2.2. Box-Behnken Experimental Design
An increase of the availability of biomass material for cellulolytic enzymes is the aim of the research on the optimization of the alkaline pre-treatment. Energetic willow was used as the raw material during the optimization step, due to low soil requirements, rapid growth and both popularity and representativeness in the group of the energetic plants. Experiments were carried out according to the Box-Behnken design for four variables (
Table 4 and
Table 5). Based on the preliminary investigations and previous literature data, the following boundary conditions were adopted for the investigated process parameters: catalyst concentration 1% and 7%, temperature 30 and 80 °C, treatment time 0.5 and 7 h, granulation 0.25 and 4 mm. The above-mentioned parameters were assigned as independent variables from X
1 to X
4, according to the values presented in
Table 4.
Glucose efficiency (G
eff), calculated from Equation (2), was assumed as the model output parameter. The granulation values of 0.25, 2 and 4 mm were selected due to the availability of grinding sieve sizes, deliberately and consciously deciding to deviate from the assumptions of the Box-Behnken experimental design. The values of glucose concentration and the sum of reducing sugars and disaccharides are presented in
Table 5. As a result of the saccharification carried out by means of enzymatic hydrolysis, the following sugars, except glucose, were identified in the enzymatic hydrolyzate: cellobiose, xylose, galactose, mannose and arabinose.
The composition of the biomass, the degree of fragmentation and the pre-treatment parameters (i.e., temperature, type and concentration of the catalyst, processing time) affect the obtained yield of the reducing sugars [
43]. Determining the optimal pre-treatment conditions with respect to the yield of sugars is an important issue in the perspective of the cost-effectiveness of lignocellulosic biomass processing. In order to select the optimal conditions of the alkaline pre-treatment, based on the performed experiments according to the data from
Table 4, the coefficients in the quadratic Equation (1) were determined. The Equation (1) was used for the estimation of the surface response area.
where β
i—coefficient in the quadratic equation.
In order to obtain a useful form of the mathematical model, given in Equation (1), the least significant variables were rejected in the consecutive iterative steps, i.e., for values of P above 0.05. The calculations revealed that the glucose yield (G
eff) can be estimated with a good accuracy (R
2 = 0.798) using the Equation (2):
Statistical parameters for the determined coefficients are presented in
Table 6.
The results of calculations, leading to the form of the model given by Equation (2) indicated that the least influence on the obtained glucose yield is described by: in the first power the pre-treatment time and the interaction of time and temperature, granulation and temperature, catalyst concentration and temperature as well as catalyst concentration and granulation; in the second power the time and granulation. The obtained experimental results are similar with the published results [
44] presenting the parameters significantly affecting the results of alkaline pre-treatment with NaOH. However, the investigations were conducted for a different lignocellulosic material (rice straw).
2.3. The Effect of Alkaline Pre-Treatment on the Composition of Energetic Willow Hydrolyzates
In order to determine the effect of the alkaline pre-treatment on the change of biomass composition, the weight loss (biomass recovery), cellulose, hemicellulose and lignin content, lignin removal and cellulose and hemicellulose recovery rates were determined in the residues after the treatment (
Table 7). It was found that an increase both of the catalyst concentration and of the temperature favors the weight loss of the processed material. The highest weight loss (40.19%) was observed for the process carried out at 80 °C and for NaOH concentration of 7% (
w/
v). The weight loss corresponds with the high removal rates of lignin (50.1%) and hemicellulose (73.2%).
The results of investigations indicate that it is not possible to completely remove lignin from the plant material as a result of alkaline treatment, even if high temperatures and high concentrations of alkaline reagents are used. Aggressive alkaline treatment of corncobs (160 °C, 10% NaOH
v/
v) removes approximately 85% of lignin [
45]. The inability to completely remove lignin from the processed material may contribute to a reduced efficiency of enzymatic hydrolysis. This may be due to limited availability of sugar polymers for enzymes and the possibility of binding of active enzyme centers by lignin, which may lead to enzyme deactivation.
The results of investigations (
Table 5 and
Table 7) indicate that an important factor influencing the result of the alkaline pre-treatment is the degree of fragmentation (granulation) of the processed material. It was found that with an increase of the degree of fragmentation, the weight loss of the processed biomass increases, despite the lower temperature and shorter treatment time (compare experiments 9 and 23,
Table 7).
It was observed that for mild alkaline pre-treatment, i.e., NaOH concentration below 4% and temperature below 40 °C, the loss of hemicellulose and lignin is insignificant, and the change in the chemical composition of the material is probably mainly due to dissolution of the extractives in solution. This is evidenced by the fact that in the case of energetic willow, after the alkaline pre-treatment, only small amounts of extractable compounds remain, i.e., non-structural biomass components. The above conclusion is consistent with the literature data suggesting the loss of biomass during the alkaline pre-treatment results mainly from the removal of lignin, extractives and hemicelluloses [
45,
46]. The increase in temperature and the increase in the concentration of the catalyst causes the increase in the removal of lignin and hemicelluloses [
47]. As on effect of alkaline treatment, hemicellulose passes into solution in the form of both simple sugars (pentoses, hexoses) and sugar oligomers [
48]. As a result, the total yield of simple sugars, which is used as a carbon source for fermentation leading to the production of biofuels, is reduced. Alkaline pre-treatment also breaks down ester bonds between lignin and hemicellulose, which results in an increase of the porosity and specific surface area of the material and consequently increases the availability of cellulose structures for cellulolytic enzymes [
49].
2.4. Enzymatic Hydrolysis of Pre-Treated Energetic Willow
After completion of the alkaline pre-treatment (delignification), the pre-treated biomass samples are further treated with biochemical methods. Enzymatic hydrolysis is intended to release monosugars from crystalline cellulose and from the remains of hemicellulose. The efficiency of this step is crucial for the conversion of the biomass to the desired products because monosugars are the source of carbon for fermentative microorganisms [
50]. The conversion of cellulose and hemicellulose is catalyzed by glucosidase and hemicellulases. Cellulose hydrolysis can be carried out using enzymes such as endoglucanases, which hydrolyse internal β-1,4-glycosidic bond, exoglucanases removing the end of a glucose chain monomers and dimers, and glycosidases, which hydrolyse dimers of glucose–cellobiose, considered as an inhibitory sugar for cellulolytic enzymes [
51]. Degradation of cellulose is complicated if the microfibers are stabilized by internal and external hydrogen bonds, and they are surrounded by hemicellulose polysaccharides, i.e., mannans and xylans. Cellulase plays an important role as it catalyzes the degradation of cellulose into fermentable sugars. Cellulose hydrolysis begins with the adsorption of cellulase on the surface of the cellulose, followed by the degradation of cellulose to sugars and cellulase desorption from the surface of the biomass. Hemicellulose structure can be broken by various enzymes. Xylan is hydrolyzed under the action of endoxylanase and β
-xylosidase, which cause its degradation to xylooligosaccharides. Enzymes such as α
-glucuronidase, α-arabinofuranoside and acetyloxyl esterase cleave side groups and heterosilyl chains [
52,
53]. Glucomannan hydrolyzates are digested by β
-mannanase and β
-mannosidase. In addition to the quality of the hydrolytic enzyme, digestibility of cellulose and hemicellulose is influenced by such factors as pH, temperature, time, and porosity of the carrier, degree of crystallization and consistency of the cellulose [
54].
The enzymatic hydrolyzates may be potentially used in fermentation processes, as a source of carbon, therefore it is crucial to remove the enzymes from the hydrolyzates after the process. This is why the enzyme immobilization was applied. Enzymes immobilized on diatomite may be separated by centrifugation following the hydrolysis [
55]. Physical adsorption of enzymes requires soaking the diatomite in the enzyme solution [
56]. Depending on the pH of the solution and the isoelectric point, the equilibrium of the immobilization may be affected. Therefore, to avoid non-linear changes, buffers such as Mc’Ilvainae or phosphate buffer must be used.
The obtained values of glucose mass, sum of reducing sugars and glucose efficiency for the raw material subjected directly to enzymatic hydrolysis were respectively 130 mg/g biomass, 210.5 mg/g biomass and 0.12. The above given results of enzymatic hydrolysis were taken as a reference for further investigations.
The results indicate that for temperature below 55 °C and catalyst concentration below 1%, enzymatic hydrolysis of the raw material results in higher yields than the hydrolysis of the material after the alkaline treatment. This is probably due to the removal of non-structural sugars during the alkaline treatment in the above-mentioned conditions. With an increase of the concentration of alkaline catalyst above 4% and temperature above 55 °C, the concentration of sugars in the hydrolyzates after enzymatic hydrolysis increases. The highest value of glucose efficiency (0.79) from energetic willow was obtained for the catalyst concentration of 7%, temperature 55 °C and pre-treatment time 7 h. Blank hydrolysis test (i.e., without enzymes, with diatomite only) did not reveal the presence of simple sugars in the liquid.
The effect of the enzyme immobilization on the glucose efficiency was investigated. For this purpose, the alkaline pre-treated energetic willow (granulation 2 mm, catalyst concentration 4%, temperature 55 °C, treatment time 3.75 h) was subjected to enzymatic hydrolysis with free enzymes and with enzymes immobilized on diatomaceous earth. The yield of glucose after hydrolysis with free and immobilized enzymes was 0.67 and 0.46, respectively. Despite the lower glucose efficiency, it was decided to apply the method with immobilization, because it allows a quick separation of enzymes from hydrolyzates and enzymes recovery. The immobilization of cellulolytic enzymes on diatomaceous earth and their application on lignocellulosic materials is not widely described in the literature. These studies are a prelude to further work aimed at material processing and enzymatic hydrolysis in a semi-continuous or continuous manner. The possibility of reusing immobilized cellulolytic enzymes on diatomite for saccharification processes of lignocellulosic biomass may be an object of further experiments. In addition, it is believed that some components resulting from alkaline pre-treatment may be adsorbed on the surface of diatomaceous earth. The group of these components include, among others, phenolic derivatives such as furfural and 5-HMF. These compounds may inhibit the subsequent fermentation to biofules carried out on the obtained hydrolyzates. It may be a subject of further research in this field.
2.5. Influence of Alkaline Pre-Treatment Parameters on the Glucose Efficiency
Based on the regression Equation (2),
Figure 1,
Figure 2,
Figure 3 and
Figure 4 are given, presenting the response of the glucose efficiency as a function of independent variables of the alkaline pre-treatment. Based on the analysis of the presented plots, the optimal process parameters for the alkaline pre-treatment were then proposed, taking into account the glucose efficiency and costs of the process.
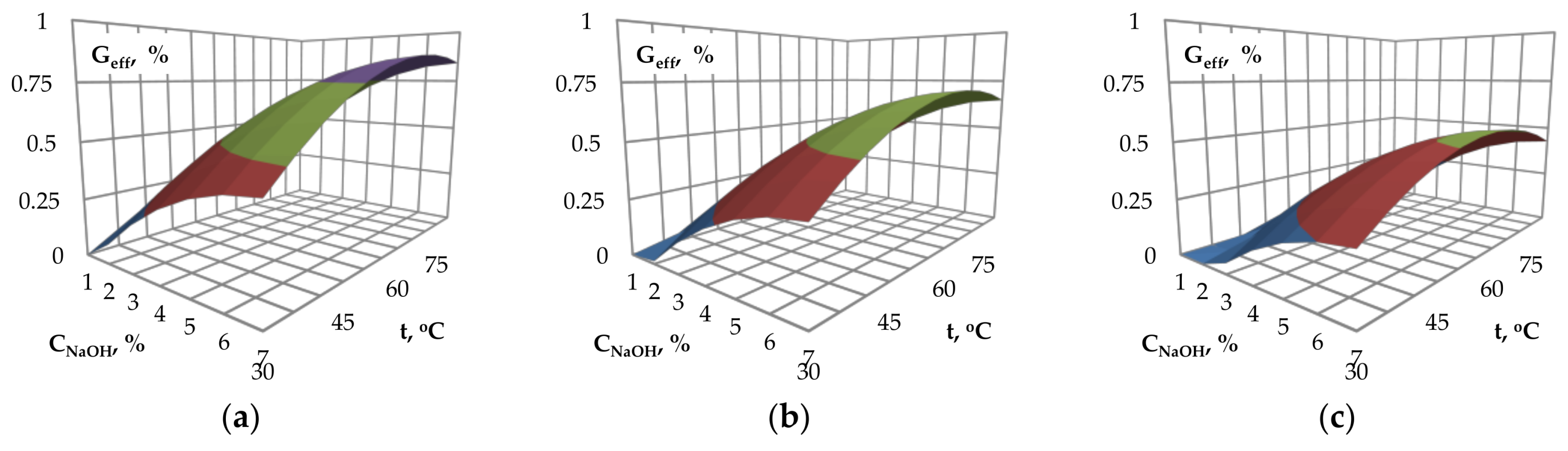
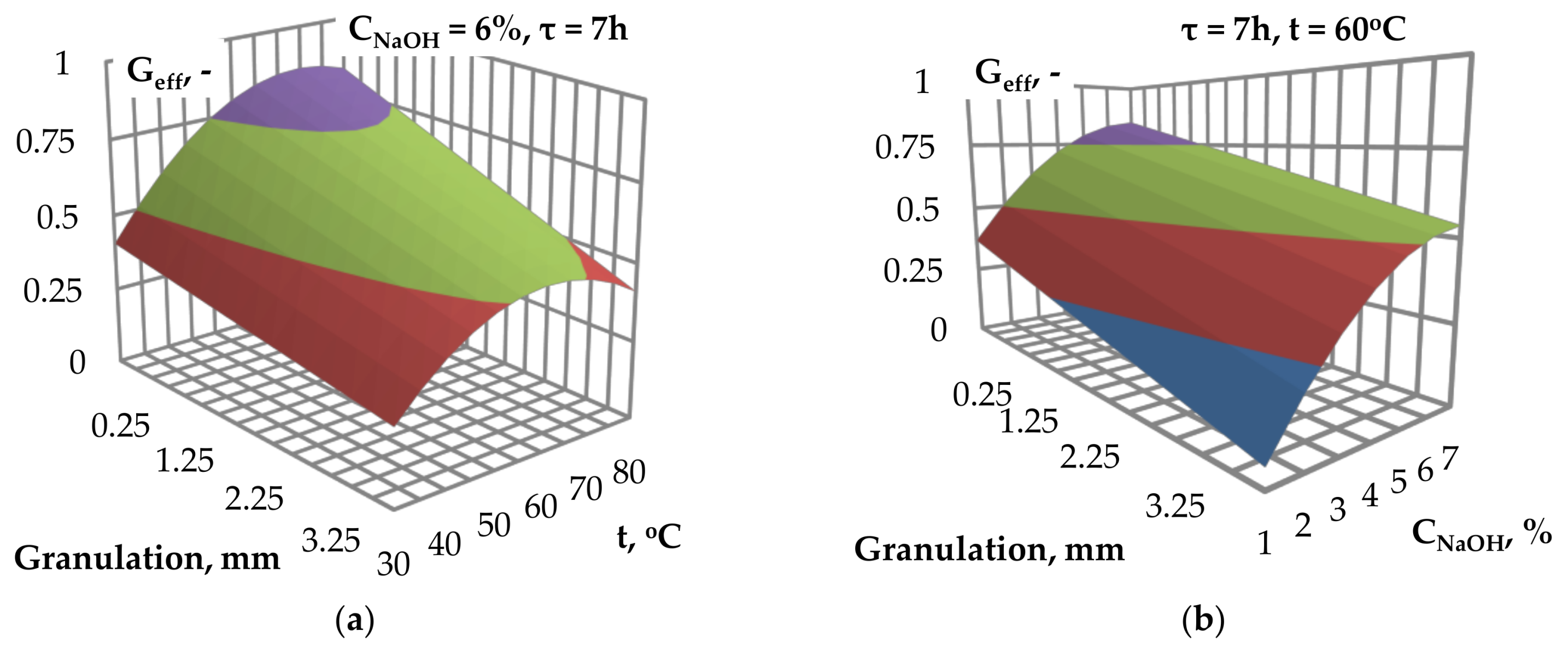
Figure 1a–c presents the values of glucose efficiency as a function of NaOH catalyst concentration and temperature. The investigated process parameters influence the glucose efficiency and optimal values of process parameters may be selected. An increase in the catalyst concentration from 1% to approximately 6% results in an increase of glucose yield. For NaOH concentration above 6%, no further increase of glucose yield was observed.
The effect of temperature on the glucose yield is similar to the effect of catalyst concentration. An increase of the temperature to approximately 75 °C, 65 °C and 55 °C, respectively for granulation of 0.25, 2 and 4 mm, results in an increase of glucose efficiency. The above-mentioned temperature values are followed by stabilization (
Figure 1a) or slight reduction (
Figure 1b,c) of the glucose efficiency.
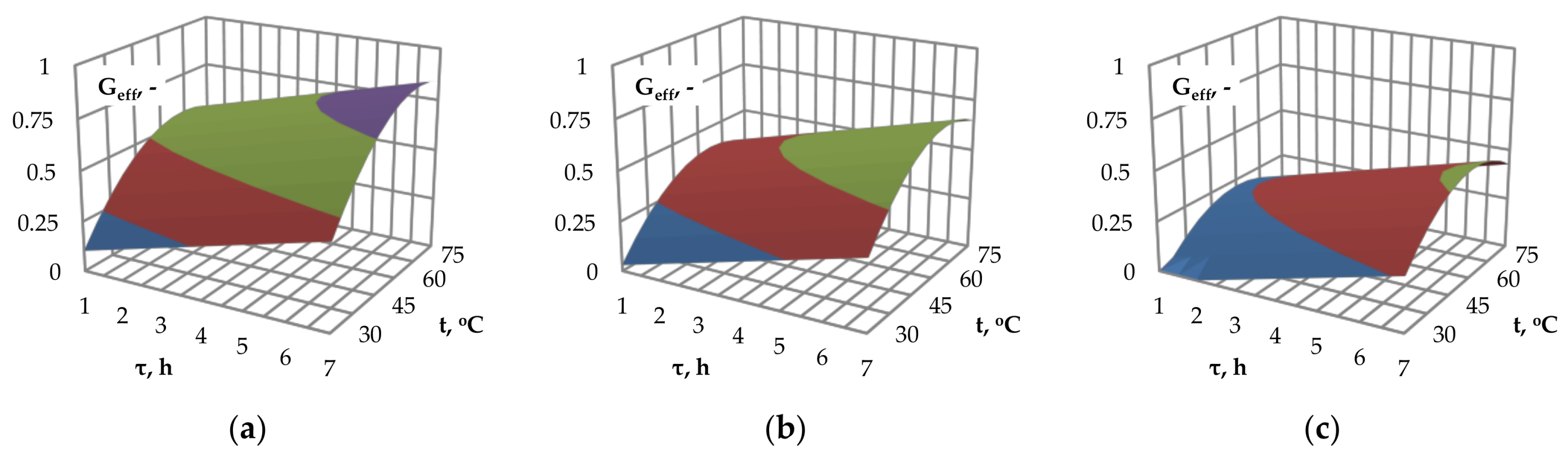
Figure 2a–c show the influence of the pre-treatment time and temperature on the glucose efficiency. In the investigated range of alkaline pre-treatment time, an increase in the processing time results in an increase of glucose efficiency. This can be explained by the lack of excessive degradation of the material during processing in the investigated temperature range.
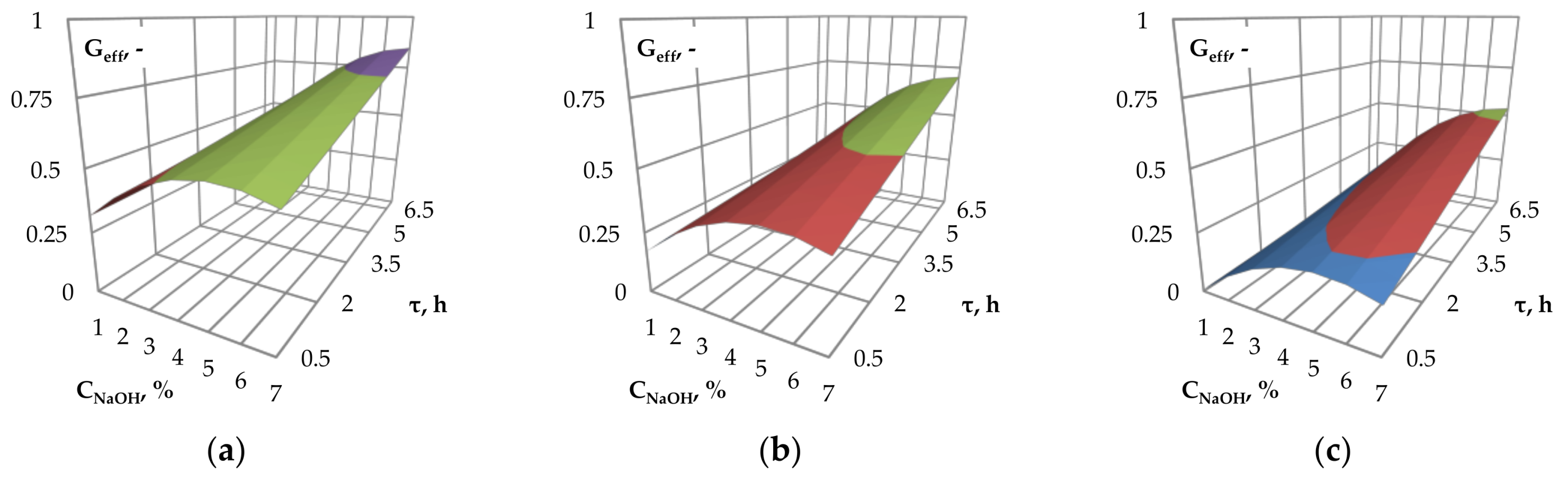
An influence of the catalyst concentration and the pre-treatment time on the glucose efficiency is presented in
Figure 3a–c. The given response surface areas as a function of the mentioned process parameters suggest that optimal values of catalyst concentration for the highest glucose efficiency can be selected. As stated before, the highest glucose yield was obtained at NaOH concentration of approx. 6–6.5%. For concentrations above the mentioned value, degradation of the lignocellulosic material may proceed [
45], resulting in no further increase in the concentration of reducing sugars in the hydrolyzate.
Figure 4a,b show the effect of granulation, temperature and catalyst concentration on the glucose efficiency. The decrease of the size of biomass particles result in an increase of glucose efficiency. This is due to an increase on the available materials surface area for the contact with catalyst and the improvements of mass and heat transfer along with the reduction of the biomass particle sizes during the alkaline pre-treatment [
5,
57,
58]. Similarly as shown in
Figure 1,
Figure 2 and
Figure 3, in the studied range of process parameters, the response surface areas in
Figure 4a,b indicate that the optimal value of catalyst concentration and the temperature of the alkaline pre-treatment can be selected for the highest glucose efficiency.
Based on the obtained model, the optimal conditions for the conversion of biomass to glucose are proposed as follows: granulation 0.25 mm, catalyst concentration 7%, temperature 80 °C, pre-treatment time 7 h. The given values of parameters correspond to the maximum values of glucose yield in the investigated range. The optimal conditions of alkaline pre-treatment of rice straw presented in the literature [
44] taking into account temperature, NaOH concentration and pre-treatment time are 81.8 °C, 2.82% and 56.7 min, respectively. Application of these conditions allowed to obtain 252.62 g of glucose from 1 kg of rice straw. In other studies on the pre-treatment of the algae mixture
Tetraselmis suecica and
Chlorella sp. with sodium hydroxide [
43], for a temperature of 120 °C, a concentration of NaOH 2% and a time of 30 min, 81 mg of reducing sugars from 1 g of dry biomass were obtained. This yield was doubled after enzymatic hydrolysis. The optimal pre-treatment conditions to maximize the glucose yield are obtained at 2.5% NaOH concentration, performing the process at 120 °C for 40 min. According to the initial content of glucose in the raw material, the best results corresponds to glucose production of 202 g from 1 kg of vineyard pruning [
59].
2.6. Optimization of the Alkaline Pre-Treatment
The obtained results show that the parameters of the alkaline pre-treatment, such as the temperature, the catalyst concentration, the pre-treatment time and the size of hydrolyzed material particles have a significant influence on the result of enzymatic hydrolysis and thus on the yield of monosugars. According to the authors of this work, the selection of conditions for alkaline pre-treatment requires optimization not only with respect to the efficiency of glucose, but also due to the costs of treatment (
Table 8). This approach is particularly justified when scaling-up, because often a small increase in the efficiency is accompanied by an enormous increase in the costs of process realization.
An exemplary economic analysis of the energetic willow alkaline pre-treatment shows that fragmentation, pre-treatment time and temperature significantly affect the costs of the process. Therefore, in order to reduce the costs of biomass processing, the optimal values of the process parameters should be lower than the maximum values from the investigated range. Therefore, it seems reasonable to compare the efficiency and relative increase in glucose efficiency for the process parameters under examination.
Figure 5 and
Figure 6 show the above-mentioned dependencies calculated on the basis of the proposed model. This approach makes it possible to indicate ranges of process parameters, above which the efficiency increase is disproportionately small compared to the increase of the process costs. Cost assessment analysis is not common in published articles.
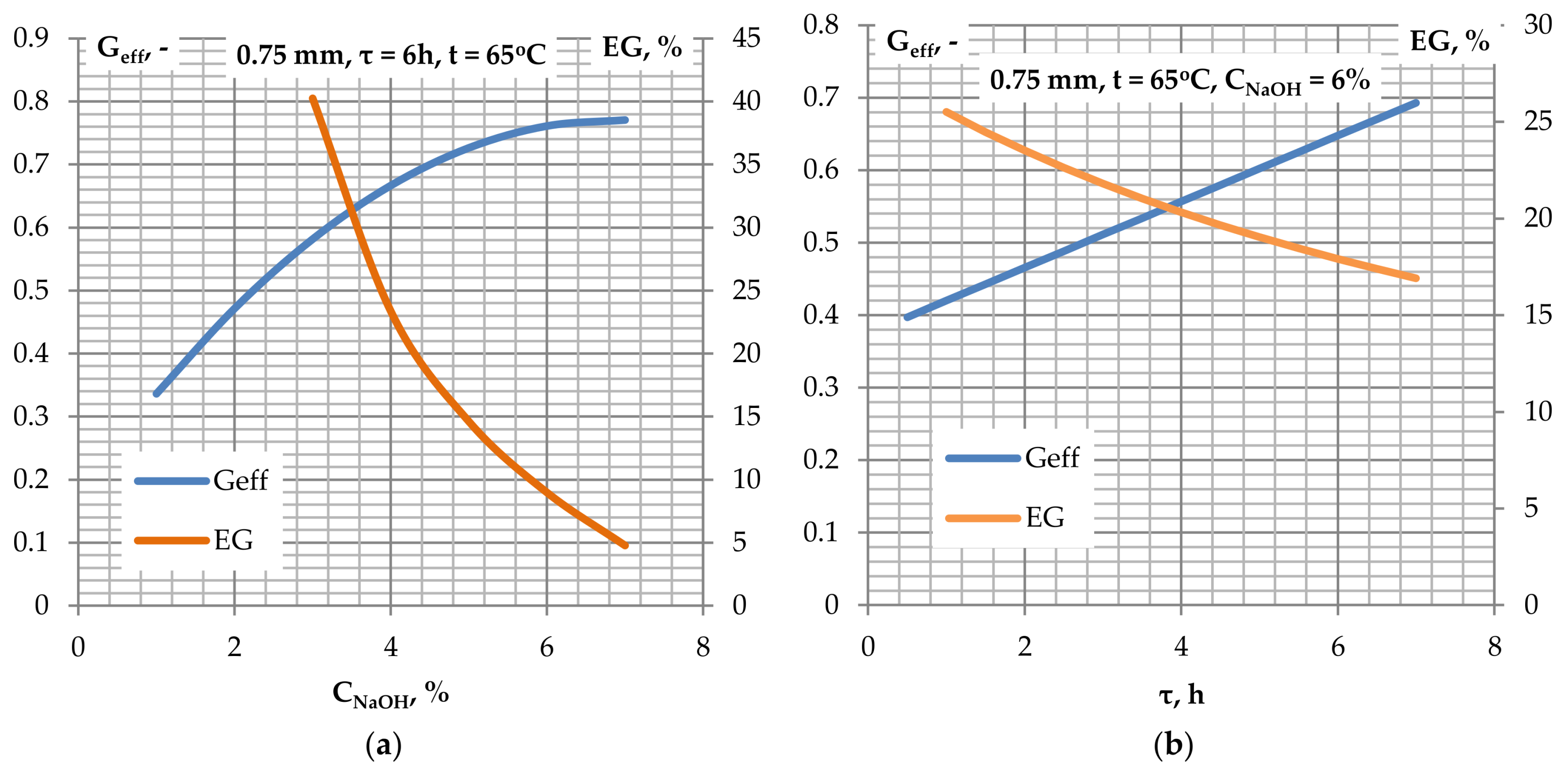
Figure 5a shows the effect of NaOH concentration on the efficiency and the efficiency gain of glucose. For NaOH concentrations above 6%, the glucose yield is practically unchanged (EG gain <5% when C
NaOH increases from 6% to 7%). Because of the small proportion of sodium hydroxide cost in the total cost, it is proposed to use C
NaOH = 7%.
Figure 5b shows the effect of the pre-treatment time on the glucose efficiency and relative glucose efficiency gain. The increase in the duration time of the alkaline treatment results in an increase in glucose efficiency. Considering the costs related to the pre-treatment time due to the need to maintain the selected temperature during the alkaline treatment, as well as decreasing values of the relative glucose efficiency gain, a duration time of 6 h is suggested as suitable for the pre-treatment.
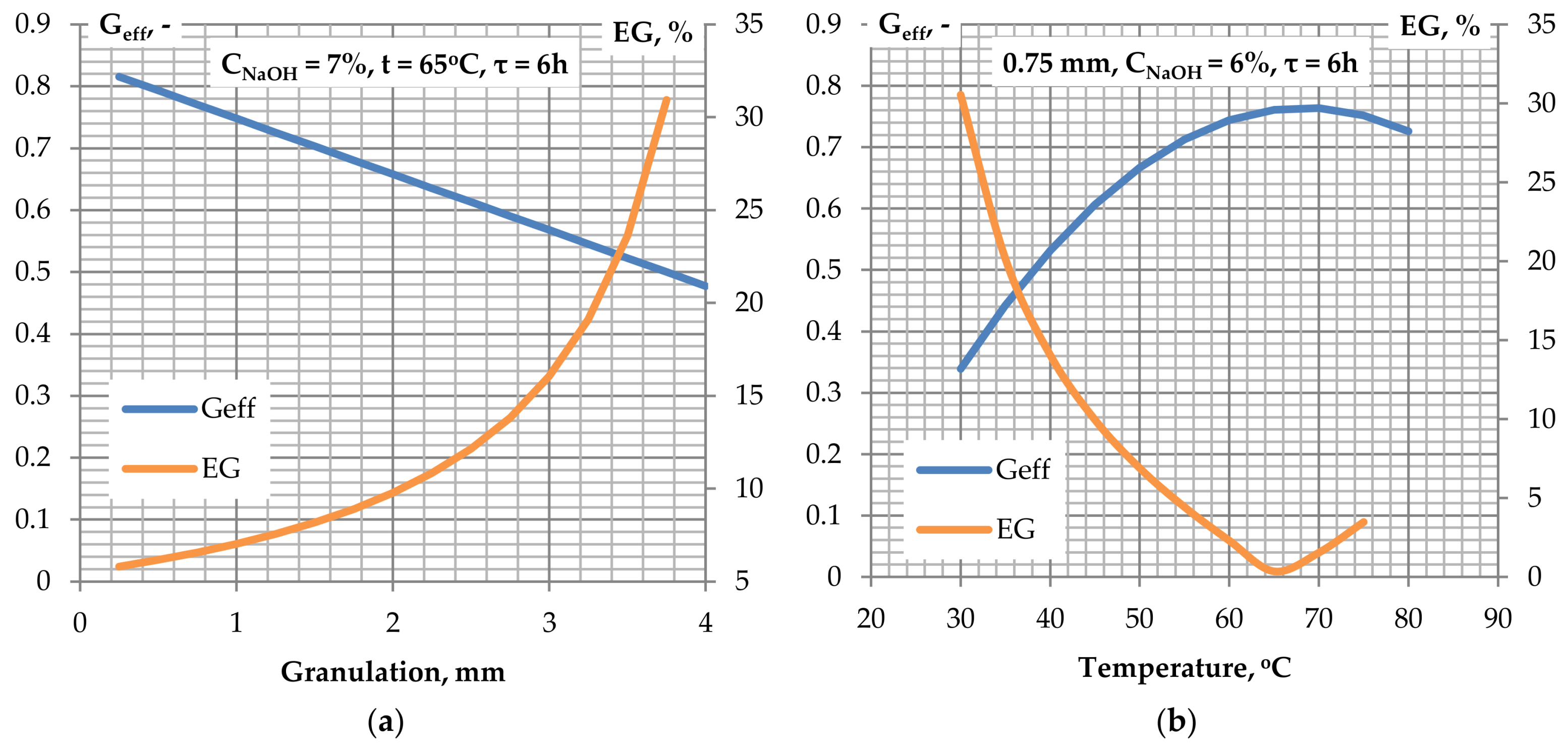
Figure 6a presents the influence of granulation on the glucose efficiency and the relativeglucose efficiency gain as a result of alkaline treatment. The highest glucose yields were obtained for granulation of 0.25 mm. However, the costs of obtaining the indicated degree of material fragmentation are relatively high. Because the efficiency gains attain the lowest values for granulation below 1 mm, keeping high values of glucose efficiency, granulation of 0.75 mm are selected to be optimal. The indicated value is economically justified, because the technical conditions of the milling process allow to obtain particles of the indicated size at the same costs as for 2 mm granulation. Material grinding to a size of 0.25 mm requires two-stage grinding, during which additional local overheating of the material and possible thermal degradation may take place.
Figure 6b shows the influence of the temperature on the glucose efficiency and relative glucose efficiency gain. The dependencies show that the highest efficiency value was obtained for a temperature of approximately 65 °C, which corresponds with the minimum on the relative glucose efficiency gain curve. The indicated temperature value is optimal for alkaline pre-treatment of biomass from
Salix viminalis L.
The results presented in
Figure 1,
Figure 2,
Figure 3,
Figure 4,
Figure 5 and
Figure 6 and economic analysis (
Table 8) for the optimal conversion of biomass from
Salix viminalis L. indicate that the following process parameters were optimal: granulation 0.75 mm, NaOH concentration 7%, pre-treatment time 6 h, temperature 65 °C. Enzymatic hydrolysis of the material previously treated with alkali in the above-mentioned conditions gave a glucose yield and a sum of reducing sugars of 415.7 g and 487 g respectively, from 1 kg of biomass, the values correspond to the glucose efficiency of 0.797 calculated from the proposed model. For comparison, the corn stover treatment with 10% sodium hydroxide at a temperature of 140 °C and for 30 min, it was possible to obtain a concentration of reducing sugars of 440 mg/g biomass [
60]. Significantly milder conditions of grass pre-treatment (C
NaOH = 0.5%, 60 °C and t = 0.5 h) but ultrasounds-assisted, allowed to obtain approximately 260 mg/g of reducing sugars [
61]. Optimal alkaline treatment conditions proposed in this paper allow to obtain up to twice the performance of reducing sugars. However, it should be noted that the type of raw material used has a significant influence on the final result of glucose efficiency.
2.7. Influence of the Material Type on the Glucose Efficiency
The effect of the chemical composition of lignocellulosic biomass on the efficiency of bioconversion for six selected raw materials: three wood (energy willow, energetic poplar and beech) and three agricultural waste (field grass, wheat straw and corncobs) was compared. In the first series of experiments, the material was subjected to enzymatic hydrolysis, while in the second series—the alkaline treatment was first carried out, followed by the enzymatic hydrolysis. The raw materials used for the research were fragmented in a mill using a 0.75 mm sieve. The two-stage conversion process was carried out under the optimal conditions indicated above, considering the economic analysis of the process (i.e., CNaOH = 7%, temperature 65 °C, pre-treatment time 6 h). The yield of glucose for wood raw materials after enzymatic hydrolysis was 230, 234 and 324 mg from 1 g of biomass for beech, energetic willow and energetic poplar, respectively. The two-stage process allowed to increase the glucose efficiency by approximately 1.4 times for poplar and 1.8 times for willow and beech. A comparable increase in the glucose release was obtained after hydrolysis of corncob, i.e., 1.65. The largest increase in the biomass to glucose conversion (approximately 3 times) was obtained for grass and straw.
The obtained results of glucose efficiency are similar to other literature data [
62], e.g., for alkaline pre-treatment of corncobs (dilute NH
3, temperature 75 °C, time 96 h). Authors of the paper [
48]obtained a yield of reducing sugars of 850 mg from 1 g of wheat straw after the alkaline treatment (temperature 121 °C, time 30 min, C
NaOH = 2%). The efficiency of reducing sugars under optimal conditions proposed in the present work for grass is 1.34 times higher than for the process carried out at 60 °C, in 30 min and for C
NaOH = 0.5%. Despite a significant improvement in the yield of reducing sugars due to the use of alkaline treatment, it occurred that the yield of monosugars obtained by direct enzymatic hydrolysis of the raw material is decent. Depending on the type of raw material subjected to hydrolysis, after 16 hours of hydrolysis, the yield of reducing sugars was obtained in the range of 38.5% for straw to 82% for energetic poplar. Such high yields of glucose yield from untreated material can be the result of enzyme immobilization on diatomite. For comparison, using non-immobilized cellulolytic enzymes ensured the yield of glucose in the level of 17% for barley straw hydrolysis, when compared to the theoretical values [
8,
63,
64] and only 3.6% of glucose for switch grass [
65,
66]. Untreated rice straw hydrolysis for 72 h allowed to obtain only 20% of total glucose [
66]. Also, the direct enzymatic hydrolysis of sugarcane bagasse allowed to obtain only 13% of reducing sugars [
67].
The highest weight loss of biomass was noted for agricultural waste materials, which may be connected to their chemical composition that differs from the composition of woody lignocellulosic materials, such as beech, poplar or willow. Grasses and grains contain significant amounts of non-structural ingredients, including dyes and minerals that can easily pass into the catalyst solution. As a result, there is a significant change in their chemical composition after the pre-treatment, which results in a higher biomass loss than for trees. Additionally, the highest degree of lignin removal was also obtained for the waste agricultural materials. Significant impact on the removal of lignin from a low—lignin content material, such as agricultural waste, using alkaline pre-treatment, is confirmed in the available literature [
65]. It is different in the case of the acid treatment, which depending on the conditions, allows for even 100% removal of hemicelluloses. However, its effect on the lignin content is smaller [
66]. Acid treatment with 85% phosphoric acid at 50 °C for 5 h allowed to remove 88.5% and 91.3% hemicellulose from the straw and maize straw, respectively. However, the loss of lignin was 15.1% for maize straw and zero for maize [
67]. The use of sulfuric acid to treat wheat straw allowed for the hydrolysis of 96% hemicellulose [
68].
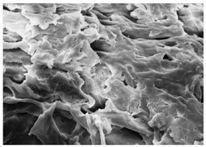
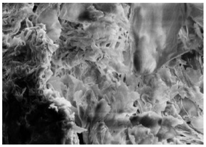
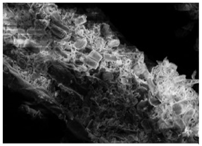
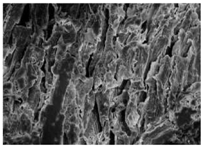
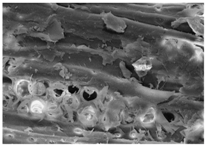
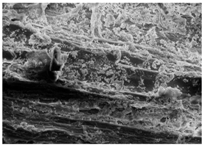
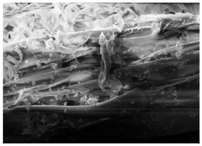
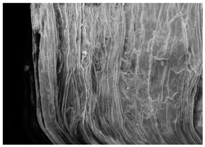
Changes in the structure of biomass resulting from alkaline and/or enzymatic treatment are presented on SEM images (
Table A2). The
Appendix B presents SEM images for raw materials (
Table A2, Figures a) and after selected pre-treatment mode: alkaline pre-treatment (
Table A1, Figures b), enzymatic hydrolysis of raw material (
Table A2, Figures c) and after a two-stage treatment i.e., alkaline pre-treatment and enzymatic (
Table A2, Figures d). Images of biomass samples after the alkaline pre-treatment revealed the loosening of materials resulting in the increased availability of cellulose and hemicellulose for hydrolytic enzymes. This is probably mainly due to the removal of lignin during the alkaline pre-treatment. SEM images taken for materials after enzymatic hydrolysis and after two-stage treatment indicated that the material degradation occurs at these stages. Also visible in the images are the characteristic structures of diatomaceous earth, which are the remains of immobilized enzymes. As can be seen from the data presented in
Table 9, the cumulative yield of simple sugars after enzymatic hydrolysis from the raw material is higher for the material after the alkaline treatment. The degree of lignin removal is also higher for these processes. This demonstrates the desirability of converting biomass to simple sugars in a two-stage process.
2.8. Influence of the Alkaline Pre-Treatment on Inhibitory Compounds Formations
During the alkaline pre-treatment of lignocellulosic biomass, delignification results in the formation of lignin derivatives, mainly phenolic compounds. During acidic hydrolysis furan derivatives, including furan and 5-HMF and acids, including acetic, levulinic and acetic acids, are formed depending on the conditions. HMF and furfural are formed during the dehydration reaction of simple sugars catalyzed by acids [
69,
70]. Most of the by-products of pre-treatment are formed due to the decomposition of hemicellulose. Usually, an increase in their concentration is related to the decrease in the concentration of simple sugars derived from hemicellulose hydrolysis [
71,
72]. The type and concentration of formed inhibitors depend on the processing conditions. The influences of temperature (140 °C and 160 °C) and the concentration of sulfuric acid (5–20 g/dm
3) on the formation of inhibitors were examined. It turned out that the higher the temperature and the higher the concentration of sulfuric acid, the higher the concentration of acetic and formic acids, HMF and furfural, with the highest concentration observed for formic acid [
69,
73]. Some of the inhibitory compounds may be adsorbed on the surface of the wood sediments. It is possible to subject such a sediment to enzymatic hydrolysis and then perform the detoxification [
74,
75,
76,
77]. It is possible to rinse the sediment several times with, for example, water [
44], for elution of the inhibitors. The effect of using sludge water on the effectiveness of inhibitor removal was investigated. It turned out that six rinsing cycles allows to remove the total content of 87% formic acid, 64% acetic acid, 86% furfural and 87% HMF [
73]. The addition of acetone to water for the purpose of elution from the inhibitor sludge, allowed to obtain a total removal of inhibitors from the sludge. Acetone dissolves organic compounds much more easily and thus removes them more effectively from the sediment. In addition, acetone can be easily recovered for being reused by vacuum distillation. The use of efficient washing of the sludge after the treatment avoids the need for its detoxification. Another situation occurs when the acid is used for direct hydrolysis of biomass. Then, it may be necessary to use the detoxification process [
71,
78].
The effect of the alkaline pre-treatment carried out under the optimal conditions in the presence of inhibitors in hydrolyzates was also examined. It turned out that a typical inhibitor present in the investigated hydrolyzates is acetic acidIts concentration after enzymatic hydrolysis prior and after the alkaline pre-treatment varied and was equal to 32.1 and 3.1 mg/g
biomass for grass, 10.4 and 7.7 mg/g
biomass for wheat, 8.7 and 3.0 mg/g
biomass for corn cobs, 3.7 and 2.34 mg/g
biomass for poplar, 11.0 and 3.35 mg/g
biomass for willow and 10.0 and 6.7 mg/g
biomass for beech. As can be seen, the effect of the pre-treatment has a significant impact on the concentration of acetic acid in the hydrolyzates. Similar observations were obtained as a result of another work concerning the alkaline pre-treatment catalyzed with NaOH, KOH and Ca(OH)
2 followed by enzymatic hydrolysis. It turned out that acetic acid was present in small concentrations only [
77].
The direct application of enzymatic hydrolysis forthe lignocellulosic material does not result in the passage of the vast majority of known inhibitors to the hydrolyzate solution. When cellulolytic enzymes were applied on untreated rice straw supported with ultrasounds and surfactant, it turned out that only acetic acid was present in the hydrolyzate solution [
77].