Dosimetric Predictors of Toxicity after Prostate Stereotactic Body Radiotherapy: A Single-Institutional Experience of 145 Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Treatment Protocol
2.3. Adverse Events and Risk Factors
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cox, J.D.; Stetz, J.; Pajak, T.F. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC). Int. J. Radiat. Oncol. Biol. Phys. 1995, 31, 1341–1346. [Google Scholar] [CrossRef] [PubMed]
- Youden, W.J. Index for rating diagnostic tests. Cancer 1950, 3, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Jackson, W.C.; Silva, J.; Hartman, H.E.; Dess, R.T.; Kishan, A.U.; Beeler, W.H.; Gharzai, L.A.; Jaworski, E.M.; Mehra, R.; Hearn, J.W.D.; et al. Stereotactic Body Radiation Therapy for Localized Prostate Cancer: A Systematic Review and Meta-Analysis of Over 6000 Patients Treated on Prospective Studies. Int. J. Radiat. Oncol. Biol. Phys. 2019, 104, 778–789. [Google Scholar] [CrossRef] [PubMed]
- Kishan, A.U.; Dang, A.; Katz, A.J.; Mantz, C.A.; Collins, S.P.; Aghdam, N.; Chu, F.I.; Kaplan, I.D.; Appelbaum, L.; Fuller, D.B.; et al. Long-term Outcomes of Stereotactic Body Radiotherapy for Low-Risk and Intermediate-Risk Prostate Cancer. JAMA Netw. Open 2019, 2, e188006. [Google Scholar] [CrossRef] [PubMed]
- Vargas, C.E.; Schmidt, M.Q.; Niska, J.R.; Hartsell, W.F.; Keole, S.R.; Doh, L.; Chang, J.H.; Sinesi, C.; Rodriquez, R.; Pankuch, M.; et al. Initial toxicity, quality-of-life outcomes, and dosimetric impact in a randomized phase 3 trial of hypofractionated versus standard fractionated proton therapy for low-risk prostate cancer. Adv. Radiat. Oncol. 2018, 3, 322–330. [Google Scholar] [CrossRef]
- Dincer, S.; Uysal, E.; Berber, T.; Akboru, M.H. The efficacy and tolerability of ultra-hypofractionated radiotherapy in low-intermediate risk prostate cancer patients: Single center experience. Aging Male 2021, 24, 50–57. [Google Scholar] [CrossRef]
- Alayed, Y.; Davidson, M.; Quon, H.; Cheung, P.; Chu, W.; Chung, H.T.; Vesprini, D.; Ong, A.; Chowdhury, A.; Liu, S.K.; et al. Dosimetric predictors of toxicity and quality of life following prostate stereotactic ablative radiotherapy. Radiother. Oncol. 2020, 144, 135–140. [Google Scholar] [CrossRef]
- Henderson, D.R.; Murray, J.R.; Gulliford, S.L.; Tree, A.C.; Harrington, K.J.; Van As, N.J. An Investigation of Dosimetric Correlates of Acute Toxicity in Prostate Stereotactic Body Radiotherapy: Dose to Urinary Trigone is Associated with Acute Urinary Toxicity. Clin. Oncol. 2018, 30, 539–547. [Google Scholar] [CrossRef]
- Wang, K.; Chen, R.C.; Kane, B.L.; Medbery, C.A.; Underhill, K.J.; Gray, J.R.; Peddada, A.V.; Fuller, D.B. Patient and Dosimetric Predictors of Genitourinary and Bowel Quality of Life After Prostate SBRT: Secondary Analysis of a Multi-institutional Trial. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 1430–1437. [Google Scholar] [CrossRef]
- Jackson, W.C.; Dess, R.T.; Litzenberg, D.W.; Li, P.; Schipper, M.; Rosenthal, S.A.; Chang, G.C.; Horwitz, E.M.; Price, R.A.; Michalski, J.M.; et al. A multi-institutional phase 2 trial of prostate stereotactic body radiation therapy (SBRT) using continuous real-time evaluation of prostate motion with patient-reported quality of life. Pract. Radiat. Oncol. 2018, 8, 40–47. [Google Scholar] [CrossRef]
- Helou, J.; D’Alimonte, L.; Quon, H.; Deabreu, A.; Commisso, K.; Cheung, P.; Chu, W.; Mamedov, A.; Davidson, M.; Ravi, A.; et al. Stereotactic ablative radiotherapy in the treatment of low and intermediate risk prostate cancer: Is there an optimal dose? Radiother. Oncol. 2017, 123, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Dess, R.T.; Jackson, W.C.; Suy, S.; Soni, P.D.; Lee, J.Y.; Abugharib, A.E.; Zumsteg, Z.S.; Feng, F.Y.; Hamstra, D.A.; Collins, S.P.; et al. Predictors of multidomain decline in health-related quality of life after stereotactic body radiation therapy (SBRT) for prostate cancer. Cancer 2017, 123, 1635–1642. [Google Scholar] [CrossRef] [PubMed]
- Musunuru, H.B.; Davidson, M.; Cheung, P.; Vesprini, D.; Liu, S.; Chung, H.; Chu, W.; Mamedov, A.; Ravi, A.; D’Alimonte, L.; et al. Predictive Parameters of Symptomatic Hematochezia Following 5-Fraction Gantry-Based SABR in Prostate Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.S.; Wang, J.P.; Gomez, C.L.; Shao, W.; Xu, X.; King, C.; Low, D.A.; Steinberg, M.; Kupelian, P. Plan quality and dosimetric association of patient-reported rectal and urinary toxicities for prostate stereotactic body radiotherapy. Radiother. Oncol. 2016, 121, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Kole, T.P.; Tong, M.; Wu, B.; Lei, S.; Obayomi-Davies, O.; Chen, L.N.; Suy, S.; Dritschilo, A.; Yorke, E.; Collins, S.P. Late urinary toxicity modeling after stereotactic body radiotherapy (SBRT) in the definitive treatment of localized prostate cancer. Acta Oncol. 2016, 55, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Seymour, Z.A.; Chang, A.J.; Zhang, L.; Kirby, N.; Descovich, M.; Roach, M.; Hsu, I.C.; Gottschalk, A.R. Dose-volume analysis and the temporal nature of toxicity with stereotactic body radiation therapy for prostate cancer. Pract. Radiat. Oncol. 2015, 5, e465–e472. [Google Scholar] [CrossRef]
- Glowacki, G.; Majewski, W.; Wojcieszek, P.; Grabinska, K.; Chawinska, E.; Bodusz, D.; Wozniak, G.; Urbanczyk, H.; Kaletka, A.; Miszczyk, L. Acute toxicity of robotic ultrahypofractionated radiotherapy CyberKnifeTM in prostate cancer patients. Neoplasma 2015, 62, 674–682. [Google Scholar] [CrossRef]
- Gurka, M.K.; Chen, L.N.; Bhagat, A.; Moures, R.; Kim, J.S.; Yung, T.; Lei, S.; Collins, B.T.; Krishnan, P.; Suy, S.; et al. Hematuria following stereotactic body radiation therapy (SBRT) for clinically localized prostate cancer. Radiat. Oncol. 2015, 10, 44. [Google Scholar] [CrossRef]
- Gomez, C.L.; Xu, X.; Qi, X.S.; Wang, P.C.; Kupelian, P.; Steinberg, M.; King, C.R. Dosimetric parameters predict short-term quality-of-life outcomes for patients receiving stereotactic body radiation therapy for prostate cancer. Pract. Radiat. Oncol. 2015, 5, 257–262. [Google Scholar] [CrossRef]
- Kim, D.W.; Cho, L.C.; Straka, C.; Christie, A.; Lotan, Y.; Pistenmaa, D.; Kavanagh, B.D.; Nanda, A.; Kueplian, P.; Brindle, J.; et al. Predictors of rectal tolerance observed in a dose-escalated phase 1-2 trial of stereotactic body radiation therapy for prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2014, 89, 509–517. [Google Scholar] [CrossRef]
- Macias, V.A.; Blanco, M.L.; Barrera, I.; Garcia, R. A Phase II Study of Stereotactic Body Radiation Therapy for Low-Intermediate-High-Risk Prostate Cancer Using Helical Tomotherapy: Dose-Volumetric Parameters Predicting Early Toxicity. Front. Oncol. 2014, 4, 336. [Google Scholar] [CrossRef] [PubMed]
- Elias, E.; Helou, J.; Zhang, L.; Cheung, P.; Deabreu, A.; D’Alimonte, L.; Sethukavalan, P.; Mamedov, A.; Cardoso, M.; Loblaw, A. Dosimetric and patient correlates of quality of life after prostate stereotactic ablative radiotherapy. Radiother. Oncol. 2014, 112, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Alayed, Y.; Davidson, M.; Liu, S.; Chu, W.; Tseng, E.; Cheung, P.; Vesprini, D.; Cheung, H.; Morton, G.; Musunuru, H.B.; et al. Evaluating the Tolerability of a Simultaneous Focal Boost to the Gross Tumor in Prostate SABR: A Toxicity and Quality-of-Life Comparison of Two Prospective Trials. Int. J. Radiat. Oncol. Biol. Phys. 2020, 107, 136–142. [Google Scholar] [CrossRef] [PubMed]
| Variables | Values | SD | |
|---|---|---|---|
| Age (years) | 73.0 | 5.8 | |
| iPSA (ng/mL) | 16.13 | 23.3 | |
| ISUP Grade | |||
| 1 | 24 | ||
| 2 | 42 | ||
| 3 | 30 | ||
| 4 | 30 | ||
| 5 | 19 | ||
| T stage | |||
| 1a | 2 | ||
| 1c | 41 | ||
| 2a | 40 | ||
| 2b | 17 | ||
| 2c | 18 | ||
| 3a | 14 | ||
| 3b | 12 | ||
| 4 | 1 | ||
| Risk group | |||
| Low | 24 | ||
| Intermediate | 72 | ||
| High | 49 | ||
| Hormonal therapy | |||
| Yes | 111 | ||
| No | 34 | ||
| BED (Gy, α/β = 1.5) | 202–252 |
| Acute | Late | |||
|---|---|---|---|---|
| Grade 2 | Grade 3 | Grade 2 | Grade 3 | |
| RTOG GU | 14 (9.7%) | 0 (0.0%) | 14 (9.7%) | 2 (1.4%) |
| GI | 7 (4.8%) | 0 (0.0%) | 9 (6.2%) | 2 (1.4%) |
| Miction pain | 2 (1.4%) | 0 (0.0%) | 1 (0.7%) | 0 (0.0%) |
| Frequency | 9 (6.2%) | 0 (0.0%) | 12 (8.3%) | 1 (0.7%) |
| Urine incontinence | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Retention | 4 (2.8%) | 0 (0.0%) | 3 (2.1%) | 0 (0.0%) |
| Hematuria | 0 (0.0%) | 0 (0.0%) | 2 (1.4%) | 2 (1.4%) |
| Stricture | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Proctitis | 2 (1.4%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Fecal incontinence | 1 (0.7%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Diarrhea | 2 (1.4%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Rectal hemorrhage | 3 (2.1%) | 0 (0.0%) | 10 (6.9%) | 2 (1.4%) |
| Toxicity | Risk Factor | HR | 95% CI | Univariate | |
|---|---|---|---|---|---|
| Acute | |||||
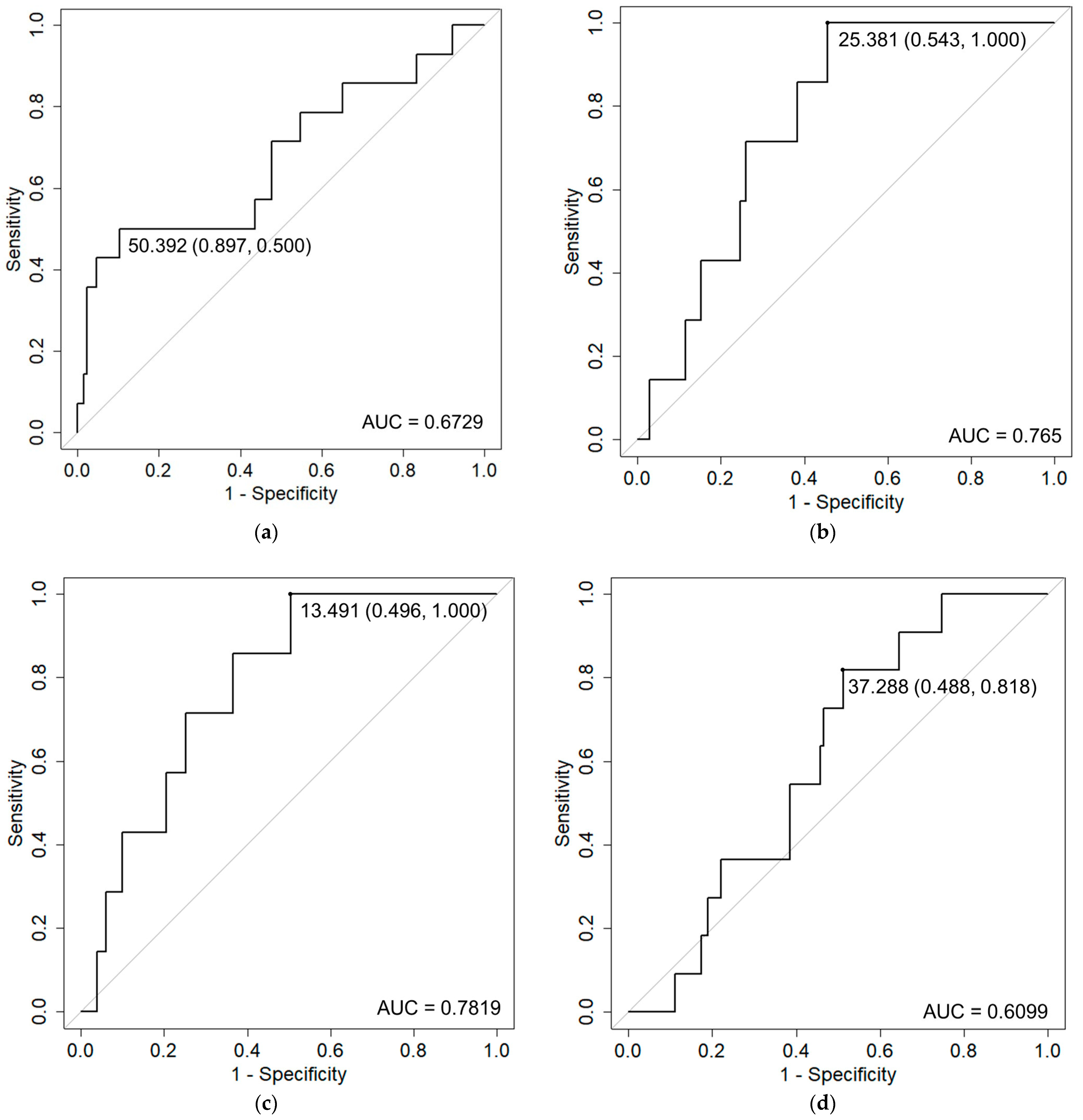
| GU | Prostate volume | 1.03 | (1.02–1.04) | 0.000006 | |
| GI | Rectum D10 cc | 1.26 | (1.1–1.45) | 0.0011 | |
| Rectum V30 Gy | 1.22 | (1.09–1.37) | 0.00083 | ||
| Late | |||||
| GU | na | ||||
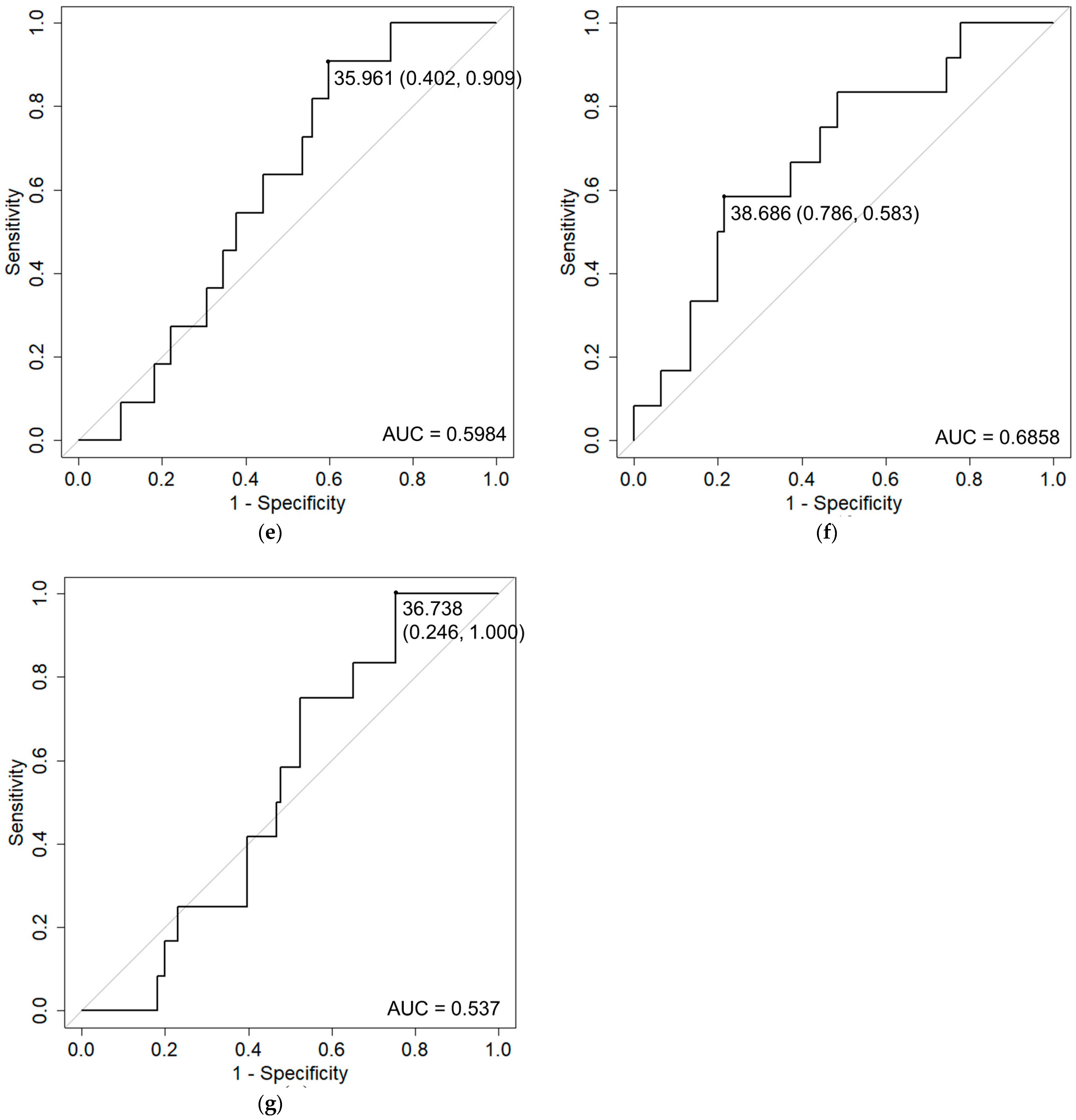
| GI | Rectum D0.1 cc | 1.45 | (1.15–1.83) | 0.0018 | |
| Rectum D1 cc | 1.45 | (1.15–1.82) | 0.0015 | ||
| Frequency | Bladder Dmax | 1.63 | (1.26–2.1) | 0.00019 | |
| Rectal hemorrhage | Rectum D0.1 cc | 1.33 | (1.11–1.6) | 0.0021 | |
| Author | Year | Treatment | n | Median Follow-Up (Months) | Response Variable | Explanatory Variable |
|---|---|---|---|---|---|---|
| Dincer et al. [6] | 2021 | 35–36.25 Gy /5 fr | 44 | 52 | Acute ≥ G2 GU toxicity | PTV ≥ 85 cc |
| Alayed et al. [7] | 2020 | 35–40 Gy /5 fr | 258 | Urinary QOL Bowel QOL Late G2 GU Late G2 GI | Bladder Dmean Bladder V38 Gy Rectal V35 Gy Bladder D2 cc Rectal V38y | |
| Henderson et al. [8] | 2018 | 36.25 Gy/5 fr | 50 | na | Acute IPSS | Bladder trigone Dmax |
| Wang et al. [9] | 2018 | 38 Gy/4 fr | 259 | na | 1 m incontinence 2 y urinary incontinence 1 m urinary obstruction/irritation 2 y urinary obstruction/irritation 1 m bowel QOL 2 y bowel QOL | Baseline QOL Baseline QOL Prior TURP CTV * Baseline QOL Baseline QOL Baseline QOL Rectum D25% Rectum D50% Rectum Dmax * |
| Jackson et al. [10] | 2018 | 37 Gy/5 fr | 66 | 36 | Urinary incontinence QOL Urinary bother Bowel QOL Sexual QOL | Baseline QOL Bladder Dmax Baseline QOL Baseline QOL |
| Helou et al. [11] | 2017 | 35 Gy/5 fr 40 Gy/5 fr | 82 177 | 38 | Late ≥ G2 GU toxicity | Prescription dose (40 Gy > 35 Gy) Pretreatment IPSS |
| Dess et al. [12] | 2017 | 35, 36.25 Gy /5 fr | 713 | na | 4 or 5 domains of QOL | Baseline depression Baseline bowel QOL |
| Musunuru et al [13] | 2016 | 35–40 Gy /5 fr | 258 | 29.7 | ≥G2 rectal bleeding | Rectal V38 Gy Anticoagulant usage Hemorrhoids |
| Qi et al. [14] | 2016 | 40 Gy/5 fr | 86 | na | Urinary irritation QOL | Bladder V85%, 90%, 95%, 100% Bladder D2 cc, 10 cc |
| Kole et al. [15] | 2016 | 35–36.25 Gy /5 fr | 216 | 48 | Late urinary flare (transient increase in IPSS) | Young age Bladder D12.7% |
| Seymour et al. [16] | 2015 | 38 Gy/4 fr | 56 | 35.49 | Late ≥ G2 GU Overall GU | Prostate volume Homogeneity index Dmax of urethra IPSS Prostate volume Urethral V44 Gy Bladder V19 Gy |
| Glowacki et al. [17] | 2015 | 36.25 Gy/5 fr | 132 | 8.5 | Acute ≥G2 GU toxicity ≥G1 GU | Diabetes PTV |
| Gurka et al. [18] | 2015 | 35–36.25 Gy /5 fr | 208 | 48 | Hematuria | Alpha antagonist usage Procedures for benign prostatic hypertrophy |
| Gomez et al. [19] | 2015 | 40 Gy/5 fr | 86 | 12 | Urinary QOL Bowel QOL | PTV Bladder V100% Rectal V90%, 100% |
| Kim et al. [20] | 2014 | 45, 47.5, 50 Gy /5 fr | 91 | 24.5 | G3 GI G2 GI | Rectum V39 Gy, 0 Gy Rectum V24 Gy |
| Macias et al. [21] | 2014 | 43.84–45.2 Gy /8 fr | 45 | 13.8 | Acute ≥ G1 GI | Rectum V28 Gy Age |
| Eliaset al. [22] | 2014 | 35 Gy/5 fr | 84 | 50.8 | Urinary QOL Bowel QOL Sexual QOL | Bladder volume Rectal D1cc Penile bulb V35 Gy |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujii, K.; Nakano, M.; Kawakami, S.; Tanaka, Y.; Kainuma, T.; Tsumura, H.; Tabata, K.-i.; Satoh, T.; Iwamura, M.; Ishiyama, H. Dosimetric Predictors of Toxicity after Prostate Stereotactic Body Radiotherapy: A Single-Institutional Experience of 145 Patients. Curr. Oncol. 2023, 30, 5062-5071. https://doi.org/10.3390/curroncol30050383
Fujii K, Nakano M, Kawakami S, Tanaka Y, Kainuma T, Tsumura H, Tabata K-i, Satoh T, Iwamura M, Ishiyama H. Dosimetric Predictors of Toxicity after Prostate Stereotactic Body Radiotherapy: A Single-Institutional Experience of 145 Patients. Current Oncology. 2023; 30(5):5062-5071. https://doi.org/10.3390/curroncol30050383
Chicago/Turabian StyleFujii, Kyohei, Masahiro Nakano, Shogo Kawakami, Yuichi Tanaka, Takuro Kainuma, Hideyasu Tsumura, Ken-ichi Tabata, Takefumi Satoh, Masatsugu Iwamura, and Hiromichi Ishiyama. 2023. "Dosimetric Predictors of Toxicity after Prostate Stereotactic Body Radiotherapy: A Single-Institutional Experience of 145 Patients" Current Oncology 30, no. 5: 5062-5071. https://doi.org/10.3390/curroncol30050383
APA StyleFujii, K., Nakano, M., Kawakami, S., Tanaka, Y., Kainuma, T., Tsumura, H., Tabata, K.-i., Satoh, T., Iwamura, M., & Ishiyama, H. (2023). Dosimetric Predictors of Toxicity after Prostate Stereotactic Body Radiotherapy: A Single-Institutional Experience of 145 Patients. Current Oncology, 30(5), 5062-5071. https://doi.org/10.3390/curroncol30050383





