1. Introduction
Fossil resources have been treated as an easy source of useful energy for thousands of years. As a result, increasing rates of consumption threaten its reserves. Besides, problems associated with climate change and greenhouse gas (GHG) emissions have rendered their application a great global problem. Therefore, it is urgent to seek alternative energy sources to meet the demand of fuel energy [
1]. In recent years, biodiesel has become an attractive alternative energy source, due to its fuel properties, cost-effectiveness, eco-friendliness, biodegradability, and renewability [
2]. It consists of fatty acid monoalkyl (methyl or ethyl) esters, obtained from vegetable oils and animal fats by esterification and transesterification with alcohols in the presence of (acid/base) catalysts [
3,
4].
The feedstock sources are the most important aspect of biodiesel production. Both edible and non-edible oil feedstocks are widely studied to produce biodiesel. Food security, shortage of agricultural land and cost of production are mainly responsible for feedstocks not being used on a commercial scale. Thus, at present, waste oil and animal fat has become an attractive replacement for conventional biodiesel feedstock sources. Furthermore, investigations show that nearly 70% of the total production cost of biodiesel is related to these feedstock oils [
5,
6]. Moreover, using waste cooking oil (WCO) as a source of fuel for commercial biodiesel production will not only reduce the raw material cost, but will also solve the problems related to its disposal in the environment [
7,
8].
The production-process optimization and improvement of biodiesel quality are other more salient issues that make it commercially viable. To get maximum biodiesel yields commercially, homogeneous acid/base catalysts have been commonly used for biodiesel production through transesterification processes. The two important factors for the wide use of homogeneous catalysts are the mild reaction condition and the fact that they are less energy intensive. However, this requires a superior quality of feedstocks to get high quality biodiesels with a maximum yield. When dealing with inferior grade feedstocks, the removal of the catalyst from the reaction mixture, soap formation, and downstream processing costs all limit their application [
8,
9]. To overcome this problem, a non-catalytic transesterification by supercritical methanol method (SCM) was used, that can handle feedstocks having impurities such as high free fatty acid (FFA) levels and moisture content. However, it involves a high capital cost, and requires an intensive temperature–pressure environment and a specialized alloyed reactor [
10].
Recently, a two-step process, hydrolysis followed by non-catalytic esterification of FFA, has been revealed as an excellent way to use waste cooking oil. Very few studies have been performed in this process for managing WCO, and more work is still needed to achieve a better quality biodiesel with less effort. As a result, the exploration of the performance under the influence of a heterogeneous catalyst in the esterification step is required for the optimization of the reaction. The use of a heterogeneous acid catalyst plays an important role in the production of biodiesel from inferior grade feedstocks. Since these catalysts are fully separable, this ensures the avoidance of product contamination. Additionally, heterogeneous catalysts are regenerable and reusable several times with good efficiency [
4,
7,
10]. Besides, solid acid catalysts have also gained attention as an alternative to heterogeneous base catalysts and classical homogeneous acid catalysts [
10,
11,
12].
There is some studies that used some solid acid catalysts such as ion exchange resins, zeolites and zeotype materials in biodiesel production. The crucial advantage of using these catalysts is the elimination of the final biodiesel washing step. However, more work in the search for new solid acid catalysts for sustainable biodiesel production is required [
13,
14,
15].
Sulfate-incorporated metal oxides, which belong to the category of solid superacids, are now receiving more attention, with the aim of replacing the conventional homogeneous base catalysts which are highly corrosive, hazardous, and environmentally polluting. Solid superacids have an advantage in the strength and type (Brönsted or Lewis) of their acidity [
16]. In addition, nowadays, functionalized mesoporous silica-based catalysts have been used in the esterification process because of their extremely large surface area, uniformity, stability, and large pore size, all of which are very beneficial in reactions involving bulky molecules [
17,
18].
To date, several heterogeneous acid catalysts have been developed for biodiesel production by transesterification of waste cooking oil, such as ZS/Si, SO
42−/TiO
2-SiO
2, carbon-based catalysts derived from starch and Zeolyte (Y756) [
19,
20,
21,
22]. However, further study on solid acid catalysts and process optimization are still required.
In this study, we investigated how the application of a mesoporous superacid catalyst sulfated zirconium oxide supported on SBA-15 (S-ZrO2/SBA-15) is expected to enhance the reaction rate and will be beneficial to the reaction involving bulky molecules. We also expected that this catalyst will be environmentally friendly, because of its good separation from the reaction mixture.
To the best of our knowledge, no work has yet used the S-ZrO2/SBA-15 catalyst in biodiesel production through an esterification from hydrolyzed waste cooking oil. In this work, a heterogeneous solid superacid catalyst S-ZrO2/SBA-15 was synthesized and applied to biodiesel production under subcritical methanol process conditions. The impact of the reaction parameters—methanol-to-oil mole ratio, temperature, time, and catalyst loading—were studied to optimize the esterification method to obtain high yields of biodiesel. The reusability of the catalyst was examined, to investigate the stability of the catalyst in the reaction.
2. Research Methodology
2.1. Chemicals
Deionized water was obtained from a Milli-Q reverse osmosis purification system (Fisher Scientific, Bedford, MA, USA). Anhydrous sodium sulfate 98.5% was purchased from Samchun Pure Chemical Co., Ltd. (Pyeongtaek, Korea). Reagent grade ethyl alcohol (C2H6O, anhydrous 99.9%), diethyl ether (C2H5)2O, >99.0%), and potassium hydroxide (KOH, >85.0%) were purchased from Daejung Chemical & Metals Co., Ltd. (Gyeonggi-do, Korea). All chemicals were of analytical grade and used without further purification. For the catalyst preparation, hydrochloric acid (HCl) and Pluronic P123 (HO(CH2CH2O)20(CH2CH(CH3) O)70(CH2CH2O)20H) were obtained from Sigma-Aldrich Co., (St. Louis, MS, USA). The zirconium oxide precursor was purchased from Daejung Chemical & Metals Co., Ltd. Tetraethyl orthosilicate (C8H20O4Si) ≥ 98%) was purchased from ACROS Organics Co., (Morris, NJ, USA). These were reagent grade chemicals.
2.2. Raw Materials
Waste cooking oil (WCO) was collected from restaurants and shops within Sangji University, Wonju City, South Korea, and processed by vacuum filtration to remove inorganic residues. Following this, it was heated at the water boiling point for half an hour to evaporate any remaining water, and a drying step using sodium sulfate was performed. Until the FFA production, the WCO was stored under ambient conditions, after which its properties were analyzed (
Table 1).
2.3. Catalyst Preparation
Sulfonated zirconium oxide supported by SBA-15 (S-ZrO2/SBA-15) was synthesized by direct impregnation of the synthesized SBA-15 with the desired amount of zirconium hydroxide, according to the following method: Pluronic P123 was dissolved in deionized water, followed by 2 M HCl solution, and stirred for 3 h at 40 °C. After full dissolution, Zr(OH)2 was added and stirred for 24 h at 40 °C. Then, the silica source tetraethyl orthosilicate (TEOS) was added to the mixture, which was stirred for 30 min at 40 °C. The product was transferred to a Teflon lined bottle, and aged in the oven for 24 h at 80 °C under static conditions. After this, the product was filtered with Whatman micro filter paper, and washed with deionized water, and dried at 105 °C for 24 h. The product was calcined for 6 h at 540 °C. Then, the calcined product was sulfated with 1.0 M H2SO4 (15.0 mL/g) at room temperature for 1 h. Finally, the sulfated material was filtered, dried at 105 °C for 24 h, and calcined again at 550 °C for 4 h.
2.4. Processing into Biodiesel
2.4.1. Hydrolysis of Feedstock Oil
A 350 mL 316 stainless steel batch reactor vessel with a glass liner was used for both the hydrolysis and esterification reactions (Hanwoul Eng. Co., Ltd., Model HR-8302, Gyeonggi-do, Korea). Usually, 150 g substrate was charged into the reaction vessel for each run at 275 °C for 45 min, and the rate of agitation was 500 rpm. For hydrolysis, water was taken into the reaction vessel according to the (WCO/water) 1:1.006 ratio (weight basis). After hydrolysis, the reaction mixture was extracted with diethyl ether. The products were separated into two different phases (FFA and glycerol) in a separating funnel. The ether was evaporated to leave an oil consisting of the hydrolyzed free fatty acids (FFA), and a dry wash was performed using anhydrous sodium sulfate to absorb the remaining water. Following this, to determine the acid value, saponification value, and the degree of hydrolysis, the extracted FFA was taken for a volumetric titration procedure.
2.4.2. Esterification of Hydrolyzed Free Fatty Acid
The extracted FFA was taken for methyl esterification under subcritical conditions. After this, a similar experimental procedure as described above was followed. A reaction under non-catalytic conditions was also performed, to compare it with the catalytic process during the esterification of FFA.
The conversion rate for the esterification process was calculated with the following formula:
where M = acid value of free fatty acid (FFA) and N = acid value of produced biodiesel.
2.4.3. Purification of Biodiesel
After completing the reaction, catalysts were separated from the reaction mixture by a filtration process. Then, the reaction mixture was placed in a separating funnel. The reaction mixture was heated to recover the non-reacted methanol for further use. Some amount of sodium sulfate was taken in a funnel to perform dry wash; this was to absorb the water, produced from the reaction, present in the biodiesel. Finally, the product was taken in order to analyze its various fuel properties.
2.4.4. Experiment Design for Esterification of Waste Cooking Oil to Optimize Biodiesel Yield
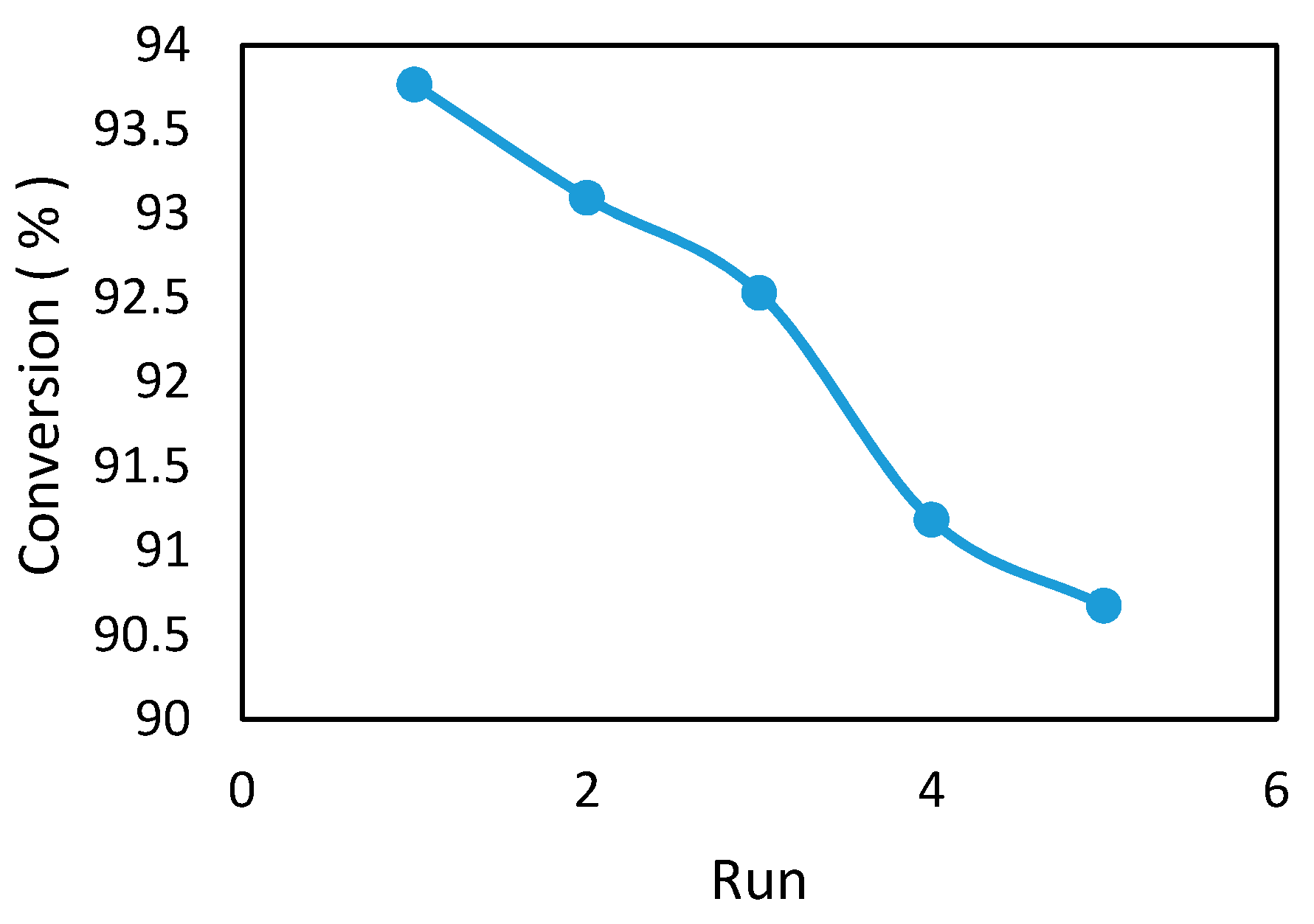
The statistical design selected for the optimization of biodiesel production from the waste cooking oil was defined by a custom full factorial design using the Minitab18 software (Eretec Inc., Gunpo-Si, Gyunggi-Do, Korea). The biodiesel yield was selected as a response for this method. The experimental range and levels of the studied factors are listed in
Table 2.The experimental matrix for the factorial design and results are shown in
Table 3. The experimental range and levels of the studied factors were chosen according to the previous work in this laboratory [
2]. The experimental range of the studied factors were established via an error and trial method.