Nevoid Basal Cell Carcinoma Syndrome: A Long-Term Study in a Family
Abstract
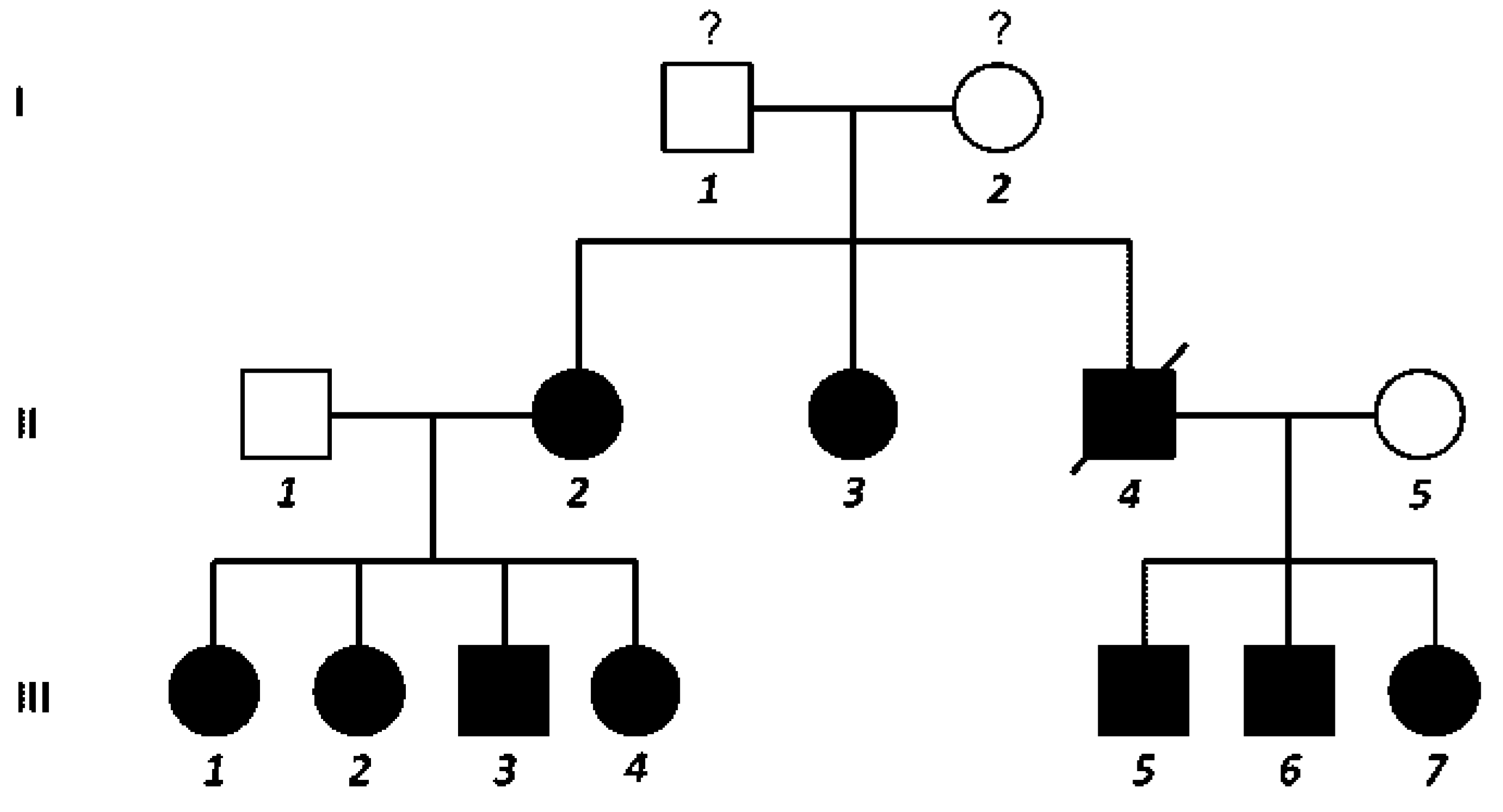
Methods
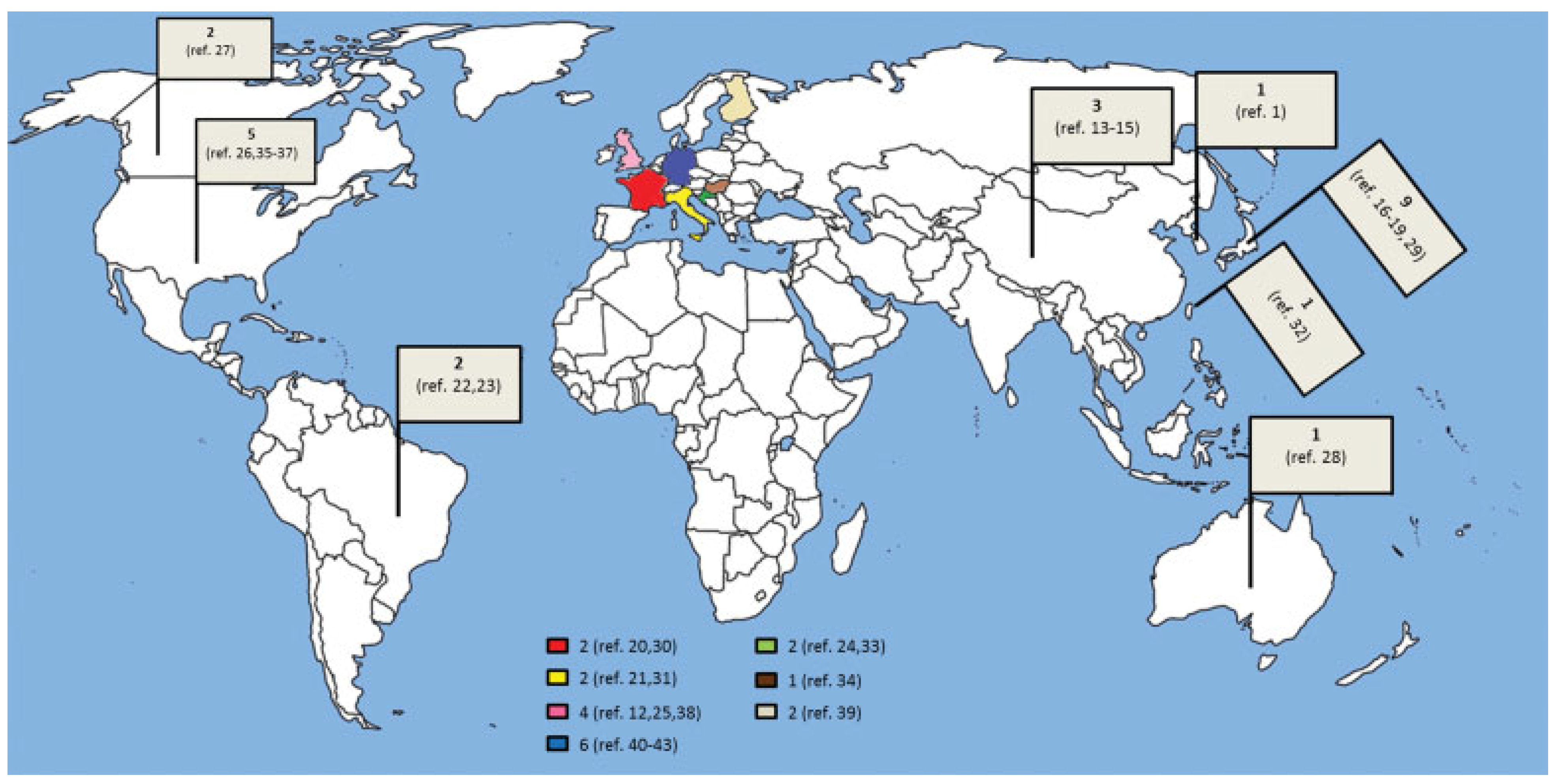
Literature Review
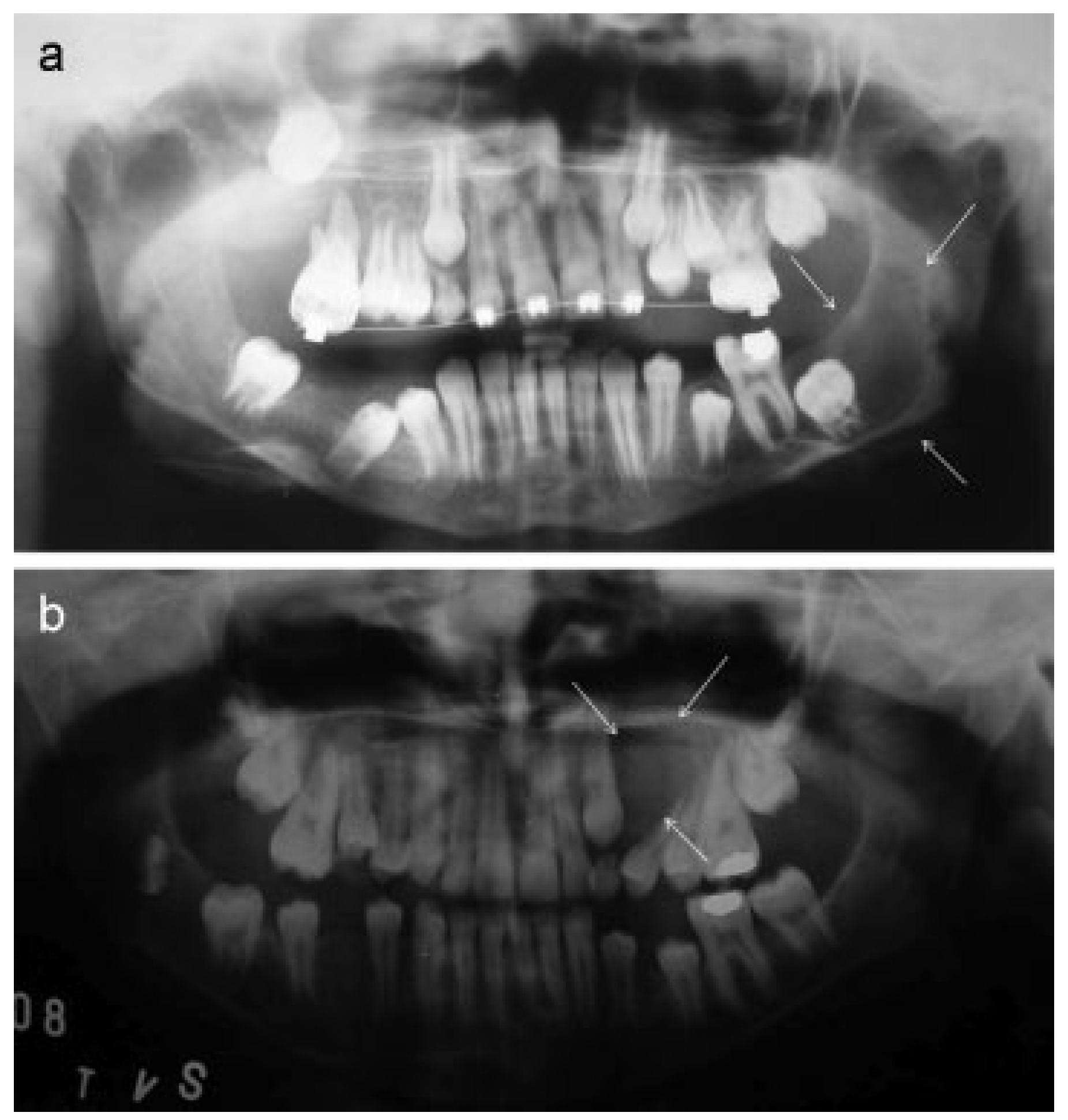
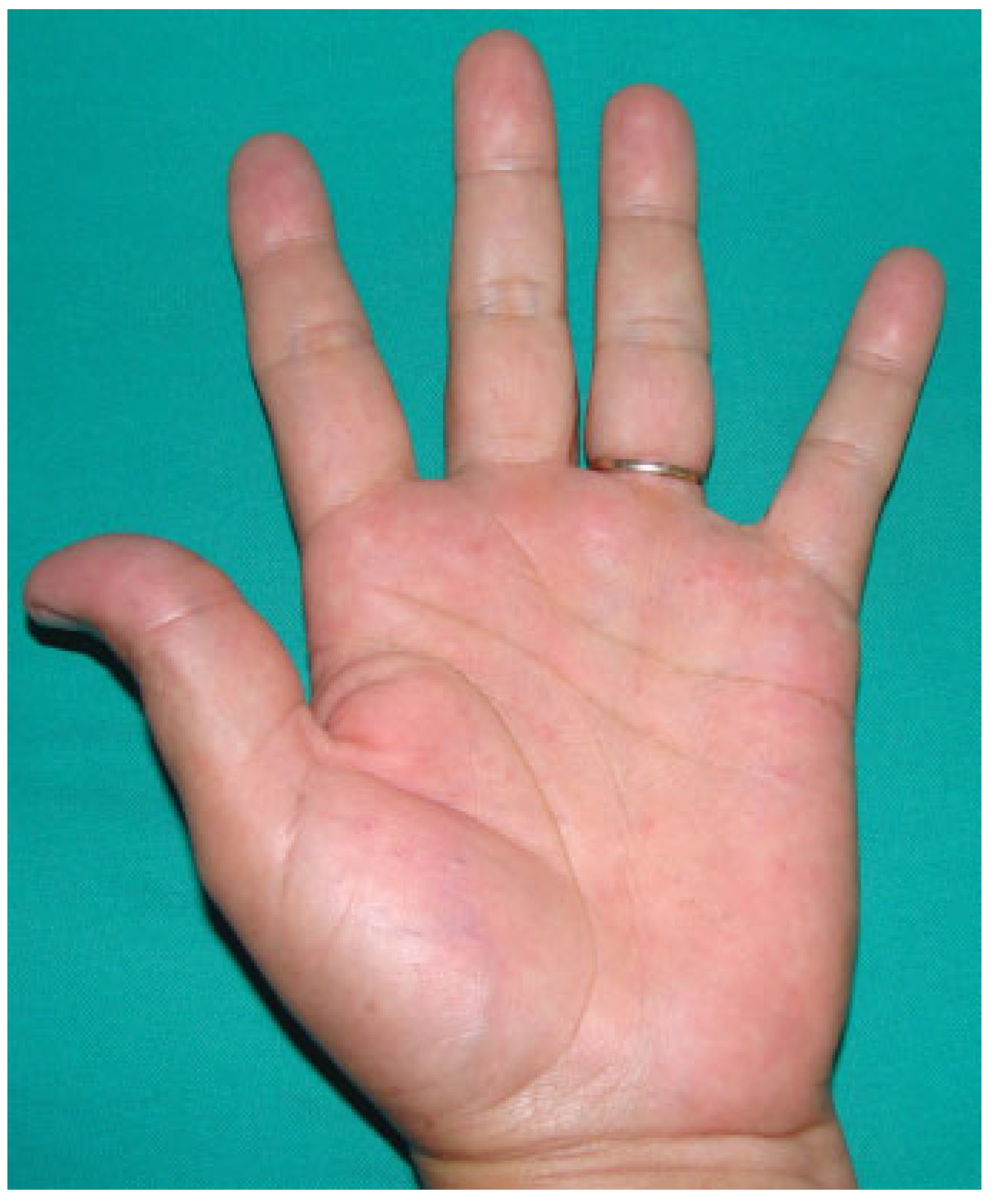
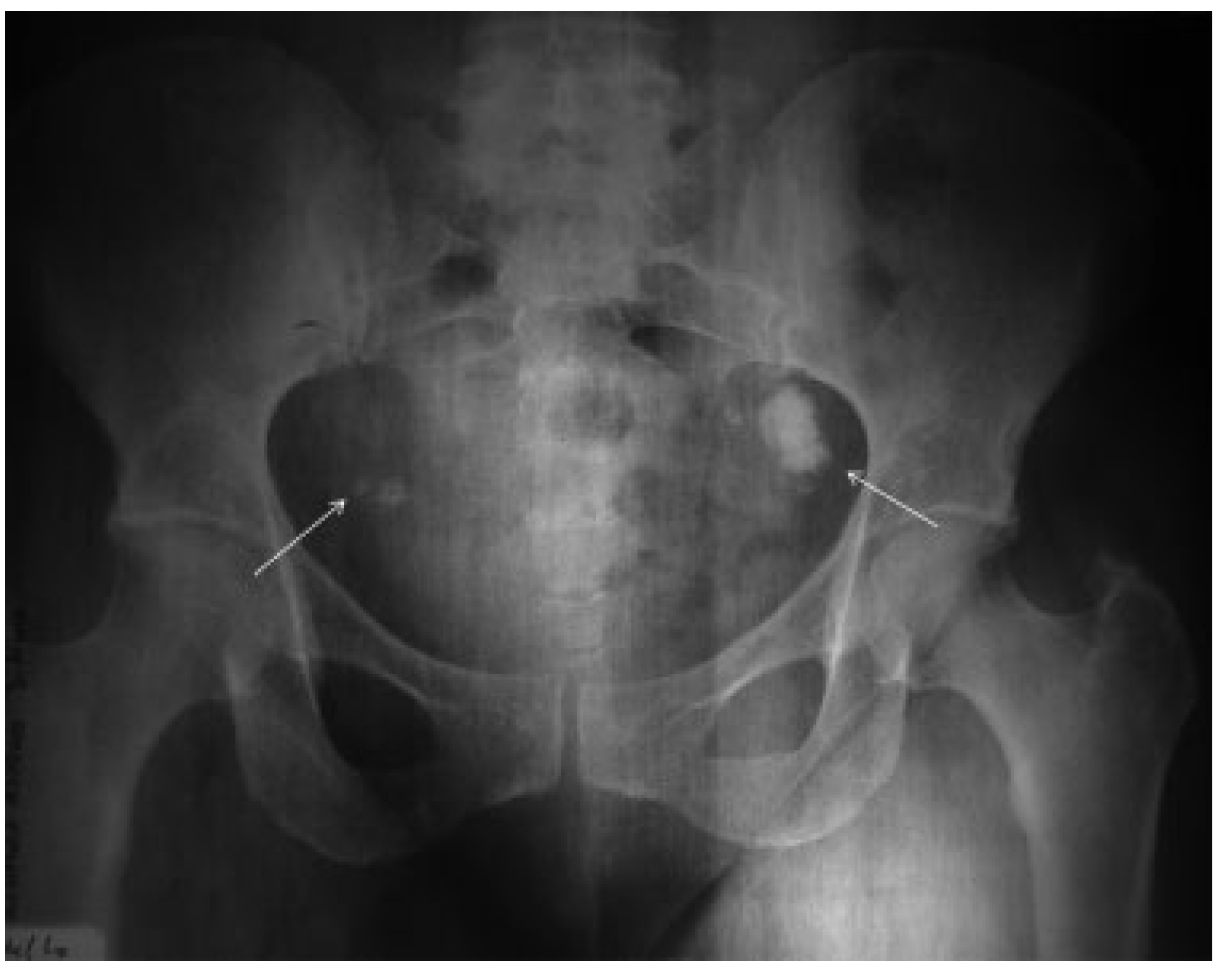
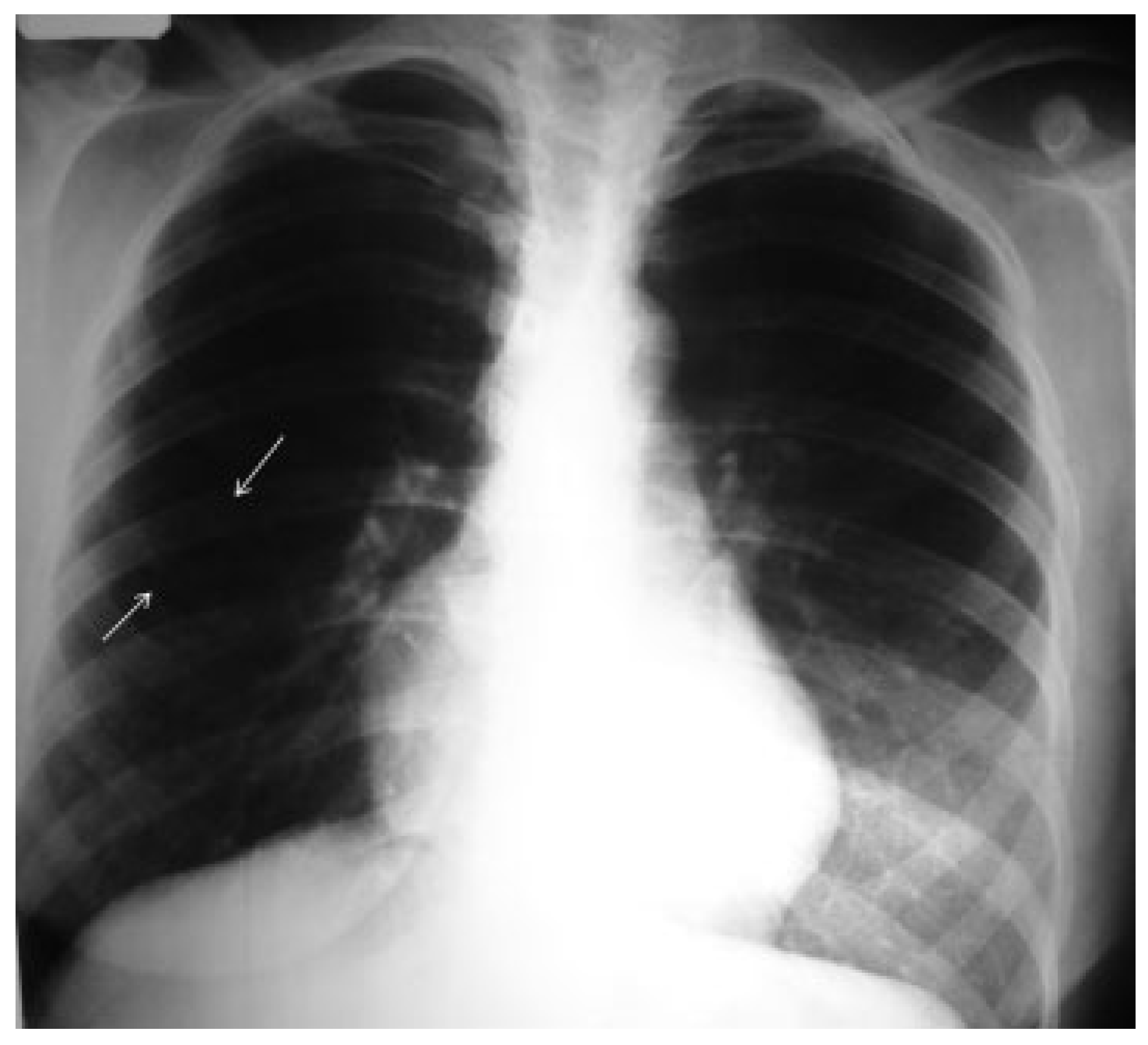
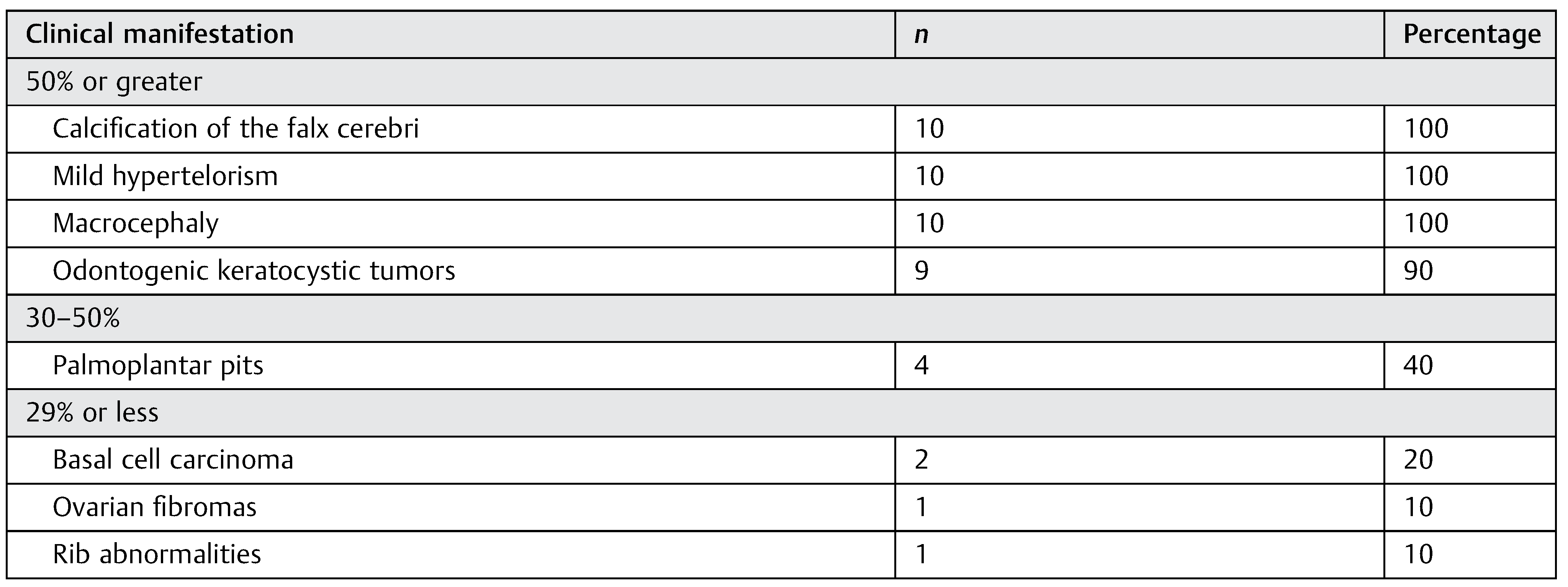
Results
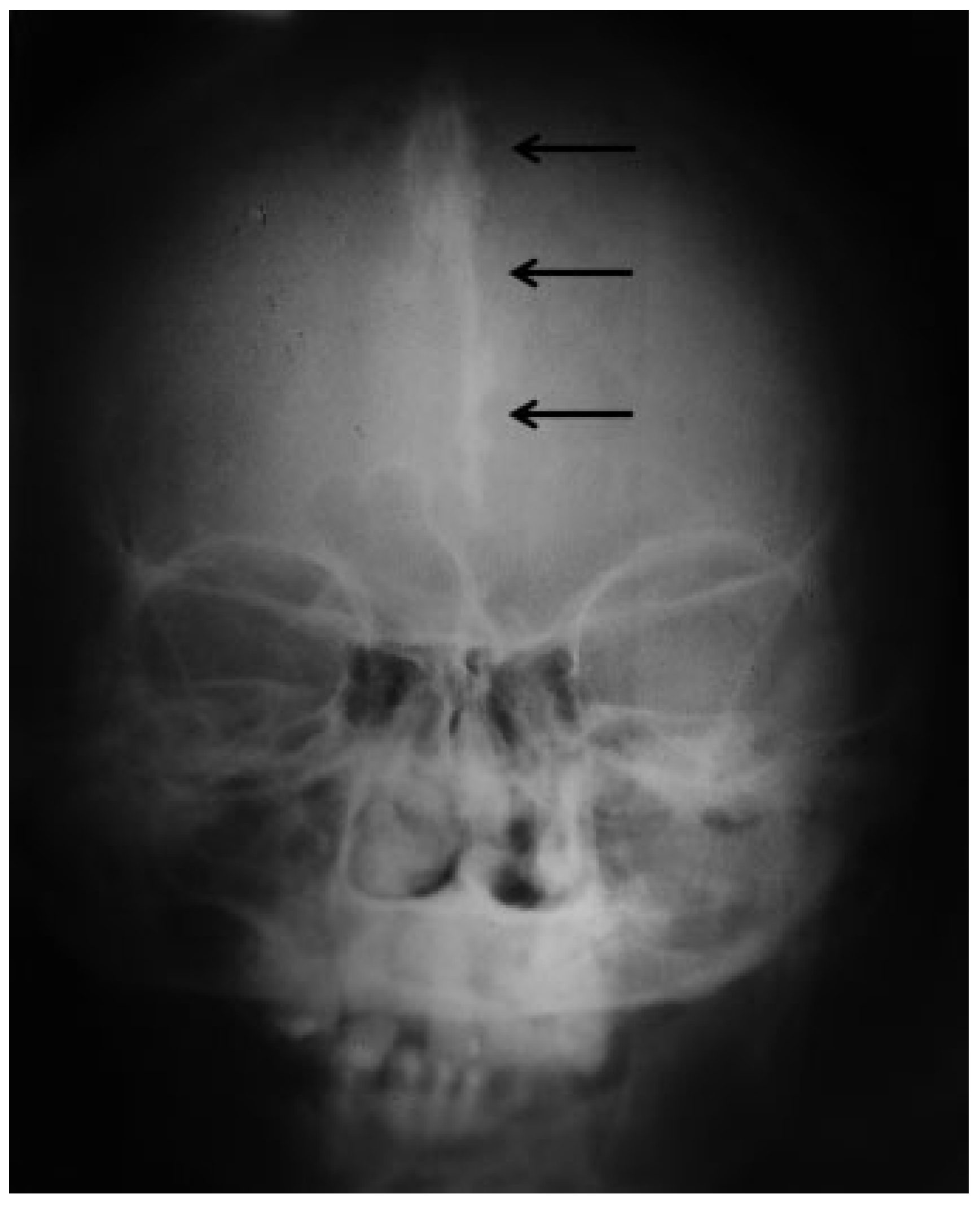
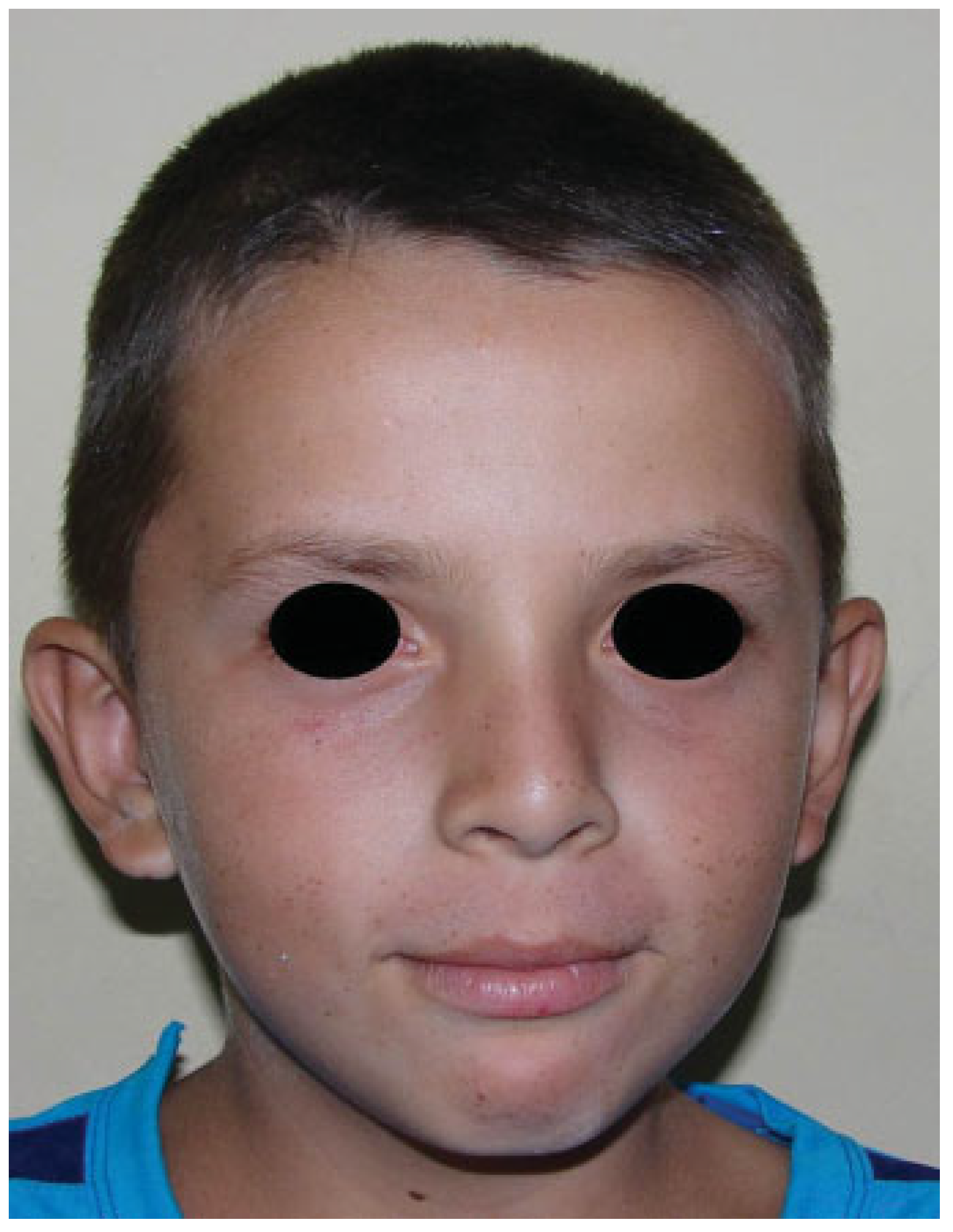
Discussion
Note
References
- Lee, Y.W.; Roh, B.H.; Ki, C.S.; et al. Identification of a novel mutation in the PTCH gene in a Korean family with naevoid basal cell carcinoma syndrome. Clin Exp Dermatol 2007, 32, 202–203. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.G.; Farndon, P.A.; Burnell, L.D.; Gattamaneni, H.R.; Birch, J.M. The incidence of Gorlin syndrome in 173 consecutive cases of medulloblastoma. Br J Cancer 1991, 64, 959–961. [Google Scholar] [CrossRef]
- Gundlach, K.K.; Kiehn, M. Multiple basal cell carcinomas and keratocysts—the Gorlin and Goltz syndrome. J Maxillofac Surg 1979, 7, 299–307. [Google Scholar] [CrossRef]
- Habibi, A.; Jafarzadeh, H. Nevoid basal cell carcinoma syndrome: a 17-year study of 19 cases in Iranian population (1991-2008). J Oral Pathol Med 2010, 39, 677–680. [Google Scholar] [CrossRef] [PubMed]
- Gorlin, R.J.; Goltz, R.W. Multiple nevoid basal-cell epithelioma, jaw cysts and bifid rib. A syndrome. N Engl J Med 1960, 262, 908–912. [Google Scholar] [CrossRef] [PubMed]
- Kimonis, V.E.; Goldstein, A.M.; Pastakia, B.; et al. Clinical manifestations in 105 persons with nevoid basal cell carcinoma syndrome. Am J Med Genet 1997, 69, 299–308. [Google Scholar] [CrossRef]
- Shafei-Benaissa, E.; Savage, J.R.; Babin, P.; et al. The naevoid basal-cell carcinoma syndrome (Gorlin syndrome) is a chromosomal instability syndrome. Mutat Res 1998, 397, 287–292. [Google Scholar] [CrossRef]
- Hahn, H.; Wicking, C.; Zaphiropoulous, P.G.; et al. Mutations of the human homolog of Drosophila patched in the nevoid basal cell carcinoma syndrome. Cell 1996, 85, 841–851. [Google Scholar] [CrossRef]
- Roudgari, H.; Farndon, P.A.; Murray, A.D.; Hardy, C.; Miedzybrodzka, Z. Is PATCHED an important candidate gene for neural tube defects? Cranial and thoracic neural tube defects in a family with Gorlin syndrome: a case report. Clin Genet 2012, 82, 71–76. [Google Scholar] [CrossRef]
- Saldanha, G. The Hedgehog signalling pathway and cancer. J Pathol 2001, 193, 427–432. [Google Scholar] [CrossRef]
- Zedan, W.; Robinson, P.A.; High, A.S. A novel polymorphism in the PTC gene allows easy identification of allelic loss in basal cell nevus syndrome lesions. Diagn Mol Pathol 2001, 10, 41–45. [Google Scholar] [PubMed]
- Evans, D.G.; Sims, D.G.; Donnai, D. Family implications of neonatal Gorlin’s syndrome. Arch Dis Child 1991, 66, 1162–1163. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, M.; Lü, Y.; Xing, Q.; Chen, W. A novel mutation of the PTCH1 gene activates the Shh/Gli signaling pathway in a Chinese family with nevoid basal cell carcinoma syndrome. Biochem Biophys Res Commun 2011, 409, 166–170. [Google Scholar]
- Fan, Z.; Li, J.; Du, J.; et al. A missense mutation in PTCH2 underlies dominantly inherited NBCCS in a Chinese family. J Med Genet 2008, 45, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Lü, Y.; Zhu, H.G.; Ye, W.M.; Zhang, M.B.; He, D.; Chen, W.T. A new mutation of PTCH gene in a Chinese family with nevoid basal cell carcinoma syndrome. Chin Med J (Engl) 2008, 121, 118–121. [Google Scholar]
- Fujii, M.; Noguchi, K.; Urade, M.; et al. Novel PTCH1 mutations in Japanese Nevoid basal cell carcinoma syndrome patients: two familial and three sporadic cases including the first Japanese patient with medulloblastoma. J Hum Genet 2011, 56, 277–283. [Google Scholar] [CrossRef]
- Abe, S.; Kabashima, K.; Sakabe, J.; et al. Coincident two mutations and one single nucleotide polymorphism of the PTCH1 gene in a family with naevoid basal cell carcinoma syndrome. Acta Derm Venereol 2008, 88, 635–636. [Google Scholar] [CrossRef] [PubMed]
- Otsubo, S.; Honma, M.; Asano, K.; Takahashi, H.; Iizuka, H. A novel germline mutation of PTCH1 gene in a Japanese family of nevoid basal cell carcinoma syndrome: are the palmoplantar pits associated with true basal cell carcinoma? J Dermatol Sci 2008, 51, 144–146. [Google Scholar] [CrossRef]
- Matsuzawa, N.; Nagao, T.; Shimozato, K.; Niikawa, N.; Yoshiura, K.I. Patched homologue 1 mutations in four Japanese families with basal cell nevus syndrome. J Clin Pathol 2006, 59, 1084–1086. [Google Scholar] [CrossRef]
- Le Brun Keris, Y.; Jouk, P.S.; Saada-Sebag, G.; et al. Prenatal manifestation in a family affected by nevoid basal cell carcinoma syndrome. Eur J Med Genet 2008, 51, 472–478. [Google Scholar]
- Pastorino, L.; Ghiorzo, P.; Nasti, S.; et al. Identification of a SUFU germline mutation in a family with Gorlin syndrome. Am J Med Genet A 2009, 149A, 1539–1543. [Google Scholar] [PubMed]
- Melo, E.S.; Kawamura, J.Y.; Alves, C.A.; Nunes, F.D.; Jorge, W.A.; Cavalcanti, M.G. Imaging modality correlations of an odontogenic keratocyst in the nevoid basal cell carcinoma syndrome: a family case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2004, 98, 232–236. [Google Scholar] [PubMed]
- Jose Tincani, A.; Santos Martins, A.; Gomes Andrade, R.; Franco Mello, E.J., Jr.; Camargo Bueno, M.A. Nevoid basal-cell syndrome: literature review and case report in a family. Sao Paulo Med J 1995, 113, 917–921. [Google Scholar][Green Version]
- Musani, V.; Gorry, P.; Basta-Juzbasic, A.; Stipic, T.; Miklic, P.; Levanat, S. Mutation in exon 7 of PTCH deregulates SHH/PTCH/SMO signaling: possible linkage to WNT. Int J Mol Med 2006, 17, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Totten, J.R. The multiple nevoid basal cell carcinoma syndrome. Report of its occurrence in four generations of a family. Cancer 1980, 46, 1456–1462. [Google Scholar][Green Version]
- Heimler, A.; Friedman, E.; Rosenthal, A.D. Naevoid basal cell carcinoma syndrome and Charcot-Marie-Tooth disease: two autosomal dominant disorders segregating in a family. J Med Genet 1978, 15, 288–291. [Google Scholar][Green Version]
- Fitzpatrick, P.J.; Thompson, G.A. Gorlin’s syndrome, or nevoid basal cell carcinoma syndrome. Can Med Assoc J 1982, 127, 465–470. [Google Scholar][Green Version]
- Wallace, D.C.; Murphy, K.J.; Kelly, L.; Ward, W.H. The basal cell naevus syndrome. Report of a family with anosmia and a case of hypogonadotrophic hypopituitarism. J Med Genet 1973, 10, 30–33. [Google Scholar][Green Version]
- Fujii, K.; Sugita, K.; Watanabe, T.; Takanashi, J.; Niimi, H. A familial case of Gorlin syndrome [in Japanese]. No To Hattatsu 1996, 28, 257–260. [Google Scholar][Green Version]
- Huault, M.; Bayonne, E.; Laxenaire, A.; Blanchard, P.; Levy, J.D. Basocellular nevomatosis. Follow-up of 3 generations [in French]. Rev Stomatol Chir Maxillofac 1998, 99, 244–249. [Google Scholar][Green Version]
- Mazzola, G.; Galioto, V.; Mazzola, U. Gorlin-Gotz syndrome in 3 patients from the same family monitored from 1993 to 1999 [in Italian]. Minerva Stomatol 2000, 49, 197–203. [Google Scholar][Green Version]
- Chung, C.H.; Wong, T.Y.; Shieh, T.Y.; Shieh, D.B.; Chao, S.C. Nevoid basal cell carcinoma syndrome—clinical manifestations and mutation analysis of a Taiwanese family. J Formos Med Assoc 2003, 102, 793–797. [Google Scholar] [PubMed][Green Version]
- Zmak, M.; Dobric, P.; Medica, I. Gorlin syndrome: a report on a family. Minerva Pediatr 1994, 46, 217–219. [Google Scholar][Green Version]
- Ujpál, M.; Szabó, G. Familial occurrence of Gorlin-Goltz syndrome [in Hungarian]. Fogorv Sz 1992, 85, 209–212. [Google Scholar] [PubMed][Green Version]
- Goldstein, A.M.; Pastakia, B.; DiGiovanna, J.J.; et al. Clinical findings in two African-American families with the nevoid basal cell carcinoma syndrome (NBCC). Am J Med Genet 1994, 50, 272–281. [Google Scholar] [CrossRef] [PubMed]
- McLoughlin, P.M.; Dickenson, A.J.; Avery, B.S. Gorlin’s syndrome and von Recklinghausen’s disease occurring in one family. Br J Oral Maxillofac Surg 1991, 29, 189–193. [Google Scholar] [CrossRef]
- Lovin, J.D.; Talarico, C.L.; Wegert, S.L.; Gaynor, L.F.; Sutley, S.S. Gorlin’s syndrome with associated odontogenic cysts. Pediatr Radiol 1991, 21, 584–587. [Google Scholar] [CrossRef]
- Evans, D.G.; Birch, J.M.; Orton, C.I. Brain tumours and the occurrence of severe invasive basal cell carcinoma in first degree relatives with Gorlin syndrome. Br J Neurosurg 1991, 5, 643–646. [Google Scholar] [CrossRef]
- Tasanen, A.; Lamberg, M.A.; Nordling, S. Skeletal anomalies and keratocysts in the basal cell nevus syndrome. Int J Oral Surg 1975, 4, 225–235. [Google Scholar] [CrossRef]
- Brumme, S.; Löwicke, G. Kinship studies in Gorlin-Goltz syndrome (basal cell nevus syndrome) [in German]. Dermatol Monatsschr 1990, 176, 487–492. [Google Scholar]
- Gartmann, H.; Groth, W.; Quinkler, C. Multiple basaloid follicular hamartomas in 2 members of a family with Gorlin-Goltz syndrome [in German]. Z Hautkr 1989, 64, 915–918. [Google Scholar] [PubMed]
- Rausch, E.; Trödhan, A.; Porteder, H. Multiple keratocysts suggesting the presence of Gorlin-Goltz syndrome: report of 3 patients in a family [in German]. Z Stomatol 1989, 86, 275–277. [Google Scholar] [PubMed]
- Lorenz, R.; Fuhrmann, W. Familial basal cell nevus syndrome. Hum Genet 1978, 44, 153–163. [Google Scholar] [PubMed]
- Aszterbaum, M.; Rothman, A.; Johnson, R.L.; et al. Identification of mutations in the human PATCHED gene in sporadic basal cell carcinomas and in patients with the basal cell nevus syndrome. J Invest Dermatol 1998, 110, 885–888. [Google Scholar] [CrossRef]
- Sasaki, R.; Miyashita, T.; Matsumoto, N.; Fujii, K.; Saito, K.; Ando, T. Multiple keratocystic odontogenic tumors associated with nevoid basal cell carcinoma syndrome having distinct PTCH1 mutations: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2010, 110, e41–e46. [Google Scholar]
- Lo Muzio, L. Nevoid basal cell carcinoma syndrome (Gorlin syndrome). Orphanet J Rare Dis 2008, 3, 32. [Google Scholar]
- Pritchard, L.J.; Delfino, J.J.; Ivey, D.M.; Sclaroff, A.; Giglio, J.A. Variable expressivity of the multiple nevoid basal cell carcinoma syndrome. J Oral Maxillofac Surg 1982, 40, 261–269. [Google Scholar]
- Manfredi, M.; Vescovi, P.; Bonanini, M.; Porter, S. Nevoid basal cell carcinoma syndrome: a review of the literature. Int J Oral Maxillofac Surg 2004, 33, 117–124. [Google Scholar]
- Rayne, J. The multiple basal cell naevi syndrome. Br J Oral Surg 1971, 9, 65–71. [Google Scholar] [CrossRef]
- Gorlin, R.J.; Vickers, R.A.; Kellen, E.; Williamson, J.J. Multiple basal-cell nevi syndrome. An analysis of a syndrome consisting of multiple nevoid basal-cell carcinoma, jaw cysts, skeletal anomalies, medulloblastoma, and hyporesponsiveness to parathormone. Cancer 1965, 18, 89–104. [Google Scholar]
- Shand, J.M.; Heggie, A.A. Cysts of the jaws and advances in the diagnosis and management of nevoid Basal cell carcinoma syndrome. Oral Maxillofac Surg Clin North Am 2005, 17, 403–414. [Google Scholar] [CrossRef]
- Woolgar, J.A.; Rippin, J.W.; Browne, R.M. A comparative histological study of odontogenic keratocysts in basal cell naevus syndrome and control patients. J Oral Pathol 1987, 16, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Avelar, R.L.; Antunes, A.A.; Santos Tde, S.; Andrade, E.S.; Dourado, E. Odontogenic tumors: clinical and pathology study of 238 cases. Braz J Otorhinolaryngol 2008, 74, 668–673. [Google Scholar] [PubMed]
- Morgan, T.A.; Burton, C.C.; Qian, F. A retrospective review of treatment of the odontogenic keratocyst. J Oral Maxillofac Surg 2005, 63, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Yagyuu, T.; Kirita, T.; Sasahira, T.; Moriwaka, Y.; Yamamoto, K.; Kuniyasu, H. Recurrence of keratocystic odontogenic tumor: clinicopathological features and immunohistochemical study of the Hedgehog signaling pathway. Pathobiology 2008, 75, 171–176. [Google Scholar] [CrossRef]
- Friedrich, R.E.; Scheuer, H.A.; Zustin, J. Expression of podoplanin in nevoid basal cell carcinoma syndrome-associated keratocystic odontogenic tumours. Anticancer Res 2012, 32, 2125–2127. [Google Scholar] [PubMed]
- Borgonovo, A.E.; Di Lascia, S.; Grossi, G.; Maiorana, C. Two-stage treatment protocol of keratocystic odontogenic tumour in young patients with Gorlin-Goltz syndrome: marsupialization and later enucleation with peripheral ostectomy. A 5-year-follow-up experience. Int J Pediatr Otorhinolaryngol 2011, 75, 1565–1571. [Google Scholar] [CrossRef]
- Antonoglou, G.N.; Sándor, G.K.; Koidou, V.P.; Papageorgiou, S.N. Non-syndromic and syndromic keratocystic odontogenic tumors: systematic review and meta-analysis of recurrences. J Craniomaxillofac Surg 2014, 42, e364–e371. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Chi, S.; Xie, J. Hedgehog signaling in skin cancers. Cell Signal 2011, 23, 1235–1243. [Google Scholar] [CrossRef]
- Mancuso, M.; Pazzaglia, S.; Tanori, M.; et al. Basal cell carcinoma and its development: insights from radiation-induced tumors in Ptch1-deficient mice. Cancer Res 2004, 64, 934–941. [Google Scholar] [CrossRef]
- Oro, A.E.; Higgins, K.M.; Hu, Z.; Bonifas, J.M.; Epstein, E.H., Jr.; Scott, M.P. Basal cell carcinomas in mice overexpressing sonic hedgehog. Science 1997, 276, 817–821. [Google Scholar] [PubMed]
- Levanat, S.; Gorlin, R.J.; Fallet, S.; Johnson, D.R.; Fantasia, J.E.; Bale, A.E. A two-hit model for developmental defects in Gorlin syndrome. Nat Genet 1996, 12, 85–87. [Google Scholar] [PubMed]
- Oliveira, D.M.; Silveira, M.M.F.; Andrade, E.S.S.; et al. Immunohistochemical Analysis of Human Homologue of Drosophila Patched (PTCH) in Dental Follicles of Impacted Third Molars. Int J Morphol 2012, 30, 105–109. [Google Scholar] [CrossRef][Green Version]
- Gorlin, R.J. Nevoid basal cell carcinoma (Gorlin) syndrome: unanswered issues. J Lab Clin Med 1999, 134, 551–552. [Google Scholar] [CrossRef] [PubMed]
- Agaram, N.P.; Collins, B.M.; Barnes, L.; et al. Molecular analysis to demonstrate that odontogenic keratocysts are neoplastic. Arch Pathol Lab Med 2004, 128, 313–317. [Google Scholar]
- Tang, J.Y.; Mackay-Wiggan, J.M.; Aszterbaum, M.; et al. Inhibiting the hedgehog pathway in patients with the basal-cell nevus syndrome. N Engl J Med 2012, 366, 2180–2188. [Google Scholar] [CrossRef]
- Girard, C.; Debu, A.; Bessis, D.; Blatière, V.; Dereure, O.; Guillot, B. Treatment of Gorlin syndrome (nevoid basal cell carcinoma syndrome) with methylaminolevulinate photodynamic therapy in seven patients, including two children: interest of tumescent anesthesia for pain control in children. J Eur Acad Dermatol Venereol 2013, 27, e171–e175. [Google Scholar]
- Kurek, A.; Grudniak, A.M.; Kraczkiewicz-Dowjat, A.; Wolska, K.I. New antibacterial therapeutics and strategies. Pol J Microbiol 2011, 60, 3–12. [Google Scholar] [CrossRef]
- Levy, J.G. Photosensitizers in photodynamic therapy. Semin Oncol 1994, 21 (Suppl. 15), 4–10. [Google Scholar]
- Agostinis, P.; Berg, K.; Cengel, K.A.; et al. Photodynamic therapy of cancer: an update. CA Cancer J Clin 2011, 61, 250–281. [Google Scholar] [CrossRef]
- Loncaster, J.; Swindell, R.; Slevin, F.; Sheridan, L.; Allan, D.; Allan, E. Efficacy of photodynamic therapy as a treatment for Gorlin syndrome-related basal cell carcinomas. Clin Oncol (R Coll Radiol) 2009, 21, 502–508. [Google Scholar] [PubMed]
- Itkin, A.; Gilchrest, B.A. delta-Aminolevulinic acid and blue light photodynamic therapy for treatment of multiple basal cell carcinomas in two patients with nevoid basal cell carcinoma syndrome. Dermatol Surg 2004, 30, 1054–1061. [Google Scholar]
- Chechia, A.; Attia, L.; Temime, R.B.; Makhlouf, T.; Koubaa, A. Incidence, clinical analysis, and management of ovarian fibromas and fibro-thecomas. Am J Obstet Gynecol 2008, 199, 473.e1–473.e4. [Google Scholar] [PubMed]
- Gorlin, R.J. Nevoid basal cell carcinoma (Gorlin) syndrome. Genet Med 2004, 6, 530–539. [Google Scholar]
- Evans, D.G.; Ladusans, E.J.; Rimmer, S.; Burnell, L.D.; Thakker, N.; Farndon, P.A. Complications of the naevoid basal cell carcinoma syndrome: results of a population based study. J Med Genet 1993, 30, 460–464. [Google Scholar]
- Morse, C.B.; McLaren, J.F.; Roy, D.; Siegelman, E.S.; Livolsi, V.A.; Gracia, C.R. Ovarian preservation in a young patient with Gorlin syndrome and multiple bilateral ovarian masses. Fertil Steril 2011, 96, e47–e50. [Google Scholar]
- Ball, A.; Wenning, J.; Van Eyk, N. Ovarian fibromas in pediatric patients with basal cell nevus (Gorlin) syndrome. J Pediatr Adolesc Gynecol 2011, 24, e5–e7. [Google Scholar] [PubMed]
- Pereyra-Rodríguez, J.J.; Bernabeu-Wittel, J.; Conejo-Mir, J. Multiple palmar pits and basal cell carcinomas. CMAJ 2010, 182, E533. [Google Scholar] [CrossRef]
- Provias, J.P.; Becker, L.E. Cellular and molecular pathology of medulloblastoma. J Neurooncol 1996, 29, 35–43. [Google Scholar]
- Amlashi, S.F.; Riffaud, L.; Brassier, G.; Morandi, X. Nevoid basal cell carcinoma syndrome: relation with desmoplastic medulloblastoma in infancy. A population-based study and review of the literature. Cancer 2003, 98, 618–624. [Google Scholar] [CrossRef]
- Campbell, R.M.; Mader, R.D.; Dufresne, R.G., Jr. Meningiomas after medulloblastoma irradiation treatment in a patient with basal cell nevus syndrome. J Am Acad Dermatol 2005, 53 (Suppl 1), S256–S259. [Google Scholar] [CrossRef] [PubMed]
- Wallin, J.L.; Tanna, N.; Misra, S.; Puri, P.K.; Sadeghi, N. Sinonasal carcinoma after irradiation for medulloblastoma in nevoid basal cell carcinoma syndrome. Am J Otolaryngol 2007, 28, 360–362. [Google Scholar] [PubMed]
- Honavar, S.G.; Shields, J.A.; Shields, C.L.; Eagle, R.C., Jr.; Demirci, H.; Mahmood, E.Z. Basal cell carcinoma of the eyelid associated with Gorlin-Goltz syndrome. Ophthalmology 2001, 108, 1115–1123. [Google Scholar] [CrossRef]
- Harris, S.A.; Large, D.M. Gorlin’s syndrome with a cardiac lesion and jaw cysts with some unusual histological features. A case report and review of the literature. Int J Oral Surg 1984, 13, 59–64. [Google Scholar]
- Kluger, N.; Marco-Baertich, I.; Guillot, B. Late onset of cardiac tumour in naevoid Basal cell carcinoma (Gorlin) syndrome. Acta Derm Venereol 2007, 87, 272–273. [Google Scholar] [CrossRef] [PubMed]
- Yan, A.T.; Coffey, D.M.; Li, Y.; et al. Images in cardiovascular medicine. Myocardial fibroma in Gorlin syndrome by cardiac magnetic resonance imaging. Circulation 2006, 114, e376–e379. [Google Scholar] [CrossRef] [PubMed]
- Bossert, T.; Walther, T.; Vondrys, D.; Gummert, J.F.; Kostelka, M.; Mohr, F.W. Cardiac fibroma as an inherited manifestation of nevoid basal-cell carcinoma syndrome. Tex Heart Inst J 2006, 33, 88–90. [Google Scholar]
- Coffin, C.M. Congenital cardiac fibroma associated with Gorlin syndrome. Pediatr Pathol 1992, 12, 255–262. [Google Scholar] [CrossRef]

 |
 |
 |
© 2016 by the author. The Author(s) 2016.
Share and Cite
de Santana Santos, T.; Vajgel, A.; Martins-Filho, P.R.S.; de Albuquerque Maranhao Filho, A.; De Holanda Vasconcellos, R.J.; Frota, R.; Filho, J.R.L. Nevoid Basal Cell Carcinoma Syndrome: A Long-Term Study in a Family. Craniomaxillofac. Trauma Reconstr. 2016, 9, 94-104. https://doi.org/10.1055/s-0035-1558454
de Santana Santos T, Vajgel A, Martins-Filho PRS, de Albuquerque Maranhao Filho A, De Holanda Vasconcellos RJ, Frota R, Filho JRL. Nevoid Basal Cell Carcinoma Syndrome: A Long-Term Study in a Family. Craniomaxillofacial Trauma & Reconstruction. 2016; 9(1):94-104. https://doi.org/10.1055/s-0035-1558454
Chicago/Turabian Stylede Santana Santos, Thiago, André Vajgel, Paulo Ricardo Saquete Martins-Filho, AlmirWalter de Albuquerque Maranhao Filho, Ricardo José De Holanda Vasconcellos, Riedel Frota, and José Rodrigues Laureano Filho. 2016. "Nevoid Basal Cell Carcinoma Syndrome: A Long-Term Study in a Family" Craniomaxillofacial Trauma & Reconstruction 9, no. 1: 94-104. https://doi.org/10.1055/s-0035-1558454
APA Stylede Santana Santos, T., Vajgel, A., Martins-Filho, P. R. S., de Albuquerque Maranhao Filho, A., De Holanda Vasconcellos, R. J., Frota, R., & Filho, J. R. L. (2016). Nevoid Basal Cell Carcinoma Syndrome: A Long-Term Study in a Family. Craniomaxillofacial Trauma & Reconstruction, 9(1), 94-104. https://doi.org/10.1055/s-0035-1558454



