Somatostatin Receptor Theranostics for Refractory Meningiomas
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. Imaging
2.3. Therapy
2.4. MRI Response Assessment
2.5. Clinical and Biological Examinations
2.6. Previous Studies
2.7. Outcomes
2.8. Data Synthesis
3. Results
3.1. Patients Characteristics
3.2. Toxicity
3.3. Study Selection
3.4. Treatment Response
3.5. Month Progression-Free Survival according to WHO-grade
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Galldiks, N.; Albert, N.L.; Sommerauer, M.; Grosu, A.L.; Ganswindt, U.; Law, I.; Preusser, M.; Le Rhun, E.; Vogelbaum, M.A.; Zadeh, G.; et al. PET Imaging in Patients with Meningioma—Report of the RANO/PET Group. Neuro-Oncol. 2017, 19, 1576–1587. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A Summary. Neuro-Oncol. 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Goldbrunner, R.; Minniti, G.; Preusser, M.; Jenkinson, M.D.; Sallabanda, K.; Houdart, E.; von Deimling, A.; Stavrinou, P.; Lefranc, F.; Lund-Johansen, M.; et al. EANO Guidelines for the Diagnosis and Treatment of Meningiomas. Lancet Oncol. 2016, 17, e383–e391. [Google Scholar] [CrossRef]
- Simpson, D. THE RECURRENCE OF INTRACRANIAL MENINGIOMAS AFTER SURGICAL TREATMENT. J. Neurol. Neurosurg. Psychiatry 1957, 20, 22–39. [Google Scholar] [CrossRef]
- Hortobágyi, T.; Bencze, J.; Varkoly, G.; Kouhsari, M.C.; Klekner, Á. Meningioma Recurrence. Open Med. 2016, 11, 168–173. [Google Scholar] [CrossRef]
- Ros-Sanjuan, A.; Iglesias-Moroño, S.; Carrasco-Brenes, A.; Bautista-Ojeda, D.; Arraez-Sanchez, M.A. Atypical Meningiomas: Histologic and Clinical Factors Associated With Recurrence. World Neurosurg. 2019, 125, e248–e256. [Google Scholar] [CrossRef] [PubMed]
- Graillon, T.; Tabouret, E.; Chinot, O. Chemotherapy and Targeted Therapies for Meningiomas: What Is the Evidence? Curr. Opin. Neurol. 2021, 34, 857–867. [Google Scholar] [CrossRef]
- Graillon, T.; Romano, D.; Defilles, C.; Saveanu, A.; Mohamed, A.; Figarella-Branger, D.; Roche, P.-H.; Fuentes, S.; Chinot, O.; Dufour, H.; et al. Octreotide Therapy in Meningiomas: In Vitro Study, Clinical Correlation, and Literature Review. J. Neurosurg. 2016, 127, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Salgues, B.; Graillon, T.; Guedj, E. [68Ga]Ga-DOTA-TOC PET/CT Fused to MRI in Post-Operative Evaluation of Olfactory Groove Meningioma: A Case on Millimetric Remnants. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 316–317. [Google Scholar] [CrossRef]
- Hung, N.D.; Dung, L.T.; Huyen, D.K.; Duy, N.Q.; He, D.-V.; Duc, N.M. The Value of Quantitative Magnetic Resonance Imaging Signal Intensity in Distinguishing between Spinal Meningiomas and Schwannomas. Int. J. Med. Sci. 2022, 19, 1110–1117. [Google Scholar] [CrossRef]
- Rachinger, W.; Stoecklein, V.M.; Terpolilli, N.A.; Haug, A.R.; Ertl, L.; Pöschl, J.; Schüller, U.; Schichor, C.; Thon, N.; Tonn, J.-C. Increased 68Ga-DOTATATE Uptake in PET Imaging Discriminates Meningioma and Tumor-Free Tissue. J. Nucl. Med. 2015, 56, 347–353. [Google Scholar] [CrossRef]
- Van Essen, M.; Krenning, E.P.; Kooij, P.P.; Bakker, W.H.; Feelders, R.A.; de Herder, W.W.; Wolbers, J.G.; Kwekkeboom, D.J. Effects of Therapy with [177Lu-DOTA0, Tyr3]Octreotate in Patients with Paraganglioma, Meningioma, Small Cell Lung Carcinoma, and Melanoma. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2006, 47, 1599–1606. [Google Scholar]
- Bartolomei, M.; Bodei, L.; De Cicco, C.; Grana, C.M.; Cremonesi, M.; Botteri, E.; Baio, S.M.; Aricò, D.; Sansovini, M.; Paganelli, G. Peptide Receptor Radionuclide Therapy with 90Y-DOTATOC in Recurrent Meningioma. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 1407. [Google Scholar] [CrossRef] [PubMed]
- Marincek, N.; Radojewski, P.; Dumont, R.A.; Brunner, P.; Müller-Brand, J.; Maecke, H.R.; Briel, M.; Walter, M.A. Somatostatin Receptor–Targeted Radiopeptide Therapy with 90Y-DOTATOC and 177Lu-DOTATOC in Progressive Meningioma: Long-Term Results of a Phase II Clinical Trial. J. Nucl. Med. 2015, 56, 171–176. [Google Scholar] [CrossRef]
- Parghane, R.V.; Talole, S.; Basu, S. Prevalence of Hitherto Unknown Brain Meningioma Detected on 68Ga-DOTATATE Positron-Emission Tomography/Computed Tomography in Patients with Metastatic Neuroendocrine Tumor and Exploring Potential of 177Lu-DOTATATE Peptide Receptor Radionuclide Therapy as Single-Shot Treatment Approach Targeting Both Tumors. World J. Nucl. Med. 2019, 18, 160–170. [Google Scholar] [CrossRef]
- Seystahl, K.; Stoecklein, V.; Schüller, U.; Rushing, E.; Nicolas, G.; Schäfer, N.; Ilhan, H.; Pangalu, A.; Weller, M.; Tonn, J.-C.; et al. Somatostatin Receptor-Targeted Radionuclide Therapy for Progressive Meningioma: Benefit Linked to 68Ga-DOTATATE/-TOC Uptake. Neuro-Oncol. 2016, 18, 1538–1547. [Google Scholar] [CrossRef]
- Müther, M.; Roll, W.; Brokinkel, B.; Zinnhardt, B.; Sporns, P.B.; Seifert, R.; Schäfers, M.; Weckesser, M.; Stegger, L.; Stummer, W.; et al. Response Assessment of Somatostatin Receptor Targeted Radioligand Therapies for Progressive Intracranial Meningioma. Nukl. Nucl. Med. 2020, 59, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Gerster-Gilliéron, K.; Forrer, F.; Maecke, H.; Mueller-Brand, J.; Merlo, A.; Cordier, D. 90Y-DOTATOC as a Therapeutic Option for Complex Recurrent or Progressive Meningiomas. J. Nucl. Med. 2015, 56, 1748–1751. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.Y.; Bi, W.L.; Weller, M.; Kaley, T.; Blakeley, J.; Dunn, I.; Galanis, E.; Preusser, M.; McDermott, M.; Rogers, L.; et al. Proposed Response Assessment and Endpoints for Meningioma Clinical Trials: Report from the Response Assessment in Neuro-Oncology Working Group. Neuro-Oncol. 2019, 21, 26–36. [Google Scholar] [CrossRef]
- Kreissl, M.C.; Hänscheid, H.; Löhr, M.; Verburg, F.A.; Schiller, M.; Lassmann, M.; Reiners, C.; Samnick, S.S.; Buck, A.K.; Flentje, M.; et al. Combination of Peptide Receptor Radionuclide Therapy with Fractionated External Beam Radiotherapy for Treatment of Advanced Symptomatic Meningioma. Radiat. Oncol. Lond. Engl. 2012, 7, 99. [Google Scholar] [CrossRef]
- Hartrampf, P.E.; Hänscheid, H.; Kertels, O.; Schirbel, A.; Kreissl, M.C.; Flentje, M.; Sweeney, R.A.; Buck, A.K.; Polat, B.; Lapa, C. Long-Term Results of Multimodal Peptide Receptor Radionuclide Therapy and Fractionated External Beam Radiotherapy for Treatment of Advanced Symptomatic Meningioma. Clin. Transl. Radiat. Oncol. 2020, 22, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Kaley, T.; Barani, I.; Chamberlain, M.; McDermott, M.; Panageas, K.; Raizer, J.; Rogers, L.; Schiff, D.; Vogelbaum, M.; Weber, D.; et al. Historical Benchmarks for Medical Therapy Trials in Surgery- and Radiation-Refractory Meningioma: A RANO Review. Neuro-Oncol. 2014, 16, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Mirian, C.; Duun-Henriksen, A.K.; Maier, A.D.; Pedersen, M.M.; Jensen, L.R.; Bashir, A.; Graillon, T.; Hrachova, M.; Bota, D.; van Essen, M.; et al. Somatostatin Receptor-Targeted Radiopeptide Therapy in Treatment-Refractory Meningioma: An Individual Patient Data Meta-Analysis. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2020. [Google Scholar] [CrossRef]
- Minutoli, F.; Amato, E.; Sindoni, A.; Cardile, D.; Conti, A.; Herberg, A.; Baldari, S. Peptide Receptor Radionuclide Therapy in Patients with Inoperable Meningiomas: Our Experience and Review of the Literature. Cancer Biother. Radiopharm. 2014, 29, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Graillon, T.; Ferrer, L.; Siffre, J.; Sanson, M.; Peyre, M.; Peyrière, H.; Mougel, G.; Autran, D.; Tabouret, E.; Figarella-Branger, D.; et al. Role of 3D Volume Growth Rate for Drug Activity Evaluation in Meningioma Clinical Trials: The Example of the CEVOREM Study. Neuro-Oncol. 2021, 23, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- O’Donoghue, J.A.; Bardiès, M.; Wheldon, T.E. Relationships between Tumor Size and Curability for Uniformly Targeted Therapy with Beta-Emitting Radionuclides. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 1995, 36, 1902–1909. [Google Scholar]
- Oddstig, J.; Bernhardt, P.; Nilsson, O.; Ahlman, H.; Forssell-Aronsson, E. Radiation-Induced up-Regulation of Somatostatin Receptor Expression in Small Cell Lung Cancer in Vitro. Nucl. Med. Biol. 2006, 33, 841–846. [Google Scholar] [CrossRef] [PubMed]
- Oddstig, J.; Bernhardt, P.; Nilsson, O.; Ahlman, H.; Forssell-Aronsson, E. Radiation Induces Up-Regulation of Somatostatin Receptors 1, 2, and 5 in Small Cell Lung Cancer in Vitro Also at Low Absorbed Doses. Cancer Biother. Radiopharm. 2011, 26, 759–765. [Google Scholar] [CrossRef]
- Claringbold, P.G.; Brayshaw, P.A.; Price, R.A.; Turner, J.H. Phase II Study of Radiopeptide 177Lu-Octreotate and Capecitabine Therapy of Progressive Disseminated Neuroendocrine Tumours. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 302–311. [Google Scholar] [CrossRef]
- Jensen, R.; Harper, J. EXTH-78. PARP INHIBITION IMPROVES RADIOTHERAPY EFFECTIVENESS IN MENINGIOMA CELL CULTURE. Neuro-Oncol. 2018, 20, vi101–vi102. [Google Scholar] [CrossRef][Green Version]
- Cullinane, C.; Waldeck, K.; Kirby, L.; Rogers, B.E.; Eu, P.; Tothill, R.W.; Hicks, R.J. Enhancing the Anti-Tumour Activity of 177Lu-DOTA-Octreotate Radionuclide Therapy in Somatostatin Receptor-2 Expressing Tumour Models by Targeting PARP. Sci. Rep. 2020, 10, 10196. [Google Scholar] [CrossRef] [PubMed]
- Garzon-Muvdi, T.; Bailey, D.D.; Pernik, M.N.; Pan, E. Basis for Immunotherapy for Treatment of Meningiomas. Front. Neurol. 2020, 11, 945. [Google Scholar] [CrossRef] [PubMed]
- Pavlakis, N.; Ransom, D.T.; Wyld, D.; Sjoquist, K.M.; Asher, R.; Gebski, V.; Wilson, K.; Kiberu, A.D.; Burge, M.E.; Macdonald, W.; et al. First Results for Australasian Gastrointestinal Trials Group (AGITG) Control Net Study: Phase II Study of 177Lu-Octreotate Peptide Receptor Radionuclide Therapy (LuTate PRRT) +/− Capecitabine, Temozolomide (CAPTEM) for Midgut Neuroendocrine Tumors (MNETs). J. Clin. Oncol. 2020, 38, 604. [Google Scholar] [CrossRef]
- Parghane, R.V.; Ostwal, V.; Ramaswamy, A.; Bhandare, M.; Chaudhari, V.; Talole, S.; Shrikhande, S.V.; Basu, S. Long-Term Outcome of “Sandwich” Chemo-PRRT: A Novel Treatment Strategy for Metastatic Neuroendocrine Tumors with Both FDG- and SSTR-Avid Aggressive Disease. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 913–923. [Google Scholar] [CrossRef] [PubMed]
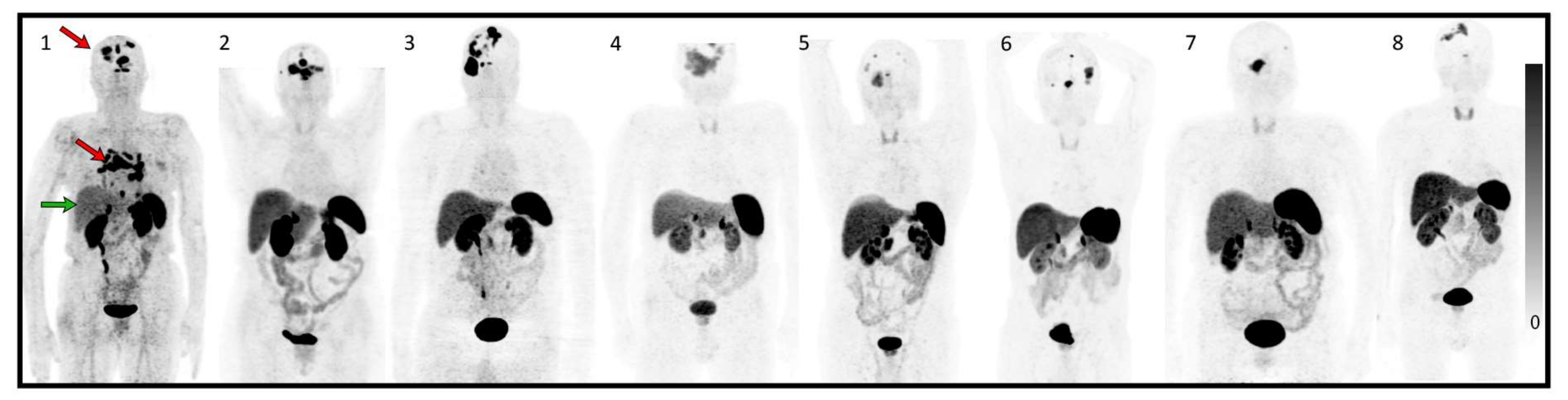
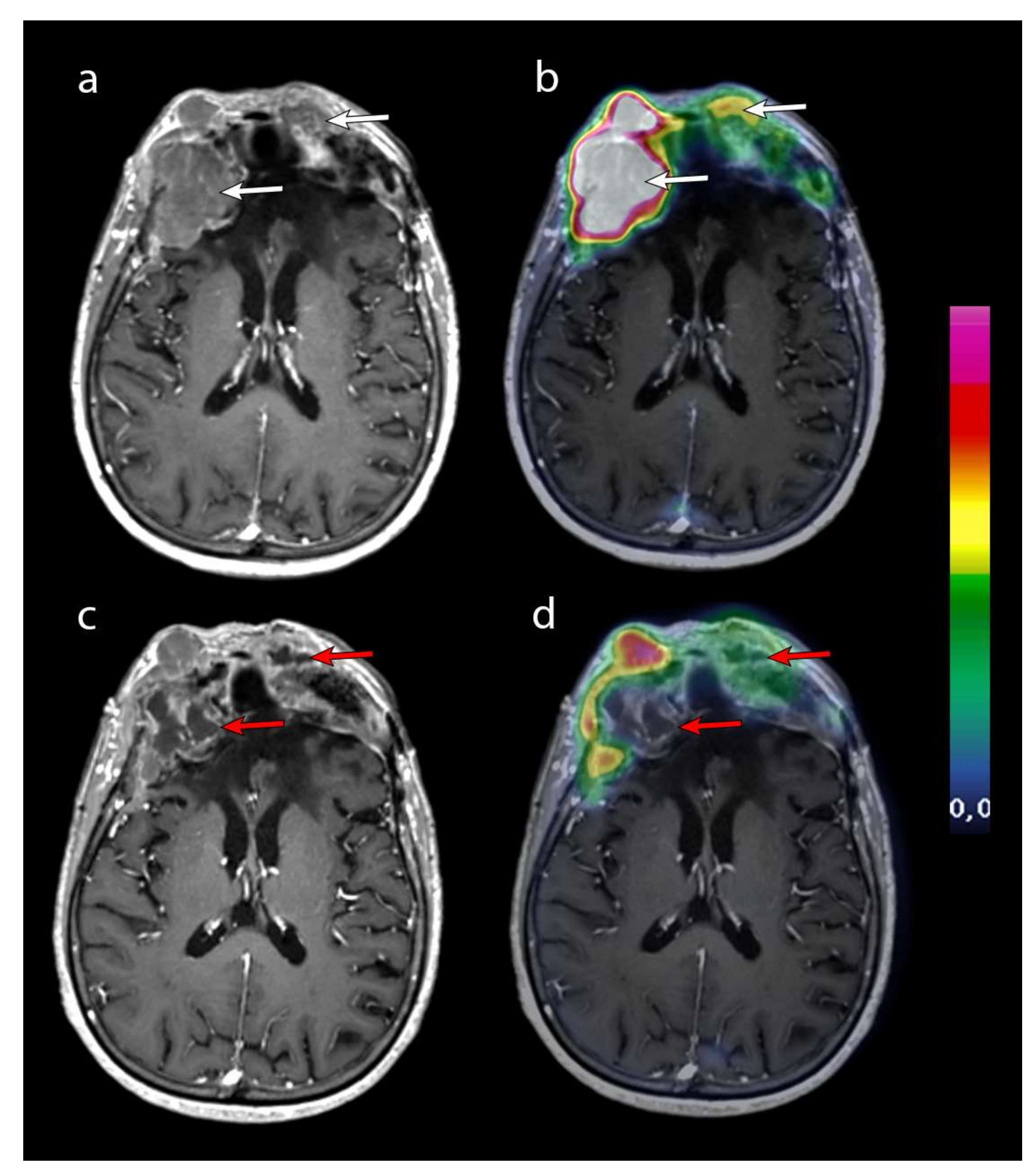
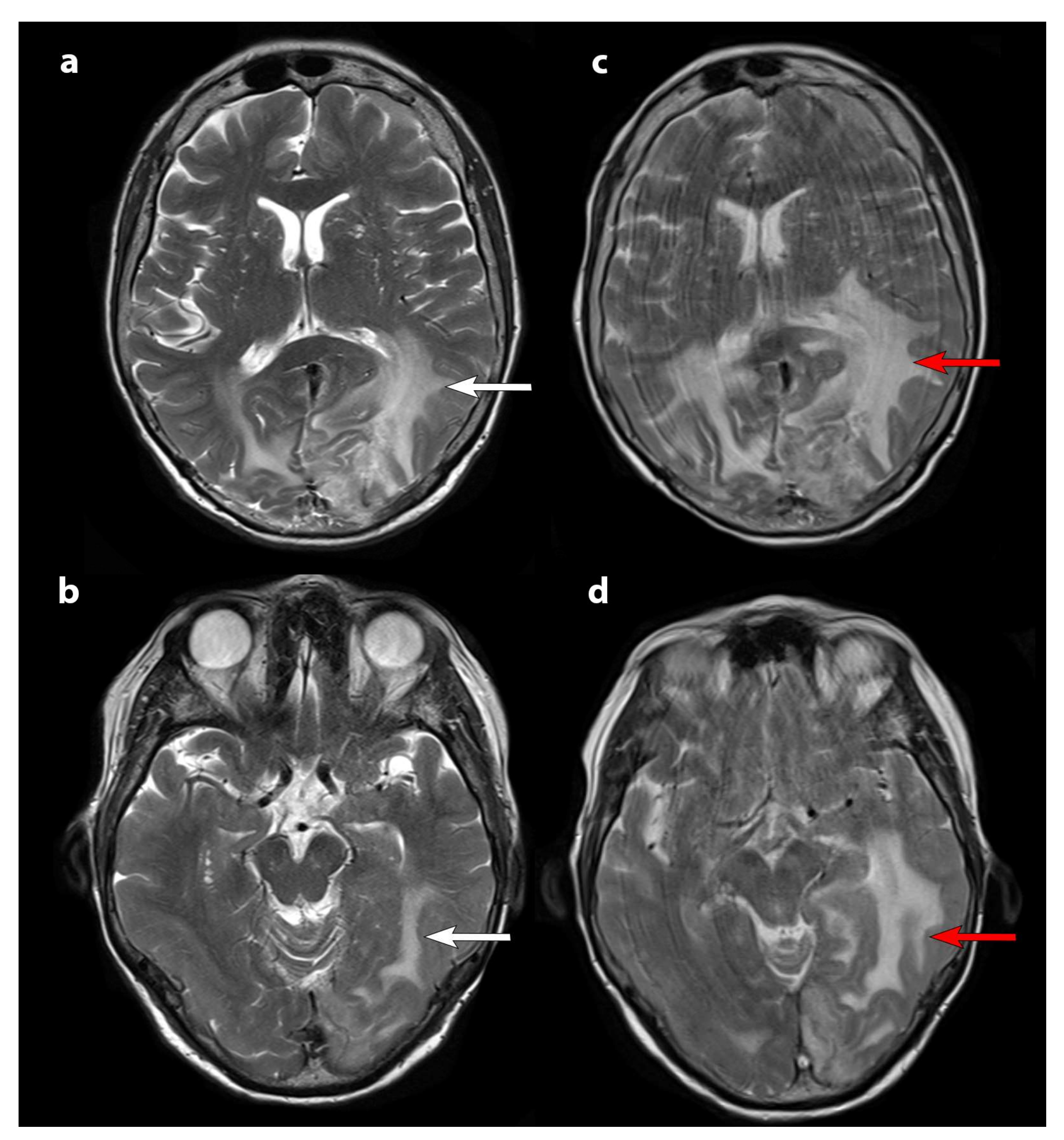
| Age | Sex | Time from Diagnosis | Localization | WHO Grade | Treatment | Ki 67 Index Primitive | Ki 67 Index Recurrence | Neurological Deficit | ECOG PS | |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 74 | F | 3 years | Multiple meningioma, metastasis | II | Surgery, EBRT, SRS, Sandostatine-Everolimus | 15–20% | N/A | motor deficit, confusion | 2 |
| 2 | 85 | F | 17 years | multiple meningioma; subcutaneous frontal lesion | II | Surgeryx3, SRSx2, EBRT | 15% | N/A | visual disturbance | 2 |
| 3 | 67 | M | 9 years | multiple meningioma | II | Surgeryx2, EBRT, SRS, Sandostatine-Everolimus | N/A | 2% | left hemiparesis | 2 |
| 4 | 72 | M | 19 years | multiple meningioma | II | Surgeryx2, EBRT, SRS, Sandostatine-Everolimus | N/A | 25% | visual disturbance; frontal syndrome | 1 |
| 5 | 60 | M | 9 years | multiple meningioma, metastasis | III; II | Surgeryx2, parotidectomy, EBRT | 30–40% | 10% | asthenia, attention deficit disorder | 1 |
| 6 | 76 | M | 15 years | multiple meningioma | II | Surgeryx5, EBRT, SRSx3 | 10–15% | 25; 20% | none | 0 |
| 7 | 73 | F | 15 years | multiple meningioma | II | Surgeryx2, EBRT, SRS | 10% | N/A | visual disturbance, ptosis, trigeminal neuralgia | 1 |
| 8 | 67 | M | 11 years | multiple meningioma | II | Surgery, EBRT | N/A | N/A | left upper limb deficiency, | 1 |
| Krenning Score | WHO Grade | SUVmax Pretreatment | Cumulative Dose (GBq) | Best Radiologic Response | PFS 2D RANO Criteria | |
|---|---|---|---|---|---|---|
| 1 | 3 | II | 7.4 | 10.6 | PD | 2 months |
| 2 | 3 | II | 26.3 | 29.6 | SD | 10 months |
| 3 | 3 | II | 45 | 29.6 | SD | 17 months |
| 4 | 2 | II | 14.5 | 29.6 | SD | 16 months |
| 5 | 2 | III; II | 12.3 | 29.6 | SD | not reached at 12 months |
| 6 | 3 | II | 16 | 29.6 | SD | not reached at 9 months |
| 7 | 3 | II | 16.8 | 29.6 | SD | N/A * |
| 8 | 3 | II | 15.4 | 29.6 | SD | not reached at 16 months |
| Current Case Series | Müther et al. 2020 [17] | Parghane et al., 2019 [15] | Seystahl et al., 2016 [16] | Gerster-Gilliéron et al., 2016 [18] | Marincek et al., 2015 [14] | Bartolomei et al., 2009 [13] | Van Essen et al., 2006 [12] | Kreissl et al., 2012 [20] Hartrampf et al. 2020 [21] | Total Analysis | |
|---|---|---|---|---|---|---|---|---|---|---|
| Radionuclide | 177Lu-DOTATATE | 177Lu-DOTATATE | 177Lu-DOTATATE | 177Lu-DOTATATE or90Y-DOTATATE | 90Y-DOTATOC | 90Y-DOTATOC or177Lu DOTATOC | 90Y DOTATOC | 177Lu-DOTATATE | EBRT + 1 cycle177Lu-DOTA(TATE)/(TOC) | |
| Patients | 8 (7 patients included in the analysis) | 7 | 5 | 20 (16 pts 177Lu-DOTATATE + 3 pts 90Y-DOTATATE + 1 combined); 19 patients included in the analysis | 15 | 34 (66 cycles 90YDOTATOC et 8 cycles 177Lu DOTATOC) | 29 treated; 28 patients included in the analysis | 5 | 10 | |
| Grade WHO-I pts | 0 | 2 (28.5%) | N/A | 4 analyzed (21%) | 9 (60%) | 5 (15%) | 14 (48%) | N/A | 6 (60%) | 29 * |
| GI PFS6 % | 0 | 100 | 100 | 100 | N/A | 78.6 | 100 | 89.7 | ||
| Grade WHO-II pts | 7 (100%) | 5 (71.4%) | N/A | 7 (35%) | 2 (13%) | 6 (18%) | 9 (31%) | N/A | 2 (20%) | 21 ** |
| GII PFS6 % | 85.7 | 20 | 57 | 50 | N/A | 14.3 (GII + III 14 patients) | 100 | 57.1 | ||
| Grade WHO-III pts | 0 | 0 | N/A | 8 (40%) | 1 (6%) | 3 (9%) | 5 (21%) | 3 (60%) | 0 | 12 ** |
| GIII PFS6 (%) | 0 | 0 | N/A | 14.3 (GII + III 14 patients) | 0 | 0 | ||||
| Unknown grade pts | 0 | 0 | 5 (100%) | 0 | 3 (20%) | 20 (59%) | 0 | 2 (40%) | 2 | 10 ** |
| Unknown grade PFS6 (%) | 100 | 100 | N/A | 100 | 100 | 100 | ||||
| All grade PFS6 (%) | 85.7 | 42.8 | 100 | 42 | 86.7 | N/A | 46.4 | 40 | 100 | 58.1 * (86 pts) |
| Radiological criteria for progression | RANO | RANO | RECIST 1.1 | Macdonald | RECIST 1.1 | N/A | SWOG | N/A | N/A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salgues, B.; Graillon, T.; Horowitz, T.; Chinot, O.; Padovani, L.; Taïeb, D.; Guedj, E. Somatostatin Receptor Theranostics for Refractory Meningiomas. Curr. Oncol. 2022, 29, 5550-5565. https://doi.org/10.3390/curroncol29080438
Salgues B, Graillon T, Horowitz T, Chinot O, Padovani L, Taïeb D, Guedj E. Somatostatin Receptor Theranostics for Refractory Meningiomas. Current Oncology. 2022; 29(8):5550-5565. https://doi.org/10.3390/curroncol29080438
Chicago/Turabian StyleSalgues, Betty, Thomas Graillon, Tatiana Horowitz, Olivier Chinot, Laetitia Padovani, David Taïeb, and Eric Guedj. 2022. "Somatostatin Receptor Theranostics for Refractory Meningiomas" Current Oncology 29, no. 8: 5550-5565. https://doi.org/10.3390/curroncol29080438
APA StyleSalgues, B., Graillon, T., Horowitz, T., Chinot, O., Padovani, L., Taïeb, D., & Guedj, E. (2022). Somatostatin Receptor Theranostics for Refractory Meningiomas. Current Oncology, 29(8), 5550-5565. https://doi.org/10.3390/curroncol29080438





