Are Extensive Open Lung Resections for Elderly Patients with Lung Cancer Justified?
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection and Perioperative Assessment
2.2. Preoperative Comorbidities
2.3. Postoperative Characteristics and Definition of Complications
2.4. Groups of Patients
2.5. Inclusion and Exclusion Criteria
- -
- Patients aged < 18 years;
- -
- Palliative surgical resections;
- -
- Patients operated with thoracoscopy (VATS-video-assisted thoracic surgery) or other surgical access from thoracotomy;
- -
- NSCLC resected by wedge resection;
- -
- Patients with preoperative diagnosed pleural carcinomatosis or carcinomatous lymphangiosis.
2.6. Data Collection and Statistical Analysis
2.7. Demographic Data
2.8. Preoperative Results
2.9. Mortality and Follow-Up
3. Results
3.1. Surgical Data
3.2. Tumor Histology and Differentiation
3.3. Tumor Staging
3.4. Mediastinal Lymph Node Staging
3.5. Postoperative Morbidity and Hospital Stay
3.6. Postoperative Mortality
3.7. Univariate Analysis
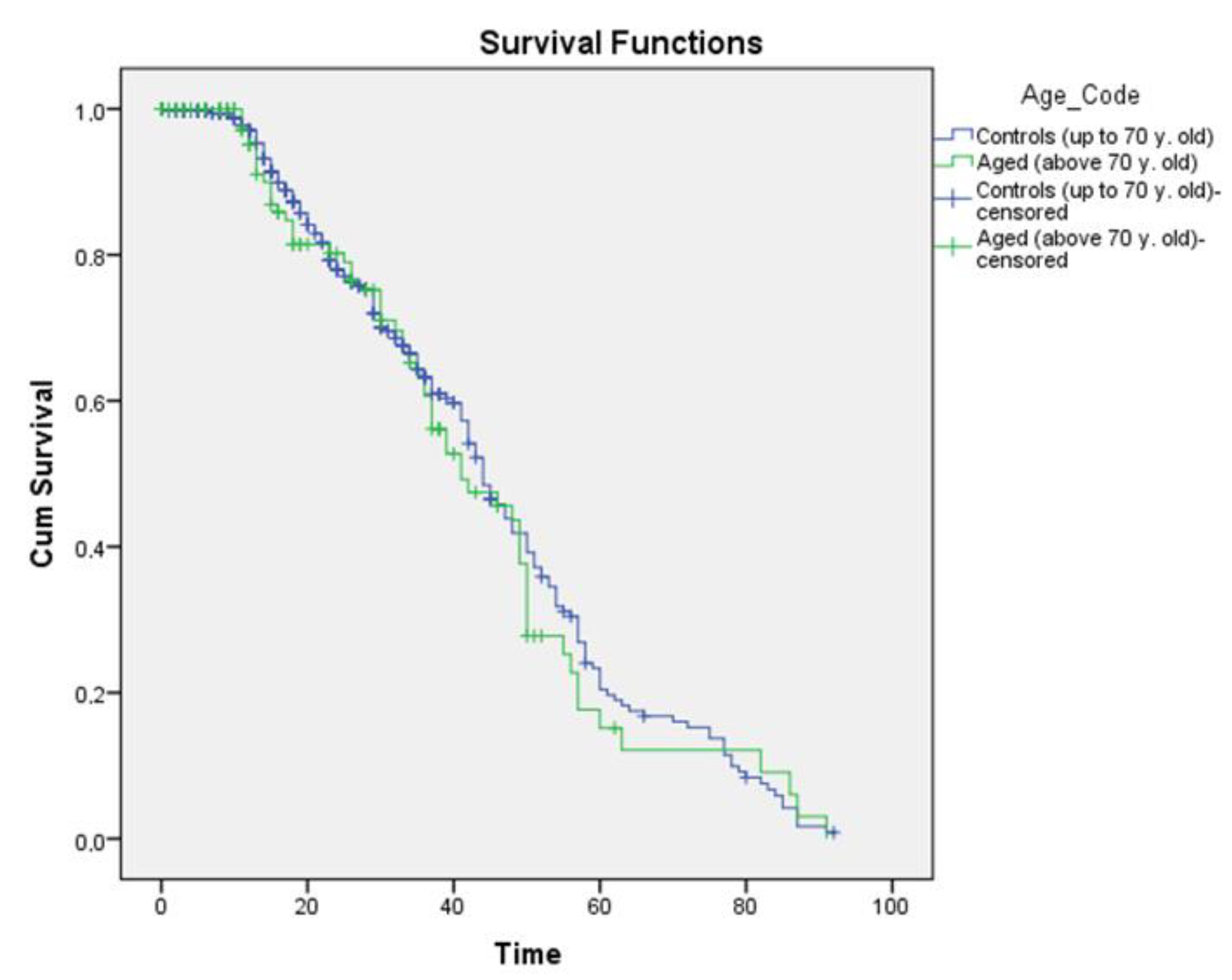
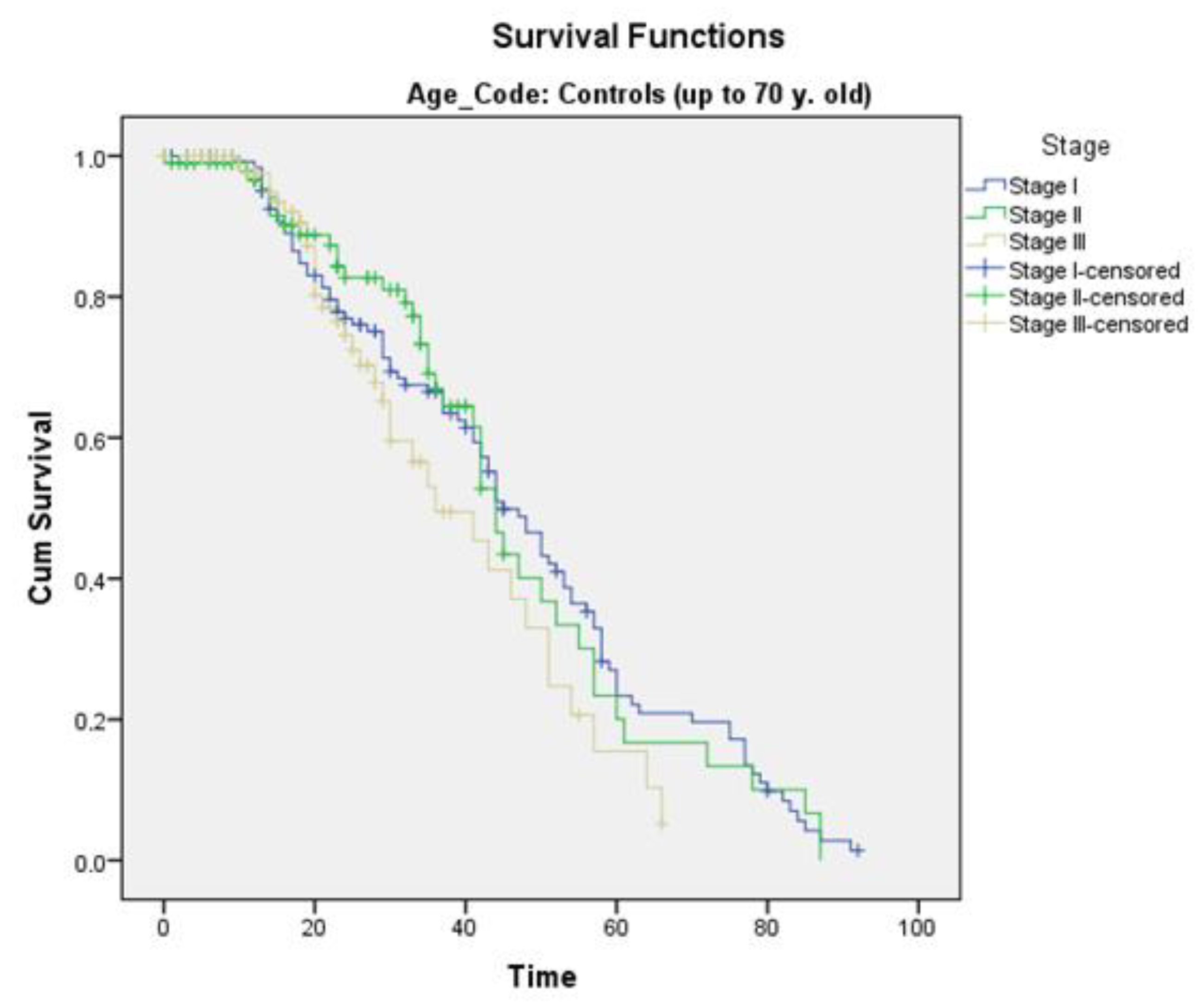
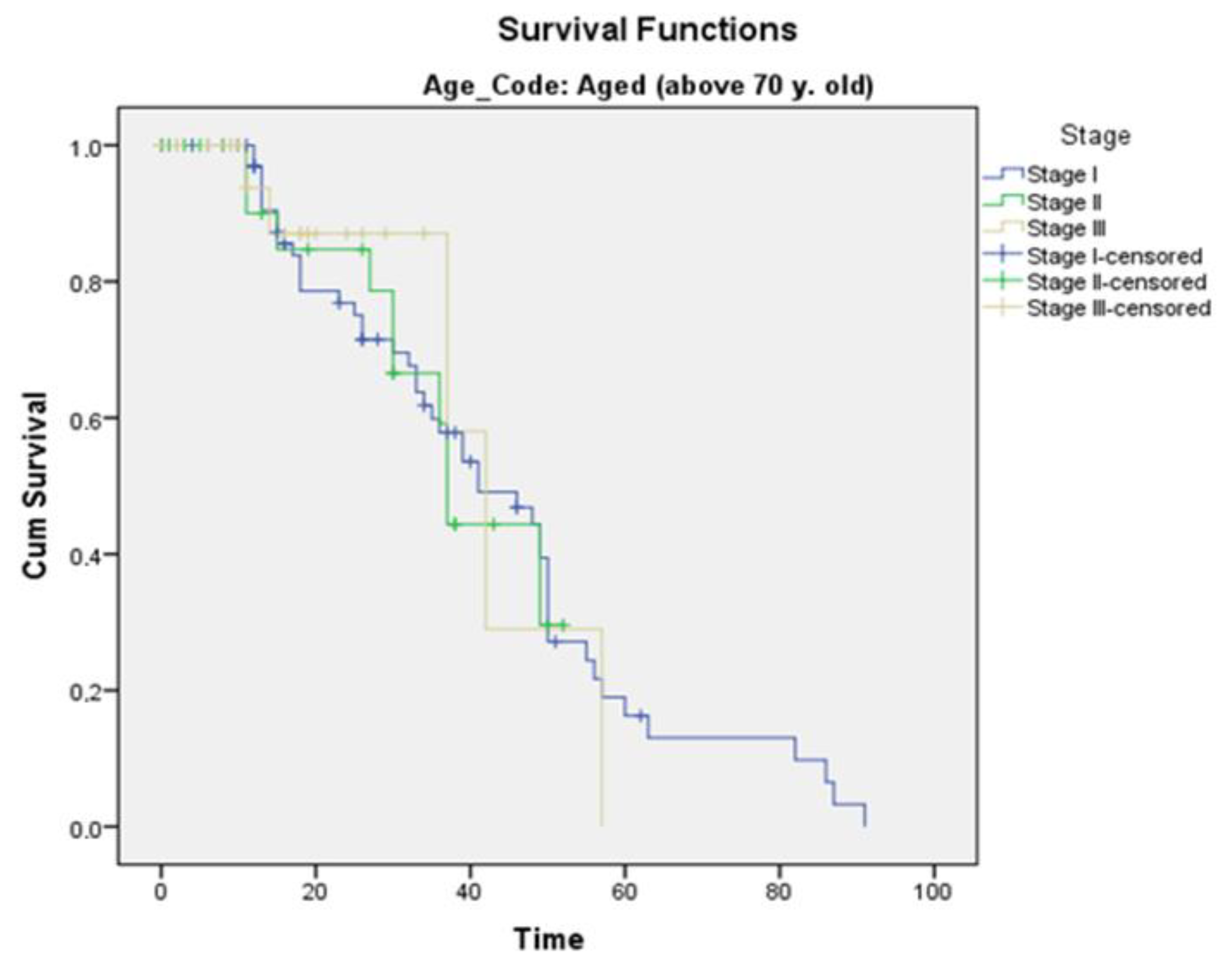
3.8. Survival
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organisation (WHO). World Health Statistics Annual; WHO: Geneva, Switzerland, 1987. [Google Scholar]
- Jemal, A.; Siegel, R.; Ward, E.; Murray, T.; Xu, J.; Thun, M.J. Cancer statistics, 2007. CA Cancer J. Clin. 2007, 57, 43–66. [Google Scholar] [CrossRef]
- Yancik, R.; Ries, A.L. Aging and cancer in America. Demographic and epidemiologic perspectives. Hematol. Oncol. Clin. N. Am. 2000, 14, 17–23. [Google Scholar] [CrossRef]
- Hassan, M.; Ehle, B.; Passlick, B.; Grapatsas, K. Lung Resections for Elderly Patients with Lung Metastases: A Comparative Study of the Postoperative Complications and Overall Survival. Curr. Oncol. 2022, 29, 357. [Google Scholar] [CrossRef]
- Grapatsas, K.; Hassan, M.; Semmelmann, A.; Ehle, B.; Passlick, B.; Schmid, S.; Le, U.-T. Should cardiovascular comorbidities be a contraindication for pulmonary metastasectomy? J. Thorac. Dis. 2022, 14, 4266–4275. [Google Scholar] [CrossRef]
- Hassan, M.; Ehle, B.; Le, U.-T.; Titze, L.; Passlick, B.; Grapatsas, K. Outcome of Repeated Resection of Pulmonary Metastases for Renal Cell Cancer. Thorac. Cardiovasc. Surg. 2022, 71, 130–137. [Google Scholar] [CrossRef]
- Grapatsas, K.; Papaporfyriou, A.; Leivaditis, V.; Ehle, B.; Galanis, M. Lung Metastatectomy: Can Laser-Assisted Surgery Make a Difference? Curr. Oncol. 2022, 29, 548. [Google Scholar] [CrossRef]
- Brunelli, A.; Charloux, A.; Bolliger, C.T.; Rocco, G.; Sculier, J.P.; Varela, G.; Licker, M.; Ferguson, M.K.; Faivre-Finn, C.; Huber, R.M.; et al. ERS/ESTS clinical guidelines on fitness for radical therapy in lung cancer patients (surgery and chemo-radiotherapy). Eur. Respir. J. 2009, 34, 17–41. [Google Scholar] [CrossRef]
- Fleisher, L.A.; Beckman, J.A.; Brown, K.A.; Calkins, H.; Chaikof, E.; Fleischmann, K.E.; Freeman, W.K.; Froehlich, J.B.; Kasper, E.K.; Kersten, J.R.; et al. ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery): Developed in collaboration with the American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Rhythm Society, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions. Circulation 2007, 116, 1971–1996. [Google Scholar]
- Mirza, S.; Clay, R.D.; Koslow, M.A.; Scanlon, P.D. COPD Guidelines: A Review of the 2018 GOLD Report. Mayo Clin. Proc. 2018, 93, 1488–1502. [Google Scholar] [CrossRef]
- Jammer, I.; Wickboldt, N.; Sander, M.; Smith, A.; Schultz, M.J.; Pelosi, P.; Leva, B.; Rhodes, A.; Hoeft, A.; Walder, B.; et al. Standards for definitions and use of outcome measures for clinical effectiveness research in perioperative medicine: European Perioperative Clinical Outcome (EPCO) definitions: A statement from the ESA-ESICM joint taskforce on perioperative outcome measures. Eur. J. Anaesthesiol. 2015, 32, 88–105. [Google Scholar]
- Seely, A.J.; Ivanovic, J.; Threader, J.; Al-Hussaini, A.; Al-Shehab, D.; Ramsay, T.; Gilbert, S.; Maziak, D.E.; Shamji, F.M.; Sundaresan, R.S. Systematic Classification of Morbidity and Mortality After Thoracic Surgery. Ann. Thorac. Surg. 2010, 90, 936–942; discussion 942. [Google Scholar] [CrossRef]
- Benker, M.; Citak, N.; Neuer, T.; Opitz, I.; Inci, I. Impact of preoperative comorbidities on postoperative complication rate and outcome in surgically resected non-small cell lung cancer patients. Gen. Thorac. Cardiovasc. Surg. 2021, 70, 248–256. [Google Scholar] [CrossRef]
- Balducci, L. Geriatric oncology: Challenges for the new century. Eur. J. Cancer 2000, 36, 1741–1754. [Google Scholar] [CrossRef]
- Scott, W.J.; Howington, J.; Movsas, B. Treatment of Stage II Non-small Cell Lung Cancer. Chest 2003, 123 (Suppl. S1), 188S–201S. [Google Scholar] [CrossRef]
- Weiss, W. Operative mortality and five year survival rates in patients with bronchogenic carcinoma. Am. J. Surg. 1974, 128, 799–804. [Google Scholar] [CrossRef]
- Dexter, E.U.; Jahangir, N.; Kohman, L.J. Resection for lung cancer in the elderly patient. Thorac. Surg. Clin. 2004, 14, 163–171. [Google Scholar] [CrossRef]
- Massard, G.; Moog, R.; Wihlm, J.M.; Kessler, R.; Dabbagh, A.; Lesage, A.; Roeslin, N.; Morand, G. Bronchogenic Cancer in the Elderly: Operative Risk and Long-Term Prognosis. Thorac. Cardiovasc. Surg. 1996, 44, 40–45. [Google Scholar] [CrossRef]
- DeMaria, J.L.C.; Cohen, H.J. Characteristics of Lung Cancer in Elderly Patients. J. Gerontol. 1987, 42, 540–545. [Google Scholar] [CrossRef]
- Riquet, M.; Medioni, J.; Manac’H, D.; Dujon, A.; Souilamas, R.; Barthes, F.L.P.; Hubsch, J.P. Non-small cell lung cancer: Surgical trends as a function of age. Rev. Mal. Respir. 2001, 18, 173–184. [Google Scholar]
- Teeter, S.M.; Holmes, F.F.; McFarlane, M.J. Lung carcinoma in the elderly population. Influence of histology on the inverse relationship of stage to age. Cancer 1987, 60, 1331–1336. [Google Scholar] [CrossRef]
- Mizushima, Y.; Noto, H.; Sugiyama, S.; Kusajima, Y.; Yamashita, R.; Kashii, T.; Kobayashi, M. Survival and Prognosis After Pneumonectomy for Lung Cancer in the Elderly. Ann. Thorac. Surg. 1997, 64, 193–198. [Google Scholar] [CrossRef]
- Ginsberg, R.J.; Hill, L.D.; Eagan, R.T.; Thomas, P.; Mountain, C.F.; DesLauriers, J.; Fry, W.A.; Butz, R.O.; Goldberg, M.; Waters, P.F. Modern thirty-day operative mortality for surgical resections in lung cancer. J. Thorac. Cardiovasc. Surg. 1983, 86, 654–658. [Google Scholar] [CrossRef]
- Romano, P.S.; Mark, D.H. Patient and hospital characteristics related to in-hospital mortality after lung cancer resection. Chest 1992, 101, 1332–1337. [Google Scholar] [CrossRef]
- Thomas, P.; Sielezneff, I.; Ragni, J.; Guidicelli, R.; Fuentes, P. Is lung cancer resection justified in patients aged beyond 70 years? Eur. J. Cardiothorac. Surg. 1993, 7, 246–251. [Google Scholar] [CrossRef]
- Damhuis, R.A.; Schutte, P.R. Resection rates and postoperative mortality in 7899 patients with lung cancer. Eur. Respir. J. 1996, 9, 7–10. [Google Scholar] [CrossRef]
- Ishida, T.; Yokoyama, H.; Kaneko, S.; Sugio, K.; Sugimachi, K. Long-term results of operation for non-small cell lung cancer in the elderly. Ann. Thorac. Surg. 1990, 50, 919–922. [Google Scholar] [CrossRef]
- Breyer, R.H.; Zippe, C.; Pharr, W.F.; Jensik, R.J.; Kittle, C.F.; Faber, L.P. Thoracotomy in patients over age seventy years: Ten-year experience. J. Thorac. Cardiovasc. Surg. 1981, 81, 187–193. [Google Scholar] [CrossRef]
- Harvey, J.C.; Ba, C.E.; Pisch, J.; Beattie, E.J. Surgical treatment of non-small cell lung cancer in patients older than seventy years. J. Surg. Oncol. 1995, 60, 247–249. [Google Scholar] [CrossRef]
- Whittle, J.; Steinberg, E.P.; Anderson, G.F.; Herbert, R. Use of Medicare Claims Data to Evaluate Outcomes in Elderly Patients Undergoing Lung Resection for Lung Cancer. Chest 1991, 100, 729–734. [Google Scholar] [CrossRef]
- Morandi, U.; Stefani, A.; Golinelli, M.; Ruggiero, C.; Brandi, L.; Chiapponi, A.; Santi, C.; Lodi, R. Results of surgical resection in patients over the age of 70 years with non small-cell lung cancer. Eur. J. Cardio-Thorac. Surg. 1997, 11, 432–439. [Google Scholar] [CrossRef]
- Au, J.; El-Oakley, R.; Cameron, E.W. Pneumonectomy for bronchogenic carcinoma in the elderly. Eur. J. Cardio-Thorac. Surg. 1994, 8, 247–250. [Google Scholar] [CrossRef]
- Thomas, P.; Piraux, M.; Jacques, L.F.; Grégoire, J.; Bédard, P.; DesLauriers, J. Clinical patterns and trends of outcome of elderly patients with bronchogenic carcinoma. Eur. J. Cardio-Thorac. Surg. 1998, 13, 266–274. [Google Scholar] [CrossRef]
- Roxburgh, J.; Thompson, J.; Goldstraw, P. Hospital mortality and long-term survival after pulmonary resection in the elderly. Ann. Thorac. Surg. 1991, 51, 800–803. [Google Scholar] [CrossRef]
- Miller, J.I.; Hatcher, C.R. Limited Resection of Bronchogenic Carcinoma in the Patient with Marked Impairment of Pulmonary Function. Ann. Thorac. Surg. 1987, 44, 340–343. [Google Scholar] [CrossRef]
- Ginsberg, R.J.; Rubinstein, L.V. Randomized trial of lobectomy versus limited resection for T1 N0 non-small cell lung cancer. Ann. Thorac. Surg. 1995, 60, 615–623. [Google Scholar] [CrossRef]
- Vogt-Moykopf, I.; Toomes, H.; Heinrich, S. Sleeve Resection of the Bronchus and Pulmonary Artery for Pulmonary Lesions. Thorac. Cardiovasc. Surg. 1983, 31, 193–198. [Google Scholar] [CrossRef]
- Shiono, S.; Abiko, M.; Sato, T. Postoperative complications in elderly patients after lung cancer surgery. Interact. Cardiovasc. Thorac. Surg. 2013, 16, 819–823. [Google Scholar] [CrossRef]
- Simonsen, D.F.; Søgaard, M.; Bozi, I.; Horsburgh, C.; Thomsen, R. Risk factors for postoperative pneumonia after lung cancer surgery and impact of pneumonia on survival. Respir. Med. 2015, 109, 1340–1346. [Google Scholar] [CrossRef]
- Wang, P.; Zhu, M.; Zhang, D.; Guo, X.; Zhao, S.; Zhang, X.; Wang, D.; Liu, C. The relationship between chronic obstructive pulmonary disease and non-small cell lung cancer in the elderly. Cancer Med. 2019, 8, 4124–4134. [Google Scholar] [CrossRef]
- Strickland, S.L.; Rubin, B.K.; Drescher, G.S.; Haas, C.F.; O’Malley, C.A.; Volsko, T.A.; Branson, R.D.; Hess, D.R. AARC Clinical Practice Guideline: Effectiveness of Nonpharmacologic Airway Clearance Therapies in Hospitalized Patients. Respir. Care 2013, 58, 2187–2193. [Google Scholar] [CrossRef]
- Agostini, P.; Naidu, B.; Cieslik, H.; Steyn, R.; Rajesh, P.B.; Bishay, E.; Kalkat, M.S.; Singh, S. Effectiveness of incentive spirometry in patients following thoracotomy and lung resection including those at high risk for developing pulmonary complications. Thorax 2013, 68, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Yamada, Y.; Koh, E.; Yoshino, I.; Sekine, Y. Severity of Chronic Obstructive Pulmonary Disease and Its Relationship to Lung Cancer Prognosis after Surgical Resection. Thorac. Cardiovasc. Surg. 2012, 61, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Zhu, J.; Li, L.; Li, D.; Du, R. The Prognostic Role of Chronic Obstructive Pulmonary Disease for Lung Cancer After Pulmonary Resection. J. Surg. Res. 2022, 275, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Riquet, M.; Manac’h, D.; Le Pimpec-Barthes, F.; Debrosse, D.; Dujon, A.; Saab, M.; Debesse, B. Operation for lung cancer in the elderly. What about octogenarians? Ann. Thorac. Surg. 1994, 58, 916. [Google Scholar] [CrossRef] [PubMed]
- Okami, J.; Higashiyama, M.; Asamura, H.; Goya, T.; Koshiishi, Y.; Sohara, Y.; Eguchi, K.; Mori, M.; Nakanishi, Y.; Tsuchiya, R.; et al. Pulmonary Resection in Patients Aged 80 Years or Over with Clinical Stage I Non-small Cell Lung Cancer: Prognostic Factors for Overall Survival and Risk Factors for Postoperative Complications. J. Thorac. Oncol. 2009, 4, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Jaklitsch, M.T.; Mery, C.M.; Audisio, R.A. The use of surgery to treat lung cancer in elderly patients. Lancet Oncol. 2003, 4, 463–471. [Google Scholar] [CrossRef]
- Suemitsu, R.; Yamaguchi, M.; Takeo, S.; Ondo, K.; Ueda, H.; Yoshino, I.; Maehara, Y. Favorable surgical results for patients with nonsmall cell lung cancer over 80 years old: A multicenter survey. Ann. Thorac. Cardiovasc. Surg. 2008, 14, 154–160. [Google Scholar]
| Variable | Control Group | Elderly Group | p-Value |
|---|---|---|---|
| n (%) | n (%) | ||
| Total (n) | n = 375 | n = 135 | |
| Sex male/female | 339/36 | 122/13 | |
| Hypertension | 101 (26.9%) | 57 (42.2%) | 0.001 |
| COPD | 67 (17.9%) | 33 (24.4%) | 0.102 |
| Diabetes mellitus | 43 (11.5%) | 15 (11.1%) | 0.526 |
| IHD-CAD | 36 (9.6%) | 21 (15.6%) | 0.45 |
| AMI | 5 (1.3%) | 1 (0.7%) | 0.499 |
| Atrial fibrillation | 14 (3.7%) | 14 (10.4%) | 0.005 |
| Brain vascular disease | 7 (1.9%) | 6 (4.4%) | 0.999 |
| TBC | 7 (1.9%) | 4 (3.0%) | 0.327 |
| Malignancy other | 26 (6.9%) | 8 (5.9%) | 0.431 |
| Systemic autoimmune diseases | 6 (1.6%) | 3 (2.2%) | 0.44 |
| Preop. chemo-radiotherapy | 26 (6.9%) | 5 (3.7%) | 0.212 |
| Total number of comorbidities | 53.30% | 74.10% | 0.001 |
| Variable | Elderly (n Patients, %) | Control (n Patients, %) | p Value |
|---|---|---|---|
| Resektion Type | p = 0.121 | ||
| Pneumonectomy | 36 (26.7%) | 141 (37.6%) | |
| Bilobectomy | 8 (5.9%) | 25 (6.7%) | |
| Lobectomy | 86 (63.7%) | 198 (52.8%) | |
| Segmentectomy | 5 (3.7%) | 11 (2.9%) | |
| Tumor differentiation | p = 0.014 | ||
| High | 17 (12.6%) | 24 (6.4%) | |
| Middle | 67 (49.6%) | 160 (42.7%) | |
| Low | 40 (29.6%) | 163 (43.5%) | |
| Undifferent | 11 (8.1%) | 28 (7.5%) | |
| Tumor histology | p = 0.037 | ||
| SCC | 80 (59.3%) | 193 (51.5%) | |
| Adenocarcinoma | 34 (25.2%) | 139 (37.1%) | |
| Large cell carcinoma | 14 (10.4%) | 35 (9.3%) | |
| other | 7 (5.2%) | 8 (2.1%) | |
| Stage | p = 0.002 | ||
| I | 75 (55.6%) | 136 (36.6%) | |
| II | 30 (22.2%) | 112 (29.9%) | |
| III | 26 (19.3%) | 109 (29.1%) | |
| IV | 4 (3.0%) | 18 (4.8%) | |
| N-Status | p = 0.002 | ||
| N0–1 | 122 (90.4%) | 294 (78.4%) | |
| N2 | 13 (9.6%) | 81 (21.6%) | |
| 30-day mortality | 7 (5.2%) | 10 (2.7%) | p = 0.168 |
| Survival | p = 0.57 | ||
| 3-y-s | 46% | 48% | |
| 5-y-s | 20.4% | 25% | |
| Mean (months) | 43.469 | 45.298 | |
| Survival pro stage (mean survival in months) | |||
| Stage I | 43.317 | 43.317 | p = 0.5 |
| Stage II | 46.442 | 41.089 | p = 0.6 |
| Stage III | 39.209 | 39.912 | p = 0.6 |
| Complication | Control | Elderly | p-Value |
|---|---|---|---|
| (n Patients, %) | (n Patients, %) | ||
| AMI | 1 (0.3%) | 1 (0.7%) | 0.46 |
| ICU-admission/Re-intubation | 13 (3.5%) | 5 (3.7%) | 1 |
| ARDS | 5 (1.3%) | 2 (1.5%) | 1 |
| Re-operation for Hemorrhage | 13 (3.5%) | 6 (4.4%) | 0.601 |
| Re-operation (other reasons) | 20 (5.3%) | 0 (0%) | 0.003 |
| Acute renal failure | 2 (0.5%) | 3 (2.2%) | 0.119 |
| Empyema | 12 (3.2%) | 0 (0%) | 0.042 |
| Bronchopleural fistula | 3 (0.8%) | 0 (0%) | 0.569 |
| TIA-CVA | 3 (0.8%) | 0 (0%) | 0.569 |
| MOF | 2 (0.5%) | 1 (0.7%) | 1 |
| Pneumonia | 3 (0.8%) | 5 (3.7%) | 0.034 |
| Atelectasis | 11 (2.9%) | 10 (7.4%) | 0.04 |
| Atrial Fibrillation | 21 (5.6%) | 12 (8.9%) | 0.22 |
| Prolonged air leak | 29 (7.7%) | 12 (8.9%) | 0.712 |
| Type of Operation/ Complications | Elderly (n, %) | Control (n, %) | p Value |
|---|---|---|---|
| Pneumonectomy | 9 (25%) | 37 (26.2%) | 0.5 |
| Bilobectomy | 4 (50%) | 5 (20%) | 0.6 |
| Lobectomy | 24 (27,9%) | 52 (26.3%) | 0.6 |
| Segmentectomy | 1 (20%) | 5 (45.5%) | 0.2 |
| Total number of complications | 38 (28.1%) | 99 (26.4%) | 0.2 |
| Variable | Pneumonectomy (n, %) | Lobectomy (n, %) | p-Value |
|---|---|---|---|
| 30-day mortality/No. Cases (%) | 1 (2.9%) | 4 (4.7%) | 0.07 |
| Major complications | 1 (2.8%) | 5 (5.8%) | 0.651 |
| Minor complications | 7/36 (19.4%) | 19/86 (22.1%) | 0.160 |
| Parameter | Hazard Ratio (95% CI) | |
|---|---|---|
| Age < 70 years old | Preoperative chemo-/ radio-therapy | 0.350 (0.172–0.715) |
| COPD | 1.661 (1.131–2.438) | |
| Age > 70 years old | COPD | 1.756 (1.004–3.073) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panagopoulos, N.; Grapatsas, K.; Leivaditis, V.; Galanis, M.; Dougenis, D. Are Extensive Open Lung Resections for Elderly Patients with Lung Cancer Justified? Curr. Oncol. 2023, 30, 5470-5484. https://doi.org/10.3390/curroncol30060414
Panagopoulos N, Grapatsas K, Leivaditis V, Galanis M, Dougenis D. Are Extensive Open Lung Resections for Elderly Patients with Lung Cancer Justified? Current Oncology. 2023; 30(6):5470-5484. https://doi.org/10.3390/curroncol30060414
Chicago/Turabian StylePanagopoulos, Nikolaos, Konstantinos Grapatsas, Vasileios Leivaditis, Michail Galanis, and Dimitrios Dougenis. 2023. "Are Extensive Open Lung Resections for Elderly Patients with Lung Cancer Justified?" Current Oncology 30, no. 6: 5470-5484. https://doi.org/10.3390/curroncol30060414
APA StylePanagopoulos, N., Grapatsas, K., Leivaditis, V., Galanis, M., & Dougenis, D. (2023). Are Extensive Open Lung Resections for Elderly Patients with Lung Cancer Justified? Current Oncology, 30(6), 5470-5484. https://doi.org/10.3390/curroncol30060414





