Effect of Time on a Hierarchical Corn Skeleton-Like Composite of CoO@ZnO as Capacitive Electrode Material for High Specific Performance Supercapacitors
Abstract
:1. Introduction
2. Experimental Methods
2.1. Chemicals and Materials
2.2. Preparation of ZnO and CoO on Nickel Foam
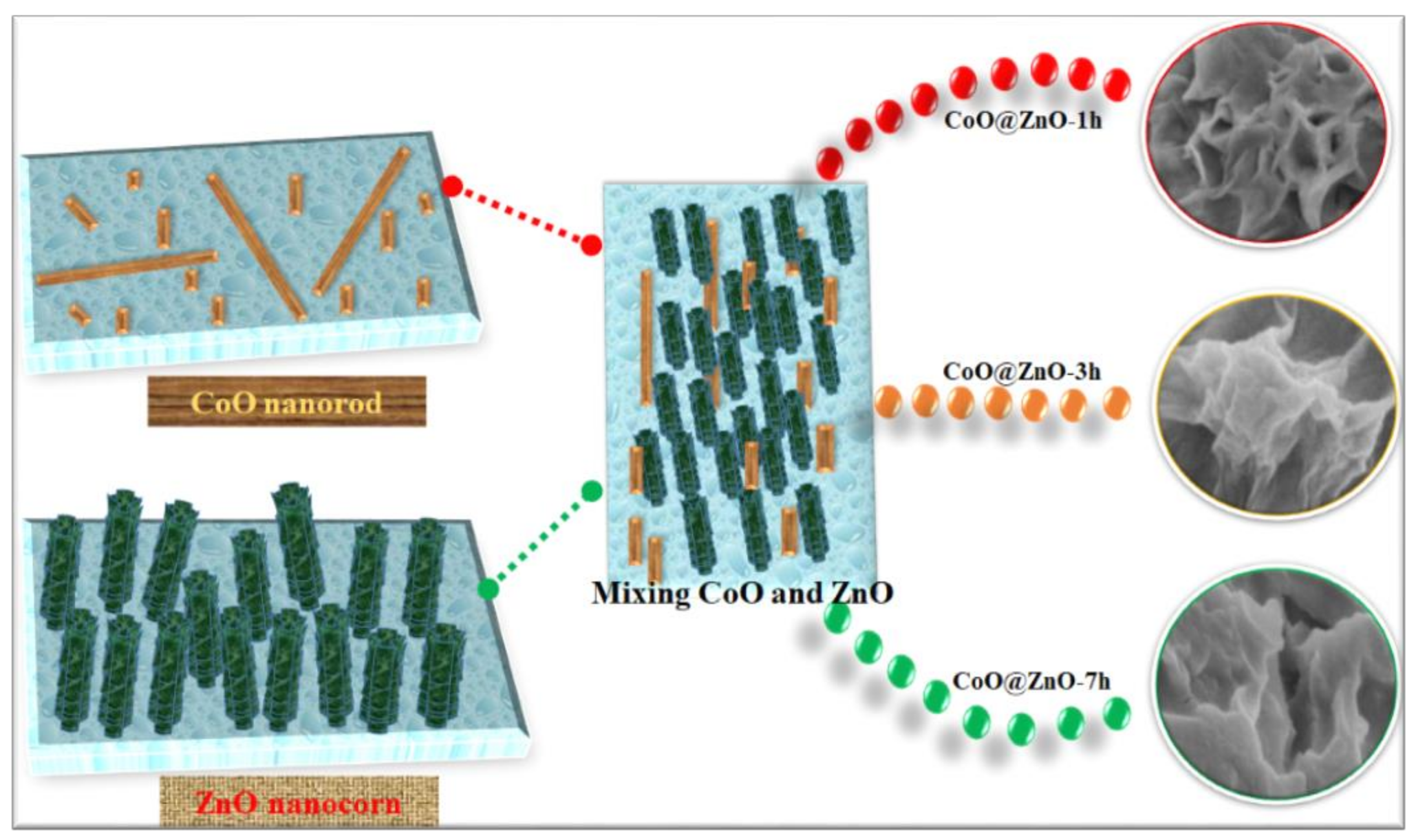
2.3. Preparation of CZ Composite Nanostructure
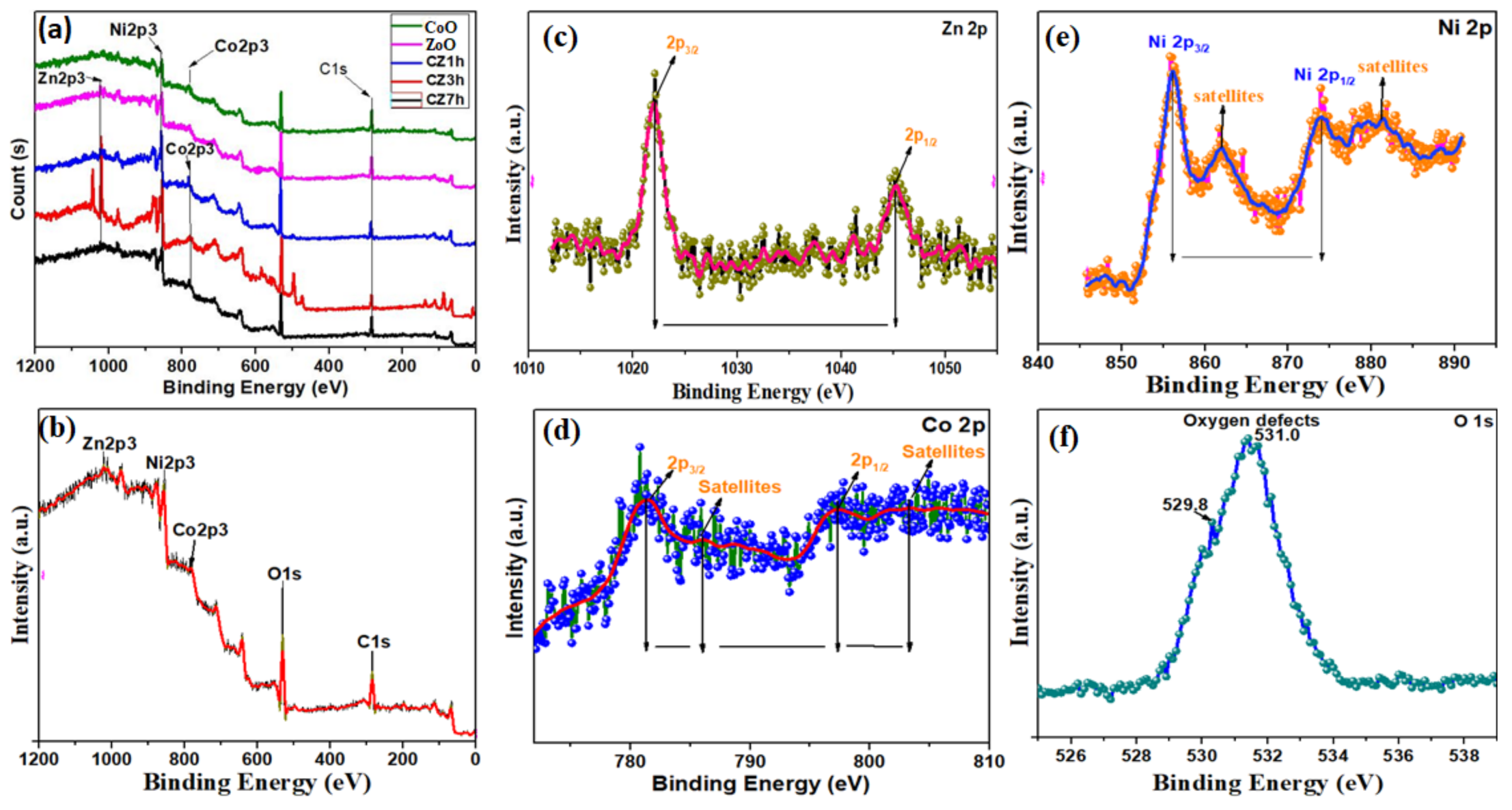
2.4. Characterization
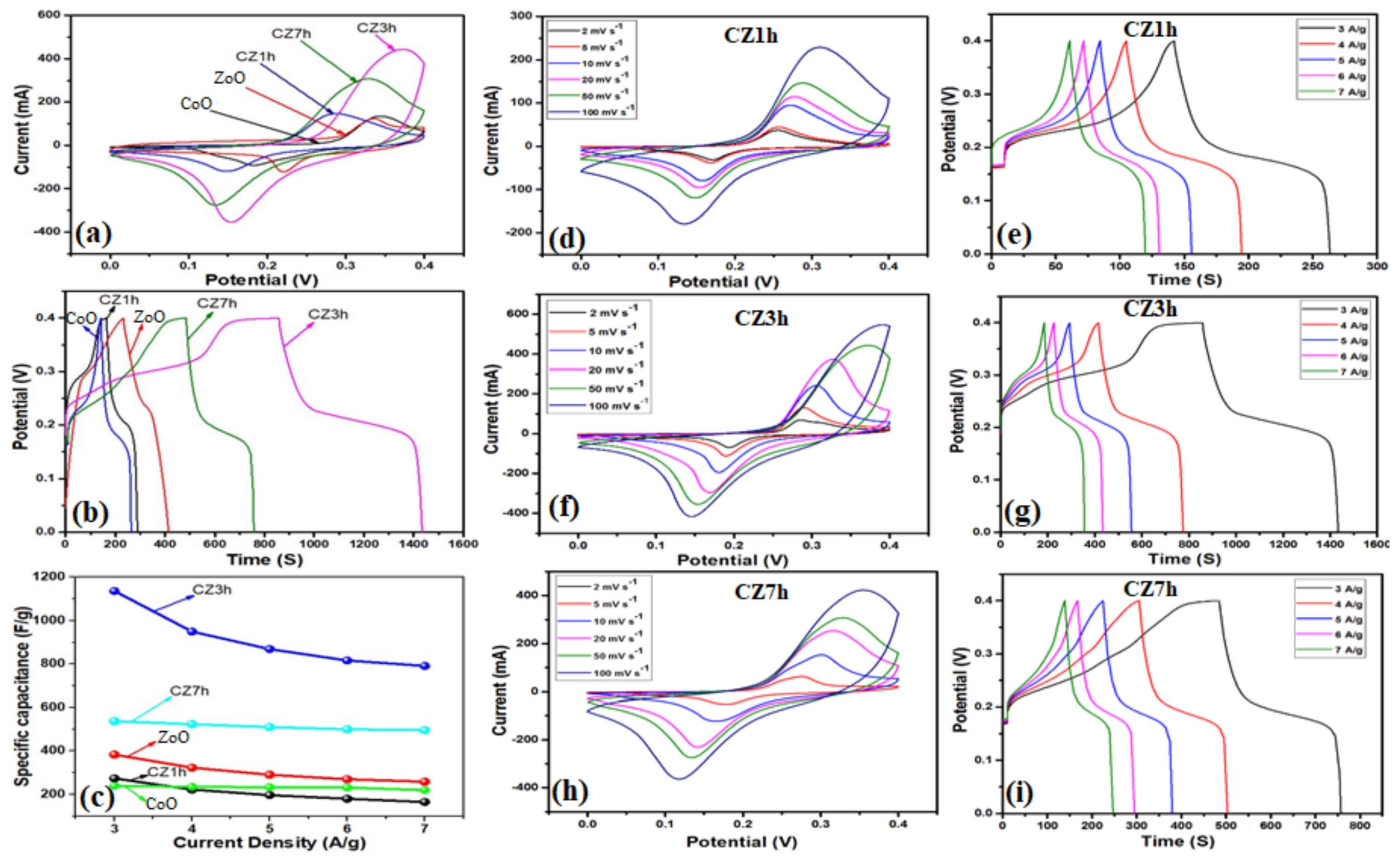
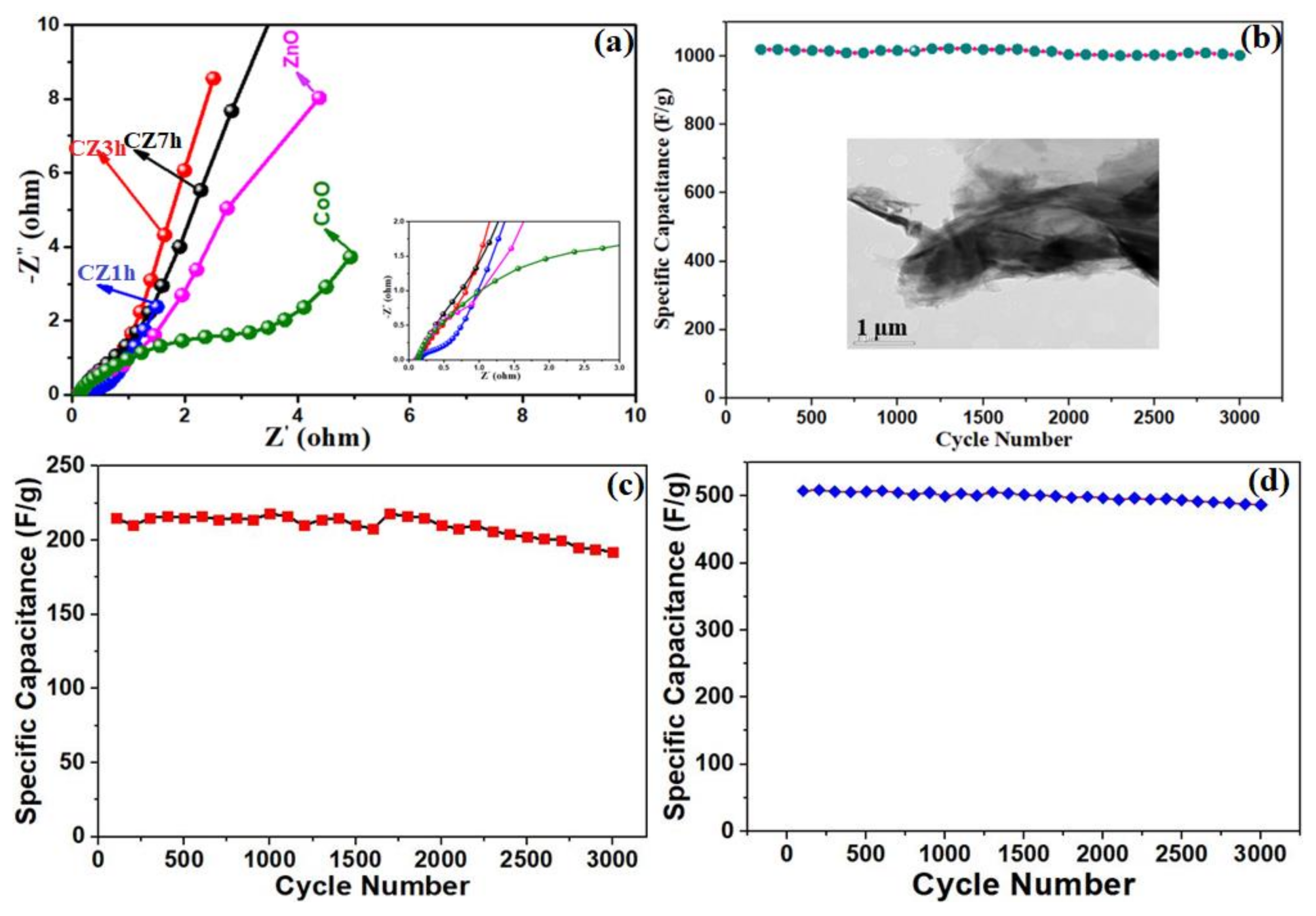
2.5. Electrochemical Characterization
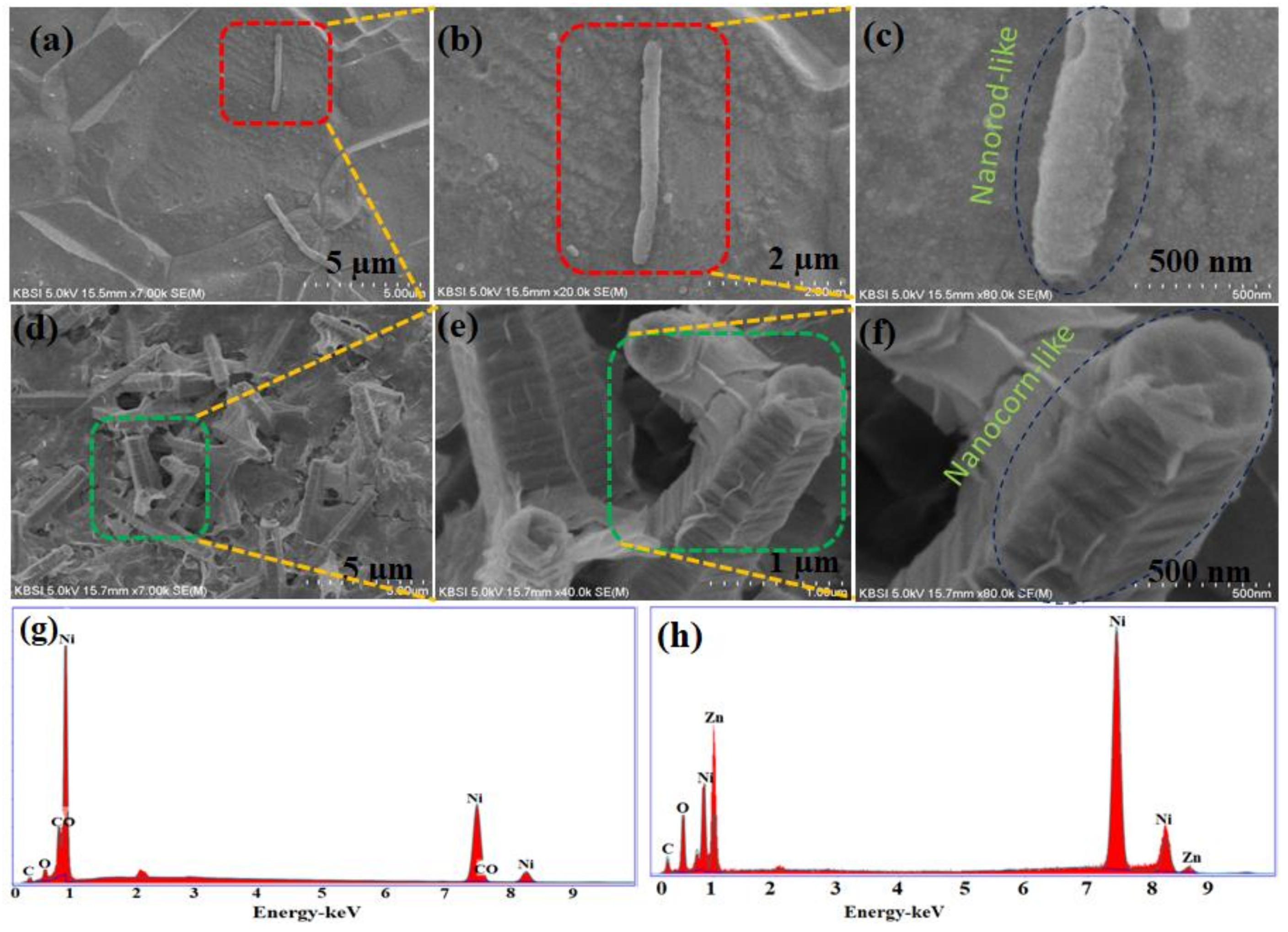
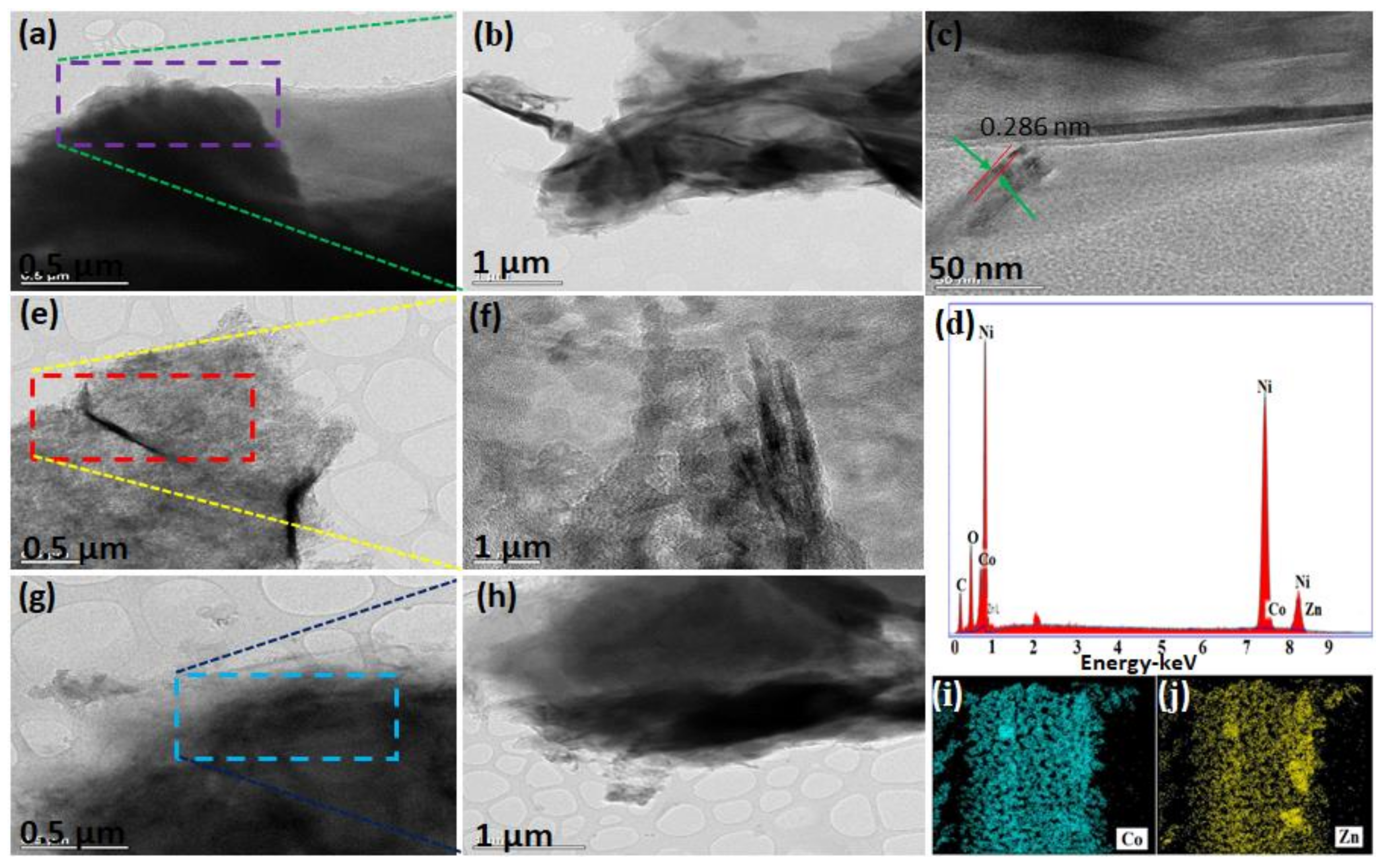
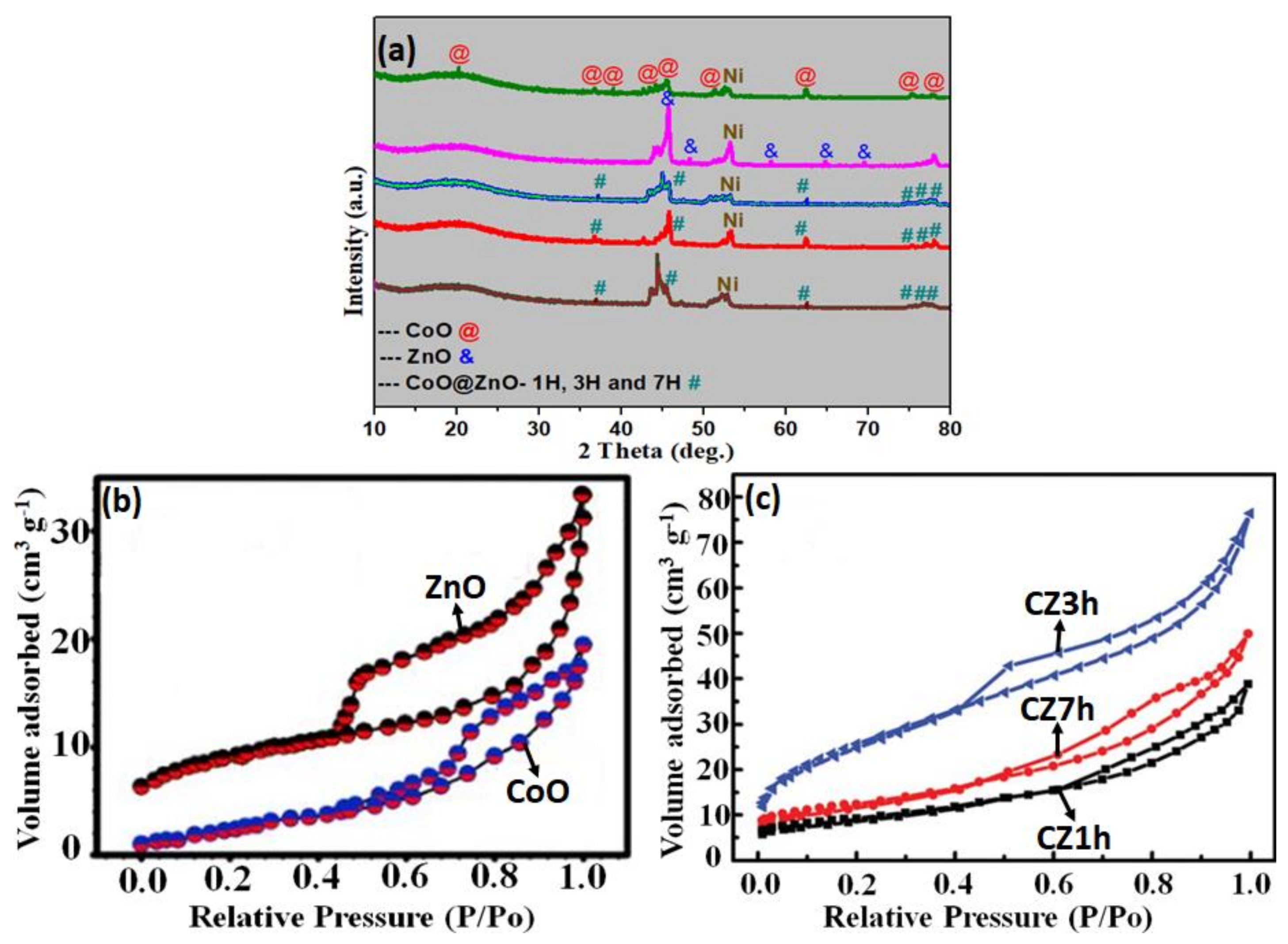
3. Results and Discussion
Characterization and Electrochemical Performance of Active Electrodes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liao, J.; Zou, P.; Su, S.; Nairan, A.; Wang, Y.; Wu, D.; Wong, C.-P.; Kang, F.; Yang, C. Hierarchical nickel nanowire@NiCo2S4 nanowhisker composite arrays with a test-tube-brush-like structure for high-performance supercapacitors. J. Mater. Chem. A 2018, 6, 15284–15293. [Google Scholar] [CrossRef]
- Cai, D.; Huang, H.; Wang, D.; Liu, B.; Wang, L.; Liu, Y.; Li, Q. High performance supercapacitor electrode based on the unique ZnO@Co3O4 core/shell heterostructures on Nickel foam. ACS Appl. Mater. Interfaces 2014, 6, 15905–15912. [Google Scholar] [CrossRef] [PubMed]
- Gobal, F.; Faraji, M. Preparation and electrochemical performances of nanoporous/cracked cobalt oxide layer for supercapacitors. Appl. Phys. A 2014, 117, 2087–20194. [Google Scholar] [CrossRef]
- Kumar, Y.A.; Rao, S.S.; Punnose, D.; Tulasivarma, C.V.; Gopi, C.V.V.M.; Prabakar, K.; Kim, H.-J. Influence of solvents in the preparation of cobalt sulfide for supercapacitor. R. Soc. Open Sci. 2017, 4, 170427. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ma, M.; Yang, J.; Su, H.; Huang, W.; Dong, X. Selective synthesis of hierarchical mesoporous spinal NiCo2O4 for high-performance supercapacitors. Nanoscale 2014, 6, 4303–4308. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wan, Z.; Yang, T.; Zhao, M.; Lv, X.; Wang, H.; Ren, X.; Mei, X. Preparation of nickel cobalt sulfide hollow nanocolloids with enhanced electrochemical property for supercapaictors applications. Sci. Rep. 2014, 6, 25151. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Sun, H.; Zhu, J.; Bi, H.; Han, Q.; Liu, X.; Wang, X. Graphene-based cobalt sulfide composite hydrogel with enhanced electrochemical properties for supercapcitors. New J. Chem. 2016, 40, 2843–2849. [Google Scholar] [CrossRef]
- Kandalkar, S.G.; Gunjakar, J.L.; Lokhande, C.D. Chemical synthesis of cobalt oxide thin film electrode for supercapacitor application. Appl. Surf. Sci. 2008, 254, 5540–5544. [Google Scholar] [CrossRef]
- Liu, S.; Mao, C.; Niu, Y.; Yi, F.; Hou, J.; Lu, S.; Jiang, J.; Xu, M.; Li, C. Facile synthesis of novel networked ultralong cobalt sulfide nanotubes and its application in supercapacitors. ACS Appl. Mater. Interfaces 2015, 7, 25568–25573. [Google Scholar] [CrossRef] [PubMed]
- Yedluri, A.K.; Kim, H.-J. Wearable super-high specific performance supercapacitors using a honeycomb with folded silk-like composite of NiCo2O4 nanoplates decorated with NiMoO4 honeycombs on nickel foam. Dalton Trans. 2018, 47, 15545–15554. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.-J.; Zhang, J.Z.; Shi, G.W.; Liu, Y.-M. One step hydrothermal synthesis of two-dimentional cobalt sulfide for high performance supercapacitors. Mater. Lett. 2014, 131, 45–48. [Google Scholar] [CrossRef]
- Lin, J.-Y.; Chou, S.-W. Cathodic deposition of interlaced nanosheet-like cobalt sulfide films for high-performance supercapacitors. RSC Adv. 2013, 3, 2043–2048. [Google Scholar] [CrossRef]
- Cheng, J.; Lu, Y.; Qiu, K.; Yan, H.; Xu, J.; Han, L.; Liu, X.; Luo, J.; Kim, J.K.; Luo, Y. Hierarchical core/shell NiCo2O4@NiCo2O4 Nanocactus arrays with dual-functionalities for high-performance supercapacitors and Li-ion batteries. Sci. Rep. 2015, 5, 12099. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Tetard, L.; Zhai, L.; Thomas, J. Supercapacitor electrode materials: nanostructures from 0 to 3 dimensions. Energy Environ. Sci. 2015, 8, 702–730. [Google Scholar] [CrossRef]
- Dong, W.; Wang, X.; Li, B.; Wang, L.; Chen, B.; Li, C.; Li, X.; Zhang, T.; Shi, Z. Hydrothermal synthesis and structure evolution of hierarchical cobalt sulfide nanostructures. Dalton Trans. 2011, 40, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Ma, L.; Huang, S.; Mei, J.; Li, E.; Yuan, G. Large areal mass and high scalable and flexible cobalt Oxide/Graphene/Bacterial cellulose electrode for supercapacitors. J. Phys. Chem. C 2016, 120, 28480–28488. [Google Scholar] [CrossRef]
- Tao, F.; Zhao, Y.-Q.; Zhang, G.-Q.; Li, H.-L. Electrochemical characterization on cobalt sulfide for electrochemical supercapacitors. Electrochem. Commun. 2007, 9, 1282–1287. [Google Scholar] [CrossRef]
- Li, Z.; Han, J.; Fan, L.; Guo, R. Template-free synthesis of Ni7S6 hollow spheres with mesoporous shells for high performance supercapacitors. CrystEngComm 2015, 17, 1952–1958. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, S.-X.; Dong, L.; Lu, Q.-L.; Zhu, J.; Nan, C.-W. One-step synthesis of surface-enriched nickel cobalt sulfide nanoparticles on grapheme for high-performance supercapacitors. Energy Storage Mater. 2017, 6, 180–187. [Google Scholar] [CrossRef]
- Kumar, R.; Kim, H.-J.; Park, S.; Srivastava, A.; Oh, I.-k. Graphene-wrapped and cobalt oxide-intercalated hybrid for extremely durable super-capacitor with ultrahigh energy and power densities. CARBON 2014, 79, 192–202. [Google Scholar] [CrossRef]
- Patil, D.S.; Pawar, S.A.; Shin, J.C. Core-shell structure of Co3O4@CdS for high performance electrochemical supercapacitor. Chemical Engineering Journal. 2018, 335, 693−702. [Google Scholar] [CrossRef]
- Yedluri, A.K.; Kim, H.-J. Wearable super-high specific performance supercapacitors using a honeycomb with folded silk-like composite of NiCo2O4 nanoplates decorated with NiMoO4 honeycombs on nickel foam. Dalton Trans. 2018, 47, 15545–15554. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Sarkar, K.K.; Mandal, K.; Khan, G.G. High-performance supercapacitor electrode based on cobalt oxide-manganese dioxide-nickel oxide ternary 1D hybrid nanotubes. ACS Appl. Mater. Interfaces 2016, 8, 20786–20792. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; Hu, Z.; Liu, Y.; Liu, P. Design synthesis of hierarchical NiCo2S4@NiMoO4 core/shell nanospheres for high-performance supercapacitors. New J. Chem. 2015, 39, 8430. [Google Scholar] [CrossRef]
- Jiang, J.; Shi, W.; Song, S.; Hao, Q.; Fan, W.; Xia, X.; Zhang, X.; Wang, Q.; Liu, C.; Yan, D. Solvothermal synthesis and electrochemical performance in super-capacitor of Co3O4/C flower-like nanostructure. J. Power Sources 2014, 248, 1281–1289. [Google Scholar] [CrossRef]
- Selvam, S.; Balamuralitharan, B.; Jegatheeswaran, S.; Kim, M.Y.; Karthick, S.N.; Raj, J.A.; Boomi, P.; Sundrarajan, M.; Prabakar, K.; Kim, H.J. Electrolyte-imprinted grpahene oxide-chitosan chelate with copper crosslinked composite electrodes for intense cyclic-stable flexible supercapacitors. J. Mater. Chem. A 2017, 5, 1380–1386. [Google Scholar] [CrossRef]
- Gu, Z.; Zhang, X. NiCo2O4@MnMoO4 core-shell flowers for high performance supercapacitors. J. Mater. Chem. A 2016, 4, 8249. [Google Scholar] [CrossRef]
- Bai, J.; Wang, K.; Feng, J.; Xiong, S. ZnO/CoO ZnCo2O4 Hierarchical Bipyramid Nanoframes: Morphology Control, Formation Mechanism, and Their Lithium Storage Properties. ACS Appl. Mater. Interfaces 2015, 7, 22848–22857. [Google Scholar] [CrossRef] [PubMed]
- Yedluri, A.K.; Kim, H.-J. Wearable super-high specific performance supercapacitors using a honeycomb with folded silk-like composite of NiCo2O4 nanoplates decorated with NiMoO4 honeycombs on nickel foam. Dalton Trans. 2018, 47, 15545–15554. [Google Scholar] [CrossRef] [PubMed]
- Niu, H.; Zhou, D.; Yang, X.; Li, X.; Wang, Q.; Qu, F. Towards three-dimensional hierarchical ZnO nanofiber@Ni(OH)2 nanoflake core-shell heterostructures for high-performance asymmetric supercapacitors. J. Mater. Chem. A 2015, 3, 18413. [Google Scholar] [CrossRef]
- Du, J.; Zhou, G.; Zhang, H.; Cheng, C.; Ma, J.; Wei, W.; Chen, L.; Wang, T. Ultrthin porous NiCo2O4 nanosheet arrays on flexible carbon fabric for high-performance supercapacitors. ACS Appl. Mater. Interfaces 2013, 5, 7405–7409. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.; Li, J.; Hou, L.; Zhang, X.; Shen, L.; Lou, X.W. Ultrathin mesoporous NiCo2O4 nanosheets supported on Ni foam as advanced electrodes for supercapcitors. Adv. Funct. Mater. 2012, 22, 4592−4597. [Google Scholar] [CrossRef]
- Pujari, R.B.; Lokhande, A.C.; Kim, J.H.; Lokhande, C.D. Bath temperature controlled phase stability of hierarchical nanoflakes CoS2 thin films for supercapacitor applications. RSC Adv. 2016, 6, 40593–40601. [Google Scholar] [CrossRef]
- Xiao, X.; Han, B.; Chen, G.; Wang, L.; Wang, Y. Praparation and electrochemical performnaces of carbon sphere@ZnO core-shell nanocomposites for supercapacitor applications. Sci. Rep. 2017, 7, 40167. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Kvit, A.; Cai, Z.; Ma, Z.; Gong, Z. A free standing cellulose nnaofibril-reduced grapheme oxide-molybdenum oxynitride aerogel film electrode for all-solid-state supercapacitors with ultrahigh energy density. J. Mater. Chem. A 2017, 5, 12528–12541. [Google Scholar] [CrossRef]
- Mahajan, H.; Bae, J.; Yun, K. Facile synthesis of ZnO-Au composites for high performance supercapacitors. J. Alloys Compd. 2018, 758, 131. [Google Scholar] [CrossRef]
- Xu, J.; Liu, S.; Liu, Y. Co3O4/ZnO nanoheterostructure derived from core-shell ZIF-8@ZIF-67 for supercapacitors. RSC Adv. 2016, 6, 52137. [Google Scholar] [CrossRef]
- Gai, Y.; Shang, Y.; Gong, L.; Su, L.; Hao, L.; Dong, F.; Li, J. A self-template synthesis of porous ZnCo2O4 microspheres for high-performance quasi-solid-state asymmetric supercapacitors. RSC Adv. 2017, 7, 1038–1044. [Google Scholar] [CrossRef]
- Xu, L.; Zhao, Y.; Lian, J.; Xu, Y.; Bao, J.; Qiu, J.; Xu, L.; Xu, H.; Hua, M.; Li, H. Morphology controlled preparation of ZnCo2O4nanostructures for asymmetric supercapacitor with ultrahigh energy density. Energy 2017, 123, 296–304. [Google Scholar] [CrossRef]
- Mohapatra, D.; Parida, S.; Badrayyana, S.; Singh, B.K. High performance flexible asymmetric CNO-ZnO//ZnO supercapacitor with an operating voltage of 1.8V in aqueous medium. Appl. Mater. Today 2017, 7, 212–221. [Google Scholar] [CrossRef]
- Xu, J.; Gao, L.; Cao, J.; Wang, W.; Chen, Z. Preparation and electrochemical capacitance of cobalt oxide (Co3O4) nanotubes as supercapacitor material. Electrochim. Acta 2010, 56, 732–736. [Google Scholar] [CrossRef]
- Pant, B.; Park, M.; Ojha, G.P.; Park, J.; Kuk, Y.S.; Lee, E.J.; Kim, H.Y.; Park, S.J. carbon nanofibers wrapped with zinc oxide nanoflakes as promising electrode for supercapacitors. J. Coll. Interface Sci. 2018, 522, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Li, J.; Zhang, X.G. Preparation and properties of Co3O4 nanorods as supercapacitor material. J. Appl. Electrochem. 2009, 39, 1871. [Google Scholar] [CrossRef]
- Wang, Q.; Zhu, L.; Sun, L.; Liub, Y.; Jiao, L. Facile synthesis of hierarchical porous ZnCo2O4 microspheres for high-performance supercapacitors. J. Mater. Chem. A 2015, 3, 982. [Google Scholar] [CrossRef]
- Li, W.; Yang, F.; Hu, Z.; Liu, Y. Template synthesis of C@NiCo2O4 hollow microsphere as electrode material for supercapacitor. J. Alloys Compd. 2018, 749, 305–312. [Google Scholar] [CrossRef]
- Yang, M.H.; Choi, B.G. Preparation of Three-Dimensional Co3O4/graphene Composite for High-Performance Supercapacitors. Chem. Eng. Commun. 2017, 204, 723–728. [Google Scholar] [CrossRef]

| Electrode Materials | Electrolyte | Specific Capacitance at Current Density | Capacitance Retention (No. of Cycles) | Year, Ref. |
|---|---|---|---|---|
| Co3O4/ZnO | 6 M KOH | 415 F/g | 93.2% (1000) | 2016 (ref. [37]) |
| ZnCo2O4 | 6 M KOH | 542.5 F/g | 95.5% (2000) | 2017 (ref. [38]) |
| ZnCo2O4 | 2 M KOH | 776.2 F/g | 84.3% (1500) | 2017 (ref. [39]) |
| Co3O4 nanotube | 6 M KOH | 574 F/g | 95% (1000) | 2010 (ref. [40]) |
| CNO–ZnO//ZnO | 3 M KOH | 125 F/g | 92% (2000) | 2017 (ref. [41]) |
| ZnO nanoflakes | 2 M KOH | 260 F/g | 73.33% (1000) | 2018 (ref. [42]) |
| Co3O4 nanorods | 6 M KOH | 456 F/g | 65% (500) | 2009 (ref. [43]) |
| ZnCo2O4 | 2 M KOH | 647.1 F/g | 91.5% (2000) | 2015 (ref. [44]) |
| C@NiCo2O4 | 6 M KOH | 404 F/g | 87.1% (1000) | 2018 (ref. [45]) |
| Co3O4/Graphene | 6 M KOH | 328 F/g | 97.9% (2000) | 2017 (ref. [46]) |
| CZ3h | 2 M KOH | 1136 F/g | 98.3% (3000) | This work |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, Y.A.; Kim, H.-J. Effect of Time on a Hierarchical Corn Skeleton-Like Composite of CoO@ZnO as Capacitive Electrode Material for High Specific Performance Supercapacitors. Energies 2018, 11, 3285. https://doi.org/10.3390/en11123285
Kumar YA, Kim H-J. Effect of Time on a Hierarchical Corn Skeleton-Like Composite of CoO@ZnO as Capacitive Electrode Material for High Specific Performance Supercapacitors. Energies. 2018; 11(12):3285. https://doi.org/10.3390/en11123285
Chicago/Turabian StyleKumar, Yedluri Anil, and Hee-Je Kim. 2018. "Effect of Time on a Hierarchical Corn Skeleton-Like Composite of CoO@ZnO as Capacitive Electrode Material for High Specific Performance Supercapacitors" Energies 11, no. 12: 3285. https://doi.org/10.3390/en11123285
APA StyleKumar, Y. A., & Kim, H.-J. (2018). Effect of Time on a Hierarchical Corn Skeleton-Like Composite of CoO@ZnO as Capacitive Electrode Material for High Specific Performance Supercapacitors. Energies, 11(12), 3285. https://doi.org/10.3390/en11123285






