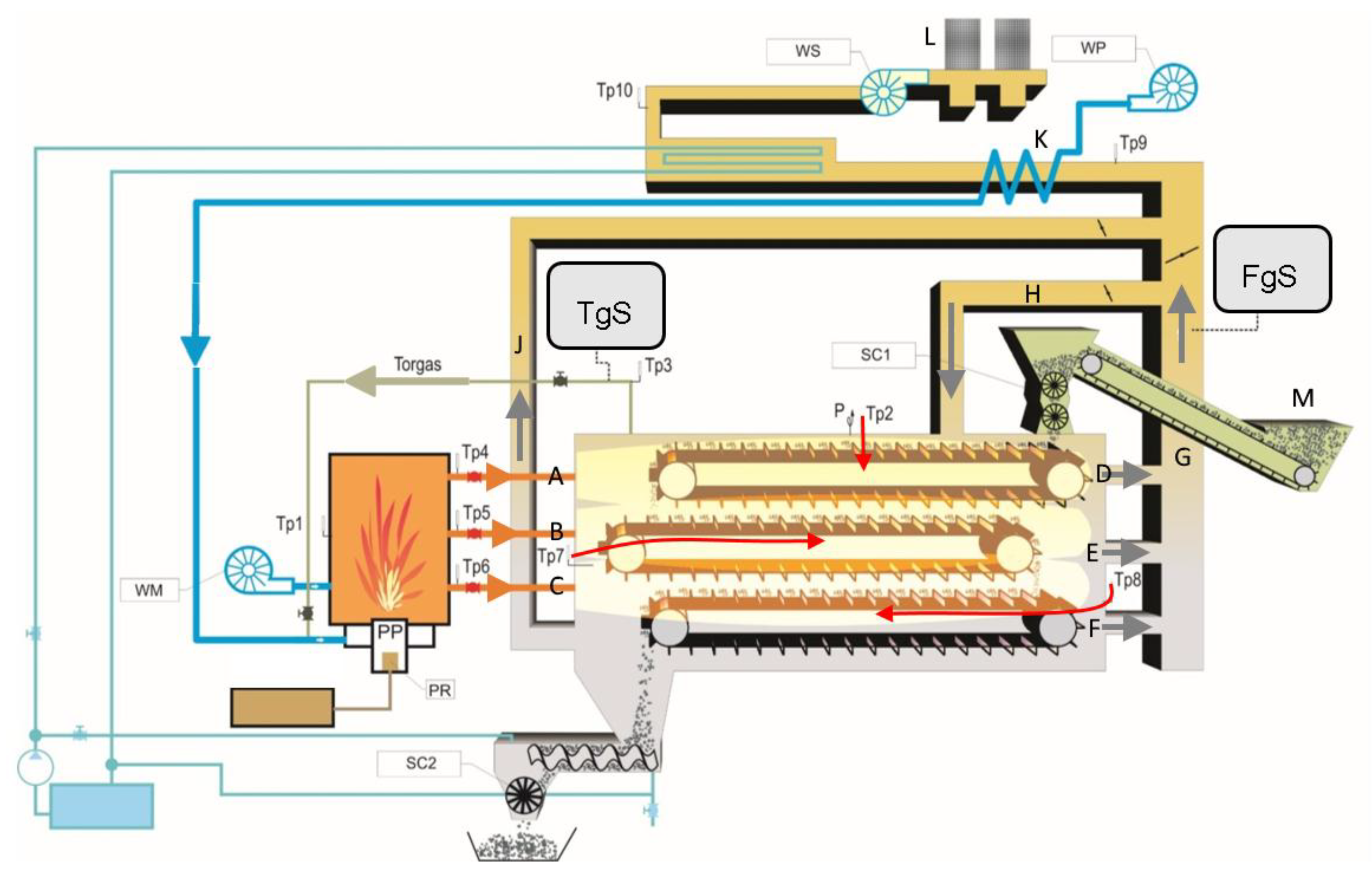
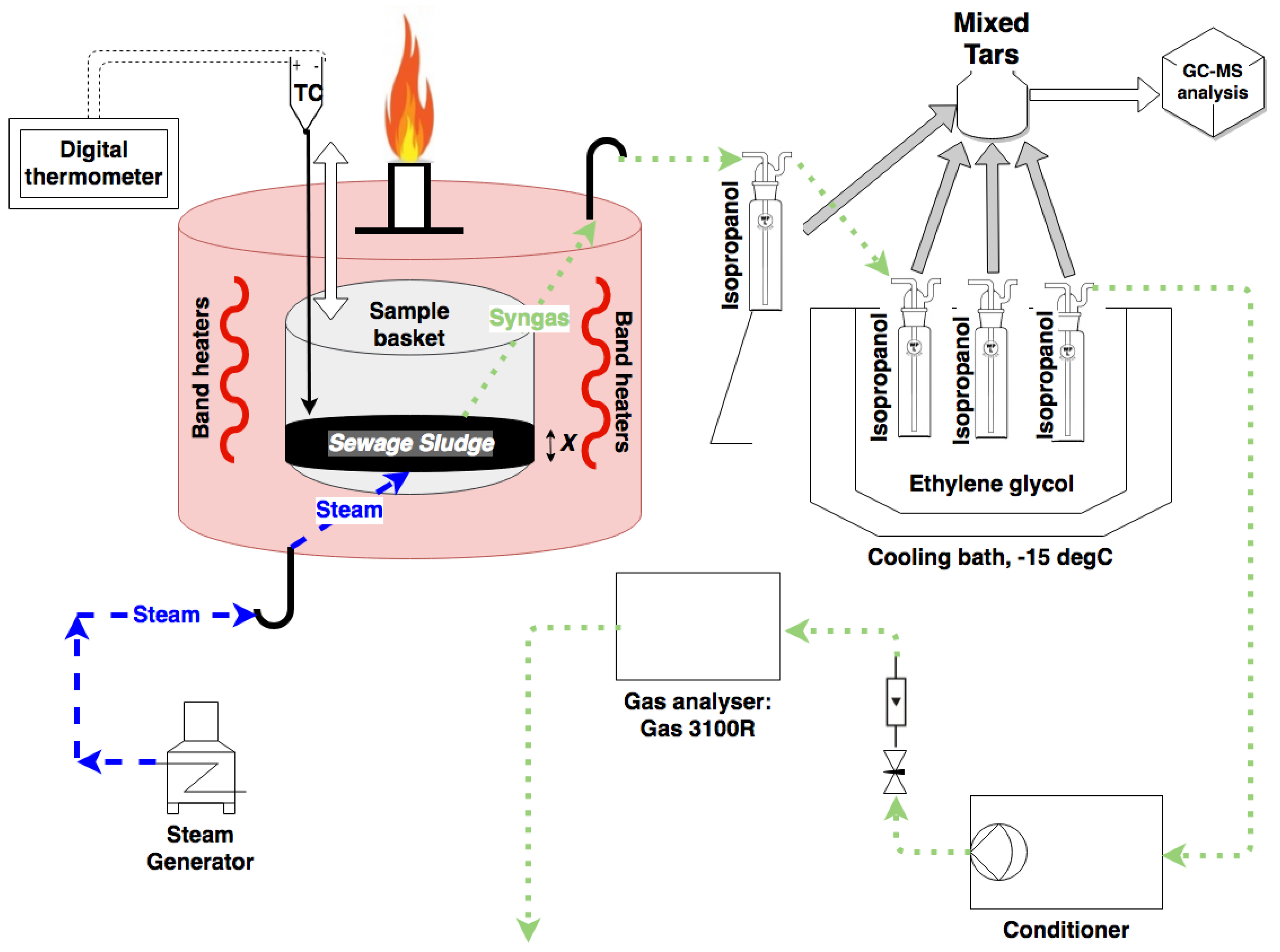
3. Materials and Methods
- Ym—mass yield, -;
- VM—respective volatile matter content of feedstock and product, %dry.
- Ye—energy yield, -;
- HHV—respective higher heating value of feedstock and product, MJ/kg.
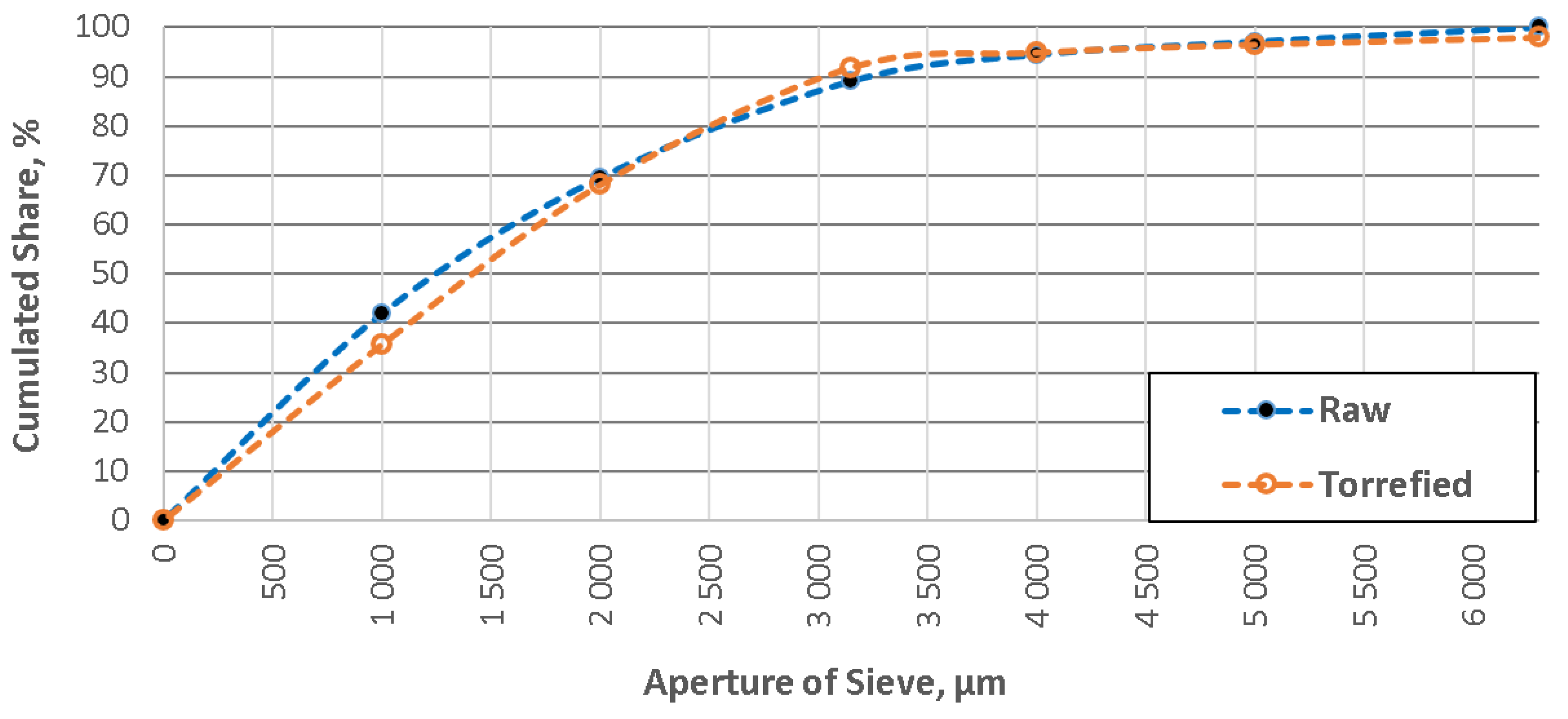
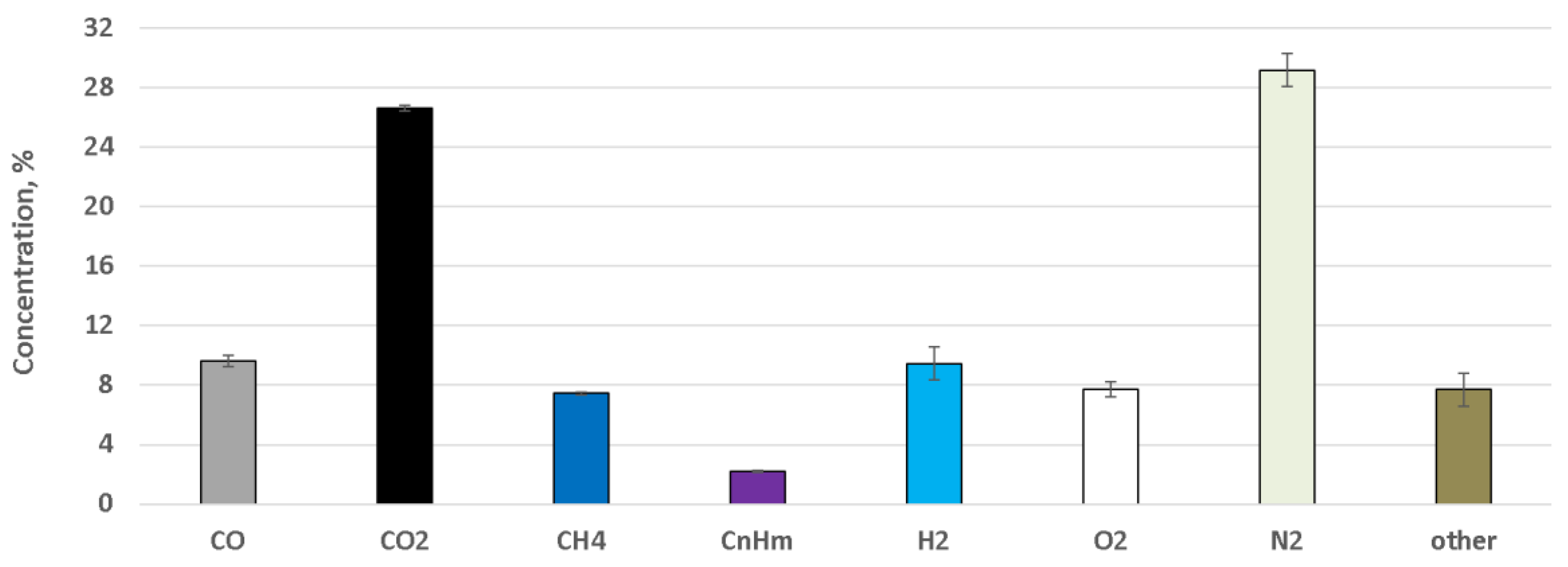
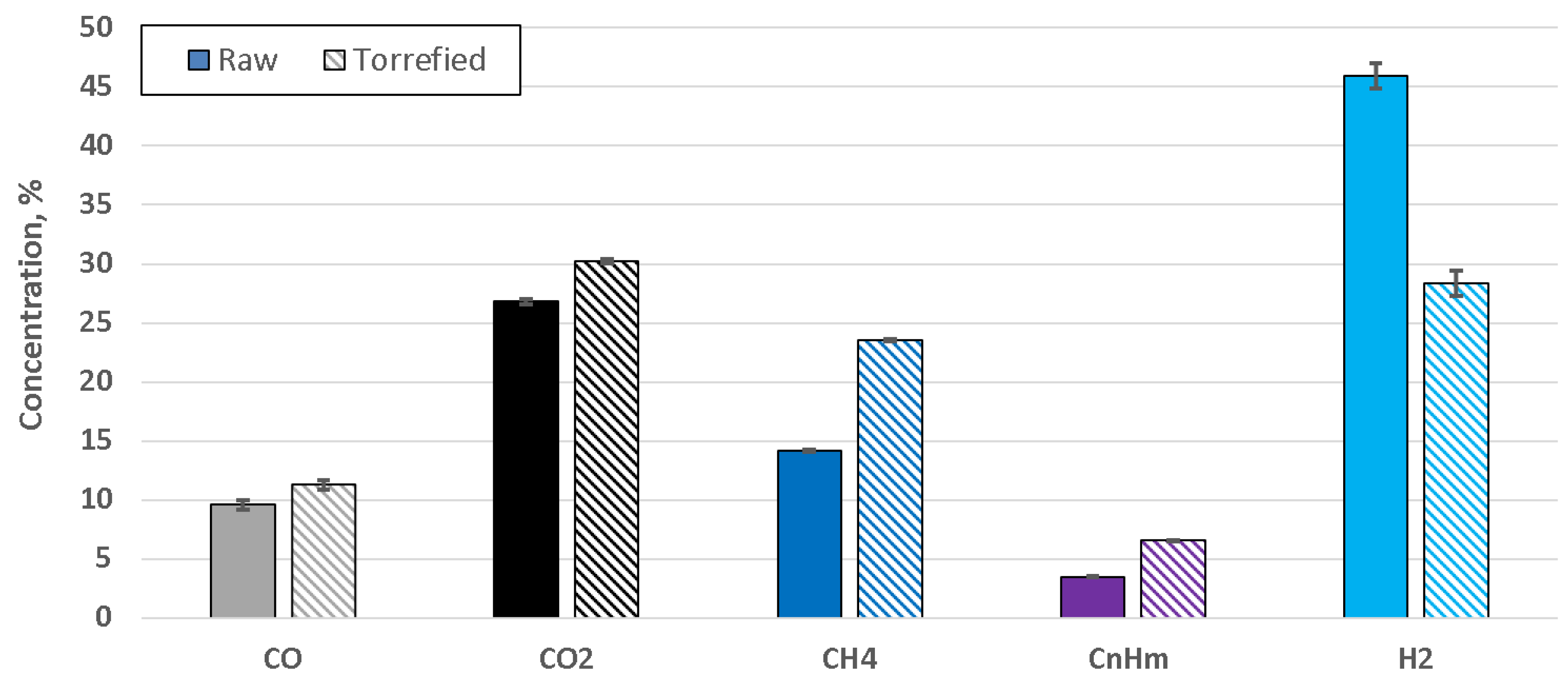
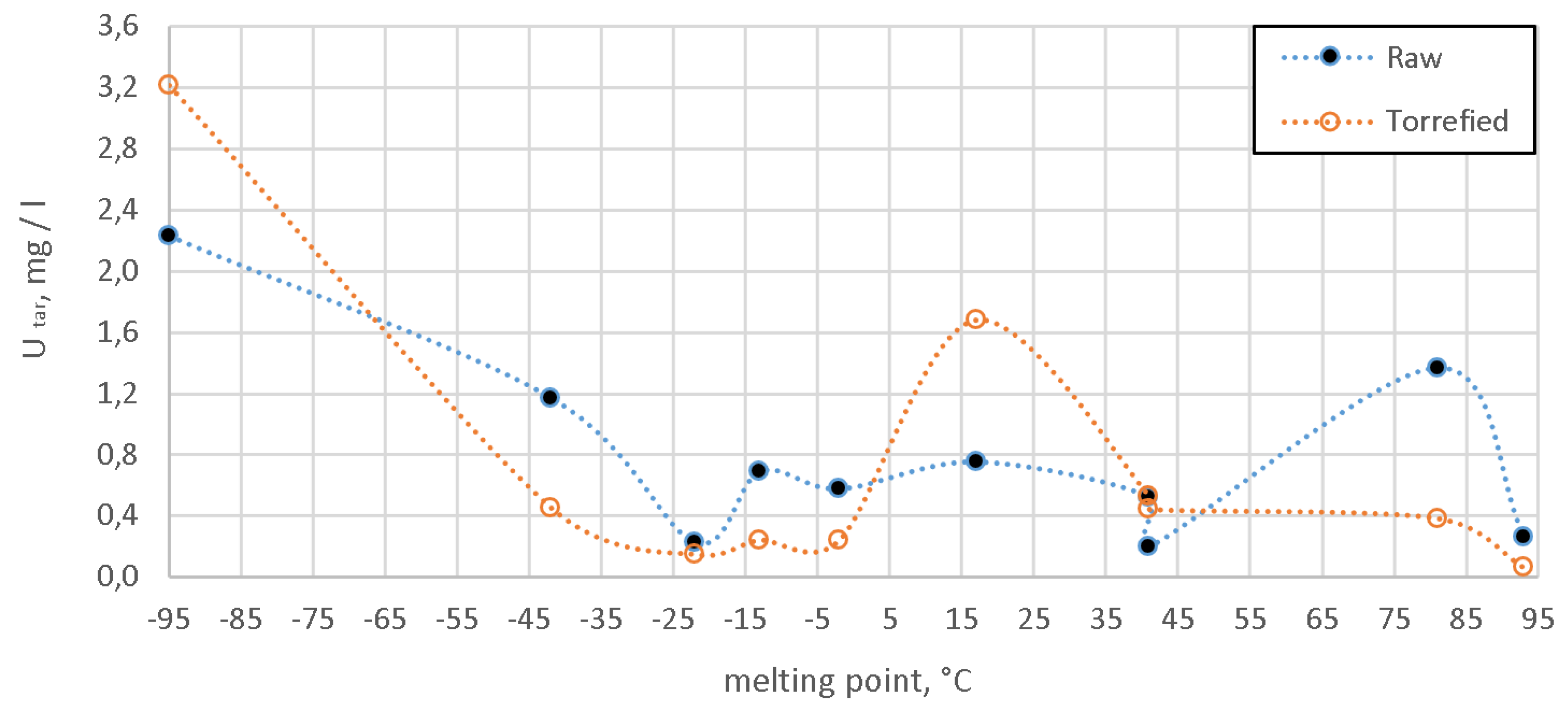
4. Results
5. Discussion
6. Conclusions
- Since the test was performed in an allothermal gasifier, investigation is needed to confirm if the gasification process can be authothermal;
- Tests performed on a pilot-scale gasifier should be performed in order to confirm if such an improvement in tar composition is indeed sufficient to significantly improve the maintenance of such a gasifier and decrease the frequency of the shutdowns.
Author Contributions
Funding
Conflicts of Interest
References
- Werle, S.; Dudziak, M. Analysis of organic and inorganic contaminants in dried sewage sludge and by-products of dried sewage sludge gasification. Energies 2014, 7, 462–476. [Google Scholar] [CrossRef]
- Kacprzak, M.; Neczaj, E.; Fijałkowski, K.; Grobelak, A.; Grosser, A.; Worwag, M.; Rorat, A.; Brattebo, H.; Almås, Å.; Singh, B.R. Sewage sludge disposal strategies for sustainable development. Environ. Res. 2017, 156, 39–46. [Google Scholar] [CrossRef]
- Lee, L.H.; Wu, T.Y.; Shak, K.P.Y.; Lim, S.L.; Ng, K.Y.; Nguyen, M.N.; Teoh, W.H. Sustainable approach to biotransform industrial sludge into organic fertilizer via vermicomposting: A mini-review. J. Chem. Technol. Biotechnol. 2018, 93, 925–935. [Google Scholar] [CrossRef]
- Cieślik, B.M.; Namieśnik, J.; Konieczka, P. Review of sewage sludge management: Standards, regulations and analytical methods. J. Clean. Prod. 2015, 90, 1–15. [Google Scholar] [CrossRef]
- Andersen, A. Disposal and Recycling Routes for Sewage Sludge Part 4: Economic Report; Office for Official Publications of the European Communities: Luxembourg, 2002. [Google Scholar]
- Werle, S.; Wilk, R.K. A review of methods for the thermal utilization of sewage sludge: The Polish perspective. Renew. Energy 2010, 35, 1914–1919. [Google Scholar] [CrossRef]
- GUS, Departament Badań Regionalnych i Środowiska. Ochrona Środowiska 2017; GUS: Warszawa, Poland, 2017; p. 551. [Google Scholar]
- Werle, S. Sewage Sludge-to-Energy Management in Eastern Europe: A Polish Perspective. Ecol. Chem. Eng. S 2015, 22, 459–469. [Google Scholar] [CrossRef]
- Pajak, T. Thermal Treatment as sustainable sewage sludge management. Environ. Prot. Eng. 2013, 39, 41–53. [Google Scholar] [CrossRef]
- Bień, J. Zagospodarowanie komunalnych osadów ściekowych metodami termicznymi. Inż. Ochr. Śr. 2012, 15, 439–450. [Google Scholar]
- Bień, J.; Bień, B. Utilisation of Municipal Sewage Sludge by Thermal Methods in the Face of Storage Disallowing. Inż. Ekol. 2015, 45, 36–43. [Google Scholar] [CrossRef]
- Jurczyk, M.P.; Pająk, T. Initial Operating Experience with the New Polish Waste-to-Energy Plants. In Waste Management; Thomé-Kozmiensky, K.J., Thiel, S., Eds.; TK Verlag: Neuruppin, Germany, 2016. [Google Scholar]
- Cyranka, M.; Jurczyk, M.; Pająk, T. Municipal Waste-to-Energy plants in Poland—Current projects. E3S Web Conf. 2016, 10, 00070. [Google Scholar] [CrossRef]
- Gorazda, K.; Tarko, B.; Wzorek, Z.; Kominko, H.; Nowak, A.K.; Kulczycka, J.; Henclik, A.; Smol, M. Fertilisers production from ashes after sewage sludge combustion—A strategy towards sustainable development. Environ. Res. 2017, 154, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Moscicki, K.J.; Niedzwiecki, L.; Owczarek, P.; Wnukowski, M. Commoditization of biomass: Dry torrefaction and pelletization-a review. J. Power Technol. 2014, 94, 233–249. [Google Scholar]
- Van der Stelt, M.J.C.; Gerhauser, H.; Kiel, J.H.A.; Ptasinski, K.J. Biomass upgrading by torrefaction for the production of biofuels: A review. Biomass Bioenergy 2011, 35, 3748–3762. [Google Scholar] [CrossRef]
- Tumuluru, J.S.; Sokhansanj, S.; Hess, J.R.; Wright, C.T.; Boardman, R.D. A review on biomass torrefcation process and product properties for energy applications. Ind. Biotechnol. 2011, 7, 384–401. [Google Scholar] [CrossRef]
- Nyakuma, B.B.; Magdziarz, A.; Werle, S. Physicochemical, Thermal and Kinetic Analysis of Sewage Sludge. Proc. ECOpole 2016, 10, 473–480. [Google Scholar] [CrossRef]
- Magdziarz, A.; Wilk, M.; Kosturkiewicz, B. Investigation of sewage sludge preparation for combustion process. Chem. Process Eng. Inz. Chem. Proces. 2011, 32, 299–309. [Google Scholar] [CrossRef]
- Magdziarz, A.; Werle, S. Analysis of the combustion and pyrolysis of dried sewage sludge by TGA and MS. Waste Manag. 2014, 34, 174–179. [Google Scholar] [CrossRef]
- Pulka, J.; Wiśniewski, D.; Gołaszewski, J.; Białowiec, A. Is the biochar produced from sewage sludge a good quality solid fuel? Arch. Environ. Prot. 2016, 42, 125–134. [Google Scholar] [CrossRef]
- Poudel, J.; Ohm, T.I.; Lee, S.H.; Oh, S.C. A study on torrefaction of sewage sludge to enhance solid fuel qualities. Waste Manag. 2015, 40, 112–118. [Google Scholar] [CrossRef]
- Poudel, J.; Karki, S.; Gu, J.H.; Lim, Y.; Oh, S.C. Effect of Co-Torrefaction on the Properties of Sewage Sludge and Waste Wood to Enhance Solid Fuel Qualities. J. Residuals Sci. Technol. 2017, 14, 23–36. [Google Scholar] [CrossRef]
- Atienza-Martínez, M.; Fonts, I.; ábrego, J.; Ceamanos, J.; Gea, G. Sewage sludge torrefaction in a fluidized bed reactor. Chem. Eng. J. 2013, 222, 534–545. [Google Scholar] [CrossRef]
- Atienza-Martínez, M.; Mastral, J.F.; Ábrego, J.; Ceamanos, J.; Gea, G. Sewage sludge torrefaction in an auger reactor. Energy Fuels 2015, 29, 160–170. [Google Scholar] [CrossRef]
- Huang, M.; Chang, C.C.; Yuan, M.H.; Chang, C.Y.; Wu, C.H.; Shie, J.L.; Chen, Y.H.; Chen, Y.H.; Ho, C.; Chang, W.R.; et al. Production of torrefied solid bio-fuel from pulp industry waste. Energies 2017, 10, 910. [Google Scholar] [CrossRef]
- Huang, Y.-F.; Sung, H.-T.; Chiueh, P.-T.; Lo, S.-L. Microwave torrefaction of sewage sludge and leucaena. J. Taiwan Inst. Chem. Eng. 2017, 70, 236–243. [Google Scholar] [CrossRef]
- Huang, Y.-F.; Sung, H.-T.; Chiueh, P.-T.; Lo, S.-L. Co-torrefaction of sewage sludge and leucaena by using microwave heating. Energy 2016, 116, 1–7. [Google Scholar] [CrossRef]
- Huang, Y.W.; Chen, M.Q.; Luo, H.F. Nonisothermal torrefaction kinetics of sewage sludge using the simplified distributed activation energy model. Chem. Eng. J. 2016, 298, 154–161. [Google Scholar] [CrossRef]
- Do, T.X.; Lim, Y.; Cho, H.; Shim, J.; Yoo, J.; Rho, K.; Choi, S.-G.; Park, B.-Y. Process modeling and energy consumption of fry-drying and torrefaction of organic solid waste. Dry Technol. 2017, 35, 754–765. [Google Scholar] [CrossRef]
- Pawlak-Kruczek, H.; Krochmalny, K.K.; Wnukowski, M.; Niedzwiecki, L. Slow pyrolysis of the sewage sludge with additives: Calcium oxide and lignite. J. Energy Resour. Technol. 2018, 140. [Google Scholar] [CrossRef]
- Pečkytė, J.; Baltrėnaitė, E. Assessment of heavy metals leaching from (bio) char obtained from industrial sewage sludge. Environ. Prot. Eng. 2015, 7, 399–406. [Google Scholar] [CrossRef]
- Council Decision. Council of the European Union 2003/33/EC—Council Decision establishing criteria and procedures for the acceptance of waste at landfills pursuant to Article 16 of and Annex II to Directive 1999/31/EC. Off. J. Eur. Communities 2003, L 11/27, 27–49. [Google Scholar]
- Wang, N.Y.; Shih, C.H.; Chiueh, P.T.; Huang, Y.F. Environmental effects of sewage sludge carbonization and other treatment alternatives. Energies 2013, 6, 871–883. [Google Scholar] [CrossRef]
- Lasek, J.A.; Kopczyński, M.; Janusz, M.; Iluk, A.; Zuwała, J. Combustion properties of torrefied biomass obtained from flue gas-enhanced reactor. Energy 2017, 119, 362–368. [Google Scholar] [CrossRef]
- Joshi, Y.; Di Marcello, M.; Krishnamurthy, E.; De Jong, W. Packed-Bed Torrefaction of Bagasse under Inert and Oxygenated Atmospheres. Energy Fuels 2015, 29, 5078–5087. [Google Scholar] [CrossRef]
- Uemura, Y.; Saadon, S.; Osman, N.; Mansor, N.; Tanoue, K.I. Torrefaction of oil palm kernel shell in the presence of oxygen and carbon dioxide. Fuel 2015, 144, 171–179. [Google Scholar] [CrossRef]
- Uemura, Y.; Omar, W.; Othman, N.A.; Yusup, S.; Tsutsui, T. Torrefaction of oil palm EFB in the presence of oxygen. Fuel 2013, 103, 156–160. [Google Scholar] [CrossRef]
- Chen, W.H.; Zhuang, Y.Q.; Liu, S.H.; Juang, T.T.; Tsai, C.M. Product characteristics from the torrefaction of oil palm fiber pellets in inert and oxidative atmospheres. Bioresour. Technol. 2016, 199, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.H.; Lu, K.M.; Liu, S.H.; Tsai, C.M.; Lee, W.J.; Lin, T.C. Biomass torrefaction characteristics in inert and oxidative atmospheres at various superficial velocities. Bioresour. Technol. 2013, 146, 152–160. [Google Scholar] [CrossRef]
- Lu, K.M.; Lee, W.J.; Chen, W.H.; Liu, S.H.; Lin, T.C. Torrefaction and low temperature carbonization of oil palm fiber and eucalyptus in nitrogen and air atmospheres. Bioresour. Technol. 2012, 123, 98–105. [Google Scholar] [CrossRef]
- Pawlak-Kruczek, H.; Wnukowski, M.; Krochmalny, K.; Kowal, M.; Baranowski, M.; Zgóra, J.; Czerep, M.; Ostrycharczyk, M.; Niedzwiecki, L. The staged thermal conversion of sewage sludge in presence of oxygen. In The Clearwater Clean Coal Conference: Proceedings of the 43rd International Technical Conference on Clean Energy; Sakkestad, B.A., Ed.; Published by Coal Technologies Associates Post Office Box 1130 Louisa, VA 23093 USA: Clearwater, FL, USA, 2018. [Google Scholar]
- Reed, T.B.; Das, A. Handbook of Biomass Downdraft Gasifier Engine Systems; Biomass Energy Foundation: Golden, CO, USA, 1988. [Google Scholar]
- Devi, L.; Ptasinski, K.J.; Janssen, F.J.J.G. A review of the primary measures for tar elimination in biomass gasification processes. Biomass Bioenergy 2002, 24, 125–140. [Google Scholar] [CrossRef]
- Royal Society of Chemistry ChemSpider | Search and Share Chemistry. Available online: http://www.chemspider.com/ (accessed on 23 November 2018).
- Asadullah, M. Biomass gasification gas cleaning for downstream applications: A comparative critical review. Renew. Sustain. Energy Rev. 2014, 40, 118–132. [Google Scholar] [CrossRef]
- Hasler, P.; Nussbaumer, T. Gas cleaning for IC engine applications from fixed bed biomass gasification. Biomass Bioenergy 1999, 16, 385–395. [Google Scholar] [CrossRef]
- Striūgas, N.; Valinčius, V.; Pedišius, N.; Poškas, R.; Zakarauskas, K. Investigation of sewage sludge treatment using air plasma assisted gasification. Waste Manag. 2017. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.D.; Baker, E.G.; Mudge, L.K. Environmental design considerations for thermochemical biomass energy. Biomass 1986, 11, 255–270. [Google Scholar] [CrossRef]
- Bui, T.; Loof, R.; Bhattacharya, S.C. Multi-stage reactor for thermal gasification of wood. Energy 1994, 19, 397–404. [Google Scholar] [CrossRef]
- Werle, S. Sewage sludge gasification: Theoretical and experimental investigation. Environ. Prot. Eng. 2013, 39, 25–32. [Google Scholar] [CrossRef]
- Schweitzer, D.; Gredinger, A.; Schmid, M.; Waizmann, G.; Beirow, M.; Spörl, R.; Scheffknecht, G. Steam gasification of wood pellets, sewage sludge and manure: Gasification performance and concentration of impurities. Biomass Bioenergy 2018, 111, 308–319. [Google Scholar] [CrossRef]
- Werle, S. Numerical analysis of the combustible properties of sewage sludge gasification gas. Chem. Eng. Trans. 2015, 1021–1026. [Google Scholar] [CrossRef]
- Werle, S.; Dudziak, M. Evaluation of the possibility of the sewage sludge gasification gas use as a fuel. Ecol. Chem. Eng. S 2016, 23, 229–236. [Google Scholar] [CrossRef]
- Szwaja, S.; Kovacs, V.B.; Bereczky, A.; Penninger, A. Sewage sludge producer gas enriched with methane as a fuel to a spark ignited engine. Fuel Process. Technol. 2013, 110, 160–166. [Google Scholar] [CrossRef]
- Werle, S. Impact of feedstock properties and operating conditions on sewage sludge gasification in a fixed bed gasifier. Waste Manag. Res. 2014, 32, 954–960. [Google Scholar] [CrossRef]
- Calvo, L.F.; García, A.I.; Otero, M. An experimental investigation of sewage sludge gasification in a fluidised bed reactor. Sci. World J. 2013. [Google Scholar] [CrossRef] [PubMed]
- Akkache, S.; Hernández, A.B.; Teixeira, G.; Gelix, F.; Roche, N.; Ferrasse, J.H. Co-gasification of wastewater sludge and different feedstock: Feasibility study. Biomass Bioenergy 2016, 89, 201–209. [Google Scholar] [CrossRef]
- Reed, G.P.; Paterson, N.P.; Zhuo, Y.; Dugwell, D.R.; Kandiyoti, R. Trace element distribution in sewage sludge gasification: Source and temperature effects. Energy Fuels 2005, 19, 298–304. [Google Scholar] [CrossRef]
- Judex, J.W.; Gaiffi, M.; Burgbacher, H.C. Gasification of dried sewage sludge: Status of the demonstration and the pilot plant. Waste Manag. 2012, 32, 719–723. [Google Scholar] [CrossRef] [PubMed]
- Kokalj, F.; Arbiter, B.; Samec, N. Sewage sludge gasification as an alternative energy storage model. Energy Convers. Manag. 2017, 149, 738–747. [Google Scholar] [CrossRef]
- Huang, Y.W.; Chen, M.Q.; Li, Q.H.; Xing, W. Hydrogen-rich syngas produced from co-gasification of wet sewage sludge and torrefied biomass in self-generated steam agent. Energy 2018, 161, 202–213. [Google Scholar] [CrossRef]
- Prins, M.J.; Ptasinski, K.J.; Janssen, F.J.J.G. More efficient biomass gasification via torrefaction. Energy 2006, 31, 3458–3470. [Google Scholar] [CrossRef]
- Dudyński, M.; Van Dyk, J.C.; Kwiatkowski, K.; Sosnowska, M. Biomass gasification: Influence of torrefaction on syngas production and tar formation. Fuel Process. Technol. 2015, 131, 203–212. [Google Scholar] [CrossRef]
- Tic, W.; Guziałowska-Tic, J.; Pawlak-Kruczek, H.; Woźnikowski, E.; Zadorożny, A.; Niedźwiecki, Ł.; Wnukowski, M.; Krochmalny, K.; Czerep, M.; Ostrycharczyk, M.; et al. Novel Concept of an Installation for Sustainable Thermal Utilization of Sewage Sludge. Energies 2018, 11, 748. [Google Scholar] [CrossRef]
- Pawlak-Kruczek, H.; Krochmalny, K.; Mościcki, K.; Zgóra, J.; Czerep, M.; Ostrycharczyk, M.; Niedźwiecki, Ł. Torrefaction of Various Types of Biomass in Laboratory Scale, Batch-Wise Isothermal Rotary Reactor and Pilot Scale, Continuous Multi-Stage Tape Reactor. Eng. Prot. Environ. 2017, 20, 457–472. [Google Scholar] [CrossRef]
- Weber, K.; Heuer, S.; Quicker, P.; Li, T.; Løvås, T.; Scherer, V. An Alternative Approach for the Estimation of Biochar Yields. Energy Fuels 2018. [Google Scholar] [CrossRef]
- Nhuchhen, D.; Basu, P.; Acharya, B. A Comprehensive Review on Biomass Torrefaction. Int. J. Renew. Energy Biofuels 2014, 2014, 506376. [Google Scholar] [CrossRef]
- Esseyin, A.E.; Steele, P.H.; Pittman, C.U., Jr. Current trends in the production and applications Torrefied Wood/Biomass—A review. Bioresources 2015, 10, 8812–8858. [Google Scholar] [CrossRef]
- Chew, J.J.; Doshi, V. Recent advances in biomass pretreatment—Torrefaction fundamentals and technology. Renew. Sustain. Energy Rev. 2011, 15, 4212–4222. [Google Scholar] [CrossRef]
- Saeed, M.A.; Andrews, G.E.; Phylaktou, H.N.; Gibbs, B.M.; Niedzwiecki, L.; Walton, R. Explosion and Flame Propagation Properties of Coarse Wood: Raw and Torrefied. In Proceedings of the Eighth International Seminar on Fire & Explosion Hazards (ISFEH8); Choa, J., Molkov, V., Sunderland, P., Tamanini, F., Torero, J., Eds.; USTC Press: Hefei, China, 2016; pp. 579–588. [Google Scholar]
| Test | Symbol | Value | Unit | Standard Procedure | |
|---|---|---|---|---|---|
| Raw | Torrefied | ||||
| Moisture content 1 | MC | 26.2 | 2.30 | % | EN ISO 18134-2:2015 |
| Volatile matter content | VM d | 58.1 | 44.40 | % | EN 15148:2009 |
| Ash content | A d | 32.5 | 45.67 | % | EN ISO 1822:2015 |
| Higher heating value | HHV | 15,700 | 10,300 | kJ/kg | EN 14918:2009 |
| Lower heating value 2 | LHV | 10,939 | 10,006 | kJ/kg | EN 14918:2009 |
| Carbon content | C d | 27.89 | 15.83 | % | EN ISO 16948:2015 |
| Hydrogen content | H d | 6.67 | 2.92 | % | EN ISO 16948:2015 |
| Nitrogen content | N d | 4.36 | 4.18 | % | EN ISO 16948:2015 |
| Sulfur content | S d | 0.29 | 0.27 | % | EN ISO 16994:2016 |
| Oxygen content | O d | 28.80 | 31.13 | % | EN ISO 16993:2015 |
| Characteristic Temperature | Raw Sewage Sludge | Torrefied Sewage Sludge | Unit |
|---|---|---|---|
| Deformation temperature (DT) | 1000 | 1010 | °C |
| Sphere temperature (ST) | 1020 | 1050 | °C |
| Hemisphere temperature (HT) | 1110 | 1110 | °C |
| Flow temperature (FT) | 1210 | 1210 | °C |
| Compound | Form. | Boil. Point | Melt. Point | Avg. Mass | Relative Area of the Peak 2 | |||
|---|---|---|---|---|---|---|---|---|
| Raw | Torrefied | |||||||
| Value | SD 1 | Value | SD 1 | |||||
| °C | °C | Da | % | % | % | % | ||
| Toluene | C7H8 | 111 | −95 | 92.14 | 21.41 | 0.64 | 32.60 | 0.39 |
| Propiononitrile | C3H5N | 97 | −92 | 55.08 3 | 0.14 | 0.03 | 1.11 | 0.02 |
| 2-Methylpyridine | C6H7N | 128 | −70 | 93.12 | 1.40 | 0.09 | 1.64 | 0.02 |
| o-Xylene | C8H10 | 140 | −48 | 106.16 | 0.10 | 0.03 | 1.40 | 0.02 |
| 3-Methyl-1H-pyrrole | C5H7N | 144 | −48 | 81.12 | 0.12 | 0.01 | 1.40 | 0.03 |
| Pyridine | C5H5N | 115 | −42 | 79.10 | 6.23 | 0.21 | 3.12 | 0.03 |
| Thiophene | C4H4S | 84 | −38 | 84.14 | 2.01 | 0.11 | 0.26 | 0.01 |
| 2-Methyl-1H-pyrrole | C5H7N | 147 | −36 | 81.12 | 0.51 | 0.01 | 0.69 | 0.02 |
| Styrene | C8H8 | 145 | −31 | 104.15 | 4.54 | 0.26 | 7.26 | 0.05 |
| 3-Methylbenzonitrile | C8H7N | 210 | −25 | 117.15 | 0.10 | 0.01 | - 5 | - |
| Benzeneacetonitrile | C8H7N | 234 | −24 | 117.15 | 0.23 | 0.01 | 0.30 | 0.01 |
| 1H-Pyrrole | C4H5N | 130 | −23 | 67.09 3 | 2.71 | 0.11 | 3.84 | 0.02 |
| 2-Methylnaphthalene | C11H10 | 242 | −22 | 142.20 | 2.24 | 0.09 | 1.93 | 0.03 |
| 1-Methylnaphthalene | C11H10 | 242 | −22 | 142.20 | 1.41 | 0.07 | 1.63 | 0.04 |
| 1-Benzofuran | C8H6O | 174 | −18 | 118.13 | 0.40 | 0.02 | 0.34 | 0.01 |
| Quinoline | C9H7N | 237 | −15 | 129.16 | 3.32 | 0.07 | 0.91 | 0.01 |
| Benzonitrile | C7H5N | 191 | −13 | 103.12 | 5.32 | 0.15 | 2.86 | 0.03 |
| 2-Methylbenzonitrile | C8H7N | 205 | −13 | 117.14 | 0.31 | 0.02 | 0.11 | 0.01 |
| 1H-Indene | C9H8 | 181 | −2 | 116.16 | 4.83 | 0.21 | 2.68 | 0.02 |
| 2-Methylquinoline | C10H9N | 247 | −2 | 143.18 | 0.21 | 0.01 | 0.13 | 0.01 |
| m-Cresol | C7H8O | 203 | 11 | 108.12 | - | - | 0.40 | 0.07 |
| Acetic acid | C2H4O2 | 118 | 17 | 60.05 3 | 0.23 | 0.05 | 3.51 | 0.01 |
| 3-Methylpyridine | C6H7N | 144 | 18 | 93.13 | 0.51 | 0.03 | 0.61 | 0.01 |
| Isoquinoline | C9H7N | 242 | 26 | 129.16 | 0.80 | 0.01 | 0.12 | 0.01 |
| 4-Methylbenzonitrile | C8H7N | 218 | 28 | 117.15 | 0.22 | 0.01 | 0.13 | 0.01 |
| Phenol | C6H6O | 182 | 41 | 94.11 | 1.81 | 0.07 | 5.56 | 0.12 |
| p-Cresol | C7H8O | 202 | 41 | 108.14 | 0.43 | 0.02 | 3.88 | 0.07 |
| 1H-Indole | C8H7N | 254 | 53 | 117.15 | 4.03 | 0.15 | 2.68 | 0.04 |
| 3-Pyridinamine | C5H6N2 | 250 | 62 | 94.11 | 0.21 | 0.03 | 1.16 | 0.01 |
| 2-Naphthonitrile | C11H7N | 157 | 67 | 153.18 | 0.72 | 0.01 | - | - |
| Naphthalene | C10H8 | 218 | 81 | 128.17 | 22.00 | 0.60 | 8.19 | 0.12 |
| Acenaphthylene | C12H8 | 280 | 93 | 152.19 | 4.21 | 0.09 | 1.05 | 0.01 |
| Phenanthrene | C14H10 | 338 | 100 | 178.23 | 3.13 | 0.01 | 0.45 | 0.03 |
| 9H-Fluorene | C13H10 | 295 | 115 | 166.22 | 0.70 | 0.02 | - | - |
| 5,5-Dimethyl-2,4-imidazolidinedione | C5H8N2 | n.a. 4 | 175 | 128.13 | 0.51 | 0.05 | 4.65 | 0.13 |
| 3,3′-Sulfanediyldipropanenitrile | C6H8N2 | n.a. | n.a. | 140.21 | 2.41 | 0.16 | 0.62 | 0.04 |
| 2-Benzothiophene | C8H6S | n.a. | n.a. | 134.20 | 1.14 | 0.04 | 0.20 | 0.01 |
| Compound | Form. | Boil. Point | Melt. Point | Avg. Mass | Concentration in the Producer Gas | |||
|---|---|---|---|---|---|---|---|---|
| Raw | Torrefied | |||||||
| Value | SD | Value | SD | |||||
| °C | °C | Da | mg/m3 | mg/m3 | mg/m3 | mg/m3 | ||
| Toluene | C7H8 | 111 | −95 | 92.14 | 2229.1 | 91.4 | 3211.0 | 61.0 |
| Pyridine | C5H5N | 115 | −42 | 79.10 | 1170.0 | 37.4 | 453.8 | 3.7 |
| 1-Methylnaphthalene | C11H10 | 242 | −22 | 142.20 | 225.6 | 7.7 | 149.4 | 3.5 |
| Benzonitrile | C7H5N | 191 | −13 | 103.12 | 688.2 | 18.2 | 243.0 | 3.5 |
| 1H-Indene | C9H8 | 181 | −2 | 116.16 | 574.7 | 25.3 | 243.6 | 0.9 |
| Acetic acid | C2H4O2 | 118 | 17 | 60.05 1 | 752.3 | 32.0 | 1680.3 | 5.2 |
| Phenol | C6H6O | 182 | 41 | 94.11 | 521.7 | 10.7 | 531.0 | 11.8 |
| p-Cresol | C7H8O | 202 | 41 | 108.14 | 198.4 | 3.7 | 444.9 | 7.9 |
| Naphthalene | C10H8 | 218 | 81 | 128.17 | 1367.6 | 47.7 | 384.7 | 8.5 |
| Acenaphthylene | C12H8 | 280 | 93 | 152.19 | 263.9 | 6.4 | 62.0 | 1.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pawlak-Kruczek, H.; Wnukowski, M.; Niedzwiecki, L.; Czerep, M.; Kowal, M.; Krochmalny, K.; Zgóra, J.; Ostrycharczyk, M.; Baranowski, M.; Tic, W.J.; et al. Torrefaction as a Valorization Method Used Prior to the Gasification of Sewage Sludge. Energies 2019, 12, 175. https://doi.org/10.3390/en12010175
Pawlak-Kruczek H, Wnukowski M, Niedzwiecki L, Czerep M, Kowal M, Krochmalny K, Zgóra J, Ostrycharczyk M, Baranowski M, Tic WJ, et al. Torrefaction as a Valorization Method Used Prior to the Gasification of Sewage Sludge. Energies. 2019; 12(1):175. https://doi.org/10.3390/en12010175
Chicago/Turabian StylePawlak-Kruczek, Halina, Mateusz Wnukowski, Lukasz Niedzwiecki, Michał Czerep, Mateusz Kowal, Krystian Krochmalny, Jacek Zgóra, Michał Ostrycharczyk, Marcin Baranowski, Wilhelm Jan Tic, and et al. 2019. "Torrefaction as a Valorization Method Used Prior to the Gasification of Sewage Sludge" Energies 12, no. 1: 175. https://doi.org/10.3390/en12010175
APA StylePawlak-Kruczek, H., Wnukowski, M., Niedzwiecki, L., Czerep, M., Kowal, M., Krochmalny, K., Zgóra, J., Ostrycharczyk, M., Baranowski, M., Tic, W. J., & Guziałowska-Tic, J. (2019). Torrefaction as a Valorization Method Used Prior to the Gasification of Sewage Sludge. Energies, 12(1), 175. https://doi.org/10.3390/en12010175







