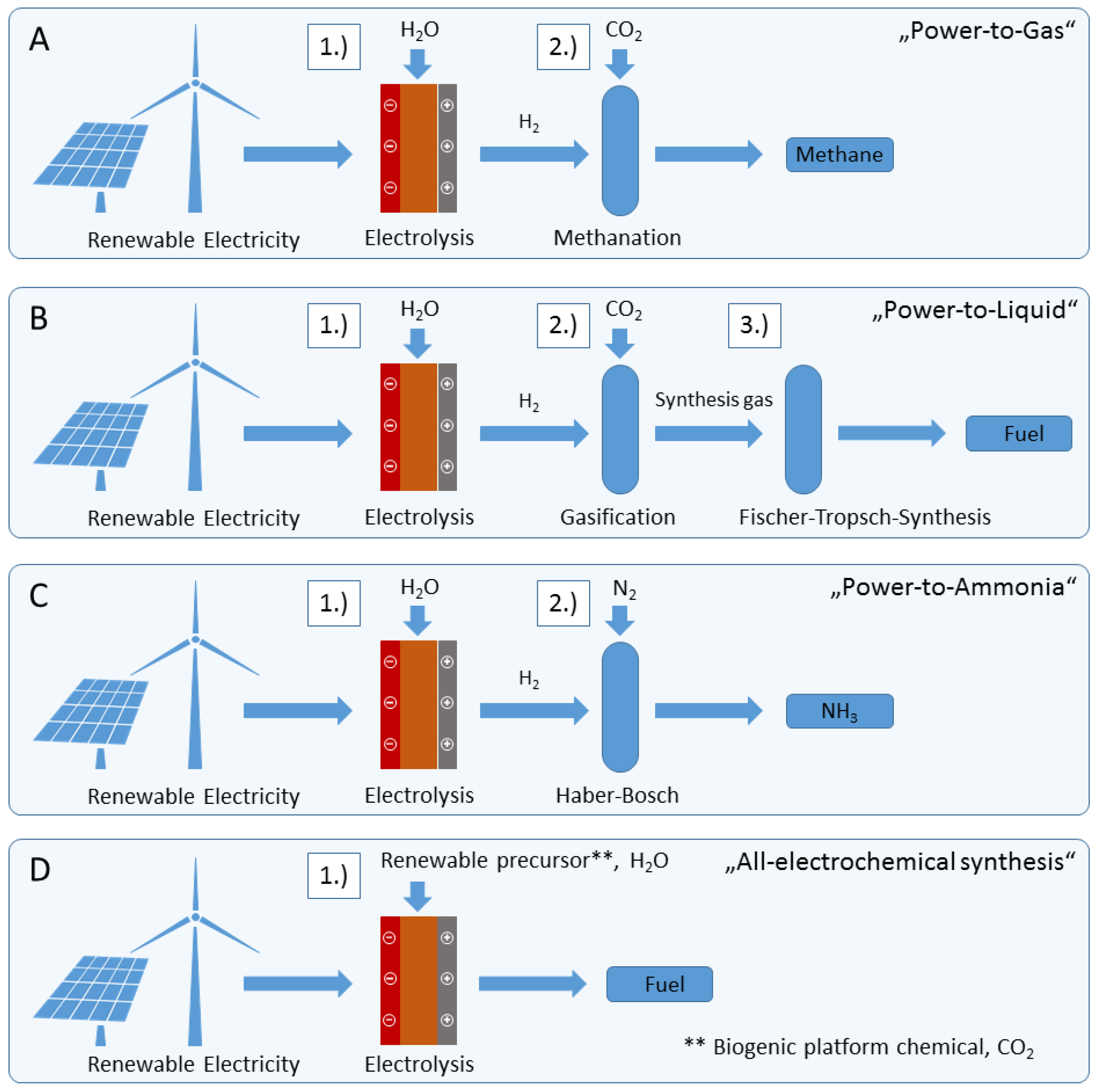
3.1. Comparison of Physico-Chemical Fuel Properties
Based on the discussed synthesis processes, five representative electrofuels are selected in this study: n-octane, methanol, methane, hydrogen, ammonia. They are compared with conventional jet fuel (Jet A-1) regarding some selected properties of importance for the utilization as potential sustainable aviation fuels; see
Table 1.
One important property of liquid fuels is the flash point, which describes the lowest temperature at which vapors of volatile fuel will ignite, when an external ignition source is present. For safety reasons, the flash point of jet fuel is above room temperature to reduce the risk of fire ignition. The flash point should not be too high, because the vapor of the fuel can no longer be ignited. Another property is the autoignition temperature, which describes the temperature at which the fuel will self-ignite in the presence of an oxidizer without an external ignition source. The lower and upper explosive limits describe the minimum and maximum of fuel concentration in air at which the mixture can burn. The vapor pressure describes the pressure of vapor above the liquid phase. If the vapor pressure is too high, the change from liquid phase to gas phase can occur and leads to an interruption of the fuel supply in the fuel line, also called vapor lock. The boiling point is related to the vapor pressure. The melting point should be low to prevent freezing of the fuel during flights at high altitudes. The chemically-stored energy of the fuel is expressed by the mass-based specific energy and the volume-based energy density. Both properties have a central influence on the range of an aircraft. The first one specifies the weight for the necessary fuel, while the latter one determines the size of the tank system.
The comparison of physical properties shows that n-octane is nearly similar to jet fuel. Specifically, the specific energy, energy density, autoignition temperature, lower and upper explosive limits are nearly the same. Other properties such as flash point, vapor pressure and boiling point could be adjusted by adding small amounts of mixable hydrocarbon-based fuels with high energy densities. Due to these reasons, a mixture based on n-octane has the potential to be used as a drop-in fuel, replacing conventional fuel in a conventional engine. Thus, only minor modifications of the aircraft are required here. The other sustainable electrofuels like methanol, methane, hydrogen and ammonia would require at least some modifications of the aircraft engine, fuel supply and tank system. The major drawback of those fuels is their low volumetric energy density, which requires larger tank systems. The data for methane, hydrogen and ammonia are based on liquid fuel, such that the tank system has to be either under high pressure or be cooled down by cryogenic tanks. For ammonia, the requirements are rather low, with only a 10 K temperature difference inside the cryogenic tank and the atmosphere at a 10-km altitude, while for cryogenic liquid methane, more than a 100 K temperature difference, and for cryogenic liquid hydrogen, more than a 200 K temperature difference are needed. (the data for a 10-km altitude ( = −50 °C, = 26,436 Pa) are: cryogenic liquid ammonia ( = −60 °C, = 21,892 Pa); cryogenic liquid methane ( = −178 °C, = 20,145 Pa); and hydrogen ( = −275 °C, = 20,447 Pa)).
3.2. Comparison of Fundamental Combustion Properties
The fuel compositions and their properties have an impact on the combustion, emission and engine performance. Some fundamental combustion properties, i.e., autoignition delay time , adiabatic flame temperature , laminar flame speed and extinction strain rate a, are computed by conducting numerical simulations with detailed reaction mechanisms for the investigated electrofuels.
For the Jet A-1 fuel combustion behavior, the Aachen surrogate (AS) [
54], defined as a mixture of n-decane (C
10H
22) 80% and 1,2,4-trimethylbenzene (C
9H
12) 20% by weight, is used as a representative surrogate mixture. For all calculations, air (O
2 + 3.76N
2) is defined as an oxidizer, except for extinction strain rate calculations. Here, pure oxygen is used for the calculation, as otherwise, NH
3 would not be comparable. This quantity is therefore for relative comparison only.
An overview of the reaction mechanisms used in this study is shown in
Table 2. For the reduction of the computation time, all hydrocarbon reactions greater than C
8 and greater than C
1 are removed in the C
8-C
16 n-alkanes [
55] and mechanisms AramcoMech 2.0 [
56,
57], respectively. The computation of the laminar flame speed for n-octane/air mixtures took around 60 CPU hours, while the computation of the other smaller fuels was much faster.
The simulations with detailed finite-rate chemistry are performed using the open-source Cantera library (Version 2.3.0) [
59], which is incorporated into a Python script in order to perform parallel computations. The autoignition delay times
were obtained through zero-dimensional perfectly-stirred reactor simulations with fixed time steps of 1 × 10
−7–1 × 10
−6 s at a constant pressure
p of 2.5 MPa and for fresh gas temperatures
of 1000–1600 K. The criteria for autoignition occurrence is defined by the maximum of heat release rate. The adiabatic flame temperature
and laminar flame speed
are computed for air/fuel equivalence ratios
of 0.6–1.6, fresh gas temperature
of 650 K and pressure
p of 2.5 MPa using the one-dimensional freely-propagating flame model with an adaptive grid refinement control and multicomponent transport. The Soret effect is neglected to reduce computation time. The extinction strain rate
a is calculated with the one-dimensional counterflow diffusion flame model and is defined as the maximum axial velocity gradient. Plug-flow boundary conditions are assumed. The mass flow rate area densities and inlet temperatures of fuel and oxidizer are set to 1 kg m
−2 s
−1 and 300 K, respectively. The distance between fuel and oxidizer inlets is set to 20 mm. In order to reduce the computation effort of the counterflow flame simulations, the scaling rules of Fiala [
60] are applied to improve the convergence behavior.
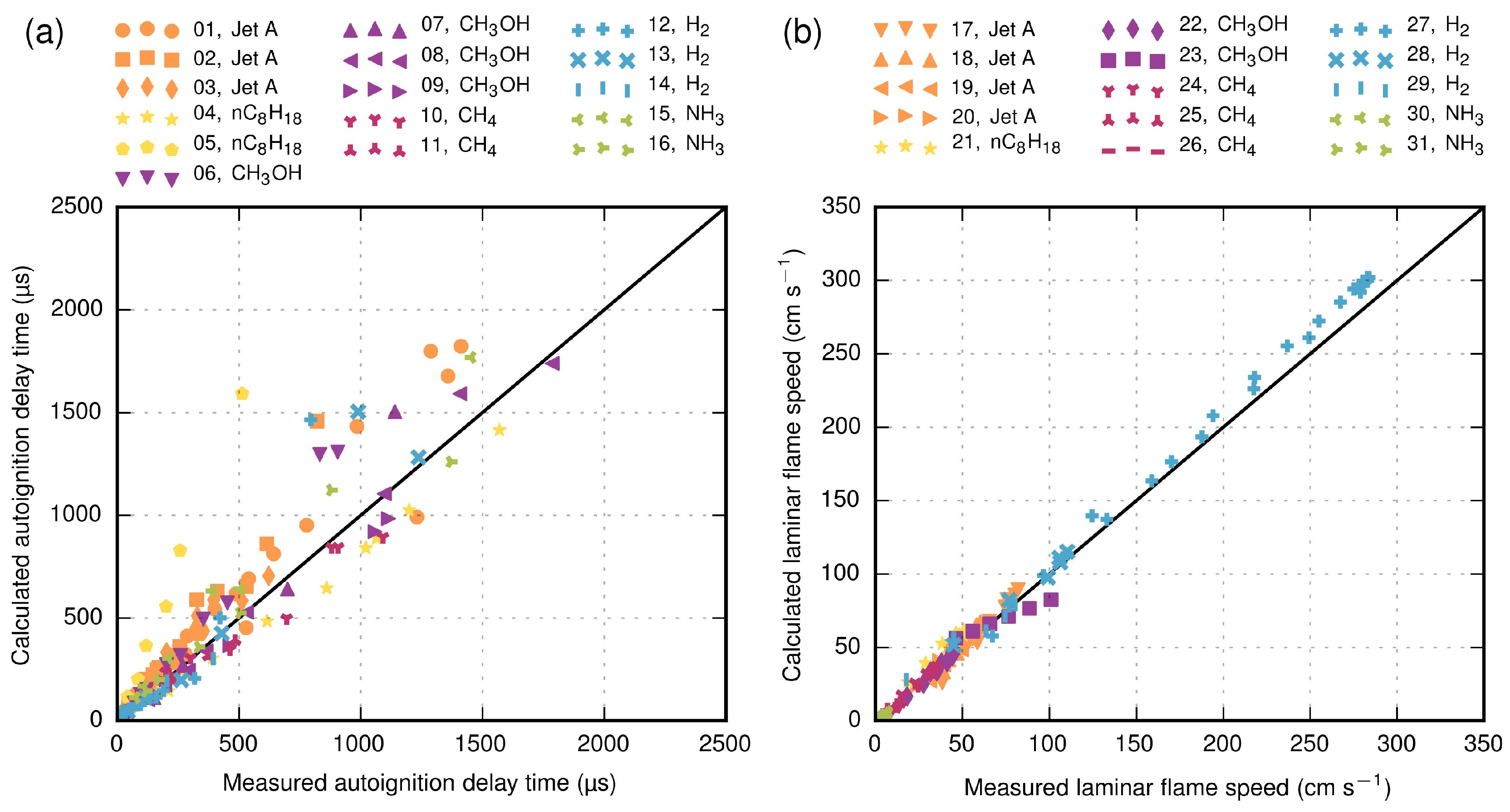
In order to make statements about the prediction quality of the computations, a validation study is conducted. Due to the lack of experimental data in the literature for the previously-mentioned conditions and mixture compositions, an indirect approach is applied in order to validate the computations. In total, 31 experimental datasets from the literature [
58,
61,
62,
63,
64,
65,
66,
67,
68,
69,
70,
71,
72,
73,
74,
75,
76,
77,
78] are used, as depicted in
Table 3, in order to validate the chosen mechanisms (
Table 2) at atmospheric conditions, as well as at elevated temperatures (up to 2085 K) and pressures (up to 5.1 MPa). The validation results are shown in
Figure 4. All mechanisms are able to predict the experimental data within a mean absolute percentage error of approximately 28% for autoignition delay times and 11% for laminar flame speeds. One exception is Dataset 05 [
66], where the deviation is greater. Further validation of the mechanisms used in this study can be found in [
54,
55,
56,
57,
58].
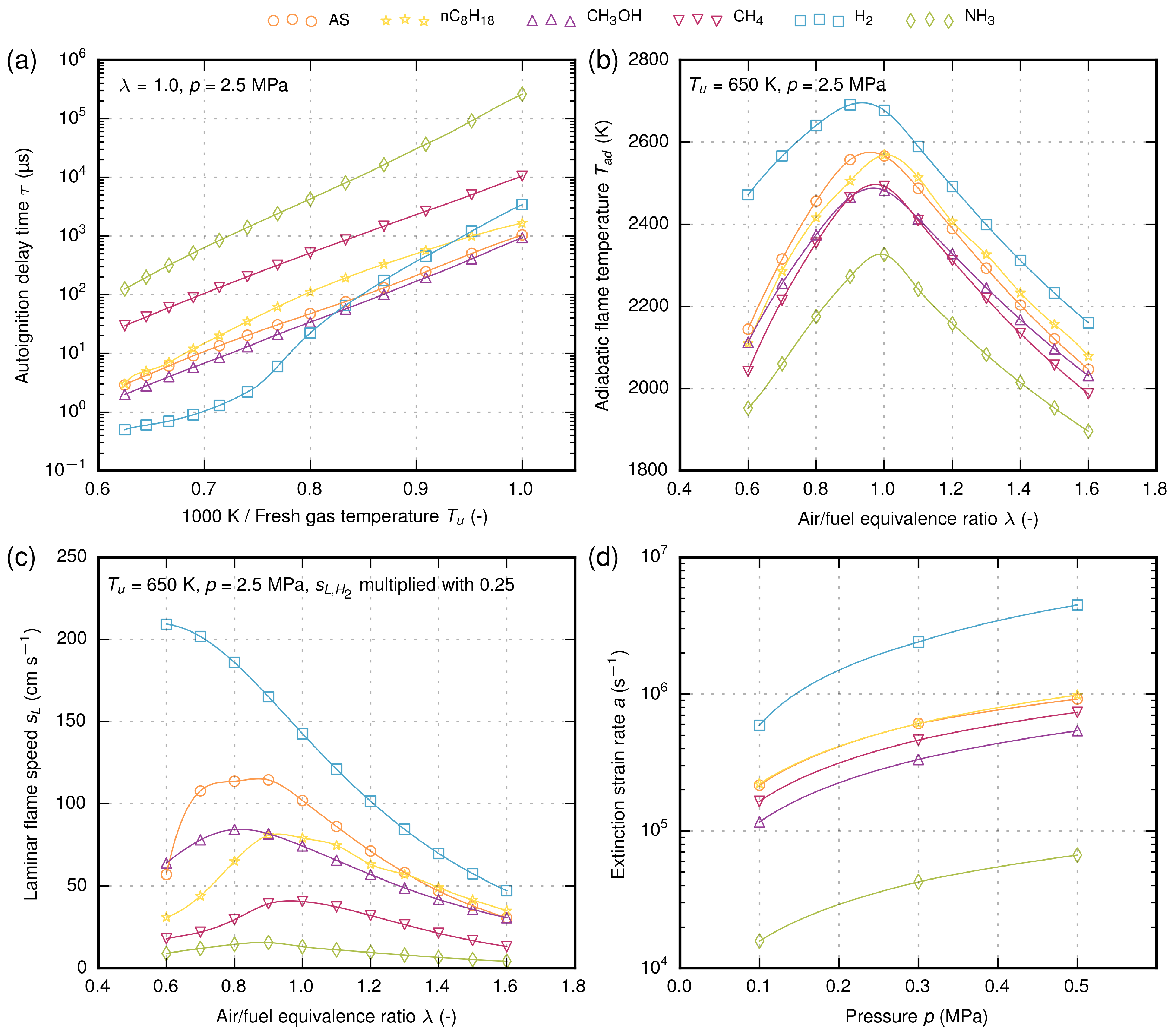
The simulation results of different fuels are depicted in
Figure 5. The autoignition delay time is plotted with logarithmic scaling as a function of inverse temperature between 1000 and 1600 K, as is common for Arrhenius-type reaction modeling. The ignition delay time can give some first hints about the reaction rate or ignition energy. For example, fuels with a high autoignition delay time need higher ignition energy to initiate the ignition as quickly as possible, which is important for engine relight during a flameout. Ammonia and methane have the highest autoignition delay time compared to n-octane, followed by jet fuel. Methanol has a slightly lower autoignition delay time, whereas the hydrogen self-ignition behavior strongly depends on the reactants’ temperature. Higher autoignition delay times could enable premixed combustion in aircraft engines.
The adiabatic flame temperature has an impact on the required air for cooling the combustion chamber and the burned hot gases. Further tendencies of emissions such as thermal nitric oxide (NO
x) and unburned hydrocarbon are specifically influenced by this property. The temperatures of jet fuel and n-octane are very similar. The same applies for methane and methanol. Ammonia shows the lowest flame temperature of all the investigated fuels, whereas hydrogen has the highest temperature. Looking at just the flame temperature, ammonia has the potential to reduce thermal NO
x emissions, but the NO
x formation could be promoted by the bounded nitrogen in the fuel molecule, which is so-called fuel NO
x. A recent study of a premixed ammonia/air flame shows that the fuel NO
x highly depends on the air/fuel equivalence ratio and can be kept rather low [
79].
The laminar flame speed plays an important role in the propagation behavior of the flame. Hydrogen has by far the highest flame speed compared to other fuels. In fuel-lean conditions (), the flame speeds of jet fuel, n-octane and methanol are similar, but the difference becomes more distinct for fuel-rich conditions (). Ammonia has the lowest laminar flame speed of all fuels.
At first glance, this property of ammonia looks rather negative. However, Verkamp et al. [
6] suggested partially dissociating ammonia into hydrogen and nitrogen before injection to enhance the combustion, or more precisely, to increase the laminar flame speed. A model for laminar flame speed of NH
3/H
2/N
2 mixtures was very recently developed by Goldmann and Dinkelacker [
80] and shows that the flame speed can be adapted in a wide range by the addition of hydrogen to ammonia. New interesting possibilities could be enabled by adjusting the ratio between ammonia and dissociated ammonia (hydrogen + nitrogen) during the operation of the engine. For example, a boost function during take-off could be implemented by a high concentration of hydrogen in the fuel, whereas during cruising, mainly ammonia could be burned.
The extinction strain rate can provide initial information about the flame stability. Fuels with higher extinction strain rates are less sensitive to the risk of flame extinction and allow higher flow velocities in the combustion chamber. Therefore, higher mass flow rates are possible, which results in an increase of thermal power. The extinction strain rate of jet fuel and n-octane is nearly the same. Furthermore, methanol and methane show similarities. Here again, hydrogen has the highest extinction strain rate, whereas ammonia has the lowest. Furthermore, here, a mixture of ammonia with hydrogen could potentially lead to reasonable flame stability.
By comparing the fundamental combustion properties, it can be seen that n-octane is most similar to jet fuel, whereas other fuels show very different behaviors. Therefore, for all these electrofuels, a modification of the combustion chamber and the whole engine would be indispensable. In all cases, hydrogen and ammonia have the maximum and minimum values, respectively. An interesting option, however, could be a mixture of hydrogen and ammonia, as this shows a promising potential to adjust the combustion properties as a function of the mixture.
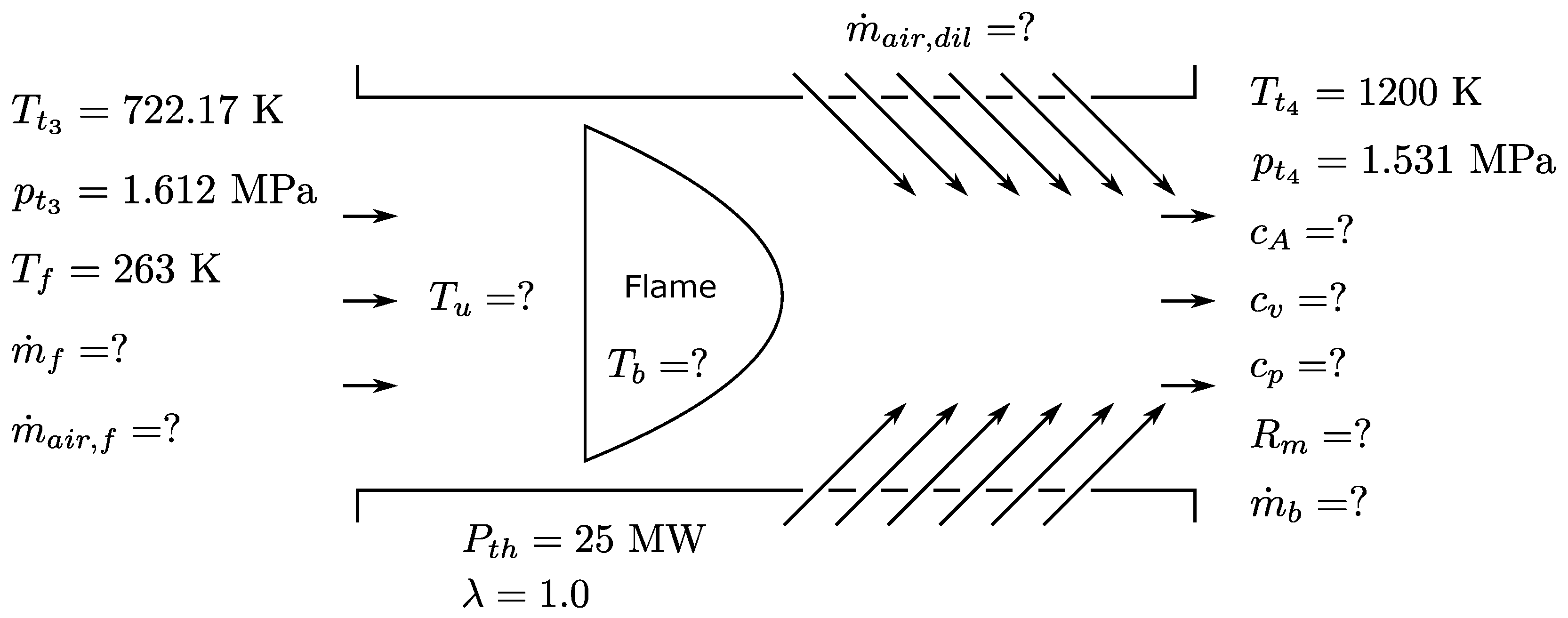
3.3. Impact of Electrofuels on the Joule Process
Alternative fuels may need other air mass flow rates, and also, other design parameters for the layout of the Joule process (compressor, burning chamber, turbine) can be different. In order to estimate these effects, several thermo-physical parameters have been determined for an aircraft engine, again at an altitude of 10 km (
= 26,436 Pa,
= 223.15 K) and a cruise speed of
= 0.8. The schematic of the combustion chamber of the aircraft engine and the relevant parameters is given in
Figure 6.
The following assumption are applied: The pressure drop across the combustion chamber
is assumed to be approximately 5% for all fuels. The turbine inlet temperature
is held constant at 1200 K, and the necessary overall air mass flow for temperature reduction
of the hot burned gases is calculated. The thermal power
of the combustion chamber is 25 MW, and premixed combustion with an air/fuel equivalence ratio
= 1.0 is assumed. The necessary mass flows of fuel
and air
for the combustion are calculated. An overall compression ratio
is assumed. The fuel in aircraft is used for cooling the engine oil, and therefore, the fuel is preheated before injection. Here, the inlet fuel temperature
of 263 K (223 K + 40 K) is chosen. Further ideal gas behavior is assumed. The thermodynamic inlet conditions for the combustion chamber and the turbine are calculated using Equations (
1)–(
7). The composition and properties of the combustion exhaust gas are calculated using Cantera [
59] and the detailed reaction mechanisms in
Table 2. The following indices are used: 0: environment, 1: turbo fan inlet, 2: compressor inlet, 3: combustion chamber inlet, 4: turbine inlet, 5: turbine exit.
Table 4 shows the calculated values of the respective Positions 0–4.
The speed of sound
(Equation (
8)) can give the first indication about the maximal mass flow at the combustor exit and with that the performance output.
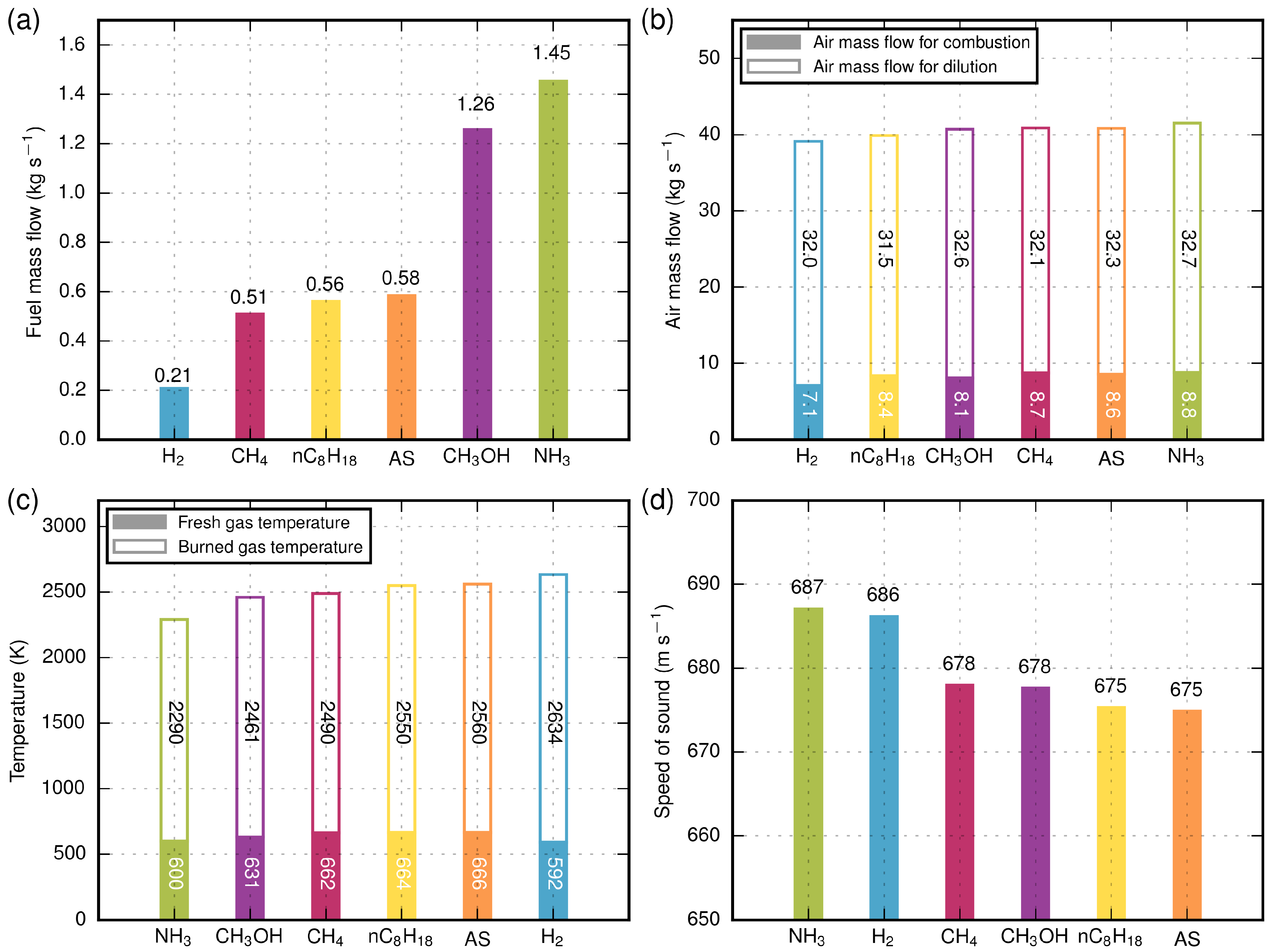
The results of the calculations are depicted in
Figure 7 and
Table 5. It can be clearly seen that the fuel consumption is different due to the specific energy of fuels. The total inlet mass flow of air for combustion and dilution is nearly the same for all fuels due to the effect of cooling capacity and adiabatic flame temperature. Hydrogen has the highest flame temperature, but also the highest specific heat capacity
of the burned gas. The heat capacity ratio
is nearly the same for all fuels. The speed of sound of all fuels is similar due to the high air content in the burned gas. Hydrogen and ammonia have a slightly higher speed of sound due to the lower mean molecular weight of products
in the burned gas. The dynamic viscosity
and thermal conductivity
are nearly the same. In summary, all input and output parameters for different electrofuels are nearly similar, except the fuel mass flow.
The detailed analysis of the necessary burner concept with details on the fuel/air supply, mixing system and flame stabilization is outside the scope of this study.
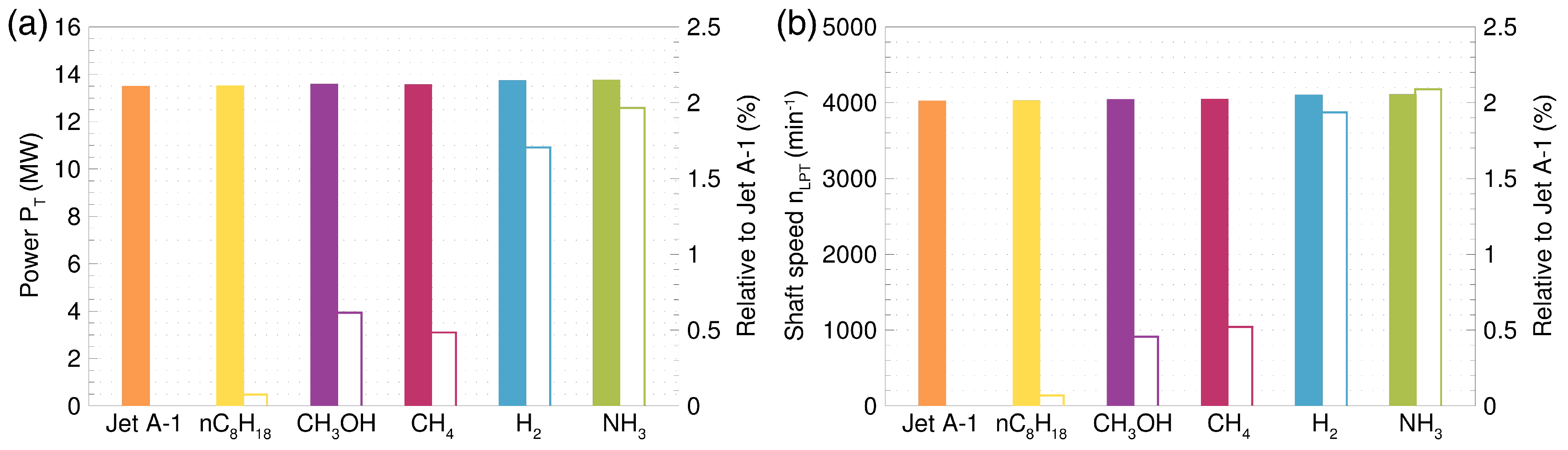
 and relative to Jet A-1
and relative to Jet A-1  .
.
 and relative to Jet A-1
and relative to Jet A-1  .
.