Abstract
Hydrothermal liquefaction (HTL) is regarded as a promising technology for the production of biofuels from biomass and wastes. As such, there is a drive towards continuous-flow processing systems to aid process scale-up and eventually commercialization. The current study presents results from a novel pilot-scale HTL reactor with a feed capacity of up to 100 L/h and a process volume of approximately 20 L. The pilot plant employs a heat exchanger for heat recovery and a novel hydraulic oscillation system to increase the turbulence in the tubular reactor. The energy grass Miscanthus and the microalgae Spirulina, both representing advanced dedicated energy crops, as well as sewage sludge as high-potential waste stream were selected to assess the reactor performance. Biomass slurries with up to 16 wt% dry matter content were successfully processed. The heat recovery of the heat exchanger is found to increase with reactor run time, reaching 80% within 5–6 h of operation. The hydraulic oscillation system is shown to improve mixing and enhance heat transfer. Bio-crudes with average yields of 26 wt%, 33 wt% and 25 wt% were produced from Miscanthus, Spirulina and sewage sludge, respectively. The yields also appeared to increase with reactor run time. Bio-crude from HTL of Spirulina was mainly composed of palmitic acid, glycerol, heptadecane and linolelaidic acid, while biocrude from sewage sludge contained mainly palmitic acid, oleic acid and stearic acid. In contrast, biocrude from HTL of Miscanthus consisted of a large number of different phenolics. An energetic comparison between the three feedstocks revealed a thermal efficiency of 47%, 47% and 33% and energy return on investment (EROI) of 2.8, 3.3 and 0.5 for HTL of Miscanthus, Spirulina and sewage sludge, respectively.
1. Introduction
Hydrothermal liquefaction (HTL) is a promising process for converting a variety of organic materials into biofuels and value-added chemicals. The process is carried out at elevated temperatures (300–400 °C) and pressures (200–300 bar) in the presence of water and results in four product phases: a bio-crude phase, a water phase, a gas phase and a solid phase. The bio-crude, which is the main desired product from the process, has a high heating value (~30–40 MJ/kg), however it contains significant amounts of oxygen (~10%–20%) requiring further upgrading for production of drop-in fuels (gasoline, diesel, kerosene, heavy fuel oil) [1,2,3,4,5].
An advantage of HTL, is the processing of aqueous slurries, avoiding the need for drying, which is a necessary step for other thermochemical technologies which require feedstock drying (gasification, pyrolysis, combustion). HTL is able to convert most types of organic material into bio-crude, which means that any type of biomass that can be suspended in water represents a suitable feedstock for HTL. Typically, slurries with 10%–20% dry matter (DM) content, in the presence or absence of catalysts, are processed, resulting in high carbon efficiencies and energy recoveries in the bio-crude. In addition, the high pressure of the system maintains the water in the liquid phase, eliminating the energy intensive step of water evaporation. HTL has been demonstrated for a variety of different feedstocks including lignocellulosic (woody biomass and wood-processing waste) [6,7,8,9,10], marine (both micro and macro algae) [11,12,13,14,15,16,17,18,19,20], waste biomasses (sewage sludge, food processing wastes and so on) [8,20,21,22,23,24] and their mixtures [25].
During the 1970–1980’s, when the first reports on HTL were published, some pilot-scale continuous processes were established such as the PERC and Albany facilities [3]. In the last 10 years, the majority of HTL work has been performed in small batch reactors, typically a few hundred millilitres in volume. Increasingly the attention is now shifting again towards scaling up HTL reactors from laboratory-scale batch systems to pilot-scale plants operating in continuous mode. During the past 5 years, a number of research groups around the world have developed and demonstrated such continuously operating pilot-scale HTL units.
The Pacific Northwest National Laboratory (PNNL) in the USA established a continuous HTL reactor system incorporating a continuous-flow stirred-tank reactor and a horizontal tubular reactor [16,24,26]. The system has been demonstrated for microalgae [27], macro-algae [16], food waste [26], sewage sludge and digested solids [24,28]. The main part of the system consists of a horizontal preheater with a capacity of 210 mL, a stirred tank reactor with a capacity of 415 mL where the slurry is heated to the reaction temperature (350 °C), a horizontal oil jacketed reactor (capacity 300 mL) for maintaining the slurry’s temperature and a high temperature-pressure filter for removing solids at the reaction conditions. The total volume of the system is approximately 1.6 L and is operated with liquid hourly space velocities (LHSV) between 1 and 2.1 (L/L/h) corresponding to approximately 1.5 L of slurry per hour.
A different design of a continuous flow pilot-scale HTL system design has been established at the Sydney University in Australia [13,29]. In this design, the pressurized slurry is pre-heated into two heat exchangers before entering the reactor which consists of four stainless steel coils immersed in a fluidized bed. The hot effluent entering the heat exchangers is cooled to 170 °C and depressurized to 10 bar by using two additional heat exchangers. The total volume of the reactor (the four coils) is approximately 2 L. The system was demonstrated for micro-algae and macro-algae by using pure water and mixtures of water with organic solvents.
In both aforementioned cases, HTL is performed at around 350 °C and 200 bar which falls below the critical point of water (374 °C, 221 bar). An altered approach is followed by Aalborg University and Steeper Energy ApS in Denmark who developed a HTL system at conditions above the critical point of water, operating at 400 °C and 300 bar [30,31]. The HTL pilot-plant at Aalborg University has a capacity of 30 kg/h slurry throughput.
There are some examples of HTL demonstration plants under development or in operation but limited information on capacities, reactor design and operation conditions are available. Companies such as Licella (North Sydney, New South Wales, Australia), Muradel Pty Ltd. (Whyalla Barson, South Australia, Australia), Southern Oil Refining (Bomen, New South Wales, Australia), Steeper Energy ApS (Aalborg, Jutland, Denmark) and Genifuel Corporation (Salt Lake City, UT, USA) are such examples. Licella has developed a catalytic HTL technology, called Cat-HTRTM, with a pilot plant at Somersby, Australia, capable of processing 7500 tonnes of slurry per year [32]. They have set up two joint ventures with Canfor (Vancouver, British Columbia, Canada) and one with Neste Corporation (Espoo, Uusimaa, Finland) and ReNew ELP (Redcar, Middlesbrough, UK) to build demonstration plants based on Cat-HTRTM technology for processing forest residues and plastic wastes respectively [33]. Southern Oil Refining has partnered up with Melbourne Water to build a demonstration plant to process 1 million tons of biosolids per annum [34]. Steeper Energy ApS has developed their HydrofractionTM technology in collaboration with Aalborg University and has now partnered with Silva Green Fuel (Norwegian-Swedish joint venture) to construct an industrial scale demonstration HTL plant processing forest waste with a capacity of 4000 L/day [35]. Genifuel Corporation, in collaboration with Metro Vancouver, is developing a pilot HTL plant by utilizing PNNL’s technology, the plant will process concentrated (up to 20% solids) primary and secondary sewage sludge [36].
To the best of our knowledge, the reactor presented in the present study, developed at Aarhus University (AU) Denmark, is the largest pilot-scale HTL reactor reported in peer reviewed literature. The reactor incorporates a patented oscillating flow technology [37] and uses a parallel hydraulic pressure release system, in order to avoid constrictions found in regular backpressure valves It has been designed and built with scalability in mind and studies have supported direct 1000-fold scalability [38]. Therefore, a unique heat exchanger design is used in the reactor which is scalable and can withstand the required operating pressures. The effectiveness of this heat exchanger is investigated in order to provide, for the first time, data on heat recoveries possible in scalable continuous HTL reactors.
The aim of the study is to investigate areas in the HTL research community, which cannot be examined using batch reactors and are lacking in the few, continuous processing studies published to date. More specifically, the objectives of the current paper are: to assess the effectiveness of a unique, scalable heat exchanger design; to investigate the effect of an oscillation unit on the HTL reactor performance; to evaluate and compare the bio-crude yields and quality to published data in order to evaluate the applicability of batch studies to continuous flow reactors and to evaluate the overall energetic performance of the reactor as a function of different feedstock slurries applied.
We investigate these research objectives by processing three different biomass feedstocks in the AU HTL pilot plant with a capacity of up to 100 L/h. Miscanthus, Spirulina and sewage sludge were selected as model feedstocks to represent 2nd, 3rd generation and waste biomass feedstocks, respectively. The feedstocks span the range from lignocellulosic feedstocks (Miscanthus), high nitrogen containing (Spirulina) and high ash (sewage sludge). This paper hence presents a broad data set of different HTL feedstocks processed at a larger scale than previously reported and discusses the overall HTL process design, performance and efficiency.
2. Materials and Methods
2.1. Materials
To test the performance of the HTL pilot reactor, three model biomass feedstocks were used: Miscanthus, Spirulina and sewage sludge. Miscanthus giganteus was harvested in early-January 2018 from the fields of the Danish Centre for Food and Agriculture (DCA), Foulum of Aarhus University (Denmark). Spirulina platensis was purchased from Inner Mongolia Rejuve Biotech Co. Ltd. (Otog, Ordos, Inner Mongolia, China) as a dry, powder. Sewage sludge was collected locally in February 2018 after the primary treatment from the wastewater treatment plant at Viborg (Energi Viborg A/S). Potassium hydroxide (KOH, 46%) and high molecular weight carboxymethyl cellulose (CMC) were bought from Sigma Aldrich. The different model biomass feedstocks were characterized in terms of moisture content, ash content and elemental composition (Table 1). Moisture content was analysed in triplicate using a Mettler Toledo HE53 Moisture analyser. The ash content was determined by placing samples in a furnace for 5 h at 550 °C. The elemental content of the samples was measured using an Elementar vario Macro Cube elemental analyser (Langenselbold, Hesse, Germany). All measurements were repeated in duplicate and a mean value is reported. The HHVs of the samples were calculated according to the equation proposed by Channiwala and Parikh [39] based on their elemental composition and ash content.

Table 1.
Proximate, ultimate analysis and HHVs of the different model biomass feedstocks.
2.2. Feedstock Pre-Treatment
In order to achieve continuous operation of HTL reactors and reduce clogging issues, a homogeneous, stable and easily pumpable slurry must be produced. Depending on the nature of the feedstock (wet/dry feedstock, presence or absence of lignin) different pre-treatment is required. For example, lignocellulosic terrestrial biomass leads to the formation of hard spherical particles upon milling or grinding. These can cause problems during feeding the slurry in the reactor such as blockages at the pump head. An additional issue is created by the tendency of lignocellulosic/terrestrial biomass to settle out if a non-homogeneous slurry is prepared [40]. Both issues, if not tackled, would result in blocking the piston of the pump in the pump shaft and the need for manually disassembling the pump and cleaning it.
In order to overcome these issues, Miscanthus was extruded; freshly harvested biomass with a natural water content of approximately 60% dry matter was roughly chopped (~10–50 mm) and extruded using a twin screw extruder (Xinda, 65 mm twin screw extruder with 2000 mm barrel length). An appropriate amount of carboxymethyl cellulose (CMC) (0.25 wt% of the slurry mass) was subsequently added as a thickener. Long, soft fibres are formed through extrusion instead of hard spherical biomass particles that are formed by milling. Typical fibres after extrusion are 0.1–8 mm in length with a length to diameter ratio of 5–25 [8]. CMC acts as a thickener, in order to increase the viscosity of slurries and avoids squeezing out water from the biomass slurry in the piston pump which would result in a dry biomass plug and blocking. Miscanthus was mixed in a paddle micer with the appropriate amount of water, CMC and catalyst (KOH 1.4 wt% of slurry mass) to create a slurry with ~15 wt% DM content. Presence of a homogeneous catalyst, such as KOH, is required when processing lignocellulosic biomass to aid its’ decomposition, reduce char formation and the amount of solids produced during HTL.
Spirulina was directly mixed with water to obtain a homogenous slurry of 16.4 wt% DM. No extrusion, catalyst or CMC were employed. Sewage sludge was processed as received from the primary treatment system at the wastewater treatment plant and processed on the same day, hours after collection. It exhibited approximately 4 wt% DM. This dry matter content is not optimal for HTL in terms of energy efficiency but was run as a base case scenario and due to practical difficulties of increasing the DM content of sewage sludge [8]. It also serves as a distinctly different data point in the study to evaluate the effect of low DM content of on the energy efficiency of the HTL plant. The slurries preparation and compositions are given in Table 2.

Table 2.
Pre-treatment and final slurry concentrations for the different model biomass feedstocks.
2.3. HTL Reactor System Description and Procedure
The AU pilot-scale HTL reactor system used in this study consists of seven sections: a feed introduction system, a heat exchanger, a trim heater, a reactor, an oscillation system, a take-off system and a product collection zone. The system is fully computer controlled through a Programmable Logic Controller (PLC) unit (NH-Technology ApS, Lystrup, Jutland, Denmark). The PLC acquires pressure and temperature sensor data and controls the actuated valves and trim heater. The process flow diagram of the AU pilot-scale HTL reactor system is given in Figure 1.
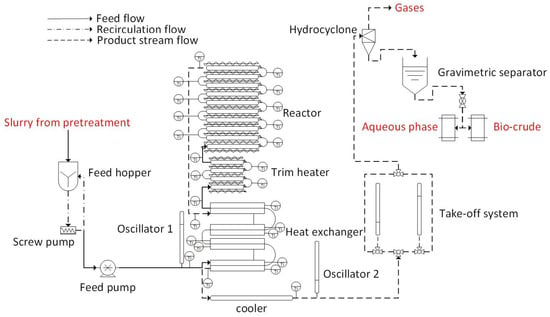
Figure 1.
Process flow diagram of the AU pilot-scale HTL reactor.
The HTL reactor is designed to have an essentially constant cross section with an internal diameter of 14.2 mm throughout the full 140 m length of the tubular system. All parts exposed to process media at temperatures higher than approximately 80 °C are constructed from a high nickel alloy (UNS N06025). In the low temperature parts, some components including valves and cylinders are made of SS316. The feeding system consists of a feed hopper and two pumps. The first pump is a progressive cavity pump and is used to provide continuous recirculation of the slurry in the hopper, in order to ensure the homogeneity of the slurry and minimize settling of the feed slurry and providing a steady flow to the second pump at slight overpressure (~2 bar). The second pump, the main high-pressure feed pump, feeds the slurry into the reactor. A Graco Check-Mate© positive displacement pump capable to deliver high-viscosity materials is used. The Graco pump can deliver pressures of up to 476 bar and flow rates of up to 600 L/h. In a typical run, a flow rate of 1 L/min (60 L/h) and a pressure of approximately 220 bar was used. The feed flow rate was determined by the number of strokes of the pump (200 cm3) over time.
The heat exchanger section consists of a double pipe counter current heat exchanger. The heat exchanger consists of a total of eight Inconel pipes, with an outer diameter (OD) of 25.4 mm and a wall thickness (WT) of 5.6 mm. They are divided in four convolutions of 2 × 6 m each, making the ‘cold’ and ‘hot’ sides of the heat exchanger. The total length of both sides, along with their connections is 49.2 m. Inlet biomass slurry is pre-heated in the ‘cold’ part of the heat exchanger by the heat of the product stream, before it is directed to the trim heater. Ten thermocouples (K-type) are employed to observe the temperature profile along the heat exchanger. Due to the high pressures in both the cold and hot tubes of the heat exchanger many common heat exchanger designs such as tube-in-tube, shell and tube or external heat transfer fluids are difficult to implement. We hence designed a unique heat exchanger where ductile cast iron (EN-GJS-500-7) heat clamps are used to clamp the hot and cold tubes together. The thermal expansion coefficient of cast iron and Inconel are very similar which ensures that the two tubes fit tightly in the clamp also at high temperature.
After the heat exchanger, the trim heater is used to heat the feed to the reaction temperature. It consists of four pipes divided in two convolutions of 2 × 3 m each, making a total length of 12.6 m. It is equipped with 32 independent electrical heaters with a heating capacity of 1 kW each, which are individually controlled by the PLC. Five thermocouples (K-type) are employed to observe the temperature profile along the trim heater, while 32 thermocouples are employed for controlling the power input to each electrical heater.
At the exit of the trim heater, biomass slurry is directed to the reactor where the main HTL reactions and biomass conversion takes place. The reactor is a section in which process conditions are maintained at constant set points. It consists of ten pipes, divided in five convolutions of 2 × 6 m each. The reactor is equipped with five independent electrical heating tapes with a heating capacity of 1 kW each, which can be individually controlled. Ten thermocouples (K-type) are employed to observe the temperature profile along the reactor, while five thermocoules are employed to control and record the temperature of each heating tape. All hot components of the HTL reactor system (heat exchanger, trim heater and reactor) are heavily insulated and enclosed in 1.5 mm thick stainless-steel casings to minimize heat losses to the surroundings.
The product stream is then directed to the ‘hot’ side of the heat exchanger where it is cooled by exchanging heat with the cold inlet biomass slurry. Subsequently, the product stream is cooled through a series of pipes (10 m of which 6 m are water cooled) before being directed to the take-off system and the product collection zone. The total volume of the system is approximately 20 L.
The hydraulic oscillation system consists of two pistons of 0.5 L capacity each. The first piston is located just after the main feed pump and before the inlet of the ‘cold’ side of the heat exchanger, the second is located after the cooling system just before the hydraulic take-off system. The aim of using the is to increase turbulence in the whole reactor system which is hypothesised to improve mixing and enhance heat transfer. One side of each piston is exposed to the HTL slurry at the reaction pressure while the other side is connected to a high pressure hydraulic oil circuit. A series of valves are controlled on the hydraulic side in order to push one piston down while the other is retracting. This is performed continuously in alternation so that the reactor contents are continuously moving backwards and forwards during their overall forward movement in the plug flow reactor. Hence, the local velocity of the slurry is increased while the overall residence time is not changed. It is hypothesised that this will avoid settling out of char and minerals in the HTL reactor due to increased velocity.
The hydraulic take-off system consists of two pistons of 0.5 L capacity each working in alternation for pressure release. The principle is similar to the oscillator system but the reactor flow is diverted via a valve to either of the pistons. The other side of the piston is connected to another hydraulic oil circuit just as the oscillator system. The piston can hence take one volume (0.5 L) from the reactor at approximately the same pressure as the overall reactor pressure by slowly retracting the piston. Once retracted each piston cylinder has an additional valve, which opens to the reactor outlet section, which is exposed to atmospheric pressure in the product collection funnel. While one cylinder is filling, the other is discharging and thereby the system allows for continuous flow. The overall system pressure is set via the main pump while the flow rate is set via the speed of the take-off pistons’ movement.
The product collection zone consists of a hydro-cyclone, which facilitates the separation of the gaseous and liquid streams and a 90 L separation funnel for collection and subsequent separation of the liquid products (bio-crude and aqueous phase).
Before each run, water was pumped, with recirculation, into the reactor system with the desired pressure (200–240 bar) to heat up the reactor. Pressurization of the water is performed by the high-pressure feed pump and can be adjusted with a back-pressure regulator instead of the above described take-off system. The advantage of the take-off system is that there are no restrictions throughout the entire reactor system and a constant inner diameter of 14.2 mm is maintained. This avoids blocking of small restrictions such as in back pressure regulators with particulate matter in the HTL feed and product stream. Once, the desired reaction temperature is reached, recirculation is stopped, the take-off system is started and the feedstock is switched to the biomass slurry. Initially, HTL products are directed into an overflow barrel until visual observation of steady bio-crude production. At this point, HTL products are directed to the product collection zone. Samples of approximately 500 mL, corresponding to the capacity of the take-off pistons, are collected directly from the product pipe at regular time intervals at a sampling temperature of ~50–60 °C and stored at 5 °C for further analysis.
At the end of each run the reactor system is cleaned up by pumping a solution of water and KOH and hydrogen peroxide (~1 wt% each) at the reaction temperature and pressure. Subsequently, water is pumped in recirculation at lower temperatures and pressures for several hours.
2.4. Residence Time and Energy Efficiency Calculations
The residence time (τ) of the biomass slurry in the different sections of the reactor can be calculated by Equation (1):
where V is the internal volume of the process section (heat exchanger, trim heater, reactor and so on.) and is the volumetric flow rate of the feed. However, the density of the feed is changing throughout the length of the process as the various process units experience different temperature profiles. Equation (1) can hence be rewritten in terms of length (L) and cross-sectional area (A) of the process unit (the volume of the process unit in Equation (1)) and the mass flow rate () and density (ρ) of the feed (the volumetric flow rate in Equation (1)):
By taking into account the change of the density of the feed through the length of each process unit due to the differences in temperature profile Equation (2) can be rearranged to:
The density of the feed was calculated by taking into account the percentage of solids into the slurry and by assuming a solids density of 500 kg/m3. It was further assumed that the density of the slurry follows an analogue change as the density of water at any given temperature and pressure. The density of water at corresponding temperatures and pressures has been acquired from the NIST database.
The heat recovery (HR) by the heat exchanger was calculated according to Equation (4) by taking into account the temperature inside the reactor (Treactor) and the temperature at the exit of the feed flow of the heat exchanger (THEX), just upstream the trim heater:
The thermal efficiency (ηth) or energy recovery (ER) was calculated according to Equation (5) by taking into account the energy content of the bio-crude product and the energy content of the feedstock in terms of higher heating values (HHV):
The total energy efficiency of the system (ηtot) was calculated, in a similar procedure, according to Equation (6) by considering the energy content of the product bio-crude, the energy content of the feedstock and the electrical energy (Pel) consumed by the main energy consuming units of the system (trim heater, reactor and feed pump):
Finally, EROI was calculated according to Equation (7) by taking into account the electrical energy (Pel) consumed by the main energy consuming units of the HTL system (trim heater, reactor and feed pump). It should be noted that this EROI does not take into account some smaller energy requirements of the reactor such as the PLC, hydraulic pumps, ventilation and so on, it also does not include any energy required upstream of the process such as biomass harvesting, chopping, transportation and so on.
2.5. Sample Work-Up and Analysis
Collected samples, containing liquid products (bio-crude and aqueous phase) and solids were stored at 5 °C. At this temperature, bio-crude has a high viscosity and can be easily gravimetrically separated from the aqueous phase by decanting the aqueous phase. This alleviates the need of using solvents such as dichloromethane for the recovery of the bio-crude as it is commonly practiced in batch HTL experiments. Aliquots of aqueous phase (~50 mL) were filtered and subsequently analysed for total organic carbon (TOC) using Hach-Lange cuvette tests (LCK 387). The filter was dried and subsequently weighed to determine the solids content in the aqueous phase. After the removal of water from the bio-crude, its yield was determined gravimetrically by weighing and taking into account the DM content of each biomass slurry, according to the following Equation (8):
Ash and elemental content of the bio-crudes were measured with the same methods described previously in Section 2.1. Moisture analysis of the bio-crudes was performed by FT-IR by using a newly developed method for determination of water content in bio-crudes from HTL of biomass [41]. FT-IR was performed with a Techno Nicolet 380 with attenuated total reflectance. Bio-crudes were analysed in duplicate by either placing a drop of bio-crude (e.g., Spirulina) or smearing bio-crude (e.g., Miscanthus) on the sampling probe. The spectral region of 4000–400 cm−1 was obtained with 32 scans at a resolution of 2 cm−1. Raw data was pre-processed with a Savitzky-Golay filter and subjected to partial least squares regression as demonstrated in our previous work.
The volatile and semi-volatile components of the bio-crudes were analysed with GC-MS with prior derivatization to improve chromatographic performance, volatility and response of nucleophilic compounds. Bio-crude (10 mg) was dissolved in dichloromethane (900 µL) and N-methyl-N-(trimethylsilyl)trifluoroacetamide (100 µL). Samples were vortexed and placed on a shaker board for 60 min. Analysis was performed using an Agilent 7890B GC coupled to a quadrupole mass filter MS (Agilent, 5977A). The injection port was operated at 280 °C in 20:1 split mode with helium (1 mL min−1) and the injection volume was 1 µL. Analytes were separated on an HP-5ms (30 m × 0.25 mm × 0.25 µm). The column oven program started at 40 °C (hold time 1 min) progressing at 4 °C min−1 to 180 °C and at 20 °C min−1 to 320 °C (hold time 6 min). Analytes were identified with authentic standards, mass spectral library (NIST11) or based on literature references.
3. Results and Discussion
3.1. Reactor Performance
In order to investigate the effectiveness of the heat exchanger design and effect of oscillation the temperature profiles inside the reactor and at the exit of the heat exchanger, were analysed. The temperature profiles for the three selected feedstocks along with information about the start and the end of feed, start and stop of oscillation and energy consumption by the trim heater are depicted in Figure 2. Miscanthus (Figure 2a) was run without oscillation while the hydraulic oscillator was started and stopped at regular time intervals for Spirulina and sewage sludge runs as indicated on Figure 2b,c. Total duration of the runs with the biomass slurries was approximately 6 h for Miscanthus and Spirulina and 5 h for sewage sludge.
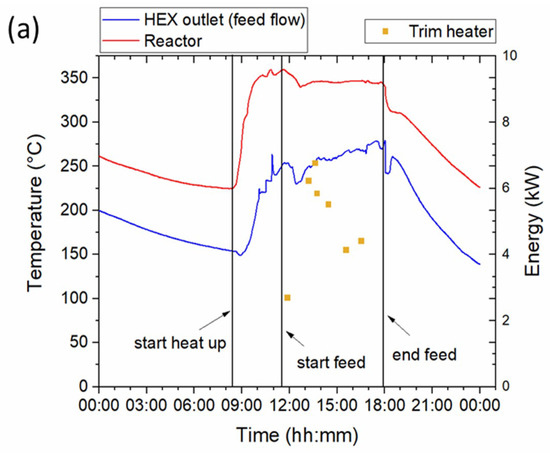
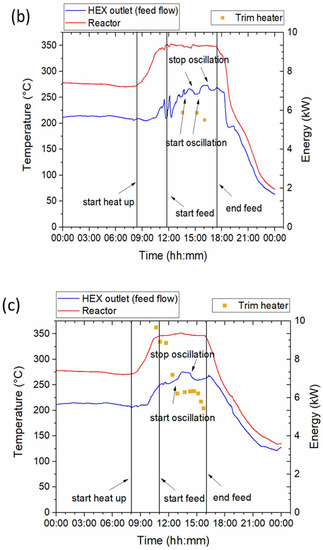
Figure 2.
Temperature profiles at the heat exchanger outlet (feed flow) and inside the reactor and energy consumption by the trim heater at 220 bar for HTL runs of (a) Miscanthus, (b) Spirulina and (c) sewage sludge.
The size of the HTL system and the presence of large quantities of water require high heat energy input to heat up the system from ambient to the reaction temperature (350 °C). Hence, the system was pre-heated overnight, under recirculation of pure water, to 250–275 °C in order to save time on the days of the runs. During heat up, from the pre-heated set point to the reaction temperature, the same procedure was followed while recirculation was stopped once the desired temperature was reached and subsequently the feed slurry was introduced in the reactor.
The temperature in the reactor is maintained constant at approximately 350 °C throughout the duration of the run, indicating steady state operation. However, this is not the case for the temperature in the heat exchanger. The temperature in the heat exchanger outlet increases constantly until the end of the feeding, indicating that the heat exchanger does not reach steady state operation during the duration of the runs (5–6 h). This phenomenon is clearer in the case of Miscanthus (Figure 2a) where no oscillation was used but is also evident in the case of Spirulina (Figure 2b). This had a direct effect on the heat recovered by the heat exchanger. As shown in Table 3, the heat recovery (HR) for the slurries with higher solids content (Miscanthus and Spirulina) are less than 70% at the beginning of the feeding and they steadily increase, reaching close to 80% in the final hour of operation. As expected, the energy consumed by the trim heater follows the reverse trend, with significantly less power requirements during higher heat recoveries. During the last hour of operation, when HR is at its maximum, 4.5 kW to 5.5 kW are required to heat the biomass slurries to the reaction temperature by the trim heater. Longer run times have been observed to further increase the heat recovery to values above 80%, leading to a further reduction of energy input to the trim heater (data not included).

Table 3.
Heat recovery (HR %) by the heat exchanger for HTL runs of the three biomass feedstocks.
The effect of oscillation is more evident in the sewage sludge experiment (Figure 2c) where the hydraulic oscillation system was operated for approximately one hour. As shown, the oscillation system was started around 13:00 and stopped at 14:20. The effect of oscillation is apparent in the heat exchanger graph and is characterized by a spike in temperature. The oscillators increase turbulence in the whole reactor system, leading to improved mixing and enhanced heat transfer. This is evident by the increase in HR during the time intervals the oscillation system was on (Table 3). The average heat recovery for the whole duration of the runs was approximately 75% for all cases. Further discussion on the energy aspects of the system is included in Section 3.3 Energy considerations.
The heating profile, of the selected biomass slurry Miscanthus, along with its residence time distribution during steady state operation (last hour of experimental run), in the different unit operations (heat exchanger, trim heater, reactor and so on) is shown in Figure 3. The temperature profile of the two other biomass feedstocks are very similar and are shown in electronic supplementary Figure S1. The residence time is calculated by assuming an ideal plug flow reactor. In reality, different mixing and flow patterns are expected in the system. Typically, the incoming feed is heated from 20 °C to 260–275 °C in the heat exchanger in 2.5 min. Subsequently it is heated to the reaction temperature (350 °C) in the trim heater in 1 min. This temperature is maintained, with some minor fluctuations (330–355 °C), in the reactor for 5.6 min. The product stream is then directed to the return side of the heat exchanger, where it transfers heat to the incoming feed and is cooled from the reaction temperature to ~80–85 °C in 3.2 min. Finally, the product stream passes through the cooling section where it is cooled to approximately 60 °C in 1.5 min before entering the product collection zone.
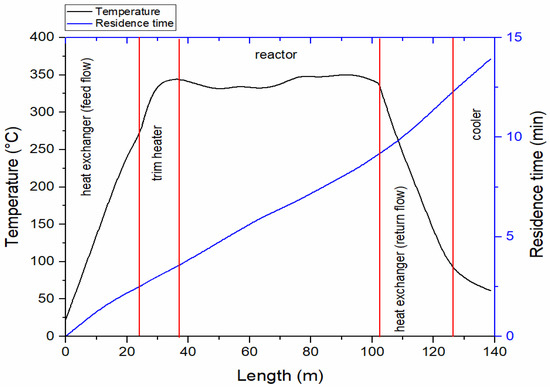
Figure 3.
Temperature profiles and residence time distribution in the different HTL process units (heat exchanger-feed flow, trim heater, reactor, heat exchanger-return flow and cooler) at 220 bar for HTL of Miscanthus.
The feedstock slurries are hence heated at a rate of approximately 100 °C/min in the heat exchanger, then initially faster in the trim heater, (see Figure 3) but on average at 75 °C/min until it reaches reaction temperature. This represents a relatively fast heating rate; similar to those achieved using batch reactors submerged into fluidized sand baths. The effect of heating rate and residence times is an area of on-going investigation. Savage et al. have reported maximum bio-crude yields using very high heating rates of approximately 300 °C/min for microalgae [42] and also reported beneficial effects of fast heating rates on the liquefaction of the model compounds starch, casein and sunflower oil [43]. Biller et al. reported higher yields of bio-crude on a continuous flow reactor when higher flow rates were used, which resulted in higher heating rates and lower residence times [44]. At the same time, the degree of de-oxygenation of the bio-crude was lower when higher flow rates were employed. It was argued, that there is a trade-off between high yields and low oxygen content in the bio-crude. These reports are interesting in relation to the current study as the oxygen contents are shown to be comparatively high (Table 4) and the yields low. Batch studies are not directly applicable to continuous studies and the lower yields are likely due to low dry matter contents, inefficient separation of bio-crude and water and losses. The oxygen contents in the present study are relatively high, compared to other continuous HTL studies and this could be due to the high heating rate and low residence times employed. The aspect of heating rates and residence times hence deserves further investigation using the current reactor by systematically varying the flow rate.

Table 4.
Yields, ultimate analysis and HHVs of bio-crudes from HTL of Miscanthus (MIS), Spirulina (SP) and sewage sludge (SS).
3.2. Bio-Crude Yields and Analysis
Liquid products were sampled every 30 to 60 min in duplicate to obtain 12 bio-crude samples per run. Bio-crudes appeared as black, viscous liquids that could be separated from the water phase as two distinct phases were created. Spirulina bio-crude had an apparent lower viscosity than bio-crudes from Miscanthus and Sewage sludge. Figure 4 depicts bio-crude yields and energy recoveries (ER) in the bio-crudes for the three feedstocks during 5–6 h of continuous operation. Table 4 shows the ultimate analysis and HHVs of the bio-crudes, while Table 5 includes bio-crude yields, proximate analysis, ERs and carbon distribution between the bio-crude and process water.
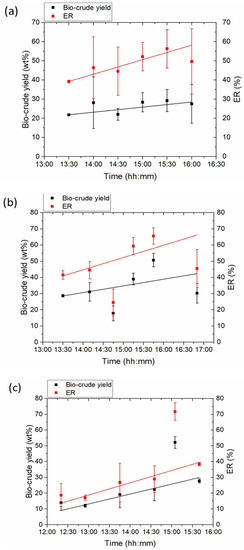
Figure 4.
Bio-crude yields and energy recovery (ER) in the bio-crude on biomass input for HTL runs of (a) Miscanthus, (b) Spirulina and (c) sewage sludge over time (straight lines indicate linear fit of the data).

Table 5.
Proximate analysis, energy recovery (ER) and carbon partitioning in bio-crudes from HTL of Miscanthus (MIS), Spirulina (SP) and sewage sludge (SS).
Bio-crude yields from all three feedstocks varied over time and between samples within the same time interval (Figure 4). These differences in yields are attributed to the changes in temperature in the pipes after the exit of the heat exchanger (return flow) and to the take-off system. An extensive pipe network exists between the exit of the heat exchanger and the product collection system. This pipe network experiences gradual heating through time as hotter product is generated and passes through. As a result, bio-crude becomes less viscous as temperature increases and has a lower tendency to stick to pipe walls, valves and fittings.
In general, bio-crude yields appeared to increase over the reactor run time, indicating that steady-state bio-crude production was not achieved during the reactor run time (5–6 h). A similar behaviour was observed with the effectiveness of the heat exchanger, which increased steadily (Figure 3). Both results suggest that the reactor should be operated for longer times (>6 h) in order to approach steady-state conditions. The increase in bio-crude yields over time had a direct effect on the ER in the bio-crude which showed a similar increase over time.
Bio-crude yields (see equation 8) from HTL of Miscanthus vary between 18.6 wt% and 37.6 wt% with a mean value, based on the 12 collected samples, of 26.2 wt% (Figure 4a, Table 4). HHVs of Miscanthus bio-crudes were found to vary between 26.6 MJ/kg and 36.6 MJ/kg with a mean value of 30.7 MJ/kg (Table 4). Accordingly, ERs in the bio-crudes varied between 35% and 63.4% with a mean value of 48.1% (Table 5). These yields were within the range of reported yields in literature from HTL of Miscanthus in small batch reactors. Biller et al. found bio-crude yield of 24.1 wt% from HTL of Miscanthus in 20 mL batch reactors [8] while Lappa et al. found yields between 30 wt% and 44.6 wt%, depending on the pre-treatment, in 50 mL batch reactors [6]. Nonetheless a significant difference, not process related, between batch and pilot-scale HTL systems has to be noted. In pilot-scale systems, bio-crude is gravimetrically separated from water and solid products without the use of any organic solvents. On the other hand, the use of organic solvents for the separation of bio-crude in batch systems can result in the extraction of additional organics from the water phase and/or the solids leading to an increase in the measured bio-crude yields [45]. Zhu et al. found bio-crude yield of 29.4 wt% during HTL of pine sawdust in a continuous flow HTL reactor [2] while higher yield of 45.3 wt% from HTL of a mixture of woody biomass is reported by Steeper Energy in their pilot scale continuous flow HTL reactor [31]. The higher bio-crude yield in the latter study is partly due to the recirculation of process water and part of the bio-crude product in the feed slurry. No recirculation of process water or bio-crude was applied in the present study.
Similarly, bio-crude yields from HTL of Spirulina are shown in Figure 4b. Yields were found to vary between 14.6 wt% and 53.6 wt% with a mean value, of 32.9 wt% (Table 4). HHVs of Spirulina bio-crudes were found to vary between 29.9 MJ/kg and 36.3 MJ/kg with a mean value of 33.2 MJ/kg (Table 4). Accordingly, ERs in the bio-crudes varied between 18.7% and 69.2% with a mean value of 46.8% (Table 5). Spirulina bio-crude was less viscous and with better flow properties at room temperature and had a higher average yield and HHV than Miscanthus bio-crude. However, the average ER was less for Spirulina bio-crude due to the higher HHV of raw Spirulina compared to raw Miscanthus (Table 1). Comparing the yields of bio-crude from our current pilot-scale campaign with literature batch results, similar values are obtained. Vardon and co-workers reported a bio-crude yield of 32.6 wt% with an HHV of 33.2 MJ/kg from HTL of Spirulina in a 2-L batch reactor [20]. Huang et al. produced bio-crude with 34.5 wt% yield and an HHV of 34 MJ/kg from HTL of Spirulina in a batch reactor [9], while Jena et al. found a maximum of 40 wt% bio-crude yield with an HHV of 35 MJ/kg from HTL of Spirulina in a 1.8-L batch reactor [46]. Researchers at PNNL continuously liquefied microalgae slurries of up to 35 wt% DM and found bio-crude yields ranging from 38–64 wt% depending on the lipid content of the algae feedstock [27].
Finally, bio-crude yields from HTL of sewage sludge are shown in Figure 4c. In general, bio-crudes from HTL of Sewage sludge exhibited lower yields and HHVs than the other two feedstocks. The low yields are attributed to the different composition of Sewage sludge (e.g., higher ash content, Table 1) but also to the much lower dry matter (DM) content of the sewage sludge slurry compared to the other feedstock slurries (Table 2). Due to the low amount of biomass per mass of water, losses are a much more significant factor compared to high dry matter content experiments. It has been shown that higher bio-crude yields are produced when higher solids loadings are used both in batch [15] and in continuous systems [13,24]. The lower HHVs are attributed to the higher ash content of bio-crudes from HTL of Sewage sludge (Table 5). Solids content measurement showed about 20 wt% of the bio-crude mass to be consisted of insoluble solids (data not shown). The lower yields and lower HHVs in bio-crudes from sewage sludge resulted in a significantly lower average energy recovery (33.6%) than from the other two feedstocks (48.1% and 46.8%). In general, higher bio-crude yields from HTL of sewage sludge are reported in literature. Biller and co-workers reported yields of 35.4 wt% from HTL of 10 wt% dry matter slurries in batch reactors [8], while a similar yield of 37 wt% from HTL of 12 wt% dry matter slurries in a continuous flow HTL system was reported by researchers in PNNL [24]. It is expected that the yields of bio-crude for all three feedstocks can be increased in future campaigns by simple measures such as re-circulating the process water in the case of Miscanthus and increasing the dry matter content in the case of sewage sludge. More complex and systematic approaches will also be investigated such as varying flow rates and hence heating rates and residence times, changing the rate of oscillation, recycling bio-crude and improving product separations.
Figure S2 in supplementary data shows the GC-MS chromatograms of the bio-crudes from HTL of Miscanthus, Spirulina and sewage sludge. Lignocellulosic biomass is well known to produce a wide range of phenolics mainly from lignin but also from carbohydrates, which also produce alkylated cyclopent-2-enones [47,48]. The most abundant compounds from Miscanthus bio-crude were phenol and catechol derivatives along with series of alkylated hydroquinones and chrom-2-enones previously identified from lignocellulosics [49]. A number of alkylated cyclopent-2-enones were also present although in lower abundance compared to other studies. The high water content of the bio-crude also resulted in the presence of lactic acid and glycolic acid, which are dissolved in the aqueous phase. Small organic acids and small alcohols are known to be abundant in the aqueous phase from HTL of lignocellulosics [50,51]. The dominating presence of phenolics and cyclopent-2-enones will lead to a diverse range of saturated 5 and 6 membered ring structures of hydrocarbons upon upgrading [52]. These compounds present a medium reactivity toward hydrodeoxygenation along with medium hydrogen consumption, which will thus influence the economics of the process [53].
The GC amenable fraction of sewage sludge bio-crude was composed of a number fatty acids ranging from C10 to C20 with the major fraction being myristic acid, palmitic acid, linoleic acid, oleic acid and stearic acid. Additionally, a number of long straight chain alcohols (C10, C12, C14, C16 and C18) were detected along with a few alkylated phenols. Even though the composition of sewage sludge varies widely depending on its origin and storage, the GC-MS results are somewhat in agreement with previously published studies. Kapusta [54] identified several phenolics and straight chain alcohols from methanol extractions while the undetected fatty acids is a combination of the solvent extraction and the column used for separation. Prajitno, et al. [55] performed thermochemical liquefaction of sewage sludge in methanol and observed a similar diversity of fatty acids. Recently, Jarvis, et al. [52] showed that the GC amenable fraction of upgraded bio-crude from sewage sludge consisted predominantly of straight chain hydrocarbons corresponding to the variety of fatty acids and alcohols observed in this study. The abundance of these compounds is advantageous as they show high reactivity for hydrodeoxygenation with low consumption of hydrogen [53].
Spirulina bio-crude contained a similar range of fatty acids for C16 and C18 with a higher abundance of unsaturated fatty acids. Furthermore, a high abundance of heptadecane, indole, 3-methylindole, phenol, p-methylphenol and m-ethylphenol was found along with a wide range of alkylated pyrroles. The abundance of these compounds is in agreement with other studies depending on whether silylation has been used to analyse the bio-crudes [12,56]. Other studies have also identified the presence of fatty amides in Spirulina bio-crudes [12]. It has previously been shown that fatty amides co-elute with fatty acids when silylation is employed [49]. However, we have observed a range of fatty amides by using a different instrumental set-up. Furthermore, the composition of Spirulina bio-crude is also in good agreement with the composition observed from upgrading of microalgae bio-crude where even nitrogen could be fully removed [52].
A carbon balance between the bio-crude and HTL water was performed in order to shed light on the partitioning of the carbon present in the feedstocks. The gas yield and composition was not analysed in the present work and only selected samples were analysed for solids yields hence the partitioning of carbon to these two phases is not known. The majority of carbon is desired to pass into the bio-crude phase in order to increase its yield and overall carbon efficiency of the process which ultimately affects the achievable reduction in emission of greenhouse gases. Carbon partitioning between the bio-crude and the HTL process water is shown in Table 5. It is calculated using the yields and elemental composition of the bio-crudes along with the total organic carbon content (TOC) of the water. Carbon partitioning to the bio-crudes followed the trend of bio-crude yields and generally showed a tendency to increase with reactor run time as the reactor was approaching steady-state conditions. On average, 44.6 wt%, 36.3 wt% and 30.6 wt% of the carbon in the original feedstock partitioned to the bio-crude for Spirulina, Miscanthus and sewage sludge, respectively. At the same time HTL water was found to contain a significant portion of the starting carbon. On average 38.3 wt%, 34.8 wt% and 32.3 wt% of the carbon in the original feedstock passed to the process water for Spirulina, sewage sludge and Miscanthus respectively. Carbon recoveries in the bio-crudes, apart for Spirulina, are lower compared to values from literature. Jazrawi et al. reported up to 50 wt% and 30 wt% of carbon retention in the bio-crude and process water respectively during continuous flow HTL of microalgae [13]. Marrone et al. found approximately 60 wt% and 40 wt% of the carbon present in primary and secondary sludge respectively to partition in the bio-crude during continuous flow HTL while 20 wt% and 40 wt% of carbon was retained in the process water [28]. The literature data shows that higher carbon recoveries to the bio-crude are possible in continuous flow HTL reactors and this is an area we aim to improve the operation by, for example, recycling process water, optimising the use of oscillation for increased mixing, increasing the DM content of slurries to the pumpable maximum, improved product separation and potentially longer residence times. In any case, HTL water contains a significant portion of the carbon present in the feedstock, mainly small organic acids [48], which needs to be further utilised either by recycling of HTL water back to the reactor or by further processing (e.g., anaerobic digestion, or hydrothermal gasification).
3.3. Energy Considerations
HTL is an energy intensive process as it involves the heating of large amounts of water, which has a very high specific heat capacity, especially at elevated temperatures. It is therefore highly relevant to perform an energy balance of the process. Energy balances in batch HTL reactors have revealed the negative effect of low dry matter content slurries and high reaction temperatures on the overall balance [15]. However, in continuous pilot-scale systems the energy balance differs significantly due to process integration and heat recovery.
The overall process efficiency of the AU HTL reactor was assessed in terms of the thermal efficiency (or the ER in the bio-crude), ηth, the total energy efficiency, ηtot and the EROI as a function of feedstock applied. The thermal efficiency was calculated by taking into account the average yields and heating values of the bio-crudes produced together with the DM concentration of the feeding slurries and flow rate. The total energy efficiency was calculated by additionally taking into account the energy consumption by the trim heater, the reactor and the main feeding pump. Consumption data for the trim heater and the reactor during the last hour of operation, when the reactor was approaching steady-state, were used for the calculations, while the energy consumption of the pump was calculated by Aspen Plus by considering flow rate of 60 L/h, discharge pressure of 220 bar and 0.6 efficiency. EROI was calculated by taking into account only the main energy requirements of the HTL process (trim heater, reactor, feeding pump). All calculations were performed by assuming one hour of operation and the results are shown in Table 6.

Table 6.
Process efficiency during HTL of Miscanthus, Spirulina and sewage sludge.
The thermal efficiency for Miscanthus and Spirulina were similar, at approximately 47%, while it was lower for sewage sludge (33%). Similarly, the total efficiency was slightly above 40% for Miscanthus and Spirulina while only 20% for sewage sludge. Accordingly, the EROI was 2.8, 3.3 and 0.5 for Miscanthus, Spirulina and sewage sludge respectively. Miscanthus and Spirulina slurries had a similar DM content (15% and 16%, respectively), leading to similar energetic performance. On the other hand, the very low DM concentration of sewage sludge (4%) together with the relatively low calorific value of the produced bio-crude resulted in a net energy loss during HTL (EROI < 1). In order to process sewage sludges efficiently through HTL an increase in their DM content is needed as described recently by Biller et al. [8] where a combination of lignocellulosic biomass assisted filtration and co-liquefaction lead to significantly higher EROI due to higher DM feedstock slurries.
It has to be noted that the calculated energy ratios take into account only the main energy consuming units of the HTL pilot-plant (main pump, trim heater, reactor). Other auxiliary units such as the pump used for recirculation of the slurry in the hopper, the hydraulic circuit or the extruder that was used for the pre-treatment of Miscanthus have been excluded from the calculations. Inclusion of these units in the calculations would unambiguously decrease these ratios. On the other hand, if a commercial HTL plant of a similar design is envisaged the heating requirements of the reactor are expected to be lower than the ones reported in the present study. In the AU pilot-plant, the power input in the reactor is set to a certain value (unlike the trim heater where the power input is automatically adjusted to the desired temperature of the slurry by the PLC) that will ensure that the temperature of the slurry will retain its temperature (350 °C) in the reactor. Since the temperature needs to be controlled within a range of a few °C, in a larger size commercial plant with better insulation, the heating requirement to maintain reaction temperature is expected to be significantly lower. One of the most significant factors affecting the EROI is the DM content of the slurry as this directly affects how much energy can be produced by unit volume slurry which has to be heated. DM contents of over 20 wt% have successfully been pumped at the pilot plant and by, for example, recycling process water and bio-crude the DM content can realistically be increased further which would lead to higher EROI.
Based on the above presented results, it is reasonable to suggest various design and operational improvements for the current and future scaled-up HTL reactors. The higher bio-crude yields along with the higher heat recoveries (Table 3) achieved towards the end of each campaign suggest that longer run times should be established in order to improve bio-crude yields and the overall energy balance of the process. This is expected to be fulfilled in the next generation design of the current pilot plant or any other up-scaled plants. Hence, heat recoveries exceeding 80% should commonly be achievable in future continuous HTL plants. In addition, the thermal heat loss per process volume is expected to be significantly lower in up-scaled plants simply due to reduced surface to volume ratio. The apparent endothermic nature of the reactor process may thus in part be due to the heat loss to the surroundings. The aspect of heating rates is interesting and certainly deserves further investigation as it can affect yields and the HHV of bio-crudes. Using batch reactors high heating rates and low residence times have been shown to increase yields quite remarkably [42]. On continuous flow reactors, the effects have not been studied and the current study does not give sufficient cause to suggest anything definitive. Achieving very fast heating rates while maintaining heat transfer is however something which is difficult to implement in practice due to the overall heat transfer coefficients limitations by conduction and convection. The residence time can more easily be varied by changing the overall reactor volume but poses a trade-off between capital cost and potentially a higher degree of de-oxygenation of the bio-crude. The flow rate can also be reduced to increase the residence time but results in lower heating rates and also has the detrimental effect of increased capital cost. The effect of heating rate and residence time should therefore be studied in more detail in continuous flow reactors in order to shed light on optimal future commercial HTL reactor designs.
The results presented above (Table 4) showed an extremely high ash content, especially for sewage sludge bio-crudes. This is unacceptably high for conventional catalytic hydrotreatment where hydrotreating catalysts are poisoned by metal contents as low as 50 ppm in the bio-crude [57]. Using the bio-crude obtained in the present study would therefore need to be distilled to obtain a lower boiling point fraction where the distillation residue would contain the majority of inorganic material and char. Alternatively solvent extraction could be carried out to obtain a fraction which is more easily hydro-treatable, for example by pentane extraction as shown recently by Bélic et al. [58]. This is a common practice in petroleum refineries where the aim is to remove the asphaltene fraction of crude oils. Dilution using a suitable solvent, for example, butanone and subsequent filtration is also an option as discussed by Jensen (2018) [57]. The first two options, distillation and solvent extraction, would result in a reduced yield of bio-crude but also in a material which is potentially easier to upgrade. The dissolving/filtration option does not reduce the bio-crude organic amount drastically but more severe conditions in the hydrotreater could be required to crack the heavier parts of the bio-crude.
Apart from these bio-crude post-treatment options, there is also the potentially of an in-situ approach where a high-pressure filter is introduced in the HTL reactor. This approach has been used at PNNL on their continuous HTL reactor where a cartridge type filter is placed in-line at the end of the reactor but still at the highest temperature (350 °C) and at reaction pressure (~200 bar) [24]. At these conditions, minerals have a low solubility in water, which leads to precipitation of salts, which can settle out in a precipitation vessel and with the combined filtration avoids any precipitates passing through the system. This can be particularly attractive for the recovery of phosphorous for future nutrient recycling applications [28]. PNNL report an ash content in the bio-crude from primary sludge of 0.4 wt% versus 20 wt% in the current study, also produced from primary sludge [24]. At the AU HTL plant a similar system has been tested using a 20 µm filtration element, which was found to be too large. Char and/or inorganic residue was recovered in the filter but the solid content in the bio-crude was still too high. We now have a new design with 5 µm filtration elements and automated back flushing of the filter/precipitation vessel under development. In-line filtration additionally leads to an easier separation of bio-crude and water as the density and viscosity of the bio-crude is reduced. Overall the application of high-pressure and temperature filtration is however technically challenging for the filtration element, valves and process control. This area is still under development for continuous HTL systems and the advantages/disadvantages of in-line filtration versus post-HTL bio-crude clean-up need to be considered.
4. Conclusions
The results presented validate the effectiveness of a custom designed heat exchanger, applicable to HTL pilot plants and future plant designs, reaching a maximum heat recovery of 80%. The effectiveness of the heat exchanger was enhanced by incorporating oscillation of the slurry in the system while no improved quality or yields of bio-crude could be shown with its use. Bio-crude yields were found to increase with reactor operational time and their average yields were 26.2 wt%, 32.9 wt% and 24.5 wt% from Miscanthus, Spirulina and sewage sludge, respectively. The corresponding chemical energy recovery in the bio-crudes was 48.1%, 46.8% and 33.6%. The results broadly confirm literature data published on batch reactors but also highlight the potential for improvement in terms of bio-crude yields and particularly oxygen content of the bio-crudes. The EROI of the system was shown to be positive when the dry matter content of the feed was high (15%) but negative for the experiment with sewage sludge (4 wt% DM), highlighting quantitatively the importance of high dry matter content slurries in HTL. The high ash content of bio-crudes was found to be an outstanding issue in the current reactor design. Further development of the overall process by introducing in-line filtration at high temperature and pressure, recycling of HTL water and bio-crude with the biomass feed along with improvement in product collection and separation are recommended. Overall, the pilot scale results confirm the promising results from batch and continuous HTL studies in terms of energy efficiency and the applicability of the heat exchanger and oscillator design.
Supplementary Materials
The following are available online at http://www.mdpi.com/1996-1073/11/10/2695/s1 as supplementary materials, Figure S1: Temperature profiles and residence time distribution in the different HTL process units (heat exchanger-feed flow, trim heater, reactor, heat exchanger-return flow and cooler) at 220 bar for HTL of (a) Spirulina and (b) sewage sludge, Figure S2: GC-MS chromatograms of bio-crudes from HTL of Miscanthus, Spirulina and sewage sludge.
Author Contributions
I.J. has designed the pilot-scale HTL reactor. K.A., P.B. and I.J. conceived, designed and performed the experiments on the pilot scale HTL reactor. K.A. and P.B. analysed the data. R.B.M. and M.G. performed FT-IR and GC-MS analysis of the bio-crudes. All of the authors were involved in preparing the manuscript.
Funding
This research was funded by the European Union’s Horizon 2020 research and innovation programme under grant agreement No. 764734 (HyFlexFuel—Hydrothermal liquefaction: Enhanced performance and feedstock flexibility for efficient biofuel production) and the Bio-Value Strategic Platform for Innovation and Research, co-funded by The Danish Council for Strategic Research and The Danish Council for Technology and Innovation, case No. 0603-00522B.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Savage, P.E.; Levine, R.B.; Huelsman, C.M. Hydrothermal Processing of Biomass for Biofuels. In Thermochemical Conversion of Biomass to Liquid Fuels and Chemicals; Crocker, M., Ed.; Royal Society of Chemistry: London, UK, 2010; pp. 192–221. [Google Scholar]
- Zhu, Y.; Biddy, M.J.; Jones, S.B.; Elliott, D.C.; Schmidt, A.J. Techno-economic analysis of liquid fuel production from woody biomass via hydrothermal liquefaction (HTL) and upgrading. Appl. Energy 2014. [Google Scholar] [CrossRef]
- Elliott, D.C.; Biller, P.; Ross, A.B.; Schmidt, A.J.; Jones, S.B. Hydrothermal liquefaction of biomass: Developments from batch to continuous process. Bioresour. Technol. 2015, 178, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.A.; Brown, R.J.; Rainey, T.J. Liquefaction biocrudes and their petroleum crude blends for processing in conventional distillation units. Fuel Process. Technol. 2017, 167, 674–683. [Google Scholar] [CrossRef]
- Pedersen, T.H.; Jensen, C.U.; Sandström, L.; Rosendahl, L.A. Full characterization of compounds obtained from fractional distillation and upgrading of a HTL biocrude. Appl. Energy 2017, 202, 408–419. [Google Scholar] [CrossRef]
- Lappa, E.; Christensen, P.S.; Klemmer, M.; Becker, J.; Iversen, B.B. Hydrothermal liquefaction of Miscanthus × Giganteus: Preparation of the ideal feedstock. Biomass Bioenergy 2016, 87, 17–25. [Google Scholar] [CrossRef]
- Zhong, C.; Wei, X. A comparative experimental study on the liquefaction of wood. Energy 2004, 29, 1731–1741. [Google Scholar] [CrossRef]
- Biller, P.; Johannsen, I.; Souza dos Passos, J.; Ottosen, L.D.M. Primary sewage sludge filtration using biomass filter aids and subsequent hydrothermal co-liquefaction. Water Res. 2017, 130, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.J.; Yuan, X.Z.; Zhu, H.N.; Li, H.; Liu, Y.; Wang, X.L.; Zeng, G. Comparative studies of thermochemical liquefaction characteristics of microalgae, lignocellulosic biomass and sewage sludge. Energy 2013, 56, 52–60. [Google Scholar] [CrossRef]
- Toor, S.S.; Rosendahl, L.; Nielsen, M.P.; Glasius, M.; Rudolf, A.; Iversen, S.B. Continuous production of bio-oil by catalytic liquefaction from wet distiller’s grain with solubles (WDGS) from bio-ethanol production. Biomass Bioenergy 2012. [Google Scholar] [CrossRef]
- Ross, A.B.; Biller, P.; Kubacki, M.L.; Li, H.; Lea-Langton, A.; Jones, J.M. Hydrothermal processing of microalgae using alkali and organic acids. Fuel 2010. [Google Scholar] [CrossRef]
- Biller, P.; Ross, A.B. Potential yields and properties of oil from the hydrothermal liquefaction of microalgae with different biochemical content. Bioresour. Technol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Jazrawi, C.; Biller, P.; Ross, A.B.; Montoya, A.; Maschmeyer, T.; Haynes, B.S. Pilot plant testing of continuous hydrothermal liquefaction of microalgae. Algal Res. 2013. [Google Scholar] [CrossRef]
- Anastasakis, K.; Ross, A.B. Hydrothermal liquefaction of the brown macro-alga Laminaria Saccharina: Effect of reaction conditions on product distribution and composition. Bioresour. Technol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Anastasakis, K.; Ross, A.B. Hydrothermal liquefaction of four brown macro-algae commonly found on the UK coasts: An energetic analysis of the process and comparison with bio-chemical conversion methods. Fuel 2015, 139. [Google Scholar] [CrossRef]
- Elliott, D.C.; Hart, T.R.; Neuenschwander, G.G.; Rotness, L.J.; Roesijadi, G.; Zacher, A.H.; Magnuson, J.K. Hydrothermal processing of macroalgal feedstocks in continuous-flow reactors. ACS Sustain. Chem. Eng. 2014. [Google Scholar] [CrossRef]
- Raikova, S.; Le, C.D.; Beacham, T.A.; Jenkins, R.W.; Allen, M.J.; Chuck, C.J. Towards a marine biorefinery through the hydrothermal liquefaction of macroalgae native to the United Kingdom. Biomass Bioenergy 2017, 107, 244–253. [Google Scholar] [CrossRef]
- Shuping, Z.; Yulong, W.; Mingde, Y.; Kaleem, I.; Chun, L.; Tong, J. Production and characterization of bio-oil from hydrothermal liquefaction of microalgae Dunaliella tertiolecta cake. Energy 2010. [Google Scholar] [CrossRef]
- Brown, T.M.; Duan, P.; Savage, P.E. Hydrothermal liquefaction and gasification of Nannochloropsis sp. Energy Fuels 2010, 24, 3639–3646. [Google Scholar] [CrossRef]
- Vardon, D.R.; Sharma, B.K.; Scott, J.; Yu, G.; Wang, Z.; Schideman, L.; Zhang, Y.; Strathmann, T.J. Chemical properties of biocrude oil from the hydrothermal liquefaction of Spirulina algae, swine manure, and digested anaerobic sludge. Bioresour. Technol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Snowden-Swan, L.J.; Zhu, Y.; Bearden, M.D.; Seiple, T.E.; Jones, S.B.; Schmidt, A.J.; Billing, J.M.; Hallen, R.T.; Hart, T.R.; Liu, J.; et al. Conceptual Biorefinery Design and Research Targeted for 2022: Hydrothermal Liquefaction Processing of Wet Waste to Fuels; PNNL-27186; Pacific Northwest National Laboratory: Richland, WA, USA, 2017.
- Déniel, M.; Haarlemmer, G.; Roubaud, A.; Weiss-Hortala, E.; Fages, J. Energy valorisation of food processing residues and model compounds by hydrothermal liquefaction. Renew. Sustain. Energy Rev. 2016, 54, 1632–1652. [Google Scholar] [CrossRef]
- Skaggs, R.L.; Coleman, A.M.; Seiple, T.E.; Milbrandt, A.R. Waste-to-Energy biofuel production potential for selected feedstocks in the conterminous United States. Renew. Sustain. Energy Rev. 2018, 82, 2640–2651. [Google Scholar] [CrossRef]
- Marrone, P.A. Genifuel Hydrothermal Processing Bench-Scale Technology Evaluation Project; The Water Research Foundation: Denver, CO, USA, 2016; ISBN 9781780408408. [Google Scholar]
- Gai, C.; Li, Y.; Peng, N.; Fan, A.; Liu, Z. Co-liquefaction of microalgae and lignocellulosic biomass in subcritical water. Bioresour. Technol. 2015, 185, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.C.; Schmidt, A.J.; Hart, T.R.; Billing, J.M. Conversion of a wet waste feedstock to biocrude by hydrothermal processing in a continuous-flow reactor: Grape pomace. Biomass Convers. Biorefinery 2017, 7, 455–465. [Google Scholar] [CrossRef]
- Elliott, D.C.; Hart, T.R.; Schmidt, A.J.; Neuenschwander, G.G.; Rotness, L.J.; Olarte, M.V.; Zacher, A.H.; Albrecht, K.O.; Hallen, R.T.; Holladay, J.E. Process development for hydrothermal liquefaction of algae feedstocks in a continuous-flow reactor. Algal Res. 2013. [Google Scholar] [CrossRef]
- Marrone, P.A.; Elliott, D.C.; Billing, J.M.; Hallen, R.T.; Hart, T.R.; Kadota, P.; Moeller, J.C.; Randel, M.A.; Schmidt, A.J. Bench-scale evaluation of hydrothermal processing technology for conversion of wastewater solids to fuels. Water Environ. Res. 2018, 90, 329–342. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Liang, X.; Jazrawi, C.; Montoya, A.; Yuen, A.; Cole, A.J.; Neveux, N.; Paul, N.A.; de Nys, R.; Maschmeyer, T.; et al. Continuous hydrothermal liquefaction of macroalgae in the presence of organic co-solvents. Algal Res. 2016. [Google Scholar] [CrossRef]
- Pedersen, T.H.; Grigoras, I.F.; Hoffmann, J.; Toor, S.S.; Daraban, I.M.; Jensen, C.U.; Iversen, S.B.; Madsen, R.B.; Glasius, M.; Arturi, K.R.; et al. Continuous hydrothermal co-liquefaction of aspen wood and glycerol with water phase recirculation. Appl. Energy 2016. [Google Scholar] [CrossRef]
- Jensen, C.U.; Rodriguez Guerrero, J.K.; Karatzos, S.; Olofsson, G.; Iversen, S.B. Fundamentals of HydrofactionTM: Renewable crude oil from woody biomass. Biomass Convers. Biorefinery 2017, 7, 495–509. [Google Scholar] [CrossRef]
- Nabi, M.N.; Rahman, M.M.; Islam, M.A.; Hossain, F.M.; Brooks, P.; Rowlands, W.N.; Tulloch, J.; Ristovski, Z.D.; Brown, R.J. Fuel characterisation, engine performance, combustion and exhaust emissions with a new renewable Licella biofuel. Energy Convers. Manag. 2015, 96, 588–598. [Google Scholar] [CrossRef]
- ChemistryViews. First Commercial-Scale Cat-HTRTM Plant. Available online: https://www.chemistryviews.org/details/news/986073/First_Commercial-Scale_Cat-HTR_Plant.html (accessed on 28 August 2018).
- Pilot Project to Turn Biosolids into Crude Oil. Available online: https://www.sustainabilitymatters.net.au/content/energy/news/pilot-project-to-turn-biosolids-into-crude-oil-763693234 (accessed on 3 September 2018).
- The Silver in Silva: The Story of Steeper Energy and SGF’s’s $59M Advanced Biofuels Project in Norway: Biofuels Digest. Available online: http://www.biofuelsdigest.com/bdigest/2018/01/16/the-silver-in-silva-the-story-of-steeper-energys-59m-advanced-biofuels-project-in-norway/ (accessed on 28 August 2018).
- Hydrothermal Processing of Wastewater Solids. Available online: http://www.genifuel.com/text/Genifuel Case Study for BlueTech Forum 2018 Innovation Showcase.pdf (accessed on 28 August 2018).
- Johannsen, I.; Adamsen, A.P.S.; Kilsgaard, B.S.; Milkevych, V. A Method and Apparatus for Producing Biofuel in an Oscillating Flow Production Line under Supercritical Fluid Conditions. U.S. Patent Publication No. WO/2016/004958, 14 January 2016. [Google Scholar]
- Johanssen, I.; More, D.; Kilsgaard, B.S. Hydrothermal liquefaction of biomass: A pilot plant. Biofuels 2016, 63–65. [Google Scholar]
- Channiwala, S.A.; Parikh, P.P. A unified correlation for estimating HHV of solid, liquid and gaseous fuels. Fuel 2002. [Google Scholar] [CrossRef]
- Berglin, E.J.; Enderlin, C.W.; Schmidt, A.J. Review and Assessment of Commercial Vendors/Options for Feeding and Pumping Biomass Slurries for Hydrothermal Liquefaction; PNNL-21981; Pacific Northwest National Laboratory: Richland, WA, USA, 2012.
- Madsen, R.B.; Anastasakis, K.; Biller, P.; Glasius, M. Rapid determination of water, total acid number and phenolics content in bio-crude from hydrothermal liquefaction of biomass using FT-IR. Energy Fuels 2018, 32, 7660–7669. [Google Scholar] [CrossRef]
- Faeth, J.L.; Valdez, P.J.; Savage, P.E. Fast hydrothermal liquefaction of Nannochloropsis sp. to produce biocrude. Energy Fuels 2013. [Google Scholar] [CrossRef]
- Gollakota, A.; Savage, P.E. Hydrothermal Liquefaction of Model Food Waste Biomolecules and Ternary Mixtures under Isothermal and Fast Conditions. ACS Sustain. Chem. Eng. 2018, 6, 9018–9027. [Google Scholar] [CrossRef]
- Biller, P.; Sharma, B.K.; Kunwar, B.; Ross, A.B. Hydroprocessing of bio-crude from continuous hydrothermal liquefaction of microalgae. Fuel 2015. [Google Scholar] [CrossRef]
- Xu, D.; Savage, P.E. Characterization of biocrudes recovered with and without solvent after hydrothermal liquefaction of algae. Algal Res. 2014, 6, 1–7. [Google Scholar] [CrossRef]
- Jena, U.; Das, K.C.; Kastner, J.R. Effect of operating conditions of thermochemical liquefaction on biocrude production from Spirulina platensis. Bioresour. Technol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, T.H.; Rosendahl, L.A. Production of fuel range oxygenates by supercritical hydrothermal liquefaction of lignocellulosic model systems. Biomass Bioenergy 2015, 83, 206–215. [Google Scholar] [CrossRef]
- Madsen, R.B.; Biller, P.; Jensen, M.M.; Becker, J.; Iversen, B.B.; Glasius, M. Predicting the Chemical Composition of Aqueous Phase from Hydrothermal Liquefaction of Model Compounds and Biomasses. Energy Fuels 2016. [Google Scholar] [CrossRef]
- Madsen, R.B.; Zhang, H.; Biller, P.; Goldstein, A.H.; Glasius, M. Characterizing Semivolatile Organic Compounds of Biocrude from Hydrothermal Liquefaction of Biomass. Energy Fuels 2017, 31, 4122–4134. [Google Scholar] [CrossRef]
- Chen, K.; Lyu, H.; Hao, S.; Luo, G.; Zhang, S.; Chen, J. Separation of phenolic compounds with modified adsorption resin from aqueous phase products of hydrothermal liquefaction of rice straw. Bioresour. Technol. 2015, 182, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Panisko, E.; Wietsma, T.; Lemmon, T.; Albrecht, K.; Howe, D. Characterization of the aqueous fractions from hydrotreatment and hydrothermal liquefaction of lignocellulosic feedstocks. Biomass Bioenergy 2015. [Google Scholar] [CrossRef]
- Jarvis, J.M.; Albrecht, K.O.; Billing, J.M.; Schmidt, A.J.; Hallen, R.T.; Schaub, T.M. Assessment of Hydrotreatment for Hydrothermal Liquefaction Biocrudes from Sewage Sludge, Microalgae, and Pine Feedstocks. Energy Fuels 2018, 32, 8483–8493. [Google Scholar] [CrossRef]
- Jensen, C.U.; Hoffmann, J.; Rosendahl, L.A. Co-processing potential of HTL bio-crude at petroleum refineries. Part 2: A parametric hydrotreating study. Fuel 2015. [Google Scholar] [CrossRef]
- Kapusta, K. Effect of ultrasound pretreatment of municipal sewage sludge on characteristics of bio-oil from hydrothermal liquefaction process. Waste Manag. 2018, 78, 183–190. [Google Scholar] [CrossRef]
- Prajitno, H.; Park, J.; Ryu, C.; Park, H.Y.; Lim, H.S.; Kim, J. Effects of solvent participation and controlled product separation on biomass liquefaction: A case study of sewage sludge. Appl. Energy 2018, 218, 402–416. [Google Scholar] [CrossRef]
- Toor, S.S.; Reddy, H.; Deng, S.; Hoffmann, J.; Spangsmark, D.; Madsen, L.B.; Holm-Nielsen, J.B.; Rosendahl, L.A. Hydrothermal liquefaction of Spirulina and Nannochloropsis salina under subcritical and supercritical water conditions. Bioresour. Technol. 2013, 131, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Jensen, C.U. PIUS—Hydrofaction Platform with Integrated Upgrading Step; Ph.D. Serien for Det Ingeniør- og Naturvidenskabelige Fakultet; Aalborg Universitetsforlag: Aalborg, Denmark, 2018. [Google Scholar]
- Bjelić, S.; Yu, J.; Iversen, B.B.; Glasius, M.; Biller, P. Detailed Investigation into the Asphaltene Fraction of Hydrothermal Liquefaction Derived Bio-Crude and Hydrotreated Bio-Crudes. Energy Fuels 2018, 32, 3579–3587. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).