Utilities, operating and maintenance as well as capital investment are the major costs of syngas production (after the raw material cost) corresponding to ~7%, 16% and 10%, respectively, of the total production cost [
19]. The syngas production cost has a great impact on the economic feasibility of gas-to-liquid (GTL) fuels and chemicals [
20] and is vital for the commercialization of GTL processes. Capital investment and operating costs (i.e., utility, maintenance, general expenses and plant overhead costs) strongly depend on the process design and the engineering. Therefore, process block diagrams (PBD) for syngas production achieving molar ratio of H
2/CO ≥ 2 at industrial scale are developed for both thermal catalytic and plasma-assisted DMR processes. Different process steps are designated and simulated in the Aspen Plus process simulator V8.8 for each PBD. The utility demand is reported while the cost of each piece of equipment is estimated. The utility demand is distinguished in cold and hot utility and expressed in kWh/kmol
syngas. The type and cost of process equipment that is required for syngas production is correlated with the maintenance/operation and capital expenditure. Eventually, the PBDs are evaluated with respect to the utility demand and required process equipment (type of equipment and cost) to define the most promising process flow diagram (PFD), which leads to the lowest syngas production cost.
2.2. Thermal Catalytic DMR Process
Purified streams of CH
4 and CO
2 are fed to the thermal catalytic reactor where the DMR reaction occurs:
The RGIBBS reactor model (minimization of Gibbs free energy) is used to simulate the thermal catalytic reactor at steady state. Chemical equilibrium is achieved at 750 °C and 1 bar. CH
4 and CO
2 conversion of 85% and 90%, respectively, are achieved at these conditions. CO
2 conversion is higher than CH
4 conversion because the reversed water-gas shift (RWGS) reaction occurs concurrently in the reactor:
Thus, slightly higher amount of CO is produced compared to the stoichiometry of the reaction. The amount of H
2 produced is also slightly higher than that dictated by the stoichiometry due to coke formation that occurs simultaneously with DMR. The coke is mainly formed through the following reactions [
32]:
The composition of the DMR reactor outlet stream after considering all the aforementioned side reactions occurring in the reactor are presented in
Table 1. The H
2/CO molar ratio in the DMR outlet stream is 0.94. A gas separator (pressure swing adsorption, absorption and membrane) could be used to separate the two gases, H
2 and CO, and afterwards, the two gas streams could be mixed again in volumetric ratio of F
H2/F
CO = 2 in order to achieve the targeted molar ratio for the methanol synthesis reaction. Pressure swing adsorption (PSA) gives high separation efficiency and is widely used in industry but low H
2 recovery [
33], especially at streams with low H
2 content, and high gas loss from the pressure release during desorption could be considerable disadvantages [
34]. Absorption of CO is a well-known process resulting in high purity and recovery of CO, but the nature of the solvent (cuprous chloride, hydrochloric acid and water) is associated with technical challenges (corrosion, safety issues, high reactivity, etc.). Next to that, high pressure and low temperature, which enhance both the absorption process and solvent recovery, have a significant impact on the operating and capital costs. Membranes are suitable for H
2 purification but practical issues, such as membrane stability, mass transfer limitations and high costs related to operation, capital investment and maintenance are considerable limitations for a large-scale, cost-efficient separation [
35]. In addition, half of the produced CO would remain unutilized in methanol synthesis thus resulting in lower resource efficiency. However, the H
2/CO molar ratio can be adjusted to the targeted ratio by co-feeding water (steam) in the DMR reactor. This reaction is known as combined steam and carbon dioxide methane reforming (SCMR), or bi-methane reforming (BMR) [
36,
37]. Since SCMR is an extremely energy intensive reaction [
38] and the focus of the current work is on the study of DMR as main source of syngas, this scenario is not investigated. However, water (steam) and the DMR outlet stream react in a WGS reactor, connected in series with the DMR reactor and operating at the same conditions (T = 750 °C; P = 1 bar) [
29] to promote heat integration and boost the energy efficiency of the process. Moreover, operating at lower temperature (i.e., 400–600 °C), would lead to CO conversion increase and consequently to higher amounts of H
2 and CO
2 and a ratio of H
2/(2CO + 3CO
2) ≤ 1 in the final syngas stream. In the latter case, a liquefaction temperature lower than −10 °C would be needed to remove the higher excess of CO
2 and yield the targeted ratio of H
2/(2CO + 3CO
2) ≥ 1 in the final syngas stream. Such decrease in the liquefaction temperature would have major impact on the energy consumption of the process. The following reaction, known as water-gas shift (WGS) reaction, takes place in the WGS reactor:
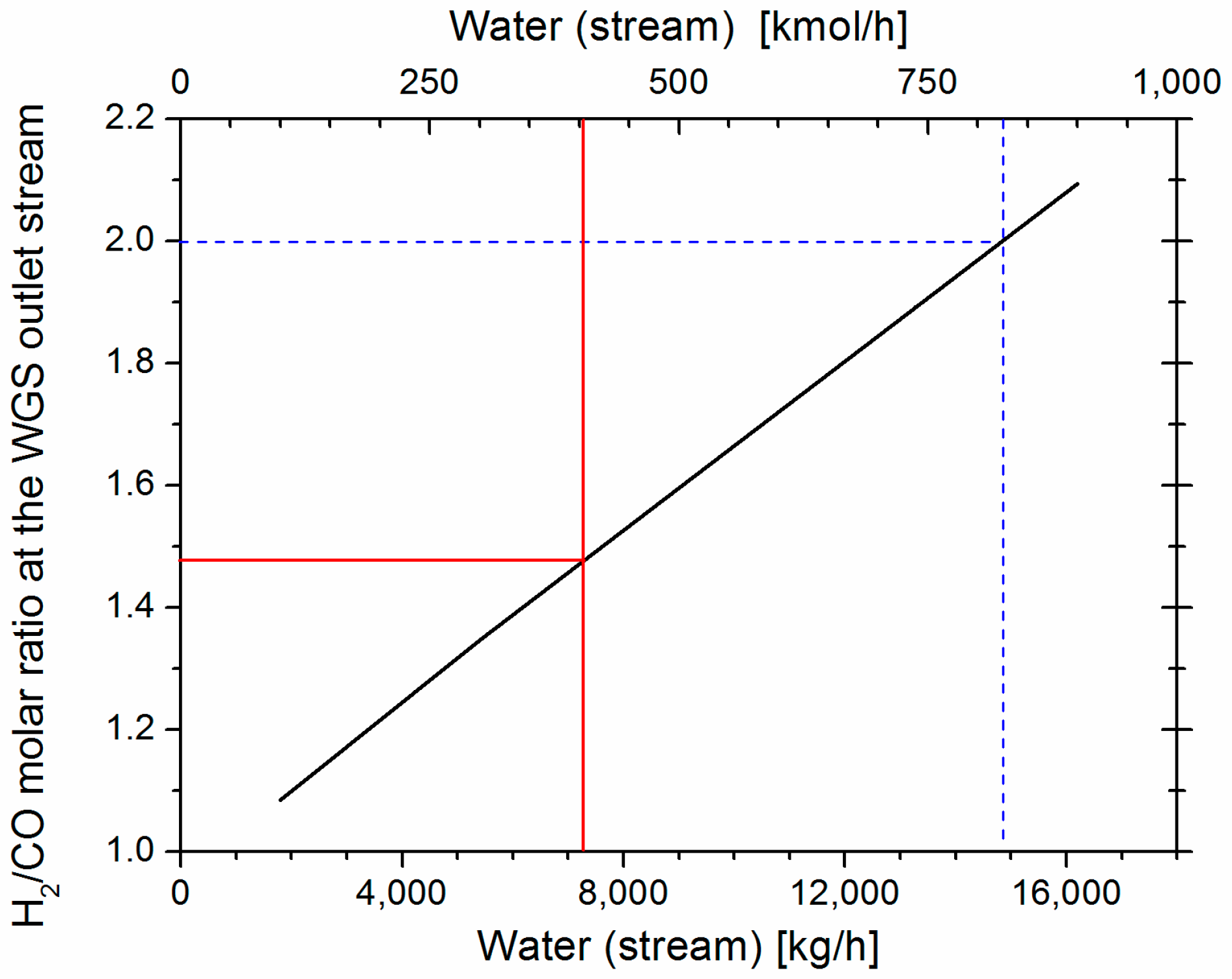
The RGIBBS reactor model is also used to simulate the WGS reactor at steady state. Chemical equilibrium is also achieved at 750 °C and 1 bar. The amount of steam that is added to promote the WGS reaction has been defined after a sensitivity analysis, setting as objective the molar ratio of H
2/CO to be equal to 2 at the WGS outlet stream (
Figure 1). Co-feeding of 14,502 kg/h of water (steam) results in the targeted ratio. However, it has been noticed that, only in the thermal catalytic process, even lower water amount (7251 kg/h) can be fed in WGS and still meet the targeted syngas composition at the end of the process. This is possible since the SMR controls the H
2/CO ratio, thus, it is not necessary the H
2/CO to be equal to 2 at the WGS outlet stream. Therefore, co-feeding of 7251 kg/h steam (2974 kg/h of fresh water and 4277 kg/h of water recycled from the compression section) are fed in the WGS.
CO and H
2O conversion of 19.5% and 41.2%, respectively, are achieved at those conditions in the WGS reactor. The composition of the WGS reactor outlet stream is presented in
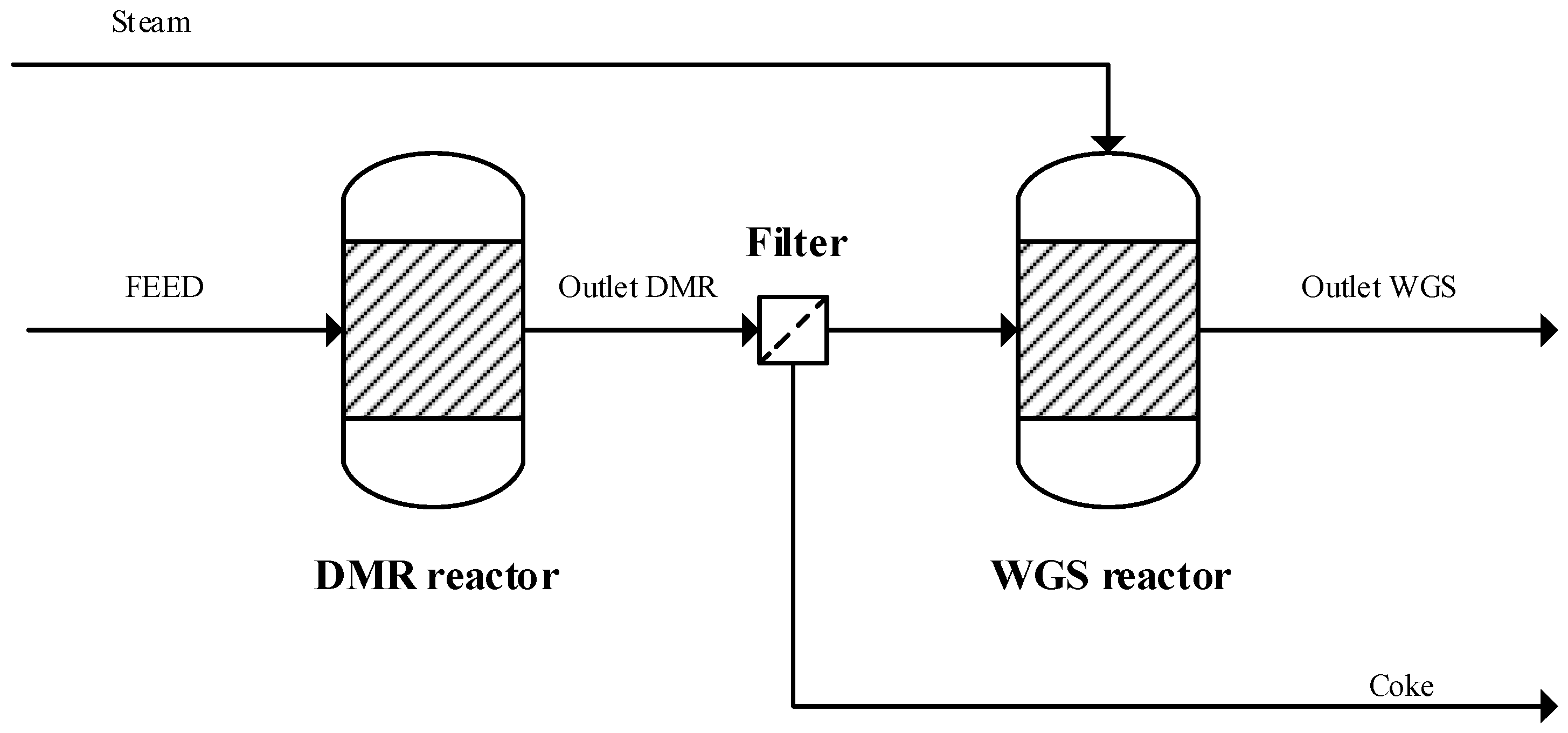
Table 1. The combined DMR and WGS constitutes the upstream part (reaction section) that is common for all thermal catalytic process alternatives. The upstream part is presented in
Figure 2.
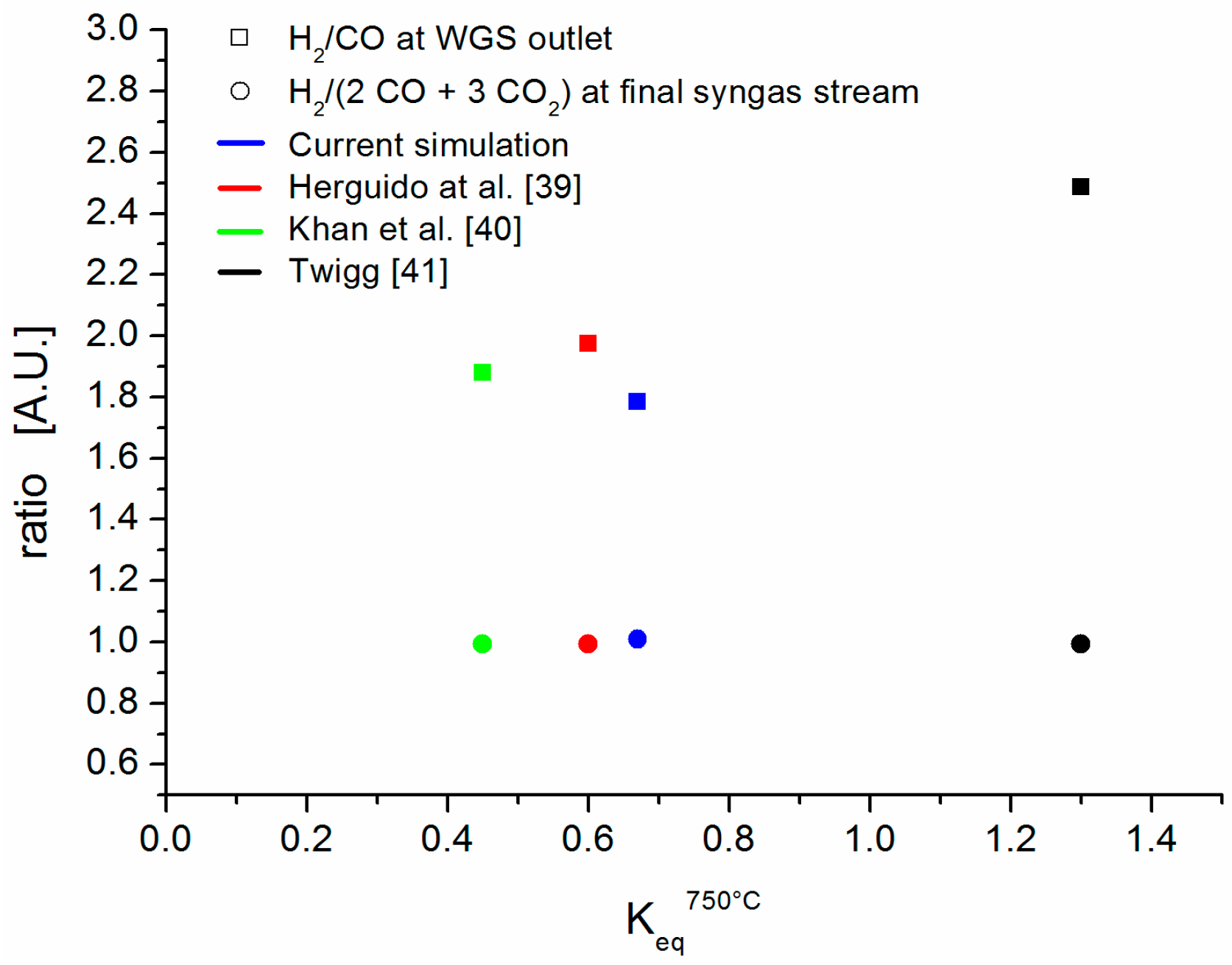
Since considerable differences have been reported between the experimental and theoretical equilibrium constants (K
eq) of the WGS reaction [
39,
40,
41], a sensitivity analysis has been performed to investigate the effect of K
eq at 750 °C on H
2/CO and H
2/(2CO + 3CO
2) (
Figure 3).
Figure 3 shows that, when K
eq varies from 0.45 to 1.3, the ratios H
2/CO and H
2/(2CO + 3CO
2) (process targets) remain practically unaffected.
The WGS reactor outlet stream flows towards the downstream part of the process in order to be purified prior to being fed into the FT process. A considerable amount of CO2 is produced after the WGS reaction; thus, it needs to be separated from the entire gas mixture to achieve the targeted stream composition of H2/(2CO + 3CO2) ≥ 1.
Absorption (both chemical and physical absorption), adsorption, membrane separation and cryogenic processes are well-studied carbon capture and storage (CCS) processes. Chemical and physical absorption (i.e., amine process and Selexol solvent, respectively) have drawn considerable attention due to their relatively high technical maturity and feasibility for large scale capacities. However, challenges, such as solvents chemical instability and high energy requirements for solvent regeneration [
42] should be taken into account. The adsorption and membrane processes are also considered promising CCS technologies due to their potential for lower energy consumption for CO
2 separation compared to absorption. However, capacity limitations, high capital investment and lower separation efficiency compared to absorption (lower CO
2 purity and recovery) are important disadvantages [
43,
44,
45,
46]. The cryogenic separation process attains high separation efficiency and compression and refrigeration systems are well-established at large scale. Nevertheless, extremely low temperature and high compression rates increase substantially the operation cost [
47] and many efforts are currently devoted to process optimization and energy requirement reduction [
48,
49].
Considering the strengths and weaknesses of the aforementioned CCS technologies as well as their technology readiness level and maturity at large scale capacities, CO2 absorption using amines and cryogenic separation are selected as downstream process sections in a thermal catalytic DMR process for syngas production. Two PBDs are developed and evaluated below.
2.2.1. Thermal Catalytic DMR Process with CO2 Absorption (Amine Process)
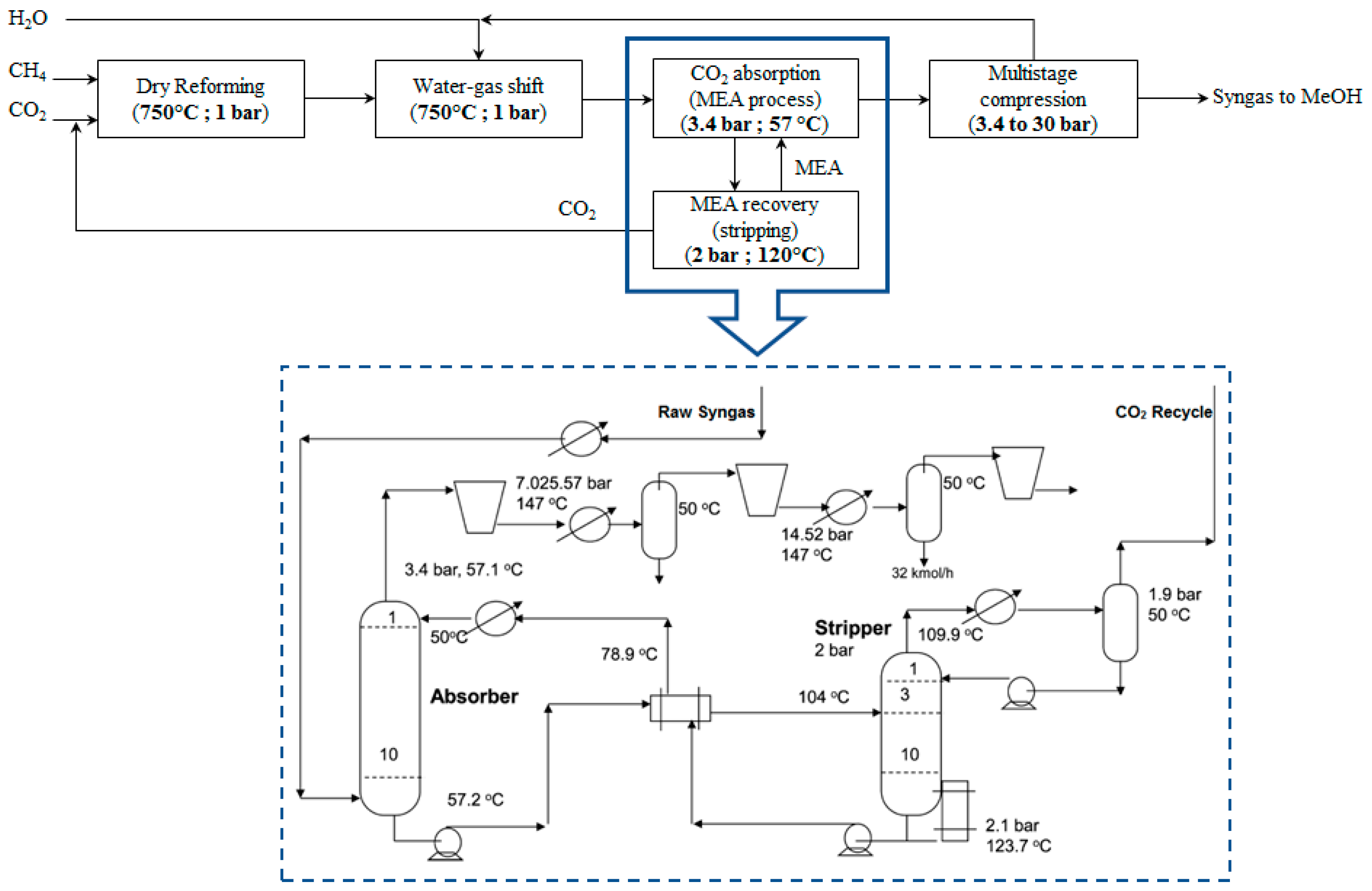
In this scenario, the excess of CO
2 is captured in an absorber using monoethanolamine (MEA) [
50]. The WGS reactor outlet stream enters from the bottom of the absorber and comes in contact with an aqueous MEA solution, which enters from the top and flows counter-currently to the WGS reactor outlet stream. CO
2 reacts exothermically with MEA and form a water-soluble salt. The released heat is absorbed by the fresh streams that enter the column. The temperature and pressure of the column are ~57 °C and 3.4 bar, respectively. The rich in salt MEA stream exits the absorber from the bottom of the column, while the free of CO
2 stream exits from the top. The rich MEA stream is preheated in a heat exchanger by the regenerated MEA stream from the stripper (heat integration to reduce the energy cost) and enters the stripper where the salt formation reaction is reversed by supplying additional heat. The temperature and pressure of the stripper column are 110–120 °C and 2 bar, respectively. In the stripper, CO
2 is degassed from the MEA stream and gets off from the top of the stripper column, whereas the regenerated MEA stream gets off from the bottom. Consequently, CO
2 and MEA are recycled back to the DMR reactor and the absorber, respectively. The PBD of the process and the process flow diagram of this downstream process alternative are presented in
Figure 4.
Absorption of CO
2 using MEA has been extensively studied and simulated [
52,
53]. The economic potential of the process has also been evaluated and compared with other peer CO
2 capture technologies [
54]. Recently, Luyben [
51] designed a DMR process for syngas production. He developed and evaluated two process alternatives: one without CO
2 recycle, thus no separation section was included, and one with CO
2 recycle, therefore an absorber with aqueous MEA solution and a stripper for solvent regeneration was included. He concluded that the scenario of CO
2 recycle results in lower total annual cost than the scenario without CO
2 recycle and notably, the total annual cost is minimized when the reactor pressure is at 4 bar. Approximately 5.6 kWh/kmol
syngas cold utility, 2.6 kWh/kmol
syngas hot utility, 2.4 kWh/kmol
syngas of electrical power and 6.6 k
$/(kmol
syngas h
−1) are required for syngas purification when adopting this process alternative.
2.2.2. Thermal Catalytic DMR Process with CO2 Liquefaction (Cryogenic Separation)
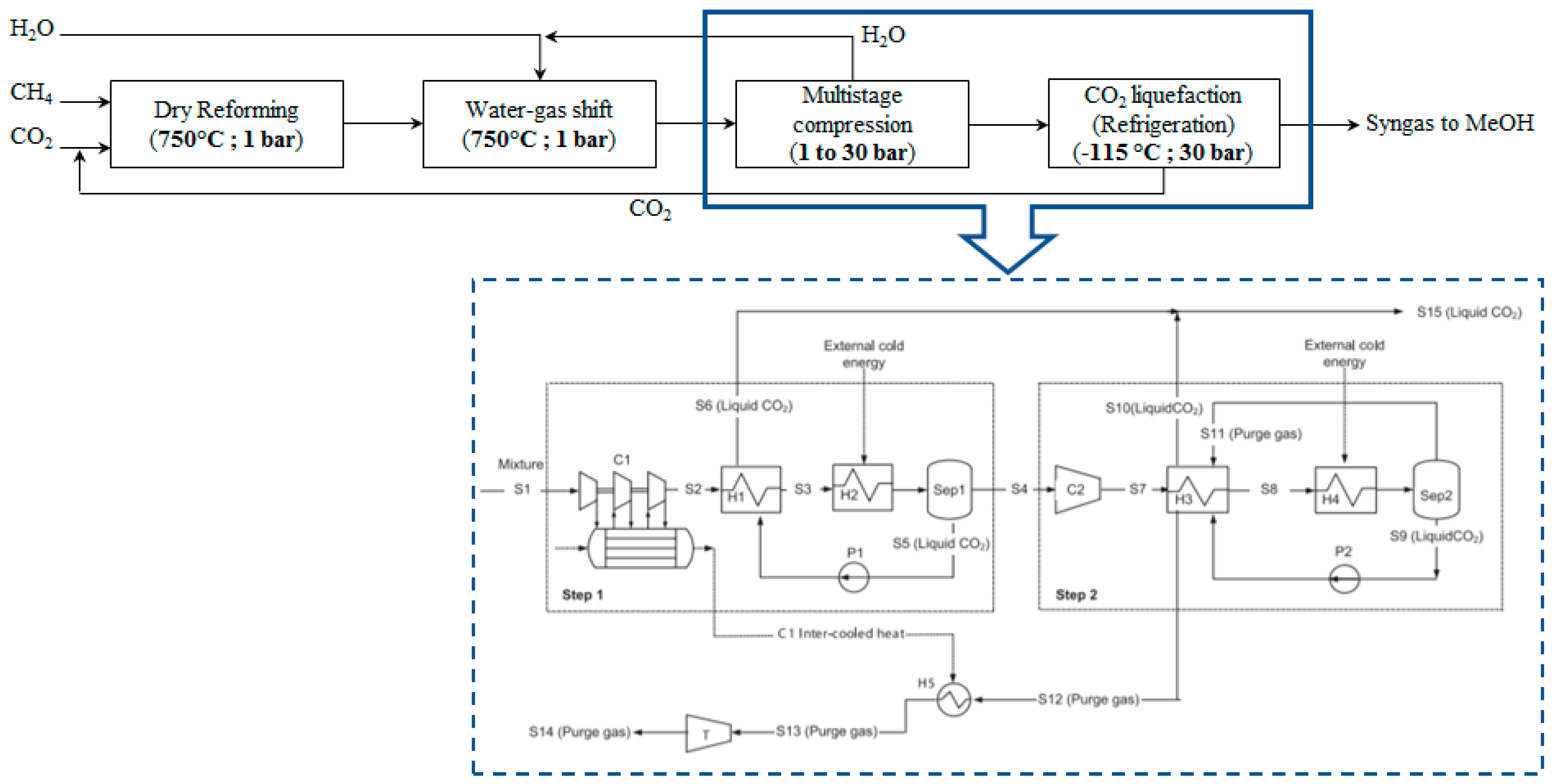
In this scenario, a cryogenic process is employed for CO
2 excess removal. The WGS reactor outlet stream is initially compressed to 30 bar, a suitable pressure for the Fischer-Tropsch process. Moreover, pressure increase facilitates the cryogenic separation as the liquefaction temperature increases too. Almost all H
2O is condensed and removed after the first compression stage; the compressed stream at 5 bar is cooled down to 20 °C and flashed in a knock-out drum (vapor–liquid separators). The stream is further compressed to 15 bar and cooled down firstly by the liquid stream coming from the knock-out drum and consequently, by an external cold utility. Part of the CO
2 is liquefied and recovered from the drum. The stream is further pressurized to 30 bar, cooled down to −115 °C and flashed. Under such conditions, almost the entire amount of CO
2 is liquefied and removed from the stream. The cold purified syngas stream can be integrated in the process and be used as cold utility. The PBD of the process and the process flow diagram of this downstream process alternative are presented in
Figure 5.
Cryogenic separation is not only used in oil and gas industry for light hydrocarbons separation [
56,
57], but also for synthetic natural gas purification [
47,
58]. Although cryogenic separation requires no chemical agents, the extreme operating conditions require high energy consumption, which is a major drawback. Especially when gases with lower boiling points (H
2, CH
4, and CO) are present, the phase transition temperature of CO
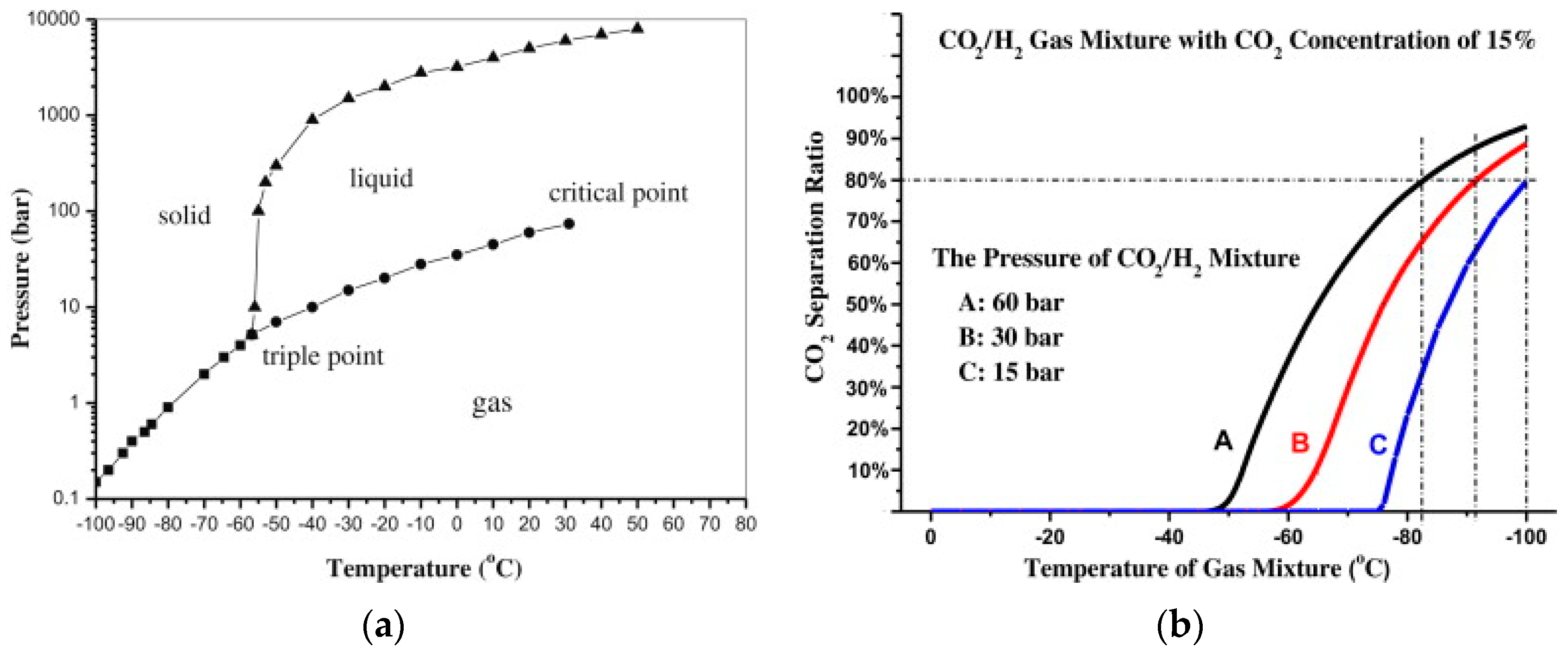
2 drops to values even lower than −80 °C and the refrigeration energy becomes the main cost driver of the separation. The effect of CO
2 concentration on the phase transition temperature is presented in
Figure 6.
Simulating this scenario in the Aspen Plus process simulator V8.8, it is found that the CO
2 phase transition temperature in the syngas stream (14.4% CO
2, 28.7% CO and 56.9% H
2) at a pressure of 30 bar is −115 °C for 99.2% recovery, result which is in accordance with
Figure 6b. Based on the simulation results, ~5.3 kWh/kmol
syngas cold utility, 3.1 kWh/kmol
syngas hot utility and 7.9 kWh/kmol
syngas electrical power are required for syngas purification. The capital investment of the CO
2 liquefaction process is lower than the MEA absorption process and similar to Selexol
TM absorption process [
55]. Xu et al. [
47] have also compared the total capital investments of a CO
2 liquefaction process over the Selexol
TM process and concluded that the first is about 40% less expensive than the second one. Thus, the capital investment of the CO
2 liquefaction process is about 4 k
$/(kmol
syngas h
−1) including the compression section and the refrigeration system needed to achieve the required operating temperatures.
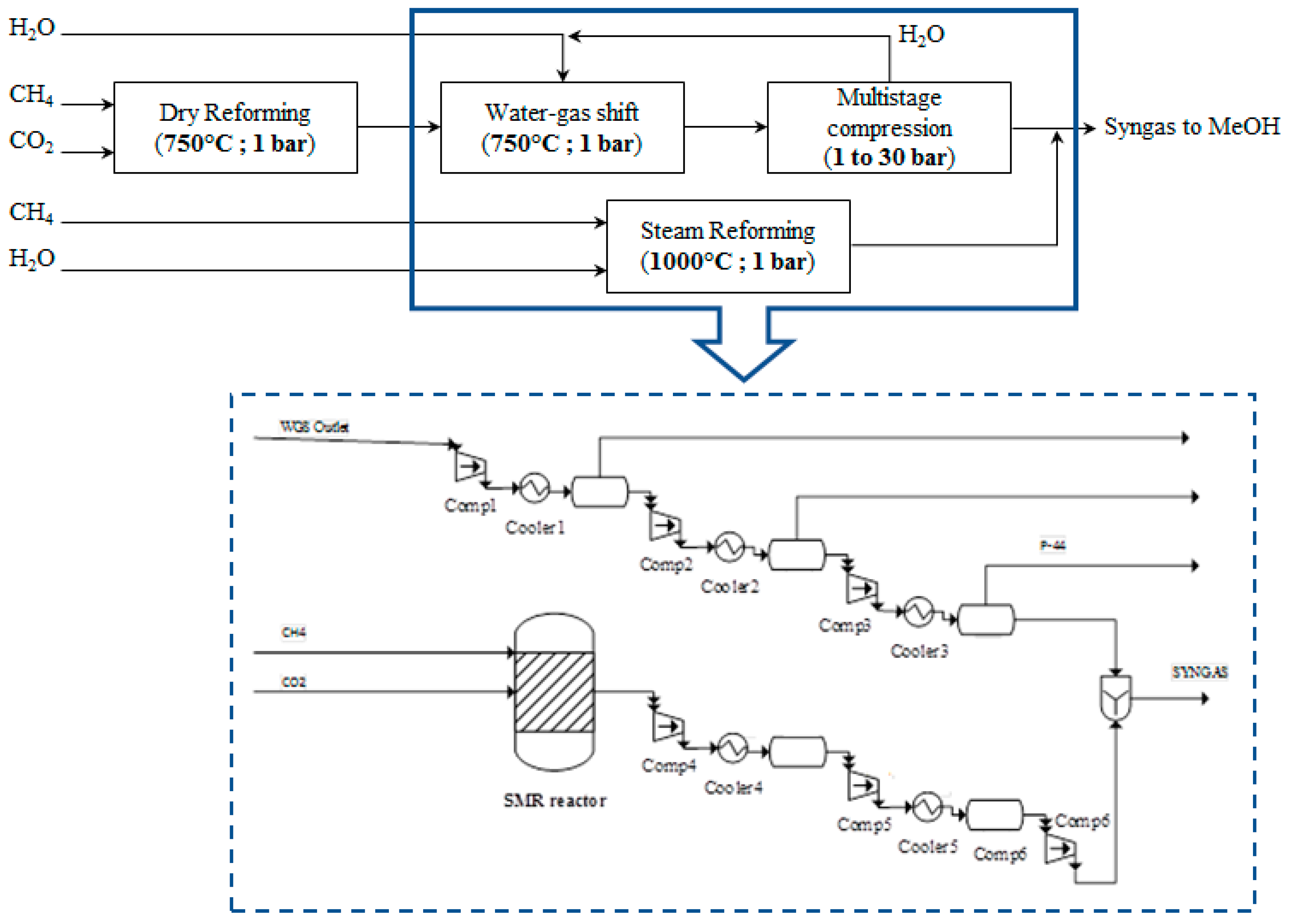
2.2.3. Thermal Catalytic DMR Process Integrated with SMR
Removal of CO
2 from the WGS outlet reactor stream increases the syngas production cost. Avoidance of implementation of CO
2 separation processes might increase the economic potential of syngas production. Additional gas conditioning units can be used instead of the energy and capital-intensive CCS technologies. In this scenario, steam methane reforming (SMR) is implemented in the process as an additional conditioning unit to tune the final syngas composition. Water (steam) reacts with CH
4 according to the following reaction:
Syngas with different ratio than the WGS reactor outlet stream is produced in the SMR reactor. The two syngas streams are mixed and the final syngas composition attains the targeted molar ratio.
The RGIBBS reactor model is also used to simulate the SMR reactor at steady state. Chemical equilibrium is achieved at 1000 °C and 1 bar. Equimolar feed stream is assumed at the inlet of SMR reactor. The amount of methane and steam that is added to promote the SMR reaction has been defined after a sensitivity analysis, setting as objective the targeted molar ratio H
2/(2CO + 3CO
2) ≥ 1 at the final syngas stream. A feed of 22,351 kg/h of methane and steam (10,528 kg/h of CH
4 and 11,823 kg/h of steam) results in achieving the targeted ratio. Conversion of 98.5% and 98.8% of CH
4 and H
2O (steam), respectively, are achieved at these conditions in the SMR reactor. The PBD and the process flow diagram of this downstream process alternative are presented in
Figure 7.
Combination of DMR, WGS and SMR also in one step, known as combined steam-CO
2 methane reforming (SCMR), has been studied elsewhere [
59,
60] as a possible route for syngas production. Gangadharan et al. [
61] concluded that a process combining dry methane reforming with steam methane reforming has comparable economic potential with the well-established SMR for syngas production and lower carbon footprint. Basini and Piovesan [
62] showed that this hybrid process is the most economical route to produce syngas with molar ratio of H
2/CO equal to 2. Based on the simulation results in the Aspen Plus process simulator V8.8, ~22.8 kW/(kmol
syngas h
−1) cold utility, 16.5 kW/(kmol
syngas h
−1) hot utility and 15.8 kWh/kmol
syngas electrical power are required for syngas purification. It is worth mentioning that there is high potential of heat integration in this process, which will radically reduce the operating cost [
63]. The capital investment of this process alternative is ~5.5 k
$/(kmol
syngas h
−1); it includes the cost of compression section, which has been estimated at 5.4 k
$/(kmol
syngas h
−1) by Luyben [
51], and the SMR reactor cost, which has been estimated at 0.06 k
$/(kmol
syngas h
−1) by Gangadharan et al. [
61].
2.2.4. Process Alternatives Evaluation for Thermal Catalytic DMR Process
In this section, the syngas purification process alternatives are evaluated based on their performance and the most competitive one is selected for the detailed design. The performance of each alternative is presented in
Table 2.
The MEA absorption process alternative requires the lowest amount of hot utility and electricity while the cold utility demand is almost the same as the CO
2 liquefaction process. The utilization of CH
4 to syngas is high too. However, it is the most capital-intensive process among the others, mainly due to distillation and stripping columns needed. Moreover, use of aqueous MEA solvents increases the environmental impact of the process. The CO
2 liquefaction process requires low cold and hot utility. Even though the cold duty is similar to the MEA absorption process, the actual cost is significantly higher because the operating temperatures are well below zero, so refrigerants as cooling media are required; in the other cases, cooling water is used since the operating temperatures are close to ambient. Considerable amount of electricity is required due to refrigeration system operation. No complex equipment is needed; therefore, the capital investment is low. Syngas losses due to purge streams, however, result in the lowest resource efficiency among the other process alternatives. The combined DMR + SMR process requires the highest cold and hot utility. In addition, ambient temperatures are targeted so cooling water can be used. High electrical power is required due to high flow rates of the process (steam is also added to promote the SMR reaction). The capital investment is relatively low since conventional equipment is used. The combined DMR + SMR process achieves the highest resource efficiency since syngas is the only product that is formed in the reformers and few separation steps are involved in the process, thus minimizing the material losses. Generally, the process design focus is on high resource efficiency (since raw material is the cost driver) and low capital investment. Low utility cost is also preferable but it is not a significant cost in syngas production cost as compared with the first two [
19,
64]. Based on the process evaluation, the combined SMR + DMR process is the most promising alternative. The economic potential of this process has been studied elsewhere [
61,
64].
2.3. Plasma-Assisted DMR Process
Purified streams of CH
4 and CO
2 are fed in the plasma reactor powered by a repetitive nanosecond pulsed power generator, where plasma-assisted dry methane reforming (P-DMR) occurs. The most important electron impact reactions, which take place inside the plasma reactor are described elsewhere [
65].
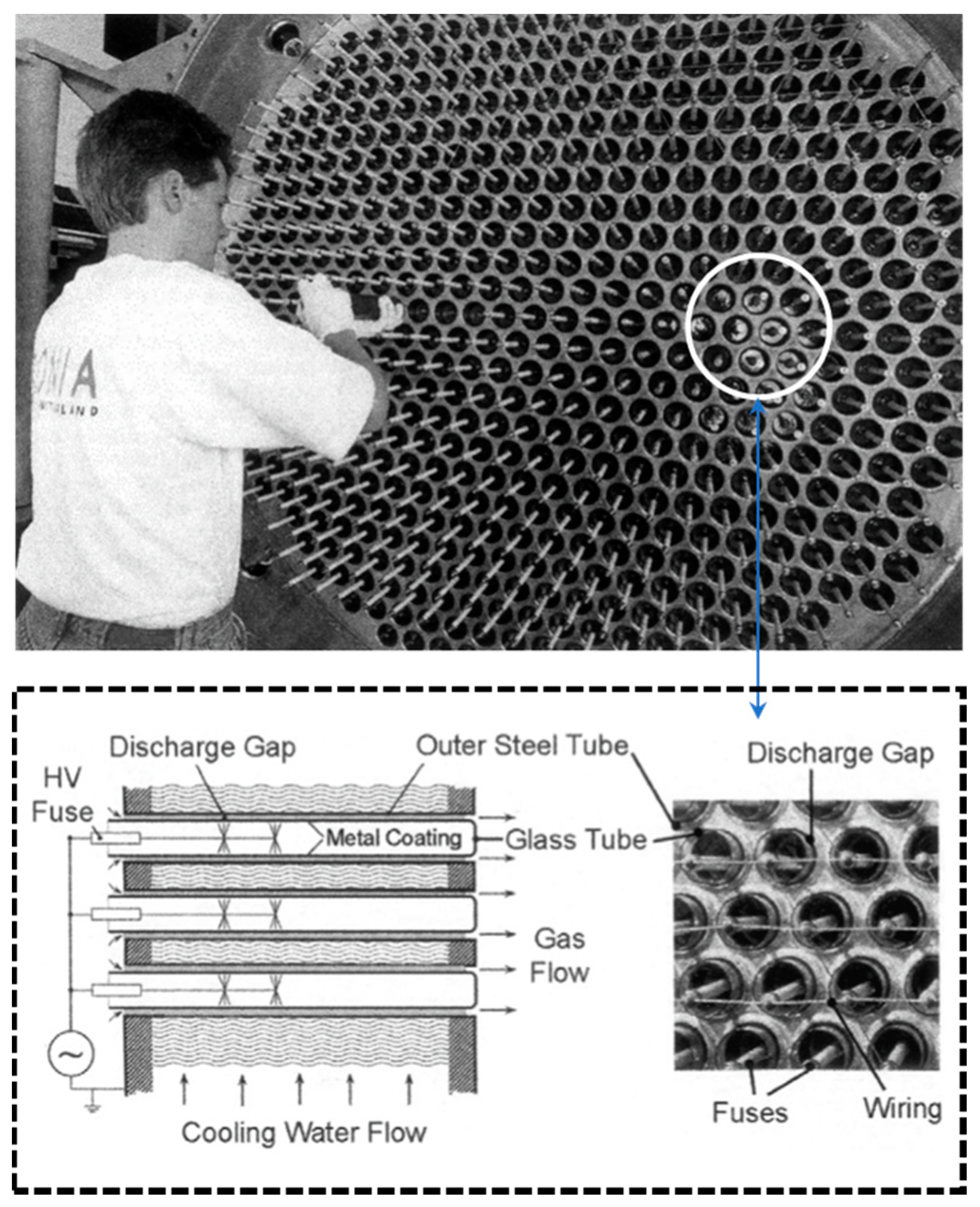
To develop the process flow diagram and evaluate the P-DMR process, the results obtained by Scapinello et al. [
31] are linearly extrapolated at industrial capacities. This is a valid assumption considering that the industrial scale plasma reactors are bundles of small scale plasma reactor units [
66], as it can be seen in
Figure 8.
Therefore, the RSTOIC reactor model (stoichiometry and molar extent is known for each reaction) is used to simulate the plasma reactor at steady state. The temperature and pressure in the plasma reactor is 200 °C and 1 bar. CH4 and CO2 conversion of approximately 51% and 42.5%, respectively are achieved at specific energy input of 10 kJ/Lfeed. CH4 conversion of is higher than that of CO2 because the dissociation energy of C-H bond (337.2 kJ/mol) is lower than the one of C=O (749 kJ/mol). Consequently, both high- and low-energy electrons may result in CH4 dissociation, while only high energy electrons may result in CO2 dissociation. Considering that the number of high energy electrons is lower than the number of low energy electrons in non-equilibrium plasma, the rate of CH4 dissociation reactions is higher than that of CO2. Thus, higher CH4 conversion is observed in the P-DMR process.
The composition of the P-DMR reactor outlet stream after considering all the important plasma activated reactions are presented in
Table 3. The H
2/CO molar ratio in the P-DMR outlet stream is 1.1. However, the molar ratio should be increased in order to reach the targeted value while unconverted reactants still need to be recovered from the stream and recycled back to the reactor.
The above-mentioned CCS technologies can be used for the unreacted CO
2 removal. Chemical absorption, membranes and adsorption technologies are mature and applicable to CH
4 removal. Even hybrid solutions (membrane and absorption based technologies) have been proposed and explicitly designed for this purpose [
67]. Once the P-DMR outlet stream is purified and the targeted molar ratio is achieved (H
2/CO ≥ 2), syngas may be fed in the FT process for methanol production.
Reactants removal can also be realized by secondary reactions; in this study, CH4 can be further converted in a SCMR reactor into syngas simply by adding steam. Except for CH4, a small amount of CO2 also reacts with H2 (RWGS reaction). Simultaneously, CO is converted to H2 (WGS reaction) resulting in H2/CO molar ratio increase. Eventually, the whole CH4 amount is converted to syngas in one reactor pass, while less CO2 needs to be removed from the syngas stream prior to FT.
Two PBDs, which correspond to the above-mentioned separation alternatives, are developed and evaluated below.
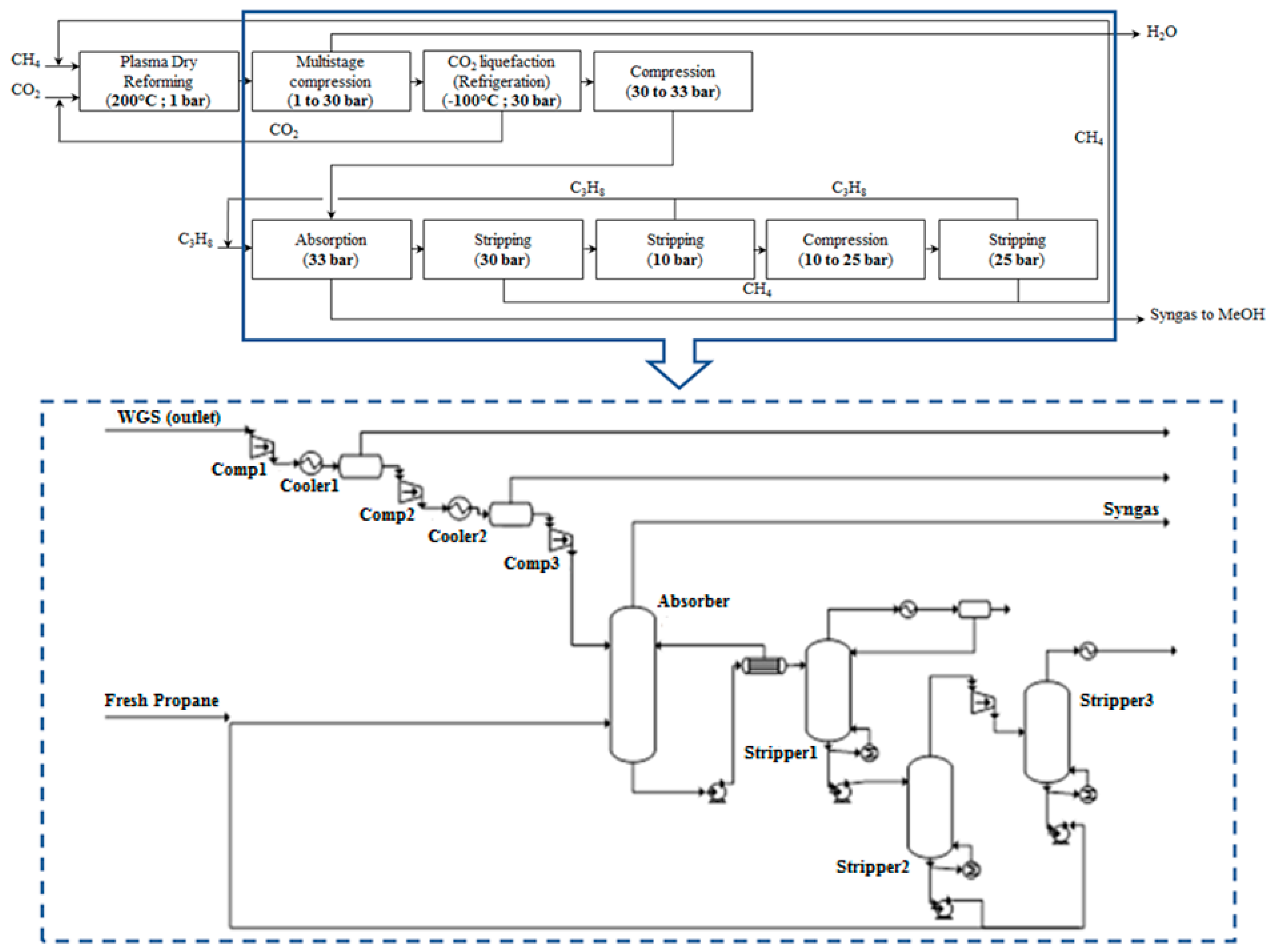
2.3.1. Plasma-Assisted DMR Process with CH4 and CO2 Removal
This process configuration is initially simulated in the Aspen Plus process simulator V8.8. The P-DMR outlet stream is compressed in two steps. First, it is compressed from atmospheric pressure to 30 bar to facilitate CO
2 removal by liquefaction; the stream is simultaneously cooled down to −100 °C. The liquid CO
2 is recovered from the bottom of a knock-out drum. The separation of CH
4 from H
2 and CO is accomplished by an absorption/stripping process [
68] at industrial scale. The cooled, free-of-CO
2 stream is further compressed to 33 bar to enhance the solubility of CH
4 in the liquid C
3H
8, which is chosen as suitable solvent; the solubility of CH
4 in liquid C
3H
8 is the highest among the organic solvents, such as C
4H
10, C
5H
12, C
6H
14 and C
8H
18 [
68]. The free-of-CO
2 stream enters the absorption column from the bottom and flows counter-currently with the solvent stream, which enters form the top. CH
4 is selectively dissolved and the rich-in-CH
4 stream leaves from the bottom of the column. The purified syngas stream escapes from the top. The solvent is regenerated in a stripping column at 30 bar where 80.5% of CH
4 is recovered. The stripper outlet stream is depressurized down to 10 bar and stripped further in a second stripping column where all the amount of CH
4 is recovered. Inevitably, ~15% of C
3H
8 is contained in the CH
4 stream; the remaining 85% of C
3H
8 is recycled back to the absorber. A third stripper, operating at 25 bar, is employed to recover the remaining amount of solvent, since C
3H
8 losses drive the economic viability of this separation process. It has been estimated that the separation is not economically viable for solvent losses higher than 6% [
68]. In this process simulation, 99% of the solvent is recovered. Eventually, CH
4 and C
3H
8, which are separated in the last stripping tower, are recycled back to the plasma reactor and absorber, respectively. The excess of CO
2 is further removed in a CO
2 liquefaction process and the targeted molar ratio (H
2/(2CO + 3CO
2) ≥ 1) is attained. The PBD and the respective process flow diagram are presented in
Figure 9.
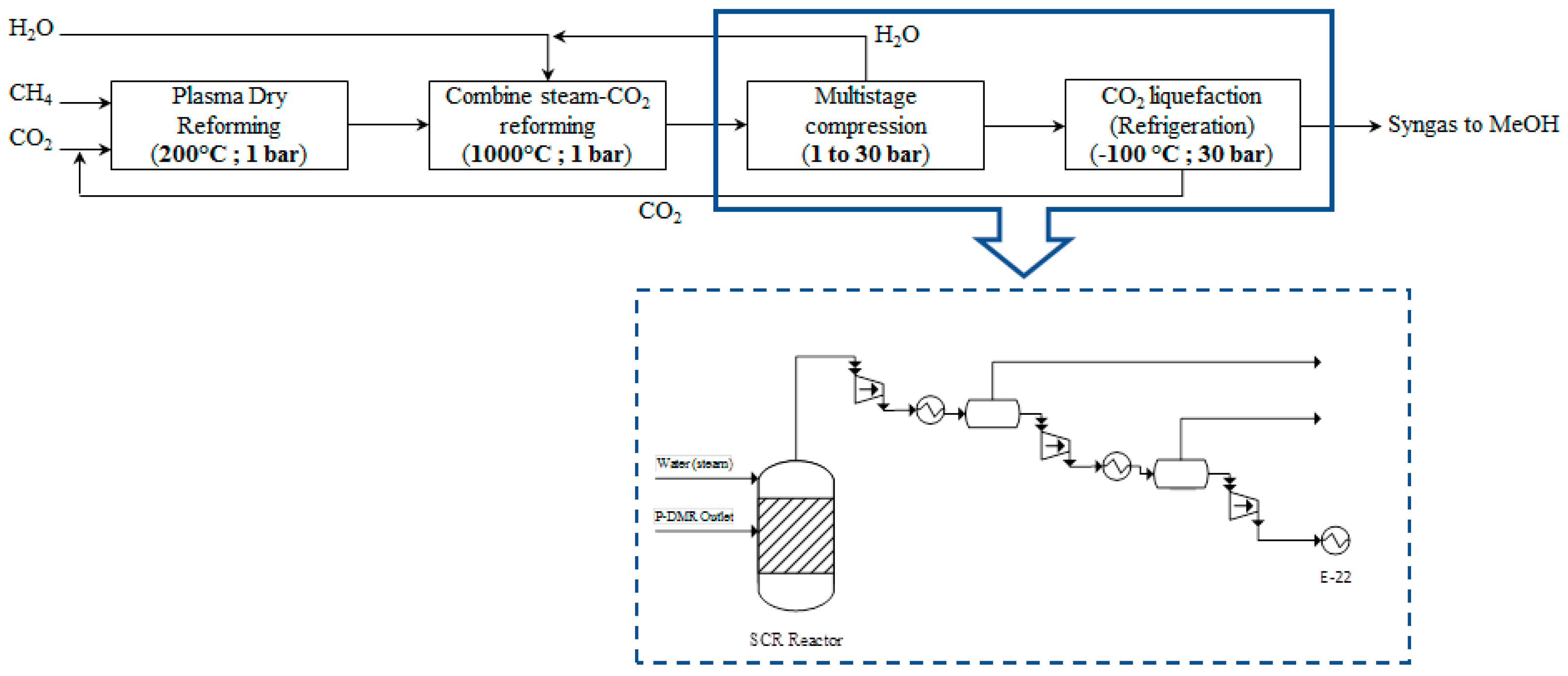
2.3.2. Plasma-Assisted DMR Process Integrated with SCMR
Steam-CO
2 combined methane reforming (SCMR) is chosen in this scenario as a process alternative to “purify” the P-DMR outlet stream by adding water, thus, SCMR is promoted according to the following reaction:
SCMR is a process with high industrial interest since syngas with H
2/CO = 2 molar ratio is produced that is suitable for gas-to-liquids [
69] and catalysts have been synthesized to boost the reaction performance [
59,
60,
70].
The RGIBBS reactor model is used to simulate the SCMR reactor at steady state. Chemical equilibrium is achieved at 1000 °C and 1 bar. The amount of steam that is added to promote the SCMR reaction has been defined after a sensitivity analysis, setting as objective the molar ratio of H
2/CO to be at least equal to 2 at the SCMR outlet stream. According to Jang et al. [
30], (CO
2 + H
2O)/CH
4 molar ratio higher than 1.2 (8.5 in this simulation), CO
2/H
2O ratio higher than 0.47 (0.6 in this simulation) and a temperature higher than 850 °C (1000 °C in the simulation) are preferable reaction conditions for syngas preparation suitable for GTL processes. Co-feeding of 18015 kg/h of water (steam) results in the targeted ratio. CH
4, CO
2 and H
2O conversion of 99.9%, 10% and 23.1%, respectively, are achieved at these conditions in the SCMR reactor. The composition of the SCMR reactor outlet stream is presented in
Table 3. The excess of CO
2 is further removed in a CO
2 liquefaction process and the targeted molar ratio (H
2/(2CO + 3CO
2) ≥ 1) is attained. The respective PBD as well as the process flow diagram are presented in
Figure 10.
2.3.3. Process Alternatives Evaluation for Plasma-Assisted DMR Process
The process alternative of CH
4 and CO
2 removal comprises eight process steps. Each process step requires unique process equipment while the feed throughputs are high; thus, the capital investment is foreseen to be high. Consequently, maintenance cost, which is assumed to be a fixed percentage of capital investment [
71], is expected to be high, too, in this scenario. Moreover, temperature and pressure swing, which aim at solvent recovery together with high flow rate streams due to solvent use, increase the electricity demand, hot and cold duty in the process. This process certainly leads to highly complex process design.
The plasma-assisted DMR integrated with SCMR seems to be simpler process than P-DMR with CH4 and CO2 removal since it comprises four process steps. The capital investment and maintenance cost are foreseen to be lower due to less pieces of equipment, while constant pressure and temperature profiles mitigate the utility demand. In addition, higher CH4 and CO2 conversion is reached per reactor pas, which leads to higher resource efficiency. Collectively, plasma-assisted DMR integrated with SCMR is the most promising process alternative and is selected for the detailed design.
2.4. Heat Integration
Heat integration is performed in both thermal catalytic and plasma-assisted DMR process to recover and efficiently utilize all the available hot and cold process streams, thus, simulate a more relevant industrial environment. The hot and cold utility demand is minimized and a more realistic energy comparison between the two processes is conducted. Eventually, the respective heat exchanger network is designed with respect to the pinch analysis principles. The following assumptions are considered:
- -
The minimum temperature difference (ΔΤmin) is set at 20 °C
- -
The streams enthalpy is based on energy balances obtained by the process simulation.
- -
For the streams where phase change occurs during heating up, a pseudo CPM is calculated comprising the required energy both for evaporation and heating; the total stream enthalpy (ΔΗ) is divided by the respective temperature difference.
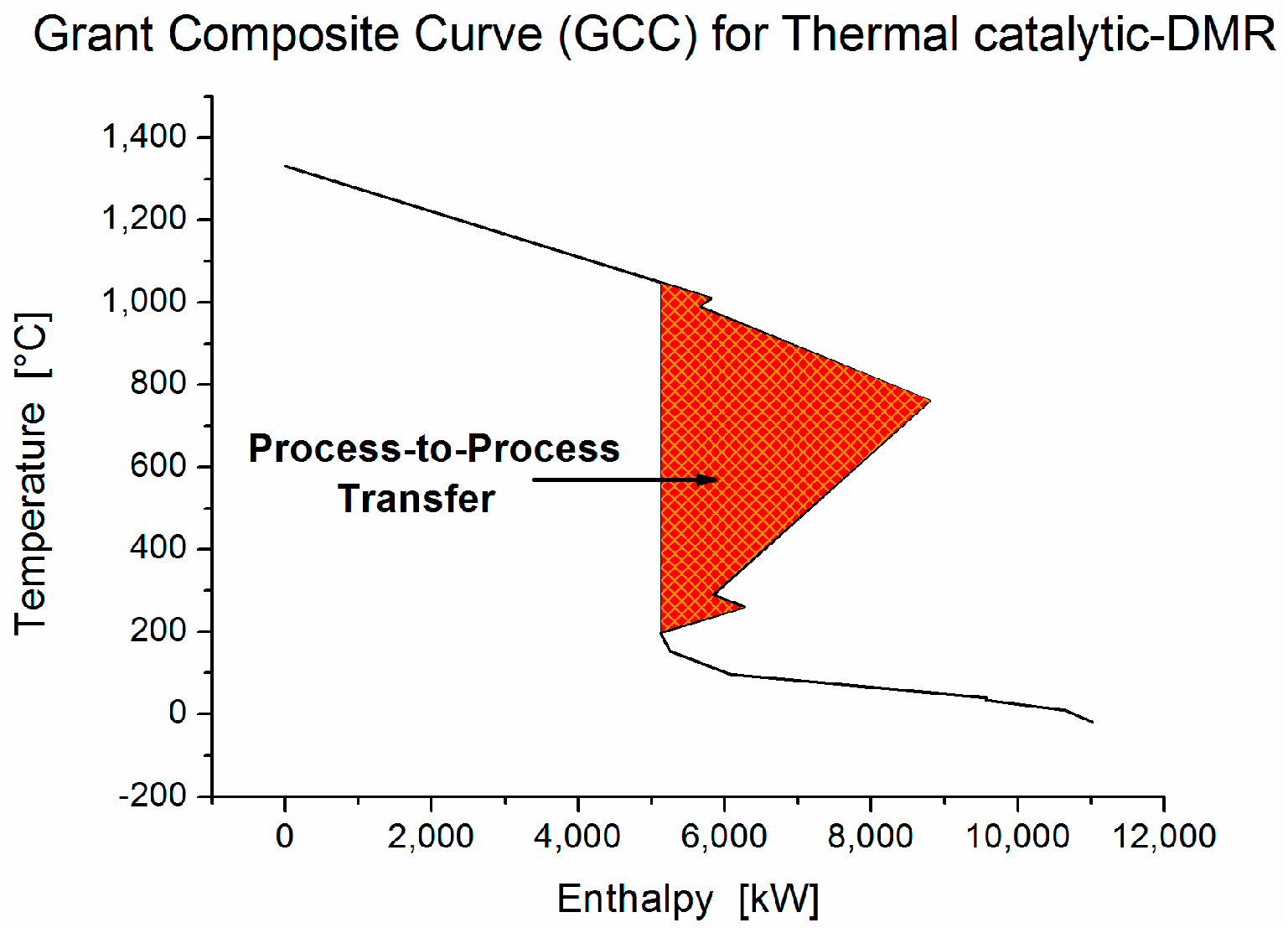
In a thermal catalytic DMR process, five cold and six hot process streams are integrated to reduce the total hot and cold utility demand (
Table 4). The total hot and cold utility demand before heat integration is 45.7 MW and 56.7 MW, respectively. The heat balances for the temperature intervals (
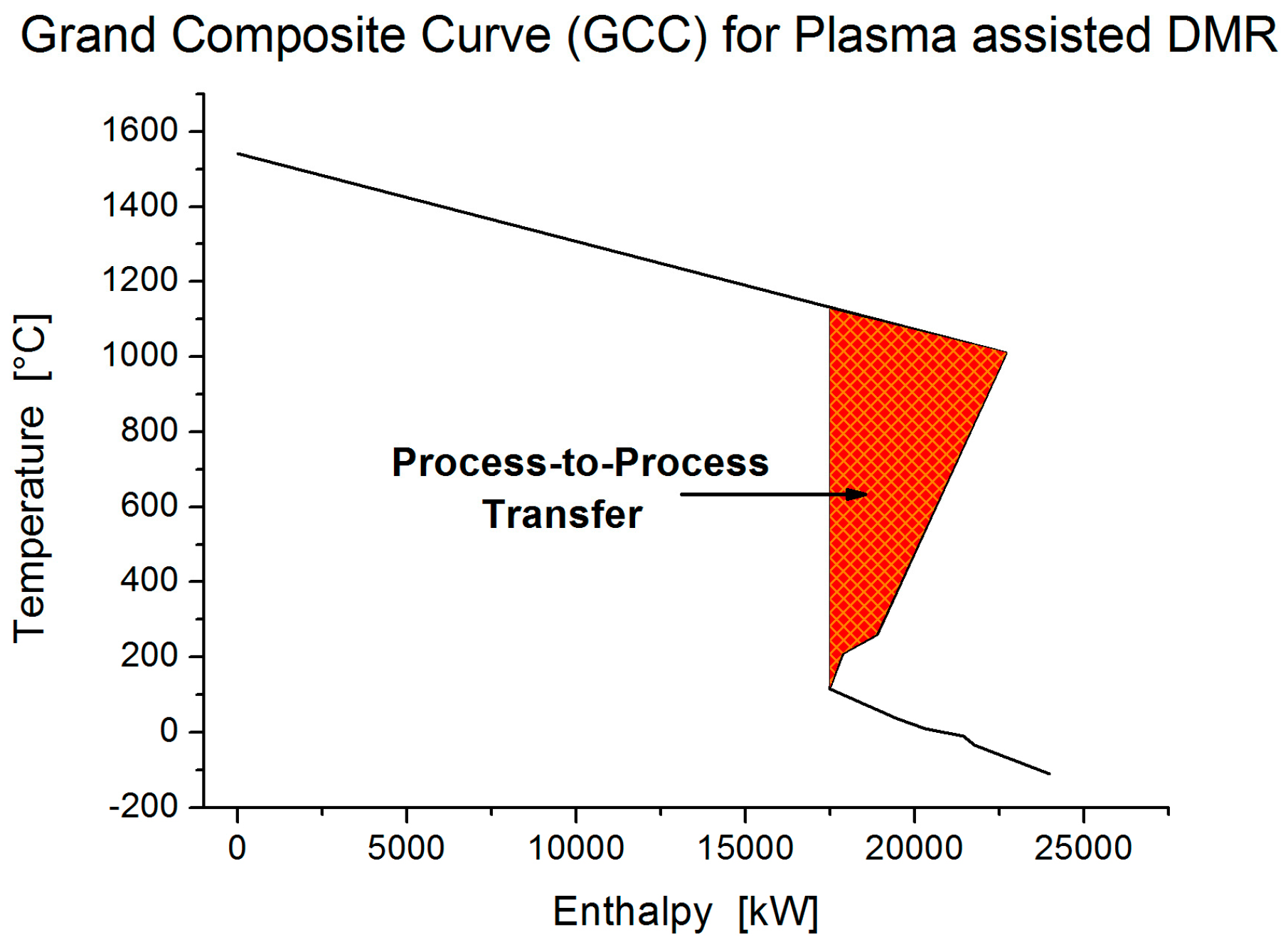
Table 5) show that high surplus of energy is accumulated in the process. This fact is confirmed by the grand composite curve (GCC) (
Figure 11) where the net heat flow against the shifted temperature is presented. A considerable energy pocket occurs in the process and proves the high potential of energy integration in thermal catalytic DMR process. The problem table algorithm for the required utilities (
Table 6) reveals that ideally no hot utility is required to preheat the process streams at steady state. Heat is required only for the endothermic DMR, WGS and SMR reactions.
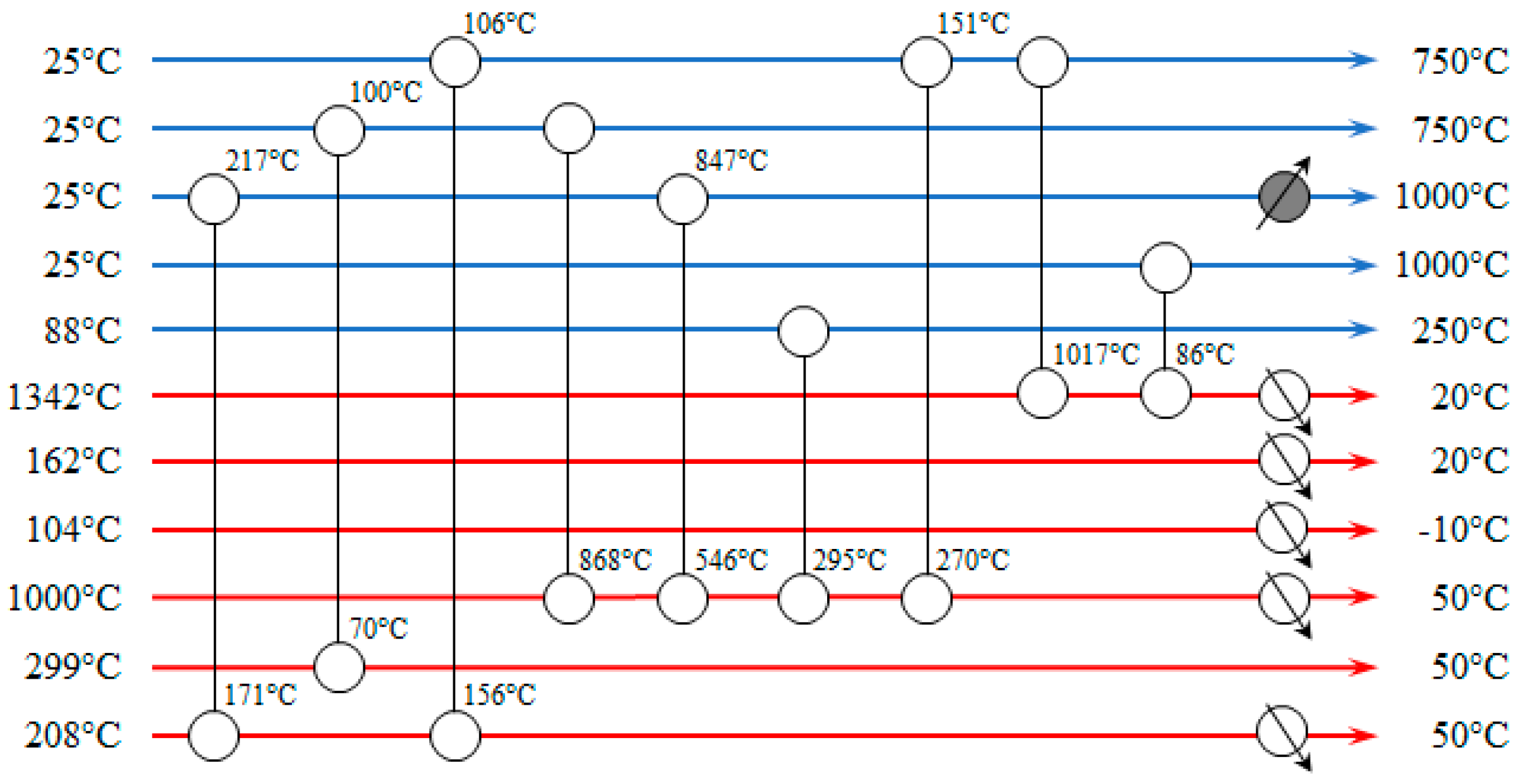
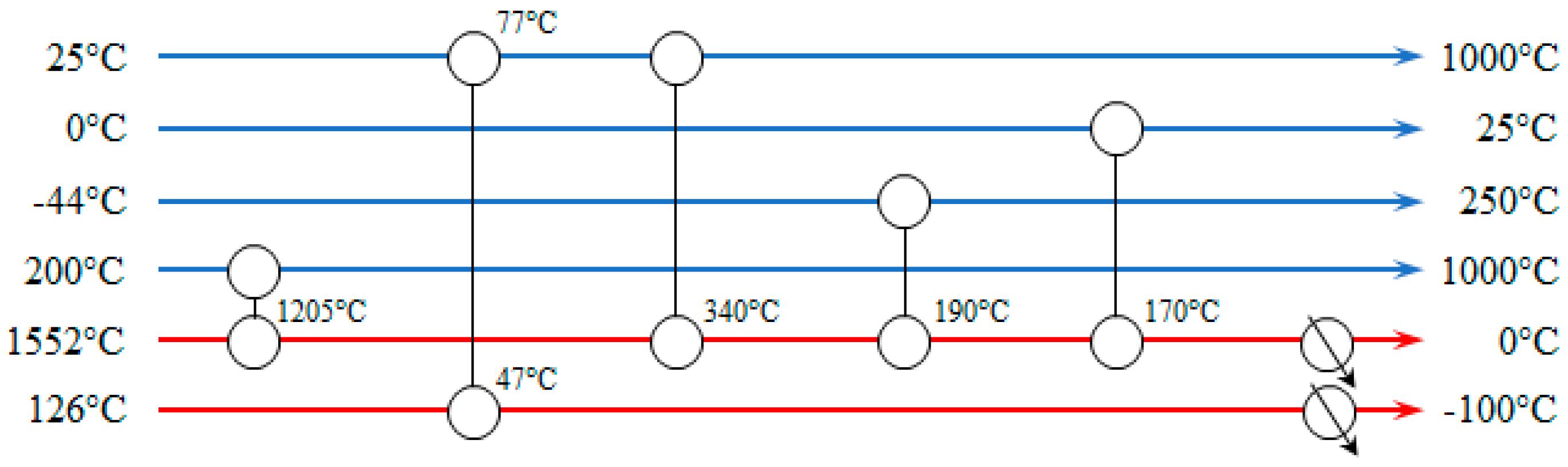
The heat exchanger network of the thermal catalytic DMR process is presented in
Figure 12. Nine process-to-process streams heat exchangers and five coolers are required for the heat integration. Ideally, no hot utility demand is required. However, in the proposed heat exchanger network the CH
4 stream to SMR reactor needs to be heated from 847 °C to 1000 °C by an external heat source, even though there is still an amount of heat in the process, which could be used to satisfy this heat deficit. It has been estimated that two more heat exchangers should have been placed in the network to achieve that resulting in higher capital investment. Instead of designing a more expensive and complicated network, it is preferred to heat the stream up to 1000 °C in the SMR.
In plasma-assisted DMR process, four cold and two hot process streams are integrated to reduce the total hot and cold utility demand (
Table 7). The total hot and cold utility demand before heat integration is 48.8 MW and 72.8 MW, respectively. The heat balances for the temperature intervals (
Table 8) show that there is a surplus of energy in the process that can be used for heating. This energy pocket gets noticeable in GCC (
Figure 13) and is high enough to satisfy the heating requirements as it is shown in the problem table algorithm (
Table 9). Eventually, no extra source of energy is required to preheat the streams at steady state; heat is required only for the endothermic SCMR reaction.
The heat exchanger network of the plasma-assisted DMR process is presented in
Figure 14. Five process-to-process streams heat exchangers and two coolers are required for the heat integration. The cold streams are heated up to the target temperature without extra hot utility. The two extra coolers cool the hot streams down to the target temperature.
Both processes manage to reduce the utilities demand after the heat integration. In
Table 10, the hot and cold utility demand before and after heat integration is presented; 95.4% and 79.7% less hot and cold utility, respectively, is required in thermal catalytic DMR; no hot utility and reduced cold utility by 67% is required in the plasma-assisted DMR process. The utility demand reduction is attained at the expense of capital investment. The number of heat exchangers increases, thus capital investment increases too. However, the objective is to attain highly energy efficient DMR processes and the heat exchangers cost is not expected to significantly raise the capital investment.
2.5. Equipment Cost Estimation
The cost of the process equipment needed for syngas production through thermal catalytic and plasma-assisted DMR is estimated. The equipment cost is estimated using cost equations, which are based on the key characteristic of each piece of equipment [
71]. The key characteristic of each piece of equipment is obtained from the process simulation.
The cost of thermal catalytic DMR, SMR, WGS and SCMR reactors is estimated by the following equation:
The reactors are fired heaters, box type reformers, made of carbon steel and operating at moderate pressure. The key characteristic of the thermal catalytic reactors is the net heat duty.
The cost of compressors is estimated by the following equation:
Reciprocating compressors are used for the gas transportation and compression. The key characteristic of the compressors is the indicated horsepower required for gas transportation and compression to the target pressure.
Shell-and-tube heat exchangers made of carbon steel are employed to build the heat exchanger network. The key characteristic is the heat exchange area and the cost is estimated by the equation:
where
;
.
The cost of coolers which operate at 20 °C is given by the same equation as the heat exchangers cost. The cost of the coolers, which operate at lower temperature (0 °C, −10 °C and −100 °C), is given by:
where F is 1, 1.55 and 4 for 0 °C, −10 °C and −100 °C, respectively.
The cost of the plasma reactor is based on the lab scale plasma reactor cost, which was used by Scapinello et al. [
31] and reached ~8 k
$. The cost also takes into account the cost of repetitive nanosecond pulsed power unit and the six-tenth power rule is used to estimate the installed cost of the plasma reactor at industrial scale. Eventually, using installation factors, the purchase equipment costs are converted into installed costs. The purchase and installed process equipment cost for thermal catalytic and plasma-assisted DMR process are presented in
Table 11 and
Table 12 respectively. Regarding the thermal catalytic process, compressors have the highest contribution to the equipment cost. SMR and DMR reactors are also significant costs. In the plasma-assisted process, the plasma reactor is the most expensive piece of equipment. Compressors and refrigeration systems are also expensive equipment units.