Abstract
In order to produce bioethanol from yellow poplar sawdust without detoxification, deacetylation (mild alkali treatment) was performed with aqueous ammonia solution. To select the optimal conditions, deacetylation was carried out under different conditions: NH4OH loading (2–10% (w/v)) and a solid-to-liquid ratio of 1:4–1:10 at 121 °C for 60 min. In order to assess the effectiveness of deacetylation, fractionation of deacetylated yellow poplar sawdust was performed using dilute acid (H2SO4, 0.5–2.0% (w/v)) at a reaction temperature of 130–150 °C for 10–80 min. The toxicity-reduced hemicellulosic hydrolyzates that were obtained through a two-step treatment at optimized conditions were fermented using Pichia stipitis for ethanol production, without any further detoxification. The maximum ethanol production was 4.84 g/L, corresponding to a theoretical ethanol yield of 82.52%, which is comparable to those of intentionally made hydrolyzates as controls.
1. Introduction
The increasing demand for alternative sources of fuel has increased public interest in the development of biofuel. Significant effort has been invested in the conversion of lignocellulosic biomass into platform chemicals as well as further upgrading to biofuels [1,2,3]. Lignocellulosic biomass, which is generally sourced from wood and agricultural wastes, is mainly composed of cellulose, hemicellulose, and lignin. Due to its recalcitrant characteristics, the biomass requires an appropriate fractionation process to produce fermentable sugars [4,5].
Generally, the fractionation methods for lignocellulosic biomass mainly contain physical, chemical, and biological methods, and their combinations [6], which have different impacts on the structure of the lignocellulosic material, and have a significant impact on the downstream processings of the biomass conversion process, which is relevant to sugar recovery, hydrolyzate detoxification, enzymatic hydrolysis and fermentation, and waste water treatment [7].
Dilute acid fractionation is an efficient chemical method that accomplishes the selective degradation of hemicellulose fractions and improves the cellulose fractions in the residual solids. When compared to other fractionation technologies, it is also an important technology in terms of cost [8]. Furthermore, this method also enhances the accessibility of cellulase to cellulose in the residual solid [9]. However, the major obstacles of dilute acid fractionation are the sugar degradation products and toxic compounds that are present in the hemicellulosic hydrolyzate, such as 5-hydroxymethylfurfural (HMF), furfural, formic acid, and acetic acid [8].
One of the well-known inhibitors is acetic acid, which is formed by the cleavage of bonded acetyl groups by deacetylation of hemicelluloses during biomass pretreatment [10,11,12]. Therefore, techniques need to be developed to reduce the toxicity of hemicellulosic hydrolyzate, so as to make high value-added products derived from biomass more economically competitive [7]. Several methods have been proposed to reduce the concentration of toxic compounds to levels that would not inhibit the fermentation process. These methods can be divided into the following three main groups: physical, chemical, and/or biological detoxification [13]. At this time, detoxification has led to a considerable loss of fermentable sugars and is therefore not a feasible method [14]. In addition, these methods usually do not provide workable results when applied alone, making it necessary to employ a combination of them, which increases operational costs [15].
In our previous studies, alkaline treatments have been studied to dissolve lignin and hemicellulose, and the de-esterification of intermolecular ester bonds [16]. However, these alkaline conditions to obtain lignin are not suitable at the biorefinery concept, because recovery of hemicellulosic sugar in hydrolyzate derived from alkaline-treated biomass is not easy for microbial fermentation, due to the high amount of lignin degradation compounds and others [17]. The loss of hemicellulosic sugars, mainly xylose, must be avoided, because these pentose sugars can be converted into higher value compounds, including ethanol, xylitol, and others [18,19], contributing to the economic feasibility of the biorefineries. To solve this problem, the deacetylation process at low severe condition prior to dilute acid fractionation could show not only significant improvement on sugars yields, but also toxicity reduction in hydrolyzate, thereby enhancing bioethanol production [20]. Therefore, several researchers have tried to remove the acetyl group with sodium hydroxide before fractionation to reduce the toxicity in hemicellulosic hydrolyzate [7,10,21,22].
In the present study, deacetylation, through mild alkaline treatment with aqueous ammonia solution, was performed to produce a toxicity-reduced hemicellulosic hydrolyzate (TRH hydrolyzate) that was suitable for ethanol fermentation, without an additional detoxification process. This treatment was performed prior to the dilute acid fractionation, with the purpose of selectively removing the toxic compounds, such as acetyl group, without degradation of the polysaccharide, to improve hemicellulosic sugar extraction yield in the dilute sulfuric acid fractionation (DSA fractionation). To evaluate the effectiveness of DSA fractionation for ethanol production, ethanol fermentation using Pichia stipitis, which has become widely known for its ability to rapidly metabolize xylose into ethanol, was conducted with TRH hydrolyzate fractionated from deacetylated yellow poplar sawdust (YPS).
2. Results and Discussion
2.1. Composition Change in the Liquid Phase Derived from Deacetylated YPS
The effectiveness of deacetylation (mild alkali treatment) was determined by the appearance of the formation of acetic acid in the liquid phase derived from YPS. Additionally, the additional effectiveness of the deacetylation might have weakened the hemicellulose structure, because untreated biomass revealed an intact structure, like a complex hierarchical structure with a compact and highly compressed surface.
Table 1 summarizes the concentrations of acetic acid and other components in the liquid phase after the deacetylation of YPS. The acetic acid formation was affected by NH4OH loading more than by the solid-to-liquid ratio (S/L ratio) when the temperature and time were fixed. The maximum acetic acid concentration of 8.16 g/L was found to be with 2% NH4OH loading and an S/L ratio of 1:4. The liquid phase contained mostly acetic acid, and negligible quantities of sugars without inhibitors (decomposed products, such as 5-HMF and furfural). De Assis Castro et al. [7] obtained the maximum formation of acetic acid in rice straw hemicellulosic hydrolyzate, which resulted in the highest hemicellulose extraction, since acetyl groups are structurally linked to hemicellulose. However, in this study, the acetic acid concentration decreased as NH4OH loading and S/L ratio increased (Table 1). The highest concentration of acetic acid was not obtained when the XMG (xylose + mannose + galactose) concentration was the highest. Ammonium acetate can be produced by saturation of acetic acid with ammonia [23].

Table 1.
Sugars and decomposed products in the liquid phase after deacetylation of yellow poplar sawdust (YPS).
2.2. Optimization of the Deacetylation Process Conditions
In order to optimize the deacetylation conditions, conditions that maximize the removal of acetyl groups, with minimum degradation of cellulose and hemicellulose, should be selected [10]. Table 2 presents the chemical composition of YPS in raw and post-deacetylation conditions. As can be seen, acetyl groups and ash were the main fractions affected by this deacetylation, which favored their removal under almost all conditions. The highest acetyl removal (100%) was achieved with an S/L ratio of 1:8 and 6% NH4OH loading at 121 °C for 60 min, and more than 80% of ash was removed; meanwhile, glucan and XMG showed losses of up to 17.99%.

Table 2.
Change of chemical composition before and after deacetylation of YPS and removal percentage.
The biomass recovery (%) is expressed as the solid remaining after deacetylation, based on the original oven-dry weight. The solid remaining was about 70% under all conditions. In particular, the highest value (77.02%) was obtained with an S/L ratio of 1:6 and 2% NH4OH loading, for which the removal of acetyl group was 91.46%. These results indicate that the purpose for deacetylation was successfully achieved, since the acetyl group was the main fraction removed from biomass. Additionally, the deacetylation removed a significant amount of ash (94.95%), which also helps to overcome the major drawback of toxic compound formation during the subsequent biomass fractionation steps. In this step, acid insoluble lignin (AIL) was hardly removed, in comparison with that of deacetylated biomass by NaOH. It can be expected that NH4OH is a weak base; thus, less delignification is achieved than that with NaOH. De Assis Castro et al. [7] shows a higher removed amount of lignin (up to 34%) via the NaOH deacetylation of rice straw.
From these results, the optimal condition of the deacetylation process was selected based on a high solid remaining (77.02%), a high removal of the acetyl group (91.46%), and low effectiveness of the carbohydrate fraction (glucan 1.05%; XMG 3.35%). Finally, the selected optimal condition was an S/L ratio of 1:6 and 2% NH4OH loading at 121 °C for 60 min. Castro et al. [7] found lower values of acetyl removal (86.5%) during the deacetylation of rice straw at 70 °C for 45 min using 80 mg of NaOH/g biomass. There, 15.1% glucan and 15.9% XMG were removed.
2.3. Optimization of Dilute Sulfuric Acid Fractionation Conditions with Deacetylated YPS for TRH Hydrolyzate Production
The DSA fractionation was performed to optimize the condition as maximum hemicellulose fractionation. Figure 1 and Figure 2 show the extraction yields of sugars and concentrations of decomposed products in hydrolyzate depending on various acid loadings, reaction times, and temperatures. Acetic acid, XMG, and glucose were released as the hemicellulose degraded [24]. The XMG was the main component present in the hydrolyzate with a concentration of 0.08–12.42 g/L. However, XMG was further degraded to furfural under higher severity, such as a high temperature and a long reaction time [25], while hexose could be converted into 5-hydroxymethyl furfural (5-HMF) [26]. Consequently, the concentration of furfural and 5-HMF consistently increased with increasing reaction severity.
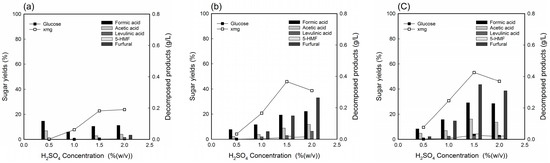
Figure 1.
The extraction yield of sugars and concentration of inhibitors on various dilute acid loading (0.5–2.0% (w/v)) fractionation of deacetylated YPS at 130 °C for reaction times of (a) 10 min, (b) 30 min, and (c) 50 min.
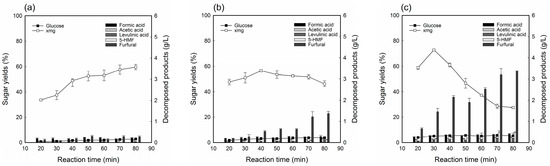
Figure 2.
Extraction yield of sugars and concentration of inhibitors on dilute acid fractionation of deacetylated YPS for 50 min at 1.5% (w/v) dilute acid loading and reaction temperature (a) 130 °C, (b) 140 °C, and (c) 150 °C.
Figure 1 shows the sugar extraction and concentration of decomposed products at 130 °C, with 0.5–2.0% (w/v) H2SO4 loading and an S/L ratio of 1:10 for (a) 10 min, (b) 30 min, and (c) 50 min. The sugar extraction yield in the hydrolyzate increased as acid loading and time increased. However, the extraction yield decreased at more than 1.5% acid loading due to a further decomposition of sugars. On the other hand, furfural and formic acid increased up to 0.4 g/L with increasing acid loading and time. Since the extraction yield of XMG was not yet high (the highest extraction yield in Figure 1c is 53.23%), additional optimization was carried out through temperature changes at 1.5% acid loading, which is now widely known to rapidly metabolize xylose into ethanol.
Figure 2 shows the change in sugar extraction yield and decomposed product concentration on 1.5% acid loading. The sugar extraction indicated a gradual increase as time increased at 130 °C (Figure 2a), but it maintained or decreased at higher temperatures (Figure 2b). Regarding the sugar solubilization, glucose concentration was not above 2.67 g/L (corresponding to extraction yield of 5.70%) in all the hydrolyzates, which indicated low cellulose degradation. In particular, the highest XMG extraction yield was 72.82% with treatment conditions at 150 °C for 30 min with 1.5% H2SO4 loading, and 1:10 S/L ratio (Figure 2c). At this time, formic acid, acetic acid, levulinic acid, 5-HMF, and furfural were 0.33, 0.18, 0.12, 0.17, and 1.45 g/L, respectively. With respect to the inhibitor compounds, all hydrolyzates derived from deacetylated YPS showed acetic acid concentrations below 0.25 g/L, which was expected, due to the effective removal of acetyl groups during deacetylation. Additionally, the decomposed products known to be fermentation-decomposed products, such as furfural, 5-HMF from pentose and hexose sugars, are generated during acid fractionation. Other studies have reported that ethanol fermentation was unsuccessful when the concentrations of HMF, furfural, and acetic acid in the hydrolyzate were more than 1, 1, and 2 g/L, respectively [12,24,27,28]. This finding implies that detoxification is rarely needed for ethanol fermentation, due to the low concentration of inhibitors present in the hydrolyzate.
In order to compare the effectiveness of the deacetylation on TRH hydrolyzate composition, DSA fractionation was performed under the highest hemicellulose extraction condition, using the deacetylated YPS obtained under the optimized alkaline conditions and non-deacetylated YPS as a reference. Table 3 shows the chemical composition of the residual solid and hydrolyzates obtained under DSA fractionation. The table shows that much lower concentrations—1.01 g/L glucose and 3.37 g/L XMG—were found in the non-deacetylated hydrolyzates. However, a higher concentration of acids and furan totaling 2.61 g/L was observed (Table 3), and the concentration of inhibitory compounds was quite different for both deacetylated and non-deacetylated YPS. Table 3 clearly shows that deacetylation could effectively remove the acetyl group in the YPS. In this condition, acetic acid, formic acid, 5-HMF, and furfural concentrations were decreased, while the XMG extraction yield was increased.

Table 3.
Chemical compositions after dilute acid fractionation of deacetylated and non-deacetylated YPS under optimal dilute acid fractionation conditions.
2.4. Ethanol Fermentation with Toxicity-Reduced Hemicellulosic Hydrolyzate
Figure 3 shows the ethanol fermentation profile with the TRH hydrolyzates to assess the toxicity reduction of the hydrolyzate. Figure 3a shows the fermentation result of TRH hydrolyzate, while Figure 3b–d represent various controls ((b): 1% (w/v) yeast extract, 2% (w/v) peptone, and an equivalent concentration of XMG in TRH hydrolyzate; (c): (b) with an equivalent concentration of acetic acid in TRH hydrolyzate; (d): (b) with an equivalent concentration of inhibitors in TRH hydrolyzate), which were intentionally made to assess the extent of microbial inhibition reduction. In comparison with the control solutions (Figure 3b,c), the maximum ethanol concentration is almost equal, and the ethanol production (Figure 3a) of TRH hydrolyzate obtained is 4.84 g/L, but the reaction time is longer due to the effectiveness of other inhibitors. However, the ethanol production rate, compared to the presence of other inhibitors, was further decreased only when the acetic acid concentration was about 1 g/L (Figure 3c).
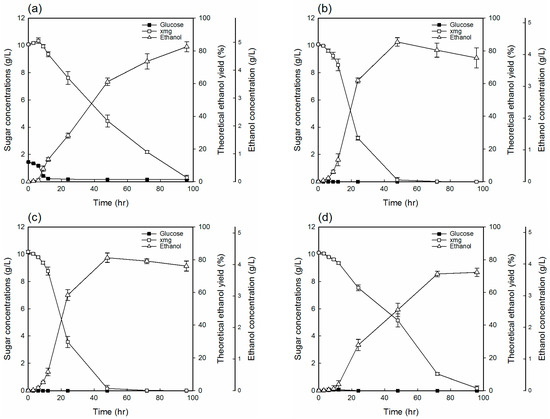
Figure 3.
Ethanol production on both hydrolyzate and various control solutions by P. stipitis: (a) toxicity-reduced hemicellulosic hydrolyzate (TRH hydrolyzate); (b) yeast extract 1%, peptone 2%, and an equivalent concentration of XMG in TRH hydrolyzate; (c): (b) with an equivalent concentration of acetic acid in TRH hydrolyzate; (d): (b) with an equivalent concentration of inhibitors in TRH hydrolyzate.
The maximum ethanol concentration obtained from toxicity-reduced hydrolyzate under optimal treatment condition was 4.84 g/L, corresponding to a theoretical ethanol yield of 82.52% (Figure 4). Most of the XMG was consumed within 72 h. Moreover, the maximum theoretical yield of ethanol obtained similar to the control solution, due to the low concentration of inhibitors in the toxicity-reduced hydrolyzate, because ethanol production from the hydrolyzate depends on the concentration of fermentation inhibitors. Therefore, it is also important to highlight that it was not necessary to ferment the deacetylated YPS hydrolyzate to any further detoxification process prior to fermentation.
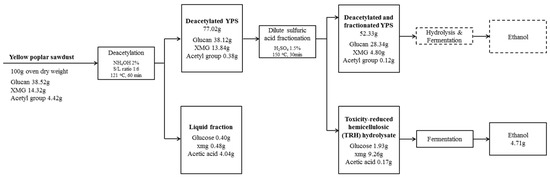
Figure 4.
Mass balance for dilute sulfuric acid fractionation of deacetylated YPS under optimized conditions.
2.5. Overall Mass Balance
The simplified flowchart and an overall mass balance of the deacetylation and consecutive fractionation of YPS, which involved microbial fermentation, are summarized in Figure 4. Most of the acetic acid and hemicellulosic sugar were separated from the YPS by deacetylation and fractionation with aqueous ammonia and sulfuric acid. Under the optimized deacetylation condition, approximately 23% of the mass fraction was solubilized into liquid hydrolyzate, which indicated 0.4 g of glucose, 0.5 g of XMG, and 4.0 g of acetic acid, while 38.1 g of glucan, 13.8 g of hemicellulose, and 0.4 g of the acetyl group were found in the 77 g of deacetylated YPS based on 100 g of raw YPS. During the deacetylation, 91.4% of the acetyl group was dissolved into a liquid fraction, which means that only 0.1% glucose and 0.3% hemicellulosic sugar (represented by XMG) were decomposed. The residual solid, i.e., deacetylated YPS, was subjected to consecutive acid fractionation under an acid concentration of 1.5 wt %, a reaction temperature of 150 °C, and a reaction time of 30 min. The TRH hydrolyzate resulting from fractionation with 77 g of deacetylated YPS had a sugar fraction including 1.9 g of glucose and 9.3 g of hemicellulosic sugar.
During the fractionation step, 66.9% of hemicellulose was dissolved into liquid hydrolyzate, while the acetyl group, ash, and water extract were mainly removed. The TRH hydrolyzate was further converted to ethanol through microbial fermentation, and 4.7 g of ethanol, which corresponds to an ethanol yield of 82.5%, was obtained. In addition, the residual solid, i.e., 52.3 g of deacetylated and fractionated YPS, which included 28.3 g of glucan and 4.8 g of XMG, could be further converted to ethanol through enzymatic hydrolysis and fermentation.
3. Materials and Methods
3.1. Raw Materials
Ground YPS was supplied by G-Biotech Co., Ltd. (Sejong, Korea). The sample particles were air-dried at 45 °C for 24 h and then used directly in deacetylation and dilute sulfuric acid fractionation (DSA fractionation) studies. The moisture content of the milled sample was 3.70%, based on the total wet biomass weight.
The chemical composition of the raw sample was 38.52% glucan, 14.32% XMG (the total sum of three oligomeric sugars xylan, mannan, and galactan), 0.65% arabinan, 18.86% acid insoluble lignin (AIL), 5.70% acid soluble lignin (ASL), 4.42% acetyl group, 3.51% acid insoluble ash (AIA), 5.55% water extract, and 1.07% ethanol extract. The mass closure of the raw sample reached 92.6% on oven-dried biomass.
3.2. Deacetylation
To obtain deacetylated YPS, the deacetylation process was carried out using aqueous ammonia solution (NH4OH) to remove the acetyl group from the biomass. Deacetylation was performed at 121 °C for 60 min with 2, 4, 6, 8, and 10% (w/v) NH4OH loading at S/L ratios of 1:4, :6, :8, and :10, respectively. After the deacetylation process, the biomass was washed continuously with deionized water until pH 7.0 was reached. The biomass was then dried and stored.
3.3. Dilute Sulfuric Acid Fractionation of Deacetylated YPS
DSA fractionation experiments were performed using sealed bomb tubular reactors, 150 mm long with an inner diameter of 10.7 mm, constructed out of stainless steel tubing (SS 316 L), capped at both ends with Swagelok fittings, to give an internal volume of 13.5 mL. The reactors were loaded with 700 mg of oven-dried deacetylated YPS. The residual moisture in the dried YPS sample was accounted for when the amount of solution to be added was quantified, yielding an S/L ratio of 1:10. The tubular reactors were submerged in the first bath (molten salt) at 240 °C for rapid preheating to the target temperature in about 0.3 min. The reactors were then quickly transferred into the second bath of silicone oil set at the desired reaction temperature. After the desired reaction time, the reactors were quickly transferred to an ice-water bath to quench the reaction for 10 min. The tubes were removed from the water bath, and the end caps and Teflon plugs were removed. The contents were separated by filtration into liquid and solid fractions. The three parameters that determined the reaction severity were the reaction temperature, reaction time, and sulfuric acid loading; these were tested over the ranges of 130–150 °C, 10–80 min, and 0.5–2.0% (w/v).
3.4. Microorganism and Inoculum Preparation
Pichia stipitis CBS 7228 was used for the fermentation of TRH hydrolyzate derived from deacetylated and DSA-fractionated YPS. The strain stock was kept on an agar plate made of 1% (w/v) yeast extract, 2% (w/v) peptone, and 2% (w/v) xylose as an additional carbon source at 4 °C. P. stipitis was inoculated in 100 mL of YPX medium (1% (w/v) yeast extract, 2% (w/v) peptone, 2% (w/v) xylose) in a 250 mL Erlenmeyer flask. The preculture was incubated at 30 °C and shaken at 150 rpm for 24 h in a shaking incubator (Vision Scientific Co., Seoul, Korea).
3.5. Ethanol Fermentation of Toxicity-Reduced Hemicellulosic Hydrolyzateml
The microorganism P. stipitis was used for ethanol fermentation. Fermentation of TRH hydrolyzate was carried out in 125 mL Erlenmeyer flasks containing 50 mL of TRH hydrolyzate (pH = 5.5) supplemented with 1% (w/v) yeast extract, 2% (w/v) peptone, and inoculated with 5% (v/v) of P. stipitis. The flasks were incubated in a rotatory shaker at 30 °C at 150 rpm for 96 h. Samples were taken at 0, 3, 6, 9, 12, 24, 48, 72, and 96 h. The samples were then centrifuged by using a 0.45 µm centrifuge filter and analyzed for ethanol and residual sugar. The theoretical ethanol yield was given by Equation (1):
3.6. Compositional Analysis
The chemical compositions of the solid and liquid samples were determined following the procedures of the National Renewable Energy Laboratory (NREL; Golden, CO, USA) laboratory analytical procedures (LAP) (NREL/TP-510-42623 for structural carbohydrates and lignins; NREL/TP-510-42618 for sugars in the liquids or in the hydrolyzates) [29,30]. The sugars were determined using high-performance liquid chromatography (Agilent 1260 Infinity, Agilent Technologies, Santa Clara, CA, USA) equipped with a refractive index detector (Agilent 1260 Infinity, Agilent Technologies, Santa Clara, CA, USA). A Bio-Rad Aminex HPX-87H column (300 mm length × 7.8 mm internal diameter) and Cation H micro-guard cartridge (30 mm length × 4.6 mm internal diameter) (Bio-Rad Laboratories Inc., Hercules, CA, USA) were used for sugar analysis. The sample was filtered using a syringe filter (0.45 µm pore size, Advanced Microdevices Pvt. Ltd., Haryana, India) before analysis, and the mobile phase was 5 mM sulfuric acid. The HPLC analysis conditions were a column temperature of 65 °C, and a mobile phase flow rate of 0.5 mL/min.
4. Conclusions
Deacetylation and DSA fractionation were optimized to maximize the fermentable sugar (mainly pentose) concentration and to minimize the decomposed products in TRH hydrolyzate, such as acetic acid and furan compounds. Significant results for the fermentation process from deacetylated and pretreated YPS were found, but not in the non-deacetylated, revealing that the fermentation ability of P. stipitis was related to the presence of inhibitors. Additionally, the reaction severity of DSA fractionation considerably decreased due to the deacetylation process. As a result, ethanol production was improved through the deacetylation and DSA fractionation of YPS, because low acetic acid was produced in TRH hydrolyzate without further detoxification.
Acknowledgments
This work was supported by the New & Renewable Energy Core Technology Program of the Korea Institute of Energy Technology Evaluation and Planning (KETEP) granted financial resource from the Ministry of Trade, Industry & Energy, Korea (No. 20153010091990).
Author Contributions
Seong Ju Kim and Tae Hyun Kim equally contributed to this work. Seong Ju Kim conducted deacetylation and fermentation experiments and created figures & tables. Tae Hyun Kim supervised experiments, interpreted the results, and wrote and reviewed manuscript. Kyeong Keun Oh designed overall study and experiments, interpreted the results, and finalized the manuscript. All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Conde-Mejía, C.; Jiménez-Gutiérrez, A.; El-Halwagi, M. A comparison of pretreatment methods for bioethanol production from lignocellulosic materials. Process Saf. Environ. Prot. 2012, 90, 189–202. [Google Scholar] [CrossRef]
- Yan, K.; Liu, Y.; Lu, Y.; Chai, J.; Sun, L. Catalytic application of layered double hydroxide-derived catalysts for the conversion of biomass-derived molecules. Catal. Sci. Technol. 2017, 7, 1622–1645. [Google Scholar] [CrossRef]
- Yan, K.; Jarvis, C.; Gu, J.; Yan, Y. Production and catalytic transform ation of levulinic acid: A platform for speciality chem icals and fuels. Renew. Sustain. Energy Rev. 2015, 51, 986–997. [Google Scholar] [CrossRef]
- Jorgensen, H.; Kristensen, J.B.; Felby, C. Enzymatic conversion of lignocellulose into fermentable sugars: Challenges and opportunities. Biofuels Bioprod. Biorefin. 2007, 1, 119–134. [Google Scholar] [CrossRef]
- Lim, W.S.; Kim, J.Y.; Kim, H.Y.; Choi, J.W.; Choi, I.G.; Lee, J.W. Structural properties of pretreated biomass from different acid pretreatments and their effects on simultaneous saccharification and ethanol fermentation. Bioresour. Technol. 2013, 139, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Long, J.; Wang, T.; Shu, R.; Zhang, Q.; Ma, L. Process intensification effect of ball milling on the hydrothermal pretreatment for corn straw enzymolysis. Energy Convers. 2015, 101, 481–488. [Google Scholar] [CrossRef]
- De Assis Castro, R.C.; Fonseca, B.F.; dos Santos, H.T.L.; Ferreira, I.S.; Mussatto, S.I.; Roberto, I.C. Alkaline deacetylation as a strategy to improve sugars recovery and ethanol production from rice straw hemicellulose and cellulose. Ind. Crop. Prod. 2017, 106, 65–73. [Google Scholar] [CrossRef]
- Mussatto, S.I. Biomass pretreatment with acids. In Biomass Fractionation Technologies for a Lignocellulosic Feedstock Based Biorefinery; Mussatto, S.I., Ed.; Elsevier Inc.: Waltham, MA, USA, 2016; pp. 169–185. [Google Scholar]
- Mosier, N.; Wyman, C.; Dale, B.; Elander, R.; Lee, Y.Y.; Holtzapple, M.; Ladisch, M. Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour. Technol. 2005, 6, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Moya, A.J.; Peinado, S.; Mateo, S.; Fonseca, B.G.; Sanchez, S. Improving bioethanol production from olive pruning biomass by deacetylation step prior acid hydrolysis and fermentation processes. Bioresour. Technol. 2016, 220, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.P.A.; Carneiro, L.M.; Roberto, I.C. Treatment of rice straw hemicellulosic hydrolyzates with advanced oxidative processes: A new and promising detoxification method to improve the bioconversion process. Biotechnol. Biofuels 2013, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Palmqvist, E.; Hahn-Hagerdal, B. Fermentation of lignocellulosic hydrolyzates. I: Inhibition and detoxification. Bioresour. Technol. 2000, 74, 17–24. [Google Scholar] [CrossRef]
- Jönsson, L.J.; Martín, C. Pretreatment of lignocellulose: Formation of inhibitory by-products and strategies for minimizing their effects. Bioresour. Technol. 2016, 199, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, D.; Amann, M.; Hirth, T.; Rupp, S.; Zibek, S. Development and optimization of single and combined detoxification processes to improve the fermentability of lignocellulose hydrolyzates. Bioresour. Technol. 2013, 133, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, L.J.; Alriksson, B.; Nilvebrant, N. Bioconversion of lignocellulose: Inhibitors and detoxification. Biotechnol. Biofuels 2013, 6, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H. Pretreatment of lignocellulosic biomass. In Bioprocessing Technologies in Integrated Biorefinery for Production of Biofuels, Biochemicals, and Biopolymers from Biomass; Yang, S.T., El-Enshasy, H.A., Thongchul, N., Martin, Y., Eds.; Wiley: New York, NY, USA, 2013; pp. 91–109. [Google Scholar]
- Guo, B.; Zhang, Y.; Yu, G.; Lee, W.H.; Jin, Y.S.; Morgenroth, E. Two-stage acidic-alkaline hydrothermal pretreatment of lignocellulose for the high recovery of cellulose and hemicellulose sugars. Appl. Biochem. Biotechnol. 2013, 169, 1069–1087. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Song, Y.; Liu, D. Enzymatic hydrolysis and simultaneous saccharification and fermentation of alkali/peracetic acid-pretreated sugarcane bagasse for ethanol and 2,3-butanediol production. Enzym. Microbiol. Technol. 2011, 49, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Gurpilhares, D.B.; Hasmann, F.A.; Pessoa, A.; Roberto, I.C. The behavior of key enzymes of xylose metabolism on the xylitol production by Candida guilliermondii grown in hemicellulosic hydrolyzate. J. Ind. Microbiol. Biotechnol. 2009, 36, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Shekiro, J.; Franden, M.A.; Wang, W.; Zhang, M.; Kuhn, E.; Johnson, D.K.; Tucker, M.P. The impacts of deacetylation prior to dilute acid pretreatment on the bioethanol process. Biotechnol. Biofuels 2012, 5, 8–21. [Google Scholar] [CrossRef] [PubMed]
- Kundu, C.; Lee, J.W. Optimization conditions for oxalic acid pretreatment of deacetylated yellow poplar for ethanol production. J. Ind. Eng. Chem. 2015, 32, 298–304. [Google Scholar] [CrossRef]
- Kundu, C.; Lee, H.J.; Lee, J.W. Enhanced bioethanol production from yellow poplar by deacetylation and oxalic acid pretreatment without detoxification. Bioresour. Technol. 2015, 178, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Brannt, W.T. A Practical Treatise on the Manufacture of Vinegar; Henry Carey Baird & Co.: Lancaster, PA, USA, 1914; pp. 316–317. [Google Scholar]
- Palmqvist, E.; Hahn-Hägerdal, B. Fermentation of lignocellulosic hydrolyzates. II: Inhibitors and mechanisms of inhibition. Bioresour. Technol. 2000, 74, 25–33. [Google Scholar] [CrossRef]
- Dunlop, A. Furfural formation and behavior. Ind. Eng. Chem. Res. 1948, 40, 204–209. [Google Scholar] [CrossRef]
- Ulbricht, R.J.; Northup, S.J.; Thomas, J.A. A review of 5-hydroxymethylfurfural (HMF) in parenteral solutions. Toxicol. Sci. 1984, 4, 843–853. [Google Scholar] [CrossRef]
- Guo, G.L.; Chen, W.H.; Chen, W.H.; Men, L.C.; Hwang, W.S. Characterization of dilute acid pretreatment of silvergrass for ethanol production. Bioresour. Technol. 2008, 99, 6046–6053. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Wua, S.; Liu, L. Enzymatic saccharification of dilute acid pretreated eucalyptus chips for fermentable sugar production. Bioresour. Technol. 2012, 110, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Tmpleton, D. Determination of Sugars, Byproducts, and Degradation Products in Liquid Fraction Process Samples; NREL/TP-510–42623; National Renewable Energy Laboratory: Golden, CO, USA, 2008.
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Tmpleton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass; NREL/TP-510–42618; National Renewable Energy Laboratory: Golden, CO, USA, 2012.
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
