Concurrent Mold, Mycobacterial, and Viral Infections in a Hematopoietic Stem Cell Transplant Recipient Undergoing Lung Transplantation for Graft-Versus-Host Disease
Simple Summary
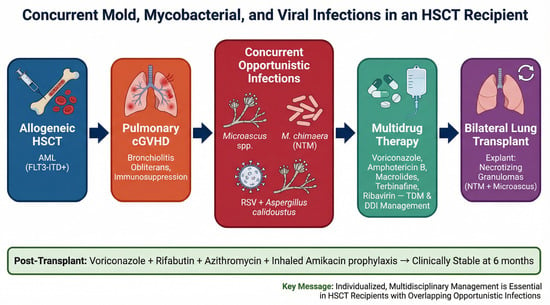
Abstract
1. Introduction
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tomblyn, M.; Chiller, T.; Einsele, H.; Gress, R.; Sepkowitz, K.; Storek, J.; Wingard, J.R.; Young, J.-A.H.; Boeckh, M.A. Guidelines for Preventing Infectious Complications among Hematopoietic Cell Transplantation Recipients: A Global Perspective. Biol. Blood Marrow Transplant. 2009, 15, 1143–1238. [Google Scholar] [CrossRef]
- Hatzimichael, E.; Tuthill, M. Hematopoietic stem cell transplantation. Stem Cells Cloning 2010, 3, 105–117. [Google Scholar]
- Rahi, M.S.; Jindal, V.; Pednekar, P.; Parekh, J.; Gunasekaran, K.; Sharma, S.; Stender, M.; Jaiyesimi, I.A. Fungal infections in hematopoietic stem-cell transplant patients: A review of epidemiology, diagnosis, and management. Ther. Adv. Infect. Dis. 2021, 8, 20499361211039050. [Google Scholar] [CrossRef]
- Pagano, L.; Caira, M.; Candoni, A.; Offidani, M.; Fianchi, L.; Martino, B.; Pastore, D.; Picardi, M.; Bonini, A.; Chierichini, A.; et al. The epidemiology of fungal infections in patients with hematologic malignancies: The SEIFEM-2004 study. Haematologica 2006, 91, 1068–1075. [Google Scholar]
- Williams, K.M.; Chien, J.W.; Gladwin, M.T.; Pavletic, S.Z. Bronchiolitis Obliterans After Allogeneic Hematopoietic Stem Cell Transplantation. JAMA 2009, 302, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, G.C.; Fazekas, T.; Lawitschka, A.; Bertz, H.; Greinix, H.; Halter, J.; Pavletic, S.Z.; Holler, E.; Wolff, D. Diagnosis and treatment of pulmonary chronic GVHD: Report from the consensus conference on clinical practice in chronic GVHD. Bone Marrow Transplant. 2011, 46, 1283–1295. [Google Scholar] [CrossRef]
- Bergeron, A.; Godet, C.; Chevret, S.; Lorillon, G.; de Latour, R.P.; de Revel, T.; Robin, M.; Ribaud, P.; Socié, G.; Tazi, A. Bronchiolitis obliterans syndrome after allogeneic hematopoietic SCT: Phenotypes and prognosis. Bone Marrow Transplant. 2012, 48, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Doucette, K.; Fishman, J.A. Nontuberculous Mycobacterial Infection in Hematopoietic Stem Cell and Solid Organ Transplant Recipients. Clin. Infect. Dis. 2004, 38, 1428–1439. [Google Scholar] [CrossRef]
- Al-Anazi, K.A.; Al-Jasser, A.M.; Al-Anazi, W.K. Infections Caused by Non-Tuberculous Mycobacteria in Recipients of Hematopoietic Stem Cell Transplantation. Front. Oncol. 2014, 4, 311. [Google Scholar] [CrossRef]
- Bergeron, A.; Mikulska, M.; De Greef, J.; Bondeelle, L.; Franquet, T.; Herrmann, J.-L.; Lange, C.; Spriet, I.; Akova, M.; Donnelly, J.P.; et al. Mycobacterial infections in adults with haematological malignancies and haematopoietic stem cell transplants: Guidelines from the 8th European Conference on Infections in Leukaemia. Lancet Infect. Dis. 2022, 22, e359–e369. [Google Scholar] [CrossRef] [PubMed]
- Griffith, D.E.; Aksamit, T.; Brown-Elliott, B.A.; Catanzaro, A.; Daley, C.; Gordin, F.; Holland, S.M.; Horsburgh, R.; Huitt, G.; Iademarco, M.F.; et al. An Official ATS/IDSA Statement: Diagnosis, Treatment, and Prevention of Nontuberculous Mycobacterial Diseases. Am. J. Respir. Crit. Care Med. 2007, 175, 367–416. [Google Scholar] [CrossRef]
- Mason, M.; Gregory, E.; Foster, K.; Klatt, M.; Zoubek, S.; Eid, A.J. Pharmacologic Management of Mycobacterium chimaera Infections: A Primer for Clinicians. Open Forum Infect. Dis. 2022, 9, ofac287. [Google Scholar] [CrossRef]
- Daley, C.L.; Iaccarino, J.M.; Lange, C.; Cambau, E.; Wallace, R.J.; Andrejak, C.; Böttger, E.C.; Brozek, J.; Griffith, D.E.; Guglielmetti, L.; et al. Treatment of Nontuberculous Mycobacterial Pulmonary Disease: An Official ATS/ERS/ESCMID/IDSA Clinical Practice Guideline. Clin. Infect. Dis. 2020, 71, 905–913. [Google Scholar] [CrossRef]
- Griffith, D.E.; Eagle, G.; Thomson, R.; Aksamit, T.R.; Hasegawa, N.; Morimoto, K.; Addrizzo-Harris, D.J.; O’dOnnell, A.E.; Marras, T.K.; Flume, P.A.; et al. Amikacin Liposome Inhalation Suspension for Treatment-Refractory Lung Disease Caused by Mycobacterium avium Complex (CONVERT). A Prospective, Open-Label, Randomized Study. Am. J. Respir. Crit. Care Med. 2018, 198, 1559–1569. [Google Scholar] [CrossRef]
- Riches, M.L.; Trifilio, S.; Chen, M.; Ahn, K.W.; Langston, A.; Lazarus, H.M.; I Marks, D.; Martino, R.; Maziarz, R.T.; A Papanicolou, G.; et al. Risk factors and impact of non-Aspergillus mold infections following allogeneic HCT: A CIBMTR infection and immune reconstitution analysis. Bone Marrow Transplant. 2015, 51, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Douglas, A.P.; Lamoth, F.; John, T.M.; Groll, A.H.; Shigle, T.L.; Papanicolaou, G.A.; Chemaly, R.F.; Carpenter, P.A.; Dadwal, S.S.; Walsh, T.J.; et al. American Society of Transplantation and Cellular Therapy Series: #8-Management and Prevention of Non-Aspergillus Molds in Hematopoietic Cell Transplantation Recipients. Biol. Blood Marrow Transplant. 2025, 31, 194–223. [Google Scholar] [CrossRef]
- Park, B.J.; Pappas, P.G.; Wannemuehler, K.A.; Alexander, B.D.; Anaissie, E.J.; Andes, D.R.; Baddley, J.W.; Brown, J.M.; Brumble, L.M.; Freifeld, A.G.; et al. Invasive Non-Aspergillus Mold Infections in Transplant Recipients, United States, 2001–2006. Emerg. Infect. Dis. 2011, 17, 1855–1864. [Google Scholar] [CrossRef]
- Bupha-Intr, O.; Butters, C.; Reynolds, G.; Kennedy, K.; Meyer, W.; Patil, S.; Bryant, P.; Morrissey, C.O. Australasian Antifungal Guidelines Steering Committee Consensus guidelines for the diagnosis and management of invasive fungal disease due to moulds other than Aspergillus in the haematology/oncology setting, 2021. Intern. Med. J. 2021, 51, 177–219. [Google Scholar] [CrossRef] [PubMed]
- Woudenberg, J.; Meijer, M.; Houbraken, J.; Samson, R. Scopulariopsis and scopulariopsis-like species from indoor environments. Stud. Mycol. 2017, 88, 1–35. [Google Scholar] [CrossRef]
- Pérez-Cantero, A.; Guarro, J. Current knowledge on the etiology and epidemiology of Scopulariopsis infections. Med. Mycol. 2019, 58, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Petanović, M.; Paradzik, M.T.; Kristof, Z.; Cvitković, A.; Topolovac, Z. Scopulariopsis brevicaulis as the cause of dermatomycosis. Acta Dermatovenerol. Croat. 2010, 18, 8–13. [Google Scholar] [PubMed]
- Chamilos, G.; Lionakis, M.S.; Kontoyiannis, D.P. Call for Action: Invasive Fungal Infections Associated With Ibrutinib and Other Small Molecule Kinase Inhibitors Targeting Immune Signaling Pathways. Clin. Infect. Dis. 2018, 66, 140–148. [Google Scholar] [CrossRef]
- Jenks, J.D.; Cornely, O.A.; Chen, S.C.; Thompson, G.R., 3rd; Hoenigl, M. Breakthrough invasive fungal infections: Who is at risk? Mycoses 2020, 63, 1021–1032. [Google Scholar] [CrossRef]
- Brunet, K.; Martellosio, J.-P.; Tewes, F.; Marchand, S.; Rammaert, B. Inhaled Antifungal Agents for Treatment and Prophylaxis of Bronchopulmonary Invasive Mold Infections. Pharmaceutics 2022, 14, 641. [Google Scholar] [CrossRef]
- Vuong, N.N.; Hammond, D.; Kontoyiannis, D.P. Clinical Uses of Inhaled Antifungals for Invasive Pulmonary Fungal Disease: Promises and Challenges. J. Fungi 2023, 9, 464. [Google Scholar] [CrossRef]
- Liu, Q.; Kong, L.; Hua, L.; Xu, S. Pulmonary Microascus cirrosus infection in an immunocompetent patient with bronchiectasis: A case report. Respir. Med. Case Rep. 2021, 34, 101484. [Google Scholar] [CrossRef]
- Glampedakis, E.; Erard, V.; Lamoth, F. Clinical Relevance and Characteristics of Aspergillus calidoustus and Other Aspergillus Species of Section Usti. J. Fungi 2020, 6, 84. [Google Scholar] [CrossRef]
- Cordonnier, C.; Rovira, M.; Maertens, J.; Olavarria, E.; Faucher, C.; Bilger, K.; Pigneux, A.; Cornely, O.A.; Ullmann, A.J.; Bofarull, R.M.; et al. Voriconazole for secondary prophylaxis of invasive fungal infections in allogeneic stem cell transplant recipients: Results of the VOSIFI study. Haematologica 2010, 95, 1762–1768. [Google Scholar] [CrossRef]
- Okamoto, K.; Santos, C.A.Q. Management and prophylaxis of bacterial and mycobacterial infections among lung transplant recipients. Ann. Transl. Med. 2020, 8, 413. [Google Scholar] [CrossRef] [PubMed]
- Shi, T.; McAllister, D.A.; O’Brien, K.L.; Simoes, E.A.F.; Madhi, S.A.; Gessner, B.D.; Polack, F.P.; Balsells, E.; Acacio, S.; Aguayo, C.; et al. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in young children in 2015: A systematic review and modelling study. Lancet 2017, 390, 946–958. [Google Scholar] [CrossRef] [PubMed]
- Hammitt, L.L.; Dagan, R.; Yuan, Y.; Cots, M.B.; Bosheva, M.; Madhi, S.A.; Muller, W.J.; Zar, H.J.; Brooks, D.; Grenham, A.; et al. Nirsevimab for Prevention of RSV in Healthy Late-Preterm and Term Infants. N. Engl. J. Med. 2022, 386, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.; Davies, S.; Labrador, J.; Loubet, P.; Martínez, S.N.; Moríñigo, H.M.; Nicolas, J.-F.; Vera, M.P.; Rämet, M.; Rebollo-Rodrigo, M.H.; et al. Safety and immunogenicity of a respiratory syncytial virus prefusion F vaccine when co-administered with adjuvanted seasonal quadrivalent influenza vaccines in older adults: A phase 3 randomized trial. Clin. Infect. Dis. 2024, 79, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Khawaja, F.; Chemaly, R.F. Respiratory syncytial virus in hematopoietic cell transplant recipients and patients with hematologic malignancies. Haematologica 2019, 104, 1322–1331. [Google Scholar] [CrossRef] [PubMed]



| Pathogens | Minimum Inhibitory Concentrations (If Applicable) |
|---|---|
| Mycobacterium chimaera | Amikacin 16 μg/mL (susceptible) Clarithromycin 1 μg/mL (susceptible) Linezolid 16 μg/mL (intermediate) Moxifloxacin 5 μg/mL (resistant) |
| Microascus spp. | Amphotericin B 1 μg/mL (no breakpoints) Caspofungin < 0.0080 g/mL (no breakpoints) Itraconazole > 16 μg/mL (no breakpoints) Voriconazole 2 μg/mL (no breakpoints) |
| Aspergillus calidoustus | Susceptibility data were not available |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Akkielah, L.; Leung, W.; Wang, S.; Ataie, L.; Xenocostas, A.; Syed, A.; Hsu, Y.-H.R.; Silverman, M.; AlMutawa, F.; Rahimi Shahmirzadi, M. Concurrent Mold, Mycobacterial, and Viral Infections in a Hematopoietic Stem Cell Transplant Recipient Undergoing Lung Transplantation for Graft-Versus-Host Disease. Curr. Oncol. 2026, 33, 145. https://doi.org/10.3390/curroncol33030145
Akkielah L, Leung W, Wang S, Ataie L, Xenocostas A, Syed A, Hsu Y-HR, Silverman M, AlMutawa F, Rahimi Shahmirzadi M. Concurrent Mold, Mycobacterial, and Viral Infections in a Hematopoietic Stem Cell Transplant Recipient Undergoing Lung Transplantation for Graft-Versus-Host Disease. Current Oncology. 2026; 33(3):145. https://doi.org/10.3390/curroncol33030145
Chicago/Turabian StyleAkkielah, Layan, Wayne Leung, Serena Wang, Lili Ataie, Anargyros Xenocostas, Asma Syed, Ying-Han R. Hsu, Michael Silverman, Fatimah AlMutawa, and MohammadReza Rahimi Shahmirzadi. 2026. "Concurrent Mold, Mycobacterial, and Viral Infections in a Hematopoietic Stem Cell Transplant Recipient Undergoing Lung Transplantation for Graft-Versus-Host Disease" Current Oncology 33, no. 3: 145. https://doi.org/10.3390/curroncol33030145
APA StyleAkkielah, L., Leung, W., Wang, S., Ataie, L., Xenocostas, A., Syed, A., Hsu, Y.-H. R., Silverman, M., AlMutawa, F., & Rahimi Shahmirzadi, M. (2026). Concurrent Mold, Mycobacterial, and Viral Infections in a Hematopoietic Stem Cell Transplant Recipient Undergoing Lung Transplantation for Graft-Versus-Host Disease. Current Oncology, 33(3), 145. https://doi.org/10.3390/curroncol33030145