From Molecular Biology to Novel Immunotherapies and Nanomedicine in Uveal Melanoma
Abstract
1. Introduction
2. Methodology
3. Molecular Biology of Uveal Melanoma
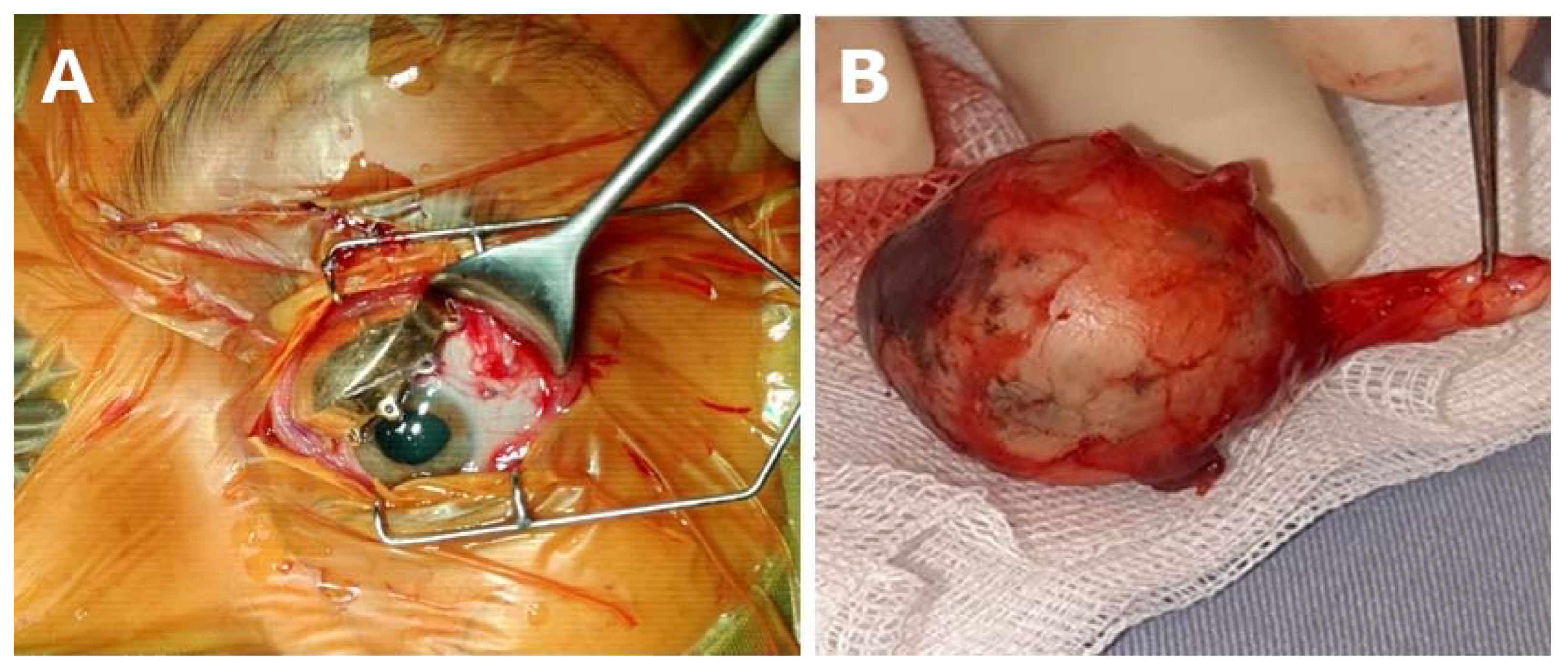
4. Current Therapy of UM
5. Novel Immunotherapy Approaches
6. Combination Immunotherapy in Development
7. Cell-Based Immunotherapies
8. Theranostics: A Step towards Personalized Medicine
9. Nanomedicine
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kaliki, S.; Shields, C.L. Uveal melanoma: Relatively rare but deadly cancer. Eye 2017, 31, 241–257. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.D.; Turell, M.E.; Topham, A.K. Uveal melanoma: Trends in incidence, treatment, and survival. Ophthalmology 2011, 118, 1881–1885. [Google Scholar] [CrossRef] [PubMed]
- Piperno-Neumann, S.; Piulats, J.M.; Goebeler, M.; Galloway, I.; Lugowska, I.; Becker, J.C.; Vihinen, P.; Van Calster, J.; Hadjistilianou, T.; Proença, R.; et al. Uveal Melanoma: A European Network to Face the Many Challenges of a Rare Cancer. Cancers 2019, 11, 817. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, P.; Romanowska-Dixon, B.; Markiewicz, A.; Zieniewicz, K.; Kozak, K.; Rogala, P.; Świtaj, T.; Dudzisz-Sledź, M. Diagnostic and therapeutic management of patients with ocular melanomas–r—Recommendations of the Polish Society of Oncology. Nowotw. J. Oncol. 2022, 72, 342–352. [Google Scholar] [CrossRef]
- Banou, L.; Tsani, Z.; Arvanitogiannis, K.; Pavlaki, M.; Dastiridou, A.; Androudi, S. Radiotherapy in Uveal Melanoma: A Review of Ocular Complications. Curr. Oncol. 2023, 30, 6374–6396. [Google Scholar] [CrossRef]
- Jackson, S.E.; Chester, J.D. Personalised cancer medicine. Int. J. Cancer 2015, 137, 262–266. [Google Scholar] [CrossRef]
- Shields, C.L.; Say, E.A.T.; Hasanreisoglu, M.; Saktanasate, J.; Lawson, B.M.; Landy, J.E.; Badami, A.U.; Sivalingam, M.D.; Hauschild, A.J.; House, R.J.; et al. Personalized Prognosis of Uveal Melanoma Based on Cytogenetic Profile in 1059 Patients over an 8-Year Period: The 2017 Harry S. Gradle Lecture. Ophthalmology 2017, 124, 1523–1531. [Google Scholar] [CrossRef]
- Broggi, G.; Russo, A.; Reibaldi, M.; Russo, D.; Varricchio, S.; Bonfiglio, V.; Spatola, C.; Barbagallo, C.; Foti, P.V.; Avitabile, T.; et al. Histopathology and Genetic Biomarkers of Choroidal Melanoma. Appl. Sci. 2020, 10, 8081. [Google Scholar] [CrossRef]
- Griewank, K.G.; Yu, X.; Khalili, J.; Sozen, M.M.; Stempke-Hale, K.; Bernatchez, C.; Wardell, S.; Bastian, B.C.; Woodman, S.E. Genetic and molecular characterization of uveal melanoma cell lines. Pigment. Cell Melanoma Res. 2012, 25, 182–187. [Google Scholar] [CrossRef]
- Onken, M.D.; Worley, L.A.; Long, M.D.; Duan, S.; Council, M.L.; Bowcock, A.M.; Harbour, J.W. Oncogenic mutations in GNAQ occur early in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2008, 49, 5230–5234. [Google Scholar] [CrossRef]
- Szalai, E.; Wells, J.R.; Ward, L.; Grossniklaus, H.E. Uveal Melanoma Nuclear BRCA1-Associated Protein-1 Immunoreactivity Is an Indicator of Metastasis. Ophthalmology 2018, 125, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Uner, O.E.; See, T.R.O.; Szalai, E.; Grossniklaus, H.E.; Stålhammar, G. Estimation of the timing of BAP1 mutation in uveal melanoma progression. Sci. Rep. 2021, 11, 8923. [Google Scholar] [CrossRef] [PubMed]
- Pašalić, D.; Nikuševa-Martić, T.; Sekovanić, A.; Kaštelan, S. Genetic and Epigenetic Features of Uveal Melanoma-An Overview and Clinical Implications. Int. J. Mol. Sci. 2023, 24, 12807. [Google Scholar] [CrossRef] [PubMed]
- Yavuzyigitoglu, S.; Koopmans, A.E.; Verdijk, R.M.; Vaarwater, J.; Eussen, B.; van Bodegom, A.; Paridaens, D.; Kiliç, E.; de Klein, A. Uveal Melanomas with SF3B1 Mutations: A Distinct Subclass Associated with Late-Onset Metastases. Ophthalmology 2016, 123, 1118–1128. [Google Scholar] [CrossRef]
- Furney, S.J.; Pedersen, M.; Gentien, D.; Dumont, A.G.; Rapinat, A.; Desjardins, L.; Turajlic, S.; Piperno-Neumann, S.; de la Grange, P.; Roman-Roman, S.; et al. SF3B1 mutations are associated with alternative splicing in uveal melanoma. Cancer Discov. 2013, 3, 1122–1129. [Google Scholar] [CrossRef]
- Decatur, C.L.; Ong, E.; Garg, N.; Anbunathan, H.; Bowcock, A.M.; Field, M.G.; Harbour, J.W. Driver Mutations in Uveal Melanoma: Associations with Gene Expression Profile and Patient Outcomes. JAMA Ophthalmol. 2016, 134, 728–733. [Google Scholar] [CrossRef]
- Ewens, K.G.; Kanetsky, P.A.; Richards-Yutz, J.; Purrazzella, J.; Shields, C.L.; Ganguly, T.; Ganguly, A. Chromosome 3 status combined with BAP1 and EIF1AX mutation profiles are associated with metastasis in uveal melanoma. Investig. Ophthalmol. Vis. Sci. 2014, 55, 5160–5167. [Google Scholar] [CrossRef]
- Barbagallo, C.; Stella, M.; Broggi, G.; Russo, A.; Caltabiano, R.; Ragusa, M. Genetics and RNA Regulation of Uveal Melanoma. Cancers 2023, 15, 775. [Google Scholar] [CrossRef]
- Wei, A.Z.; Maniar, A.B.; Carvajal, R.D. New targeted and epigenetic therapeutic strategies for the treatment of uveal melanoma. Cancer Gene Ther. 2022, 29, 1819–1826. [Google Scholar] [CrossRef]
- Field, M.G.; Durante, M.A.; Decatur, C.L.; Tarlan, B.; Oelschlager, K.M.; Stone, J.F.; Kuznetsov, J.; Bowcock, A.M.; Kurtenbach, S.; Harbour, J.W. Epigenetic reprogramming and aberrant expression of PRAME are associated with increased metastatic risk in Class 1 and Class 2 uveal melanomas. Oncotarget 2016, 7, 59209–59219. [Google Scholar] [CrossRef]
- Field, M.G.; Decatur, C.L.; Kurtenbach, S.; Gezgin, G.; van der Velden, P.A.; Jager, M.J.; Kozak, K.N.; Harbour, J.W. PRAME as an Independent Biomarker for Metastasis in Uveal Melanoma. Clin. Cancer Res. 2016, 22, 1234–1242. [Google Scholar] [CrossRef] [PubMed]
- Jindal, V. Role of immune checkpoint inhibitors and novel immunotherapies in uveal melanoma. Chin. Clin. Oncol. 2018, 7, 8. [Google Scholar] [CrossRef] [PubMed]
- Amir, A.L.; van der Steen, D.M.; van Loenen, M.M.; Hagedoorn, R.S.; de Boer, R.; Kester, M.D.; de Ru, A.H.; Lugthart, G.J.; van Kooten, C.; Hiemstra, P.S.; et al. PRAME-specific Allo-HLA-restricted T cells with potent antitumor reactivity useful for therapeutic T-cell receptor gene transfer. Clin. Cancer Res. 2011, 17, 5615–5625. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.K.; Barker, C.; Coit, D.G.; Joseph, R.W.; Materin, M.; Rengan, R.; Sosman, J.; Thompson, J.A.; Albertini, M.R.; Boland, G.; et al. NCCN Guidelines Insights: Uveal Melanoma, Version 1.2019: Featured Updates to the NCCN Guidelines. J. Natl. Compr. Cancer Netw. 2020, 18, 120–131. [Google Scholar] [CrossRef]
- Abrams, M.J.; Gagne, N.L.; Melhus, C.S.; Mignano, J.E. Brachytherapy vs. external beam radiotherapy for choroidal melanoma: Survival and patterns-of-care analyses. Brachytherapy 2016, 15, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Verma, V.; Mehta, M.P. Clinical Outcomes of Proton Radiotherapy for Uveal Melanoma. Clin. Oncol. 2016, 28, e17–e27. [Google Scholar] [CrossRef]
- Romano, M.R.; Catania, F.; Confalonieri, F.; Zollet, P.; Allegrini, D.; Sergenti, J.; Lanza, F.B.; Ferrara, M.; Angi, M. Vitreoretinal Surgery in the Prevention and Treatment of Toxic Tumour Syndrome in Uveal Melanoma: A Systematic Review. Int. J. Mol. Sci. 2021, 22, 10066. [Google Scholar] [CrossRef]
- Foti, P.V.; Travali, M.; Farina, R.; Palmucci, S.; Spatola, C.; Liardo, R.L.E.; Milazzotto, R.; Raffaele, L.; Salamone, V.; Caltabiano, R.; et al. Diagnostic methods and therapeutic options of uveal melanoma with emphasis on MR imaging-Part II: Treatment indications and complications. Insights Imaging 2021, 12, 67. [Google Scholar] [CrossRef]
- Jager, M.J.; Shields, C.L.; Cebulla, C.M.; Abdel-Rahman, M.H.; Grossniklaus, H.E.; Stern, M.-H.; Carvajal, R.D.; Belfort, R.N.; Jia, R.; Shields, J.A.; et al. Uveal melanoma. Nat. Rev. Dis. Primers 2020, 6, 24. [Google Scholar] [CrossRef]
- Binder, C.; Mruthyunjaya, P.; Schefler, A.C.; Seider, M.I.; Crilly, R.; Hung, A.; Meltsner, S.; Mowery, Y.; Kirsch, D.G.; Tehe, B.S.; et al. Practice Patterns for the Treatment of Uveal Melanoma with Iodine-125 Plaque Brachytherapy: Ocular Oncology Study Consortium Report 5. Ocul. Oncol. Pathol. 2019, 6, 210–218. [Google Scholar] [CrossRef]
- Souto, E.B.; Zielinska, A.; Luis, M.; Carbone, C.; Martins-Gomes, C.; Souto, S.B.; Silva, A.M. Uveal melanoma: Physiopathology and new in situ-specific therapies. Cancer Chemother. Pharmacol. 2019, 84, 15–32. [Google Scholar] [CrossRef] [PubMed]
- Jaradat, I.; Zewar, A.; AlNawaiseh, I.; AlRawashdeh, K.; Khurma, S.; Mehyar, M.; Abdeen, G.; Yousef, Y.A. Characteristics, management, and outcome of patients with uveal melanoma treated by Iodine-125 radioactive plaque therapy in a single tertiary cancer center in Jordan. Saudi J. Ophthalmol. 2018, 32, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Mirshahi, R.; Sedaghat, A.; Jaberi, R.; Azma, Z.; Mazloumi, M.; Naseripour, M. Ruthenium-106 plaque radiotherapy for uveal melanoma: Analysis of tumor dimension and location on anatomical and functional results. BMC Ophthalmol. 2022, 22, 309. [Google Scholar] [CrossRef] [PubMed]
- Sikuade, M.J.; Salvi, S.; Rundle, P.A.; Errington, D.G.; Kacperek, A.; Rennie, I.G. Outcomes of treatment with stereotactic radiosurgery or proton beam therapy for choroidal melanoma. Eye 2015, 29, 1194–1198. [Google Scholar] [CrossRef] [PubMed]
- Dunavoelgyi, R.; Dieckmann, K.; Gleiss, A.; Sacu, S.; Kircher, K.; Georgopoulos, M.; Georg, D.; Zehetmayer, M.; Poetter, R. Local tumor control, visual acuity, and survival after hypofractionated stereotactic photon radiotherapy of choroidal melanoma in 212 patients treated between 1997 and 2007. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Damato, B.; Kacperek, A.; Errington, D.; Heimann, H. Proton beam radiotherapy of uveal melanoma. Saudi J. Ophthalmol. 2013, 27, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Rundle, P. Treatment of posterior uveal melanoma with multi-dose photodynamic therapy. Br. J. Ophthalmol. 2014, 98, 494–497. [Google Scholar] [CrossRef]
- Shields, C.L.; Shields, J.A.; Perez, N.; Singh, A.D.; Cater, J. Primary transpupillary thermotherapy for small choroidal melanoma in 256 consecutive cases: Outcomes and limitations. Ophthalmology 2002, 109, 225–234. [Google Scholar] [CrossRef]
- Amaro, A.; Gangemi, R.; Piaggio, F.; Angelini, G.; Barisione, G.; Ferrini, S.; Pfeffer, U. The biology of uveal melanoma. Cancer Metastasis Rev. 2017, 36, 109–140. [Google Scholar] [CrossRef]
- Zewar, A.; Nawaiseh, I.; Jaradat, I.; Khzouz, J.; Alrawashdeh, K.; Abdeen, G.; Mehyar, M.; Khurma, S.; Yousef, Y.A. Management and Outcome of Uveal Melanoma in a Single Tertiary Cancer Center in Jordan. Turk Patoloji Derg. 2016, 32, 186–192. [Google Scholar] [CrossRef][Green Version]
- Berus, T.; Halon, A.; Markiewicz, A.; Orlowska-Heitzman, J.; Romanowska-Dixon, B.; Donizy, P. Clinical, Histopathological and Cytogenetic Prognosticators in Uveal Melanoma—A Comprehensive Review. Anticancer Res. 2017, 37, 6541–6549. [Google Scholar] [CrossRef] [PubMed]
- Barker, C.A.; Salama, A.K. New NCCN Guidelines for Uveal Melanoma and Treatment of Recurrent or Progressive Distant Metastatic Melanoma. J. Natl. Compr. Cancer Netw. 2018, 16, 646–650. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, C.; Kim, D.W.; Gombos, D.S.; Oba, J.; Qin, Y.; Williams, M.D.; Esmaeli, B.; Grimm, E.A.; Wargo, J.A.; Woodman, S.E.; et al. Uveal melanoma: From diagnosis to treatment and the science in between. Cancer 2016, 122, 2299–2312. [Google Scholar] [CrossRef] [PubMed]
- Grossniklaus, H.E. Progression of ocular melanoma metastasis to the liver: The 2012 Zimmerman lecture. JAMA Ophthalmol. 2013, 131, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Rowcroft, A.; Loveday, B.P.T.; Thomson, B.N.J.; Banting, S.; Knowles, B. Systematic review of liver directed therapy for uveal melanoma hepatic metastases. HPB 2020, 22, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Sajan, A.; Fordyce, S.; Sideris, A.; Liou, C.; Toor, Z.; Filtes, J.; Krishnasamy, V.; Ahmad, N.; Reis, S.; Brejt, S.; et al. Minimally Invasive Treatment Options for Hepatic Uveal Melanoma Metastases. Diagnostics 2023, 13, 1836. [Google Scholar] [CrossRef] [PubMed]
- Huppert, P.E.; Fierlbeck, G.; Pereira, P.; Schanz, S.; Duda, S.H.; Wietholtz, H.; Rozeik, C.; Claussen, C.D. Transarterial chemoembolization of liver metastases in patients with uveal melanoma. Eur. J. Radiol. 2010, 74, e38–e44. [Google Scholar] [CrossRef]
- Sato, T.; Eschelman, D.J.; Gonsalves, C.F.; Terai, M.; Chervoneva, I.; McCue, P.A.; Shields, J.A.; Shields, C.L.; Yamamoto, A.; Berd, D.; et al. Immunoembolization of malignant liver tumors, including uveal melanoma, using granulocyte-macrophage colony-stimulating factor. J. Clin. Oncol. 2008, 26, 5436–5442. [Google Scholar] [CrossRef]
- Dueland, S.; Solheim, J.M.; Espen Foss, A.; Grut, H.; Hagness, M.; Line, P.-D. Dismal survival following liver transplantation for liver-only metastases in patients with ocular malignant melanoma. Trends Transplant. 2020, 13, 1–4. [Google Scholar] [CrossRef]
- Johansson, P.A.; Brooks, K.; Newell, F.; Palmer, J.M.; Wilmott, J.S.; Pritchard, A.L.; Broit, N.; Wood, S.; Carlino, M.S.; Leonard, C.; et al. Whole genome landscapes of uveal melanoma show an ultraviolet radiation signature in iris tumours. Nat. Commun. 2020, 11, 2408. [Google Scholar] [CrossRef]
- Kaštelan, S.; Antunica, A.G.; Oresković, L.B.; Pelčić, G.; Kasun, E.; Hat, K. Immunotherapy for Uveal Melanoma —Current Knowledge and Perspectives. Curr. Med. Chem. 2020, 27, 1350–1366. [Google Scholar] [CrossRef] [PubMed]
- Wessely, A.; Steeb, T.; Erdmann, M.; Heinzerling, L.; Vera, J.; Schlaak, M.; Berking, C.; Heppt, M.V. The Role of Immune Checkpoint Blockade in Uveal Melanoma. Int. J. Mol. Sci. 2020, 21, 879. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.A.; Rodriguez, D.A.; Kurtenbach, S.; Kuznetsov, J.N.; Sanchez, M.I.; Decatur, C.L.; Snyder, H.; Feun, L.G.; Livingstone, A.S.; Harbour, J.W. Single-cell analysis reveals new evolutionary complexity in uveal melanoma. Nat. Commun. 2020, 11, 496. [Google Scholar] [CrossRef] [PubMed]
- Oliva, M.; Rullan, A.J.; Piulats, J.M. Uveal melanoma as a target for immune-therapy. Ann. Transl. Med. 2016, 4, 172. [Google Scholar] [CrossRef] [PubMed]
- Niederkorn, J.Y. Ocular immune privilege and ocular melanoma: Parallel universes or immunological plagiarism? Front. Immunol. 2012, 3, 148. [Google Scholar] [CrossRef] [PubMed]
- Rossi, E.; Schinzari, G.; Zizzari, I.G.; Maiorano, B.A.; Pagliara, M.M.; Sammarco, M.G.; Fiorentino, V.; Petrone, G.; Cassano, A.; Rindi, G.; et al. Immunological Backbone of Uveal Melanoma: Is There a Rationale for Immunotherapy? Cancers 2019, 11, 1055. [Google Scholar] [CrossRef]
- Harjunpää, H.; Guillerey, C. TIGIT as an emerging immune checkpoint. Clin. Exp. Immunol. 2020, 200, 108–119. [Google Scholar] [CrossRef]
- Stålhammar, G.; Seregard, S.; Grossniklaus, H.E. Expression of immune checkpoint receptors Indoleamine 2,3-dioxygenase and T cell Ig and ITIM domain in metastatic versus nonmetastatic choroidal melanoma. Cancer Med. 2019, 8, 2784–2792. [Google Scholar] [CrossRef]
- Fu, Y.; Xiao, W.; Mao, Y. Recent Advances and Challenges in Uveal Melanoma Immunotherapy. Cancers 2022, 14, 3094. [Google Scholar] [CrossRef]
- Woo, S.R.; Turnis, M.E.; Goldberg, M.V.; Bankoti, J.; Selby, M.; Nirschl, C.J.; Bettini, M.L.; Gravano, D.M.; Vogel, P.; Liu, C.L.; et al. Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Res. 2012, 72, 917–927. [Google Scholar] [CrossRef]
- Ruffo, E.; Wu, R.C.; Bruno, T.C.; Workman, C.J.; Vignali, D.A.A. Lymphocyte-activation gene 3 (LAG3): The next immune checkpoint receptor. Semin. Immunol. 2019, 42, 101305. [Google Scholar] [CrossRef] [PubMed]
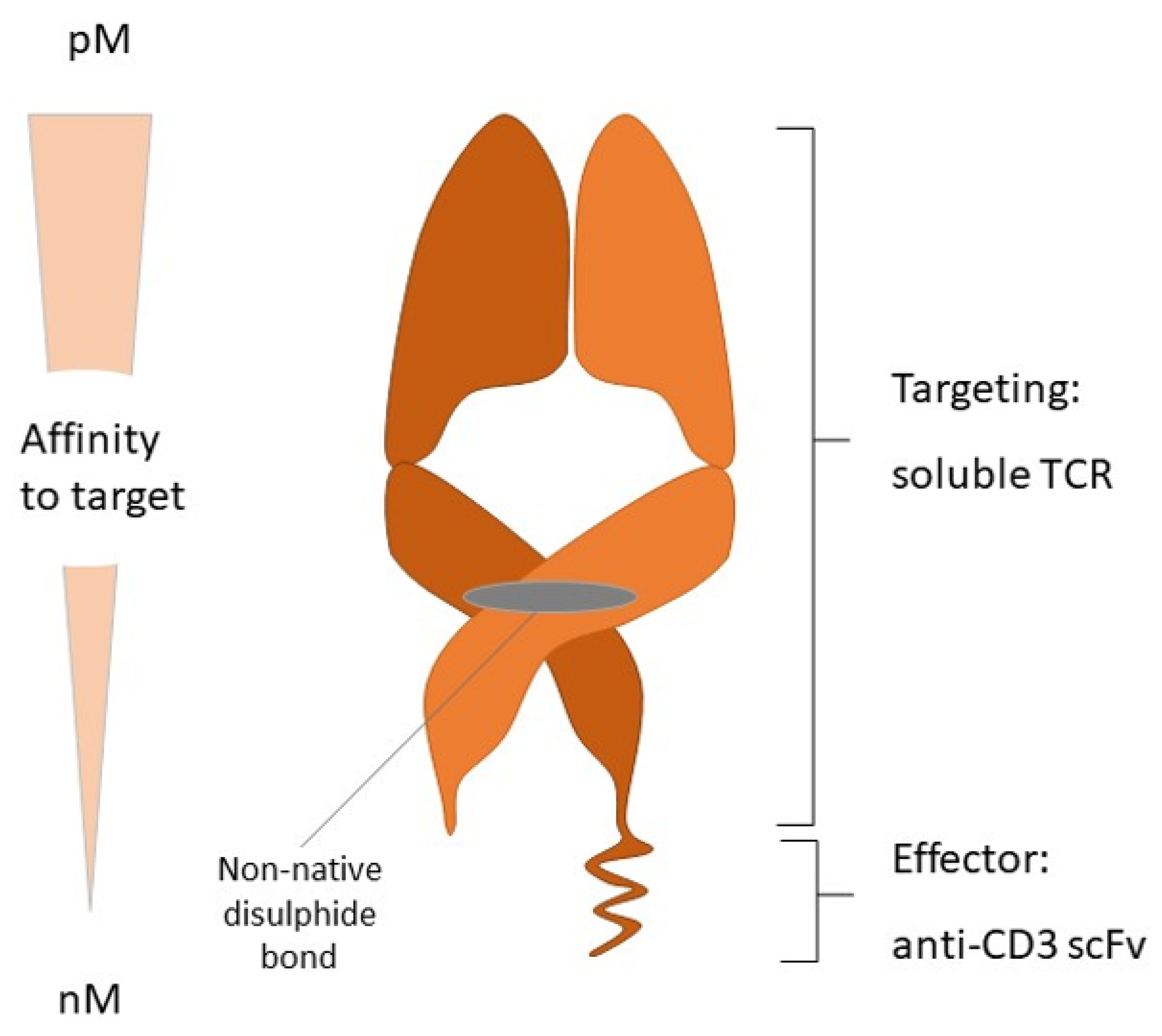
- Wang, S.; Chen, K.; Lei, Q.; Ma, P.; Yuan, A.Q.; Zhao, Y.; Jiang, Y.; Fang, H.; Xing, S.; Fang, Y.; et al. The state of the art of bispecific antibodies for treating human malignancies. EMBO Mol. Med. 2021, 13, e14291. [Google Scholar] [CrossRef] [PubMed]
- Goebeler, M.-E.; Bargou, R.C. T cell-engaging therapies—BiTEs and beyond. Nat. Rev. Clin. Oncol. 2020, 17, 418–434. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, G.S.; Oliver, S. Methods of Treating Ocular Cancer Using Anti-Met Antibodies and Bispecific Antigen Binding Molecules that Bind MET. AU20.202.202024136A1. 20 February 2020. [Google Scholar]
- Herrmann, M.; Krupka, C.; Deiser, K.; Brauchle, B.; Marcinek, A.; Ogrinc Wagner, A.; Rataj, F.; Mocikat, R.; Metzeler, K.H.; Spiekermann, K.; et al. Bifunctional PD-1 × αCD3 × αCD33 fusion protein reverses adaptive immune escape in acute myeloid leukemia. Blood 2018, 132, 2484–2494. [Google Scholar] [CrossRef]
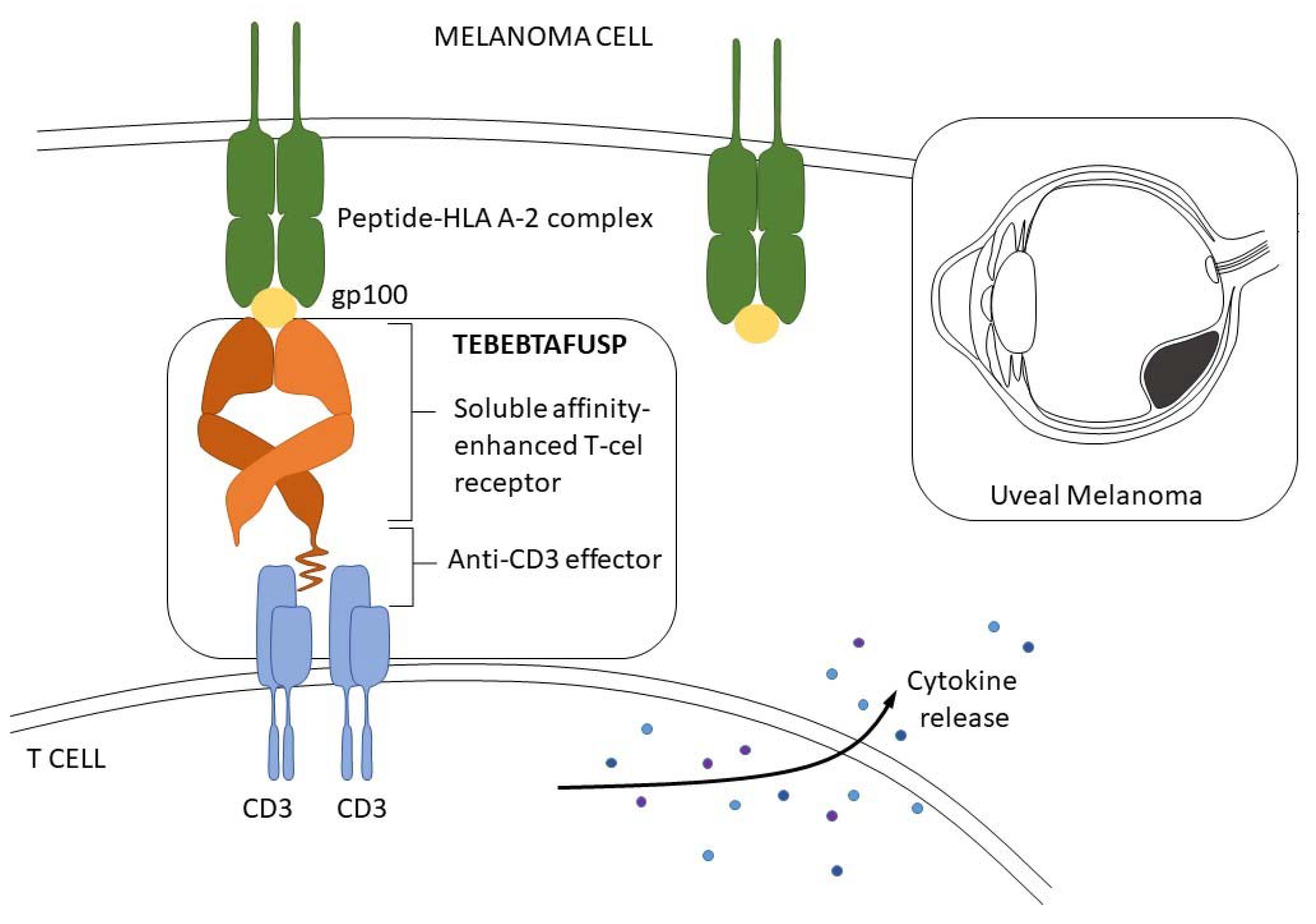
- Damato, B.E.; Dukes, J.; Goodall, H.; Carvajal, R.D. Tebentafusp: T Cell Redirection for the Treatment of Metastatic Uveal Melanoma. Cancers 2019, 11, 971. [Google Scholar] [CrossRef] [PubMed]
- Orloff, M.; Seedor, R.; Sato, T. Review of bi-specific therapies in uveal melanoma. Cancer Gene Ther. 2022, 29, 1814–1818. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, L.; Vaubel, J.; Mohr, P.; Hauschild, A.; Utikal, J.; Simon, J.; Garbe, C.; Herbst, R.; Enk, A.; Kämpgen, E.; et al. Phase II DeCOG-study of ipilimumab in pretreated and treatment-naïve patients with metastatic uveal melanoma. PLoS ONE 2015, 10, e0118564. [Google Scholar] [CrossRef]
- Kummer, M.; Schuler-Thurner, B. Immunotherapy of Uveal Melanoma: Vaccination Against Cancer. Methods Mol. Biol. 2017, 1499, 273–278. [Google Scholar] [CrossRef]
- Nathan, P.; Hassel, J.C.; Rutkowski, P.; Baurain, J.-F.; Butler, M.O.; Schlaak, M.; Sullivan, R.J.; Ochsenreither, S.; Dummer, R.; Kirkwood, J.M.; et al. Overall Survival Benefit with Tebentafusp in Metastatic Uveal Melanoma. N. Engl. J. Med. 2021, 385, 1196–1206. [Google Scholar] [CrossRef]
- Marseglia, M.; Amaro, A.; Solari, N.; Gangemi, R.; Croce, E.; Tanda, E.T.; Spagnolo, F.; Filaci, G.; Pfeffer, U.; Croce, M. How to Make Immunotherapy an Effective Therapeutic Choice for Uveal Melanoma. Cancers 2021, 13, 2043. [Google Scholar] [CrossRef]
- Schank, T.E.; Hassel, J.C. Immunotherapies for the Treatment of Uveal Melanoma—History and Future. Cancers 2019, 11, 1048. [Google Scholar] [CrossRef] [PubMed]
- Kaunitz, G.J.; Cottrell, T.R.; Lilo, M.; Muthappan, V.; Esandrio, J.; Berry, S.; Xu, H.; Ogurtsova, A.; Anders, R.A.; Fischer, A.H.; et al. Melanoma subtypes demonstrate distinct PD-L1 expression profiles. Lab. Investig. 2017, 97, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Javed, A.; Arguello, D.; Johnston, C.; Gatalica, Z.; Terai, M.; Weight, R.M.; Orloff, M.; Mastrangelo, M.J.; Sato, T. PD-L1 expression in tumor metastasis is different between uveal melanoma and cutaneous melanoma. Immunotherapy 2017, 9, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Pelster, M.S.; Gruschkus, S.K.; Bassett, R.; Gombos, D.S.; Shephard, M.; Posada, L.; Glover, M.S.; Simien, R.; Diab, A.; Hwu, P.; et al. Nivolumab and Ipilimumab in Metastatic Uveal Melanoma: Results from a Single-Arm Phase II Study. J. Clin. Oncol. 2021, 39, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Huang, X.; Chen, X.; Liu, J.; Wu, C.; Pu, Q.; Wang, Y.; Kang, X.; Zhou, L. Characterization of a novel anti-human lymphocyte activation gene 3 (LAG-3) antibody for cancer immunotherapy. MAbs 2019, 11, 1139–1148. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.R.; Odashiro, A.N.; Lim, L.A.; Miyamoto, C.; Blanco, P.L.; Odashiro, M.; Maloney, S.; De Souza, D.F.; Burnier, M.N., Jr. Current and emerging treatment options for uveal melanoma. Clin. Ophthalmol. 2013, 7, 1669–1682. [Google Scholar] [CrossRef]
- Tanaka, R.; Terai, M.; Londin, E.; Sato, T. The Role of HGF/MET Signaling in Metastatic Uveal Melanoma. Cancers 2021, 13, 5457. [Google Scholar] [CrossRef]
- Bosch, J.J.; Iheagwara, U.K.; Reid, S.; Srivastava, M.K.; Wolf, J.; Lotem, M.; Ksander, B.R.; Ostrand-Rosenberg, S. Uveal melanoma cell-based vaccines express MHC II molecules that traffic via the endocytic and secretory pathways and activate CD8 cytotoxic, tumor-specific T cells. Cancer Immunol. Immunother. 2010, 59, 103–112. [Google Scholar] [CrossRef]
- Meecham, W.J.; Char, D.H.; Kaleta-Michaels, S. Infiltrating lymphocytes and antigen expression in uveal melanoma. Ophthalmic Res. 1992, 24, 20–26. [Google Scholar] [CrossRef]
- Biswas, S.K.; Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: Cancer as a paradigm. Nat. Immunol. 2010, 11, 889–896. [Google Scholar] [CrossRef]
- Whelchel, J.C.; Farah, S.E.; McLean, I.W.; Burnier, M.N. Immunohistochemistry of infiltrating lymphocytes in uveal malignant melanoma. Investig. Ophthalmol. Vis. Sci. 1993, 34, 2603–2606. [Google Scholar]
- Lagouros, E.; Salomao, D.; Thorland, E.; Hodge, D.O.; Vile, R.; Pulido, J.S. Infiltrative T regulatory cells in enucleated uveal melanomas. Trans. Am. Ophthalmol. Soc. 2009, 107, 223–228. [Google Scholar] [PubMed]
- Chandran, S.S.; Somerville, R.P.T.; Yang, J.C.; Sherry, R.M.; Klebanoff, C.A.; Goff, S.L.; Wunderlich, J.R.; Danforth, D.N.; Zlott, D.; Paria, B.C.; et al. Treatment of metastatic uveal melanoma with adoptive transfer of tumour-infiltrating lymphocytes: A single-centre, two-stage, single-arm, phase 2 study. Lancet Oncol. 2017, 18, 792–802. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, B.F.; Odashiro, A.N.; Saraiva, V.S.; Logan, P.; Antecka, E.; Burnier, M.N., Jr. Immunohistochemical expression of melan-A and tyrosinase in uveal melanoma. J. Carcinog. 2007, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Kaur, I.; Ali, M.J.; Biswas, N.K.; Das, S.; Kumar, S.; Honavar, S.G.; Maitra, A.; Chakrabarti, S.; Majumder, P.P. Exome sequencing reveals the likely involvement of SOX10 in uveal melanoma. Optom. Vis. Sci. 2014, 91, e185–e192. [Google Scholar] [CrossRef]
- Casalou, C.; Mayatra, J.M.; Tobin, D.J. Beyond the Epidermal-Melanin-Unit: The Human Scalp Anagen Hair Bulb Is Home to Multiple Melanocyte Subpopulations of Variable Melanogenic Capacity. Int. J. Mol. Sci. 2023, 24, 12809. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, S.; Burrows, R.C.; Kalina, R.E.; George, D.; Boehm, M.; Bothwell, M.A.; Schmidt, R. Immunophenotypic Differences Between Uveal and Cutaneous Melanomas. Arch. Ophthalmol. 2002, 120, 466–470. [Google Scholar] [CrossRef]
- Kan-Mitchell, J.; Rao, N.; Albert, D.M.; Van Eldik, L.J.; Taylor, C.R. S100 immunophenotypes of uveal melanomas. Investig. Ophthalmol. Vis. Sci. 1990, 31, 1492–1496. [Google Scholar]
- Kan-Mitchell, J.; Liggett, P.E.; Taylor, C.R.; Rao, N.; Granada, E.S.; Danenberg, K.D.; White, W.L.; Van Eldik, L.J.; Horikoshi, T.; Danenberg, P.V. Differential S100 beta expression in choroidal and skin melanomas: Quantitation by the polymerase chain reaction. Investig. Ophthalmol. Vis. Sci. 1993, 34, 3366–3375. [Google Scholar]
- Thill, M.; Berna, M.J.; Grierson, R.; Reinhart, I.; Voelkel, T.; Piechaczek, C.; Galambos, P.; Jager, M.J.; Richard, G.; Lange, C.; et al. Expression of CD133 and other putative stem cell markers in uveal melanoma. Melanoma Res. 2011, 21, 405–416. [Google Scholar] [CrossRef]
- Djirackor, L.; Kalirai, H.; Coupland, S.E.; Petrovski, G. CD166high Uveal Melanoma Cells Represent a Subpopulation with Enhanced Migratory Capacity. Investig. Ophthalmol. Vis. Sci. 2019, 60, 2696–2704. [Google Scholar] [CrossRef]
- Landreville, S.; Agapova, O.A.; Kneass, Z.T.; Salesse, C.; Harbour, J.W. ABCB1 identifies a subpopulation of uveal melanoma cells with high metastatic propensity. Pigment. Cell Melanoma Res. 2011, 24, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Casalou, C.; Moreiras, H.; Mayatra, J.M.; Fabre, A.; Tobin, D.J. Loss of ‘Epidermal Melanin Unit’ Integrity in Human Skin During Melanoma-Genesis. Front. Oncol. 2022, 12, 878336. [Google Scholar] [CrossRef] [PubMed]
- Del Bufalo, F.; De Angelis, B.; Caruana, I.; Del Baldo, G.; De Ioris, M.A.; Serra, A.; Mastronuzzi, A.; Cefalo, M.G.; Pagliara, D.; Amicucci, M.; et al. GD2-CART01 for Relapsed or Refractory High-Risk Neuroblastoma. N. Engl. J. Med. 2023, 388, 1284–1295. [Google Scholar] [CrossRef] [PubMed]
- Wei, F.; Cheng, X.X.; Xue, J.Z.; Xue, S.A. Emerging Strategies in TCR-Engineered T Cells. Front. Immunol. 2022, 13, 850358. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.clinicaltrials.gov/ (accessed on 26 July 2023).
- Rohaan, M.W.; Gomez-Eerland, R.; Foppen, M.H.G.; van Zon, M.; de Boer, R.; Bakker, N.A.M.; Pronk, L.M.; Sari, A.; Mallo, H.A.; van de Wiel, B.A.; et al. 1184PD—Results of a phase I trial with MART-1 T cell receptor modified T cells in patients with metastatic melanoma. Ann. Oncol. 2019, 30, v481–v482. [Google Scholar] [CrossRef]
- Felices, M.; Lenvik, T.R.; Davis, Z.B.; Miller, J.S.; Vallera, D.A. Generation of BiKEs and TriKEs to Improve NK Cell-Mediated Targeting of Tumor Cells. Methods Mol. Biol. 2016, 1441, 333–346. [Google Scholar] [CrossRef]
- Vallera, D.A.; Felices, M.; McElmurry, R.; McCullar, V.; Zhou, X.; Schmohl, J.U.; Zhang, B.; Lenvik, A.J.; Panoskaltsis-Mortari, A.; Verneris, M.R.; et al. IL15 Trispecific Killer Engagers (TriKE) Make Natural Killer Cells Specific to CD33 Targets While Also Inducing Persistence, In Vivo Expansion, and Enhanced Function. Clin. Cancer Res. 2016, 22, 3440–3450. [Google Scholar] [CrossRef]
- Apte, R.S.; Sinha, D.; Mayhew, E.; Wistow, G.J.; Niederkorn, J.Y. Cutting Edge: Role of Macrophage Migration Inhibitory Factor in Inhibiting NK Cell Activity and Preserving Immune Privilege. J. Immunol. 1998, 160, 5693. [Google Scholar] [CrossRef]
- Rook, A.H.; Kehrl, J.H.; Wakefield, L.M.; Roberts, A.B.; Sporn, M.B.; Burlington, D.B.; Lane, H.C.; Fauci, A.S. Effects of transforming growth factor beta on the functions of natural killer cells: Depressed cytolytic activity and blunting of interferon responsiveness. J. Immunol. 1986, 136, 3916–3920. [Google Scholar] [CrossRef]
- Repp, A.C.; Mayhew, E.S.; Apte, S.; Niederkorn, J.Y. Human uveal melanoma cells produce macrophage migration-inhibitory factor to prevent lysis by NK cells. J. Immunol. 2000, 165, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, E.; Baird, A.W.; O’Farrelly, C. Microanatomy of the liver immune system. Semin. Immunopathol. 2009, 31, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Dithmar, S.A.; Rusciano, D.A.; Armstrong, C.A.; Lynn, M.J.; Grossniklaus, H.E. Depletion of NK cell activity results in growth of hepatic micrometastases in a murine ocular melanoma model. Curr. Eye Res. 1999, 19, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, H.; Howard, K.; Mellon, J.; Mayhew, E.; Rusciano, D.; Niederkorn, J.Y. Reduction of liver metastasis of intraocular melanoma by interferon-beta gene transfer. Investig. Ophthalmol. Vis. Sci. 2003, 44, 3042–3051. [Google Scholar] [CrossRef] [PubMed]
- Langbein, T.; Weber, W.A.; Eiber, M. Future of Theranostics: An Outlook on Precision Oncology in Nuclear Medicine. J. Nucl. Med. 2019, 60, 13s–19s. [Google Scholar] [CrossRef] [PubMed]
- Bhujwalla, Z.M.; Kakkad, S.; Chen, Z.; Jin, J.; Hapuarachchige, S.; Artemov, D.; Penet, M.F. Theranostics and metabolotheranostics for precision medicine in oncology. J. Magn. Reson. 2018, 291, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kwak, G.; Kim, K.; Yoon, H.Y.; Kwon, I.C. Theranostic designs of biomaterials for precision medicine in cancer therapy. Biomaterials 2019, 213, 119207. [Google Scholar] [CrossRef]
- Dal Corso, A.; Pignataro, L.; Belvisi, L.; Gennari, C. αvβ3 Integrin-Targeted Peptide/Peptidomimetic-Drug Conjugates: In-Depth Analysis of the Linker Technology. Curr. Top. Med. Chem. 2016, 16, 314–329. [Google Scholar] [CrossRef]
- Kobayashi, H.; Choyke, P.L. Target-cancer-cell-specific activatable fluorescence imaging probes: Rational design and in vivo applications. Acc. Chem. Res. 2011, 44, 83–90. [Google Scholar] [CrossRef]
- Sun, W.; Li, M.; Fan, J.; Peng, X. Activity-Based Sensing and Theranostic Probes Based on Photoinduced Electron Transfer. Acc. Chem. Res. 2019, 52, 2818–2831. [Google Scholar] [CrossRef]
- Ling, D.; Park, W.; Park, S.J.; Lu, Y.; Kim, K.S.; Hackett, M.J.; Kim, B.H.; Yim, H.; Jeon, Y.S.; Na, K.; et al. Multifunctional tumor pH-sensitive self-assembled nanoparticles for bimodal imaging and treatment of resistant heterogeneous tumors. J. Am. Chem. Soc. 2014, 136, 5647–5655. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Kim, J.; Lee, J.Y.; Matsuda, S.; Hideshima, S.; Mori, Y.; Osaka, T.; Na, K. Stimuli-responsive magnetic nanoparticles for tumor-targeted bimodal imaging and photodynamic/hyperthermia combination therapy. Nanoscale 2016, 8, 11625–11634. [Google Scholar] [CrossRef] [PubMed]
- de la Rica, R.; Aili, D.; Stevens, M.M. Enzyme-responsive nanoparticles for drug release and diagnostics. Adv. Drug Deliv. Rev. 2012, 64, 967–978. [Google Scholar] [CrossRef]
- Kim, E.J.; Bhuniya, S.; Lee, H.; Kim, H.M.; Cheong, C.; Maiti, S.; Hong, K.S.; Kim, J.S. An activatable prodrug for the treatment of metastatic tumors. J. Am. Chem. Soc. 2014, 136, 13888–13894. [Google Scholar] [CrossRef]
- Zhou, Y.; Maiti, M.; Sharma, A.; Won, M.; Yu, L.; Miao, L.X.; Shin, J.; Podder, A.; Bobba, K.N.; Han, J.; et al. Azo-based small molecular hypoxia responsive theranostic for tumor-specific imaging and therapy. J. Control. Release 2018, 288, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Lecocq, Q.; De Vlaeminck, Y.; Hanssens, H.; D’Huyvetter, M.; Raes, G.; Goyvaerts, C.; Keyaerts, M.; Devoogdt, N.; Breckpot, K. Theranostics in immuno-oncology using nanobody derivatives. Theranostics 2019, 9, 7772–7791. [Google Scholar] [CrossRef] [PubMed]
- Fornaguera, C.; García-Celma, M.J. Personalized Nanomedicine: A Revolution at the Nanoscale. Journal of Personalized Medicine. Pers. Med. 2017, 7, 12. [Google Scholar] [CrossRef]
- Vladimir, T. Handbook of Materials for Nanomedicine Lipid-Based and Inorganic Nanomaterials; Jenny Stanford Publishing: Dubai, United Arab Emirates, 2020. [Google Scholar] [CrossRef]
- Bilmin, K.; Synoradzki, K.J.; Czarnecka, A.M.; Spałek, M.J.; Kujawska, T.; Solnik, M.; Merks, P.; Toro, M.D.; Rejdak, R.; Fiedorowicz, M. New Perspectives for Eye-Sparing Treatment Strategies in Primary Uveal Melanoma. Cancers 2022, 14, 134. [Google Scholar] [CrossRef]
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. 2021, 20, 101–124. [Google Scholar] [CrossRef]
- Weng, Y.; Liu, J.; Jin, S.; Guo, W.; Liang, X.; Hu, Z. Nanotechnology-based strategies for treatment of ocular disease. Acta Pharm. Sin. B 2017, 7, 281–291. [Google Scholar] [CrossRef]
- You, S.; Luo, J.; Grossniklaus, H.E.; Gou, M.L.; Meng, K.; Zhang, Q. Nanomedicine in the application of uveal melanoma. Int. J. Ophthalmol. 2016, 9, 1215–1225. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, C.; Platania, C.B.M.; Drago, F.; Barbagallo, D.; Di Pietro, C.; Purrello, M.; Bucolo, C.; Ragusa, M. Do Extracellular RNAs Provide Insight into Uveal Melanoma Biology? Cancers 2021, 13, 5919. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wang, R.; Hardy, P. Potential of miRNA-Based Nanotherapeutics for Uveal Melanoma. Cancers 2021, 13, 5192. [Google Scholar] [CrossRef] [PubMed]
- Milán Rois, P.; Latorre, A.; Rodriguez Diaz, C.; Del Moral, Á.; Somoza, Á. Reprogramming Cells for Synergistic Combination Therapy with Nanotherapeutics against Uveal Melanoma. Biomimetics 2018, 3, 28. [Google Scholar] [CrossRef] [PubMed]
- Latorre, A.; Latorre, A.; Castellanos, M.; Lafuente-Gómez, N.; Diaz, C.R.; Crespo-Barreda, A.; Lecea, M.; Cordani, M.; Martín-Duque, P.; Somoza, Á. Albumin-based nanostructures for uveal melanoma treatment. Nanomedicine 2021, 35, 102391. [Google Scholar] [CrossRef] [PubMed]
- Bonaccorso, A.; Pepe, V.; Zappulla, C.; Cimino, C.; Pricoco, A.; Puglisi, G.; Giuliano, F.; Pignatello, R.; Carbone, C. Sorafenib Repurposing for Ophthalmic Delivery by Lipid Nanoparticles: A Preliminary Study. Pharmaceutics 2021, 13, 1956. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Yue, W.; Ibrahim, K.; Shen, J. A Long-Acting Curcumin Nanoparticle/In Situ Hydrogel Composite for the Treatment of Uveal Melanoma. Pharmaceutics 2021, 13, 1335. [Google Scholar] [CrossRef]
- Huang, T.; Xu, X.; Cheng, C.; Wang, J.; Yang, L. Cooperative phototherapy based on bimodal imaging guidance for the treatment of uveal melanoma. J. Nanobiotechnology 2023, 21, 146. [Google Scholar] [CrossRef]
- Ding, Y.; Yu, J.; Chen, X.; Wang, S.; Tu, Z.; Shen, G.; Wang, H.; Jia, R.; Ge, S.; Ruan, J.; et al. Dose-Dependent Carbon-Dot-Induced ROS Promote Uveal Melanoma Cell Tumorigenicity via Activation of mTOR Signaling and Glutamine Metabolism. Adv. Sci. 2021, 8, 2002404. [Google Scholar] [CrossRef]
- Mruthyunjaya, P.; Schefler, A.C.; Kim, I.K.; Bergstrom, C.; Demirci, H.; Tsai, T.; Bhavsar, A.R.; Capone, A.; Marr, B.; McCannel, T.A.; et al. A Phase 1b/2 Open-label Clinical Trial to Evaluate the Safety and Efficacy of AU-011 for the Treatment of Choroidal Melanoma. Investig. Ophthalmol. Vis. Sci. 2020, 61, 4025. [Google Scholar]
- Kines, R.C.; Thompson, C.D.; Spring, S.; Li, Z.; de Los Pinos, E.; Monks, S.; Schiller, J.T. Virus-Like Particle-Drug Conjugates Induce Protective, Long-lasting Adaptive Antitumor Immunity in the Absence of Specifically Targeted Tumor Antigens. Cancer Immunol. Res. 2021, 9, 693–706. [Google Scholar] [CrossRef] [PubMed]
| Evaluated Treatment | Mechanism | Clinical Trial | Status |
|---|---|---|---|
| DYP688 | N/A mechanism | NCT05415072 | Recruiting |
| Pembrolizumab 25 MG/1 ML Intravenous Solution | anti-PD-1 | NCT05282901 | Recruiting |
| Pembrolizumab Lenvatinib | anti-PD-1 + multiple kinase inhibitor | NCT05308901 | Recruiting |
| Autologous Tumor-Infiltrating Lymphocytes Melphalan Interleukin-2 | T cell therapies | NCT04812470 | Recruiting |
| Tebentafusp | ImmTAC molecule | NCT05315258 | Recruiting |
| Vaccination with IKKb-Matured Dendritic Cells | IKKb-matured dendritic cells | NCT04335890 | Active, not recruiting |
| Nivolumab + Relatimab | Anti-PD-1 + anti-LAG-3 | NCT03743766 | Active, not recruiting |
| C7R-GD2 ART Cells Cyclophosphamide Fludarabine | T cell therapies | NCT03635632 | Active, not recruiting |
| Tumor-Infiltrating Lymphocytes (TIL) | T cell therapies | NCT03467516 | Recruiting |
| Aldesleukin Autologous CD8+ SLC45A2-specific T Lymphocytes Cyclophosphamide Ipilimumab | T cell therapies + Anti-CTLA-4 | NCT03068624 | Active, not recruiting |
| Ipilimumab Nivolumab Melphalanchemosaturation via Percutaneous Hepatic Perfusion | anti-CTLA-4 + anti-PD-1 | NCT04283890 | Recruiting |
| Nivolumab | anti-PD-1 | NCT03025256 | Active, not recruiting |
| Isolated Hepatic Perfusion Ipilimumab Nivolumab | anti-CTLA-4 + anti-PD-1 | NCT04463368 | Active, not recruiting |
| Nivolumab Relatlimab | anti-PD-1 + anti-LAG-3 | NCT04552223 | Active, not recruiting |
| Autologous Dendritic Cells Loaded with Autologous Tumor RNA | T cell therapies | NCT01983748 | Active, not recruiting |
| Stereotactic Body Radiotherapy Ipilimumab Nivolumab | radiation anti-CTLA-4 + anti-PD-1 | NCT05077280 | Recruiting |
| NovacureOptune Opdivo Yervoy | anti-PD-1 + anti-CTLA-4 | NCT05004025 | Recruiting |
| FHD-286 | BRG1 and BRM enzymatic inhibitor | NCT04879017 | Active, not recruiting |
| Binimetinib Belinostat | MEK inhibitor + histone deacetylase inhibitors | NCT05170334 | Recruiting |
| SD-101 Nivolumab Ipilimumab | agonist of toll-like receptor 9 (TLR9) + anti-PD-1 + anti-CTLA-4 | NCT04935229 | Recruiting |
| Hypofractionated Linearaccelerator Radiotherapy | radiation | NCT00872391 | Recruiting |
| Endoresection Laser Diode | N/A procedure | NCT02874040 | Active, not recruiting |
| Carmustine Ethiodized Oil Transarterial Chemoembolization Medical Device Usage and Evaluation | procedure | NCT04728633 | Recruiting |
| Sunitinib Valproic Acid | tyrosine kinase inhibitor has an effect on the production of GABA | NCT02068586 | Active, not recruiting |
| AU-011 Suprachoroidal Microinjector PDT Laser | photodynamic therapy | NCT04417530 | Active, not recruiting |
| 6MHP NeoAg-mBRAF PolyICLC CDX-1140 | peptides to stimulate helper T cells/biologic vaccines anti-CD40 | NCT04364230 | Active, not recruiting |
| IDE196 Binimetinib Crizotinib | PKC inhibitor + MEK inhibitor + ALK and ROS1 inhibitor | NCT03947385 | Recruiting |
| IOA-244 Avelumab Pemetrexed Cisplatin Ruxolitinib | inhibitor of PI3Kδ + anti-PD-L1 + kinase inhibitors | NCT04328844 | Active, not recruiting |
| APG-115 + Pembrolizumab | binding of the protein inhibitor to MDM2 + anti-PD-1 | NCT03611868 | Recruiting |
| Spartalizumab | anti-PD-1, PCD-1 | NCT04802876 | Recruiting |
| PV-10 (10% Rosebengaldisodium) | T cell therapies | NCT00986661 | Active, not recruiting |
| LVGN3616 + LVGN6051 + Nab-Paclitaxel LVGN3616 + LVGN6051 + Bevacizumab + Cyclophosphamide LVGN3616 + LVGN6051 + LVGN7409 + Nab-Paclitaxel LVGN3616 + LVGN6051 + LVGN7409 + Bevacizumab + Cyclophosphamide | anti-PD-L1/PD-1 anti-VEGF-A | NCT05075993 | Recruiting |
| KZR-261 | protein secretion inhibitor | NCT05047536 | Recruiting |
| SEA-CD40 Pembrolizumab Pemetrexed Carboplatin | anti-CD-40 + anti-PD-1 | NCT04993677 | Active, not recruiting |
| Adoptive Therapy with Autologous MC2 TCR T cells | T cell therapies | NCT04729543 | Recruiting |
| IN10018 Cobimetinib | FAK inhibitor + MEK inhibitor | NCT04109456 | Recruiting |
| Defactinib VS-6766 | focal adhesion kinase inhibitor + RAF/MEK inhibitor | NCT04720417 | Active, not recruiting |
| Pembrolizumab Stereotactic Radiosurgery | anti-PD-1 + radiation | NCT02858869 | Active, not recruiting |
| Device: Sir-Spheres® | radioactive element | NCT01473004 | Active, not recruiting |
| Vorinostat | histone deacetylase inhibitor | NCT01587352 | Active, not recruiting |
| Melphalan/HDS | cytostatic | NCT02678572 | Active, not recruiting |
| Vaccination with IKKb-Matured Dendritic Cells | IKKb-matured dendritic cells | NCT04335890 | Active, not recruiting |
| Treatment Approach | Mechanism of Action |
|---|---|
| Current therapy | Ionizing radiation, surgical treatment |
| Bispecific antibodies | Immune response, engagement of immune cells |
| Monoclonal T cell receptors | T cell receptor cytotoxicity |
| Bispecific antibodies that include bispecific T cell engagers (BiTEs) Bifunctional checkpoint inhibitory T cell engagers (CiTEs) | T cell receptor cytotoxicity and inhibition of the PD–1/PD–L1 axis |
| Immune checkpoint inhibitors | Immune checkpoint inhibitory function by inhibiting molecules like programmed cell death 1 (PD-1), programmed cell death 1 ligand 1, and cytotoxic T lymphocyte antigen 4 (CTLA-4) |
| Cell-based therapies | Chimeric antigen receptor (CAR)-modified T cells (CAR-T) and T cell receptor (TCR)-engineered T cells (TCRS)—T cell receptor cytotoxicity NK cell activation |
| Theranostic | Visualization of changed area and anticancer activity by substances with known mechanism of action, photosensitization, enzymatic activity, chemotherapeutic agent release, radioactive particle decay |
| Nanomedicine | Different drug or miRNA delivery systems: organic, inorganic, topical application of anticancer agents, photosensitization |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Synoradzki, K.J.; Paduszyńska, N.; Solnik, M.; Toro, M.D.; Bilmin, K.; Bylina, E.; Rutkowski, P.; Yousef, Y.A.; Bucolo, C.; Zweifel, S.A.; et al. From Molecular Biology to Novel Immunotherapies and Nanomedicine in Uveal Melanoma. Curr. Oncol. 2024, 31, 778-800. https://doi.org/10.3390/curroncol31020058
Synoradzki KJ, Paduszyńska N, Solnik M, Toro MD, Bilmin K, Bylina E, Rutkowski P, Yousef YA, Bucolo C, Zweifel SA, et al. From Molecular Biology to Novel Immunotherapies and Nanomedicine in Uveal Melanoma. Current Oncology. 2024; 31(2):778-800. https://doi.org/10.3390/curroncol31020058
Chicago/Turabian StyleSynoradzki, Kamil J., Natalia Paduszyńska, Malgorzata Solnik, Mario Damiano Toro, Krzysztof Bilmin, Elżbieta Bylina, Piotr Rutkowski, Yacoub A. Yousef, Claudio Bucolo, Sandrine Anne Zweifel, and et al. 2024. "From Molecular Biology to Novel Immunotherapies and Nanomedicine in Uveal Melanoma" Current Oncology 31, no. 2: 778-800. https://doi.org/10.3390/curroncol31020058
APA StyleSynoradzki, K. J., Paduszyńska, N., Solnik, M., Toro, M. D., Bilmin, K., Bylina, E., Rutkowski, P., Yousef, Y. A., Bucolo, C., Zweifel, S. A., Reibaldi, M., Fiedorowicz, M., & Czarnecka, A. M. (2024). From Molecular Biology to Novel Immunotherapies and Nanomedicine in Uveal Melanoma. Current Oncology, 31(2), 778-800. https://doi.org/10.3390/curroncol31020058









