1. Introduction
Gasification is recognized as a suitable and promising technology for exploiting carbon-based resources (fossil or renewables) under environmentally favourable conditions, such as greenhouse gas (GHG) reduction [
1,
2,
3,
4] and high energy conversion rates [
5]. Moreover, this technology offers significant opportunities for rural electrification, since it enables the optimal use of local and bio-based energy, while promoting the active participation of local stakeholders [
6,
7,
8]. In such conditions, it is relevant to explore proper alternatives for optimizing the integration of local energy resources in order to reduce the implementation and operation cost of small-scale energy systems [
9].
Reported options for achieving a high conversion in gasifiers imply the use of relatively complex systems, such as additional reforming units or air separators; moreover, these alternatives also require an additional investment related to their implementation. Despite their effectiveness, their complexity and relative implementation cost may not be compatible with isolated, small-scale gasification systems that also require performance improvement. Common auxiliary services in gasifiers, for instance, air-separation units, water-gas shifting (WGS) reactors, or reformers, can be significant in the total cost of a gasification facility. Their relative costs calculated from reported direct capital costs are 1.09, 0.48, and 1.22, respectively, when compared with the cost of the gasification unit that is the comparison parameter (cost factor = 1) [
10].
Regarding operational issues, auxiliary units such as those for gas compression in fluidized-bed units are the leading energy consumption concern for these type of reactors [
11]. Even if such kinds of experimental components can improve the syngas quality, they are usually more expensive than commercial models [
12]. Hence, seeking alternatives aimed at reducing the cost syngas improvement is a relevant topic for making the technology available for the final stakeholder. Concerning the techno-economic point of view, it is possible to affirm that low-cost and straightforward syngas shifting proposals are relevant for consolidating the cost-effectiveness of gasification. Improving syngas quality is a relevant issue for small-scale energy systems, since electricity generation is quite sensitive to fuel gas quality [
13]. Moreover, efficiency is one of the factors that determine the final cost of electricity in such types of systems [
14].
Although including syngas reforming (chemical processes) or carbon capture (physical processes) increases the implementation costs of a gasification-based energy system, cost-effectiveness could be achieved once the technology is scaled up [
15], which may not be applicable in remote gasification systems. However, it is also noted that gasifiers without additional equipment are suitable for small-scale use, due to their simplicity and low cost. Hence, it is relevant to find syngas improvement options able to combine simplicity, effectiveness, and inexpensiveness.
Regarding other available alternatives for enhancing the syngas quality such as CO
2 or N
2 capture, it can be seen that they can raise the efficiency of downstream elements, including generator or chemical reactors, due to the reduction of gas transportation/handling capacities. Even though carbon capture appears as an emerging, yet not wholly mature, concept in energy systems, post-combustion CO
2-removal systems that are based on absorption and adsorption seem to be the most common alternatives to avoid extensive modifications of the infrastructure already implemented [
16]. Despite this affirmation, pre-combustion CO
2 capture, which requires gasification, has not yet been discarded, and its combination with other pre-treatment stages could be relevant [
17]. In addition, gasification technology could be significant for municipal solid waste (MSW) treatment, for instance, since it can operate despite considerable variations in the feedstock composition [
18].
This work describes a novel alternative for increasing the CO/CO
2 of syngas based on CO
2 adsorption combined with WGS (a combination of physical and chemical phenomena). The patented proposal considers that both techniques handle similar operating conditions to ease their coupling. In this case, both phenomena were induced in a single structure that encloses the CO
2 adsorption and the catalysed chemical reactions [
19].
It has been reported that WGS processes require a temperature range that is dependent on the catalyst; hence, it is necessary to reach proper operating conditions in order to enable the catalytic activity. In such a context, high- and low-temperature conditions could be equally exploited by selecting the proper catalyst. For instance, high-temperature WGS has been reported in the range of 350–500 °C for Fe/Cr catalyst, while low temperature ranges (150–300 °C) are appropriate for Cu/Zn catalyst operation [
20]. This parameter could be close to 350 °C for Pd-Ag catalysts [
21], 500 °C for Ni-Mg-Al-Ca catalysts [
22], or in the range between 300 °C and 450 °C for Co-Mo catalysts [
23]. Ranges between 180 °C and 300 °C for Ni-Ce-based catalysts have also been reported [
24]. Nevertheless, according to [
25], WGS is possible at 150 °C with Cu-ZnO-Al
2O
3 catalysts. On the other hand, CO
2 capture over zeolite at atmospheric pressure has been achieved with Zeolite 13X, which has been recognised as a suitable adsorbent, since it removes 20.1% of CO
2 at 25 °C and 1 bar [
26].
Catalyst supported over zeolite could have adsorbing activity in addition to its primary function of boosting WGS; moreover, it can find application in reducing potential CO2 emissions. Hence, it is possible to suggest that adsorption is the most compatible CO2 capture technology in cases where reforming is suitable. The current analysis evaluates both effects in a pilot-scale updraft gasifier equipped with an upper chamber filled with zeolite-based packed material. The analysis of syngas composition and its comparison with reported gasification systems were the evaluation parameters.
In the current work, quality criterion is based on the relative amount of CO
2 in the syngas stream. The presence of CO
2 is undesirable in fuel gases, since it implies a reduction in the global energy content [
27]. However, in gasification, CO
2 is a resulting element of oxidation, and can deliver energy for reduction reactions, such as hydrogen formation. Additionally, the heat produced during its formation allows gasification temperatures above 1000 °C, which boosts the formation of CO [
28]. Unlike CO
2, CO can be oxidised to deliver energy, and it is recognized as one of the components of syngas that can act as an energy carrier.
Hence, CO
2 content requires being controlled due to the multiple effects that its formation can cause on the performance of a gasifier. Moreover, it should be compared with CO, since its composition could decrease when the amount of CO
2 increases [
29]. The CO/CO
2 ratio has been considered to be an indicator of the direction that an oxidation process takes between gasification and combustion [
30], and as a suitable variable for controlling tasks in gasifiers [
31]. The objective of this research is to present an alternative for improving atmospheric gasifiers’ performance based on CO
2 adsorption and WGS, maintaining low complexity for construction and operation by inducing both phenomena in a single structure. The presented alternative is expected to become a suitable solution for small-scale gasification systems that generally require the improvement of their energy performance in order to reduce the final energy cost.
2. Materials and Methods
The experimental facility is a pilot-scale device consisting of a thermally isolated updraft gasifier equipped with an upper chamber. This chamber was filled with packed, adsorbing-catalytic material for assessing its performance. In addition to forming a gas reactor that hosts chemical reactions, this packed bed can also act as a CO
2 adsorption facility because of its use of zeolite, as described below in this section. The catalytic material is based on acid-activated Raney nickel as the active component, supported over zeolite. This material was selected based on its effectiveness and stability [
32,
33]. Additionally, zeolite was chosen as the support material due to its properties as a molecular sieve for CO
2 adsorption [
34,
35]. The gasifier is equipped with a control system that allows the CO/CO
2 ratio to be maintained during operation.
The packed-bed configuration is applied in unit operations and chemical reactions engineering as a relatively simple solution for increasing the superficial contact that is demanded by specific physical and chemical phenomena, such as adsorption and catalytic processes. Since packed beds are commonly formed by bulk solids, they allow having empty spaces surrounding the solid structures that offer the superficial contact. During industrial operations, such empty spaces are occupied by working fluids while they pass through the packed-bed structure. Moreover, the contact between the solid superficies and the working fluids is generated. In this case, the zeolite-based material that composes the upper packed bed has been provided with active catalytic material along its surface. Moreover, it has also been put through a surface preparation process for activating potential adsorption properties of zeolite. In consideration of this, the upper packed bed is expected to host physical and chemical phenomena that increase the energy vector contents in the syngas.
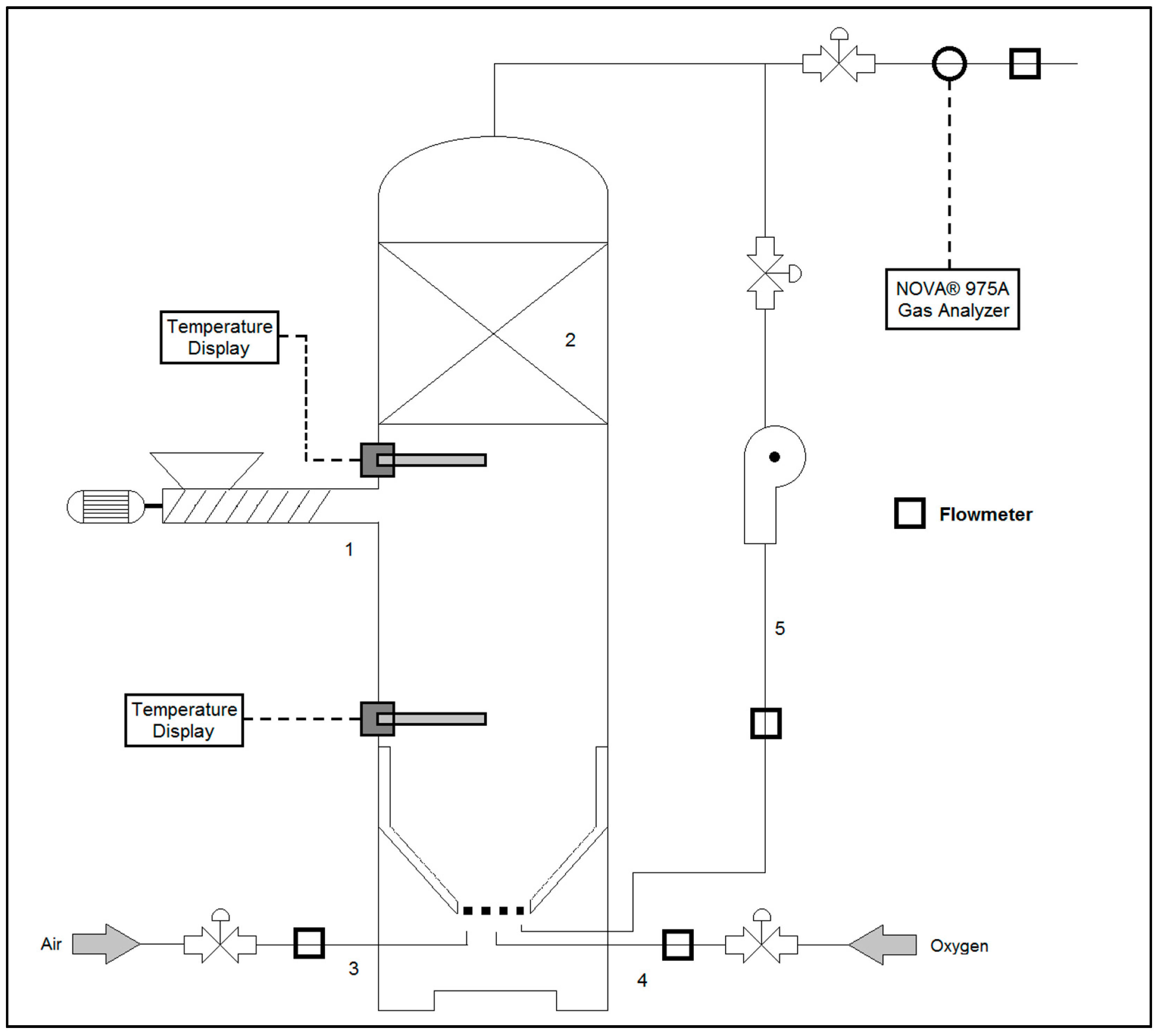
Regarding the gasifier configuration, the system scheme is presented in
Figure 1. It includes an inlet point for the biomass feedstock (1) located in the lower part of the packed-bed chamber (2). Biomass feedstock is added to the gasifier by a screw-type automatic solid feeder. An air inlet point (3) in the lower part of the gasification throat, and an oxygen inlet point connected to a liquid O
2 vessel (4) are also part of the research facilities for evaluating the effect of the oxygen input on the reactor performance. The gasifier is also equipped with a gas reflux pipeline (5) that can re-inject reformed syngas to the lower part of the gasification throat for modifying the gas retention time. The reduction zone temperature and the temperature in the lower part of the packed bed were registered with K-type thermocouples. The general characteristics of the experimental facilities are stated in
Table 1. Gaseous compound concentrations (O
2, CO, CO
2, CH
4, and H
2) were registered with a NOVA
® 975A in-line gas analyser (NOVA ANALYICAL SYSTEMS
®, Hamilton, ON, Canada). In addition, an overall image of the experimental facility is presented in
Figure 2.
The experiments consisted of performing continuous operation of the gasifier in order to gather the syngas quality information once steady-state conditions had been achieved. Unvarying conditions were ensured by implementing a 20-h stabilization period, and the experiments were designed to last 90 min without significant variations in the monitored operating variables. The gasifier operates in atmospheric pressure ranges.
Two types of experiments were carried out in steady-state conditions in order to assess the effect of packed-bed material under different oxygen contents. The first one utilized only air as the oxidizing agent, and the second one, air was combined with highly-concentrated oxygen. Each experiment was repeated three times in order to ensure their repeatability.
All experiments were performed with and without the inclusion of the packed material in the upper chamber to assess its effect. The first set of experiments was developed by using a blend of oxygen (99%
v/
v O
2) and air. The second round of experiments was carried out exclusively with ambient air. Biomass feed rate and reflux flow rate were maintained under similar conditions. The assays considered the operating conditions in
Table 2 and, moreover, thermal images of the upper chamber were registered to complement the temperature measurement
The configuration of the gasifier takes account of the fact that gasification performed in counter-current (as in updraft gasifiers) implies that the drying, pyrolysis, and gasification stages are clearly differentiated by the vertical temperature profile achieved during operation. Moreover, the contact between gaseous and solid phases favours the formation of energy vectors that are mainly generated through endothermic reactions [
36]. A description of the chemical reactions that compose the gasification process is presented in
Table 3. It is recognised that the operation generates a temperature gradient from the reduction zone (high temperature) to the upper part of the packed bed (low temperature). Such a phenomenon is caused by the heat exchange between the ascending syngas and the descending solid biomass. Hence, the location of the packed bed it expected to exploit catalytic and CO
2 adsorption processes that occur at low temperatures in order to improve the syngas quality.
Regarding the bulk material used for the upper packed bed, it is worth mentioning that the average particle size was 0.75 cm and was mainly composed of zeolite particles previously prepared by catalytic material impregnation (Raney nickel) and thermal activation. During the experimental assays, the temperature of the syngas that reaches the lower part of the packed bed was also registered with the purpose of verifying if this condition is appropriate for the catalytic and CO2 adsorption phenomena.
Concerning the feedstock blends prepared for the experiments, it should be mentioned that co-gasification between MSW and charcoal was proposed with the aim of exploiting the presence of MSW humidity as a water steam source. In such a context, the purpose of adding a significant proportion of a dry fuel, such as charcoal, is to ensure water evaporation inside the gasification reactor. Operating with blends of MSW and charcoal aims to obtain more significant syngas calorific values. Such an effect has been reported, since co-gasification allows improvement of the syngas quality due to the combination of volatiles and water steam in the gas phase, during gasification [
38].
Feedstocks with considerable humidity and volatile carbon content (suitable for tar generation) can be combined with feedstocks with high fixed carbon content (characterized by more significant heating values and significant content of reactants proper for reduction reactions) with the purpose of enhancing the tar conversion into syngas [
39]. In addition, wet-basis humidity in MSW can behave as steam at reaction conditions and react with carbonous surfaces to boost hydrogen production, as in steam gasification [
40,
41,
42]. Samples were prepared with an optimal 3:2 mass proportion (charcoal and MSW), correspondingly. This figure was chosen with the purpose of maximizing the energy vector content in syngas [
38,
43,
44].
Biomass feedstock batches were prepared by blending chopped MSW and vegetable charcoal made of palm oil kernel shell. Particle size was fixed at approximately 1 cm effective diameter. This particle size was selected since it has been reported to increase syngas yield (3.2%) and cold gas efficiency (CGE) (3.4%) during gasification, if compared to larger diameters [
4]. The feedstock for these assays (MSW) was obtained from the rural community San Pedro del Laurel, Ecuador, which is a place that would benefit from the implementation of the gasification facility. MSW was sampled according to the standard method NT ENVIR 001 [
45]. Proximate and ultimate analyses of batch samples were performed in all cases with standard laboratory methods.
Even if different biomass characteristics are expected to affect the output properties after a thermal decomposition process [
46], it is also possible to achieve homogenous properties for the output products by controlling the operation variables [
40]. Results were compared with reported figures of different gasification facilities found in the literature.
The laboratory tests performed as part of this research followed the standard procedures listed in
Table 4, together with the laboratory equipment.
3. Results
As was mentioned, the test feedstock was composed of vegetable charcoal and MSW. Considering MSW can contain several components suitable for being differentiated by primary sorting, this initial activity was undertaken, and the results are reported in
Table 5.
Sorting results show that kitchen waste is the dominant component in MSW. Regarding proximate and ultimate composition, the data generated for MSW and kernel shell charcoal are presented in
Table 6 and
Table 7, respectively. Data obtained from the literature is used for the purpose of comparison.
Results of the experimental assays are presented in
Table 8. The figures present cold gas efficiency (CGE) values as well. This factor relates the inlet feedstock heating value with the outlet syngas heating value for explaining the thermal efficiency of the process [
50].
To compare syngas compositions with reported data from literature sources, data were homogenised to a nitrogen-free basis, and the CO/CO
2 figures were calculated. The values are presented in
Table 9.
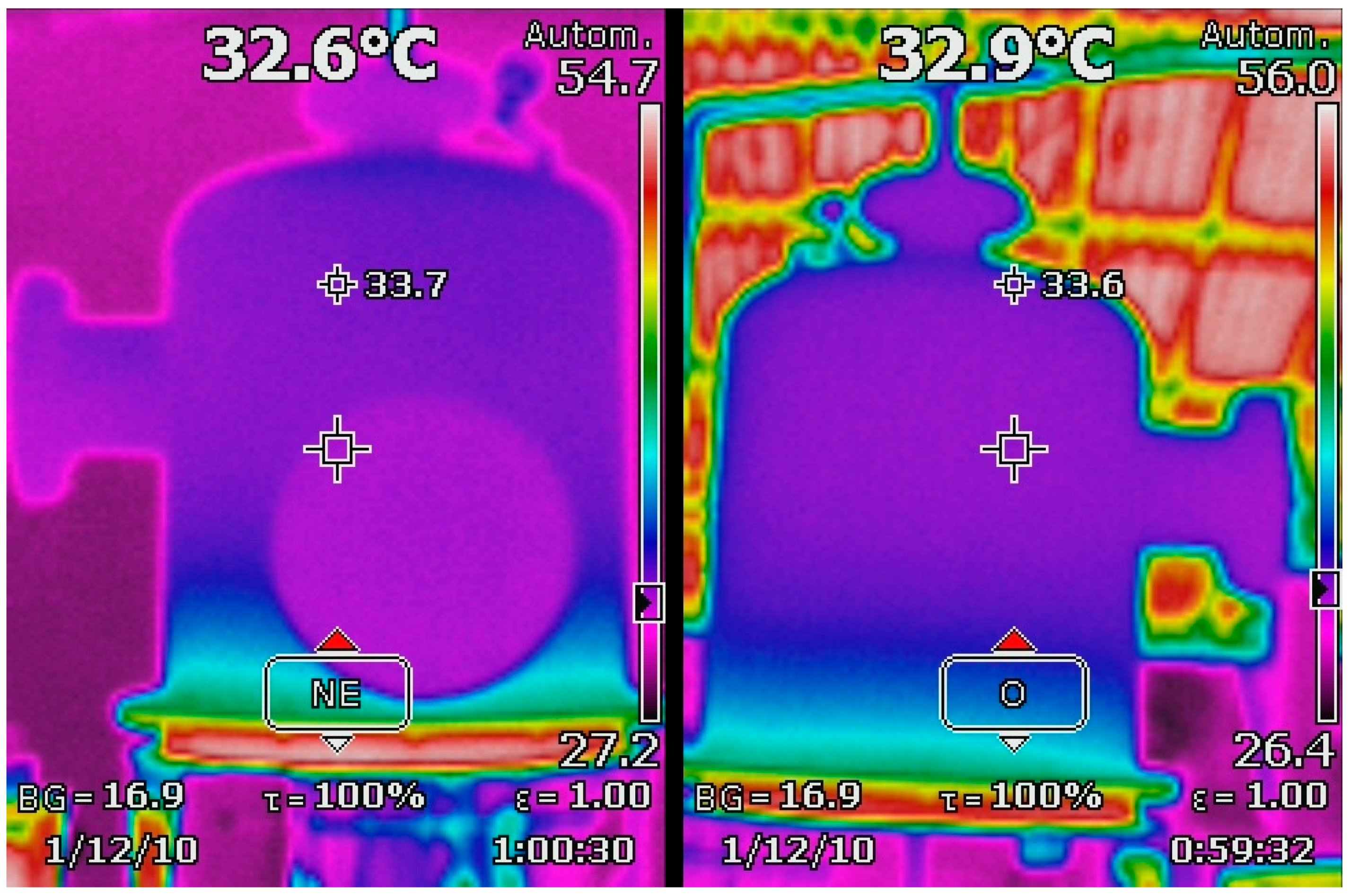
Even if the packed bed activity is considered to be a relevant research topic by itself, in addition to its study in the context of the gasification case presented, thermal images were recorded reflecting the upper temperature range in order to show any potential activity to be reported. Thermal images of the upper chamber during O
2 + air operation (front and side) with packed material addition are shown in
Figure 3.
The implementation cost figures are presented for the purpose of comparing the economic incidence of this alternative if compared with other syngas enhancement methods. Even if the figures were obtained in a pilot facility, relative cost factors are shown in
Table 10. It is relevant to mention that such figures, in the case of implementation costs, were obtained from the construction report carried out during the building stage of the experimental gasification facility. Since the upper chamber and the remaining parts of the gasification reactor have similar structures, the cost factors were obtained by using unit costs and relative dimensions. The baseline cost (cost factor = 1) refers to the gasifier without the upper chamber required for implementing the shifting proposal.
In the case of the operating costs, the variation in the cost factor is calculated by considering additional worker-hours, plus supplies required for preparing the packed material.
4. Discussion
Regarding the solid fuel that is used in these assays, a certain similarity with other reported figures can be noticed. Even if kitchen waste is not the only component of MSW, in this case, several properties of MSW, such as volatile carbon, are similar to the reported values of other types of biomass with a single component. In contrast, MSW presents a low fixed carbon content. Hence, it requires being blended with charcoal to improve its performance as fuel [
43,
44]. In this case, the charcoal stream is 1.5 times greater than the humid MSW and 18.13% of the total solid input was composed of free water in such conditions. Hence, the energy input contributed by the charcoal exceeds 37 times the energy requirement that the free water would require for evaporating. These figures show that it is possible to consider that evaporation was, indeed, one of the phenomena involved in gasification during the registered assays. Moreover, such consideration supports the consideration adopted about neglecting drying-type pre-treatment stages in MSW gasification. The presented figures show that co-gasification is a suitable alternative for MSW since it enables its embedded water to be used as one of the reactants, rather than as a component to be removed.
In addition, fuel-processing figures could be related to the water contained in the inlet solid fuel. Steam co-gasification processes are commonly associated with significant hydrogen production rates and with the presence of significant amounts of fixed carbon [
51]. Therefore, it is expected to generate proper conditions for exploiting the water presence by adding charcoal and wet biomass blends instead of considering pre-treatment stages based on drying. The effect of avoiding this type of pre-treatment is in accord with this consideration, and has been reported for other types of organic materials, such as rubber [
52]. In this case, the combination is expected to demonstrate the suitability of considering local MSW as a proper energy source to be converted in the gasifier. Steam gasification and methane reforming processes consider water steam as one of the reactants together with solid carbon and gaseous methane, correspondingly. Moreover, it has been affirmed that this type of gas-phase reaction is the one that generates the most significant hydrogen production rate [
38].
Concentrated oxygen addition increases the partial pressure of this component in the reacting gas stream, which also reduces the partial pressure of inert gases during gasification, such as nitrogen. Even if air can act as an oxidising agent, O
2 addition is expected to reduce heat losses due to inert gases heating, and to increase the reaction rate due to the augmentation of the concentration of one of the reactants. Both effects can increase gasification temperature; hence, H
2 and CO formation can be boosted in comparison with gasification fed with air only. Similar effects of adding O
2 have been reported previously [
53].
The design of the gasifier coupled an initial gasification stage formed by an updraft configuration with gas treatment in series. The gas treatment was specifically designed to improve the syngas quality, i.e., to increase the content of energy vectors, such as H2, CO, and CH4. Accepting that enhancing the energy vectors contents in the syngas is a suitable treatment to improve the gasifier’s performance, it should be mentioned that a packed-bed able to host catalytic processes and carbon capture related to CO2 appears to be a suitable alternative when compared with other gas-shift options. The presence of the packed-bed allowed the achievement of promising figures related to syngas quality compared with other proposals with the same aim.
Even if the lower part of the gasifier initiated the solid-gas conversion, as was verified by the high temperature recorded, an extra stage for gas reforming and CO2 retention was also considered in order to enhance syngas quality. Both effects were induced in the same packed bed by putting the catalytic material over a support base that can accomplish this primary purpose together with CO2 adsorption. Despite the fact that generating a heterogeneous behaviour along a single structure such as a packed bed could be a non-desirable effect (CO2 adsorption with molecular sieve and catalysed chemical changes are mutually exclusive surface phenomena, and are planned to occur in different zones of the same packed-bed), the construction and operation of an atmospheric gasifier coupled with a packed-bed structure is, a priori, more accessible than a pressurized gasifier case. If both technological alternatives could deliver similar outputs, it would be expected that avoiding high-pressure conditions would result in a low-complexity option.
The comparative analysis of results shows that the chemical phenomenon can be achieved by inducing effects such as gas retention time growth by the presence of a porous medium, and also due to the catalytic activity provided by the active material adsorbed over its surface (Raney nickel) [
54,
55]. The occurrence of the chemical phenomena responsible for the gaseous compounds reforming was generated by the presence of the nickel-based catalyst [
56], and boosted by the physical effects induced by the porous media (gas retention time increase and augmentation of the interfacial area exposed to the catalyst), as has been verified in packed-bed reactors.
Additionally, it can be confirmed that wet MSW co-gasification enhances hydrogen formation even if feedstock composition is variable. This is shown by the relatively high figures for the standard deviation of all proximate analysis components when compared with the reported results of experiments carried out with fixed types of biomass and assumed low variability in their composition figures. The statement is also supported by the fact that hydrogen content was significant, although the ultimate composition of this element in feedstock was relatively low. Accepting that MSW is a poor feedstock material in terms of quality compared with other bio-based materials such as the ones used in this work for comparison purposes, the validation of the above-presented technology could be assessed with a more suitable solid fuel in order to reduce the test differences related with the inputs.
Although the effect of the presence of the packed bed in syngas does indeed show an improvement in its quality, it is necessary to determine the predominant phenomenon that leads to achieving this outcome. Even if the catalytic activity is recognised as a reduction of the activation energy of a chemical reaction and it can be verified by the increase of chemical conversion rate [
57], effectiveness, efficiency, and cost are recognised as the selection criteria [
58]. Although Raney nickel was used in this work due to its cost effectiveness and availability, it is suitable to be reproduced with other catalytic materials adsorbed in the packed bed to vary the CO/CO
2 ratio, such as minerals containing calcium hydroxide [
42]. On the other hand, CO
2 capture has been successfully proved at temperature ranges between 30 °C and 100 °C for silica-based adsorbents [
59]. Moreover, low-temperature adsorption has also been achieved with ceramics [
60]. Hence, it would be consistent to consider that it is also possible with zeolite-based materials (aluminosilicate-based substances as ceramics), as in this case. The temperature range achieved in the lower part of the packed bed shows that both catalytic reforming and CO
2 adsorption over zeolite are suitable in the operating conditions that were achieved. According to Cunha et al. [
61], water conversion into hydrogen through steam gasification reactions start being verified at around 120 °C with nickel-based catalysts, even if full conversion would require temperatures in the range of 200 °C. Moreover, CO
2 adsorption over zeolite has also been reported at atmospheric pressure levels and temperature ranges around 100 °C. Zhao et al. [
3], for instance, reported that zeolite can adsorb CO
2 over its surface with efficiencies above 5% under relatively low pressure and temperature levels. Considering such reported records together with the temperature registers, it is possible to affirm that the packed bed has activity in the syngas quality obtained from the gasifier. Such activity could be noticed due to the temperature drop registered during gasification. Even if the inner temperature sensor showed that it was possible to achieve temperature figures above 120 °C, the thermal images show that it dropped to approximately 33 °C in the upper sections of the packed bed. Even if this phenomenon can be related to the syngas shifting, it is affirmed that its specific performance requires further study.
Regarding the experimental oxygen contents, even if the energy carriers’ content were increased in syngas, it is also shown that the remnant oxidizing agent is significant if compared with the reported results of other syngas quality improvement alternatives. This fact leads to the consideration that several operational parameters, such as oxygen feed and reflux ratio, need to be optimised in further studies and tests. From the chemical reaction engineering perspective, the reported operation could have been performed with an excess of one of the reactants, or the retention time could have been larger for increasing the conversion rate of oxygen. Hence, further experiments could consider a larger reflux ratio, which would imply a higher retention time; moreover, the effect of reducing the oxygen feed rate should be assessed for determining whether such variation affects the performance of the operation. It is also expected that a variation of this type enhances the obtained figures of cold gas efficiency in all cases, which is related to the energy vectors content increase [
62].
Concerning the economic feasibility of the presented syngas improvement alternative, the relative costs figures show an increase of 4.3% if compared with a gasifier without the upper chamber required. In addition, CGE can be increased in at least 24.6% while CO/CO
2 ratio can be increased up to 3.5 times. Based on this figure and the above-mentioned effectiveness, it is possible to affirm that including a packed bed in the upper part of the gasifier is a suitable alternative that can be combined with other proposals without increasing the implementation costs. In addition, the link between performance and profitability [
63] shows that the CGE improvement (achieved by including the upper packed bed in experimental assays) should be validated in real operation, based on the presented results.