Liver Cancer-Specific Isoform of Serine Protease Inhibitor Kazal for the Detection of Hepatocellular Carcinoma: Results from a Pilot Study in Patients with Dysmetabolic Liver Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Circulating Biomarkers
2.3. Statistical Analysis
3. Results
3.1. Patients’ Characteristics
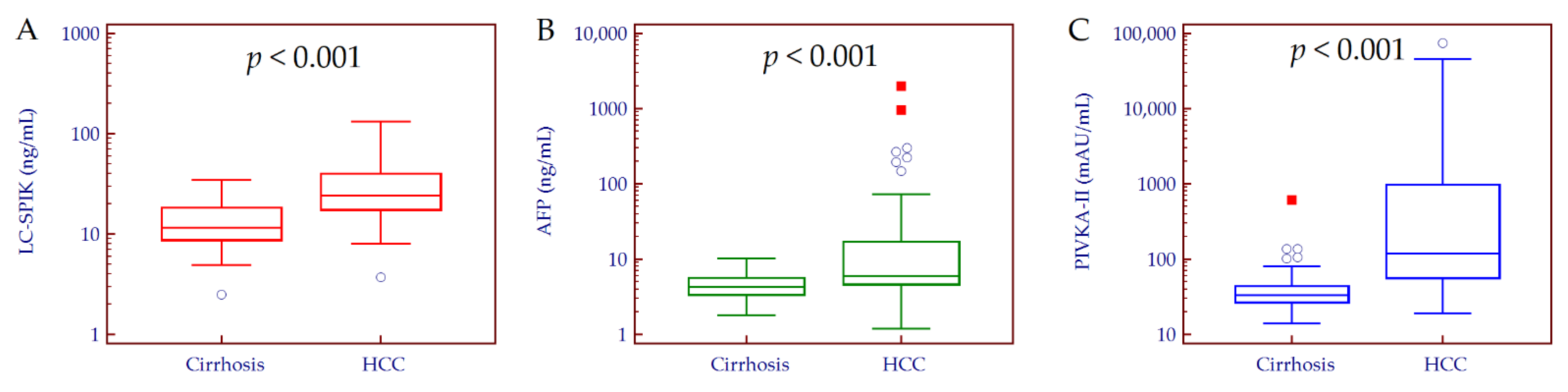
3.2. Circulating Biomarkers Values
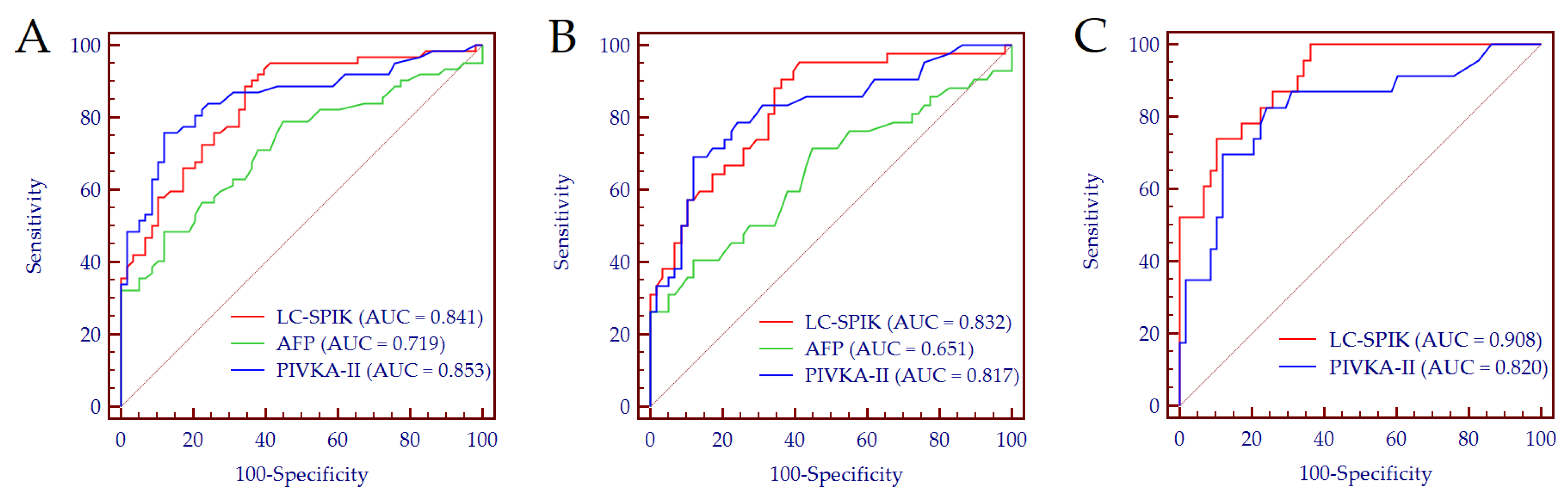
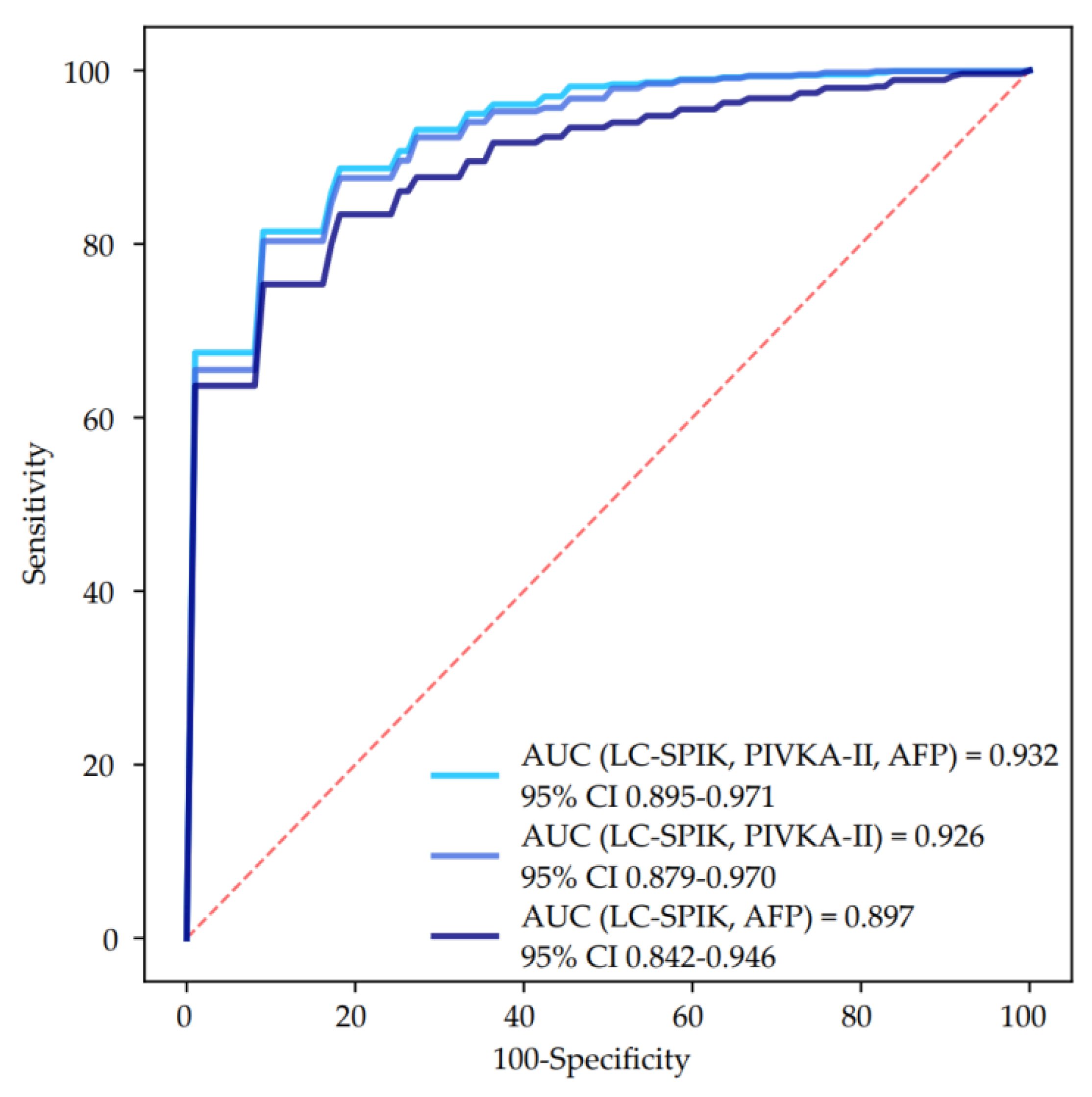
3.3. Biomarkers Performance for HCC Detection
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- The Global Cancer Observatory-IARC. Available online: https://gco.iarc.fr/today (accessed on 24 May 2022).
- Villanueva, A. Hepatocellular Carcinoma. N. Engl. J. Med. 2019, 380, 1450–1462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caviglia, G.P.; Ciruolo, M.; Olivero, A.; Carucci, P.; Rolle, E.; Rosso, C.; Abate, M.L.; Risso, A.; Ribaldone, D.G.; Tandoi, F.; et al. Prognostic Role of Serum Cytokeratin-19 Fragment (CYFRA 21-1) in Patients with Hepatocellular Carcinoma. Cancers 2020, 12, 2776. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [CrossRef] [PubMed] [Green Version]
- Marrero, J.A.; Kulik, L.M.; Sirlin, C.B.; Zhu, A.X.; Finn, R.S.; Abecassis, M.M.; Roberts, L.R.; Heimbach, J.K. Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology 2018, 68, 723–750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campion, D.; Tucci, A.; Ponzo, P.; Caviglia, G.P. Non-invasive biomarkers for the detection of hepatocellular carcinoma. Minerva Biotecnol. 2019, 31, 11–22. [Google Scholar] [CrossRef]
- Rizzo, A.; Ricci, A.D. PD-L1, TMB, and other potential predictors of response to immunotherapy for hepatocellular carcinoma: How can they assist drug clinical trials? Expert Opin. Investig. Drugs. 2022, 31, 415–423. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, S.; Tovoli, F.; Barbera, M.A.; Garuti, F.; Palloni, A.; Frega, G.; Garajovà, I.; Rizzo, A.; Trevisani, F.; Brandi, G. Metronomic capecitabine vs. best supportive care in Child-Pugh B hepatocellular carcinoma: A proof of concept. Sci. Rep. 2018, 8, 9997. [Google Scholar] [CrossRef]
- Lu, F.; Lamontagne, J.; Sun, A.; Pinkerton, M.; Block, T.; Lu, X. Role of the inflammatory protein serine protease inhibitor Kazal in preventing cytolytic granule granzyme A-mediated apoptosis. Immunology 2011, 134, 398–408. [Google Scholar] [CrossRef] [Green Version]
- Lu, F.; Shah, P.A.; Rao, A.; Gifford-Hollingsworth, C.; Chen, A.; Trey, G.; Soryal, M.; Talat, A.; Aslam, A.; Nasir, B.; et al. Liver Cancer-Specific Serine Protease Inhibitor Kazal Is a Potentially Novel Biomarker for the Early Detection of Hepatocellular Carcinoma. Clin. Transl. Gastroenterol. 2020, 11, e00271. [Google Scholar] [CrossRef] [PubMed]
- Caviglia, G.P.; Ribaldone, D.G.; Abate, M.L.; Ciancio, A.; Pellicano, R.; Smedile, A.; Saracco, G.M. Performance of protein induced by vitamin K absence or antagonist-II assessed by chemiluminescence enzyme immunoassay for hepatocellular carcinoma detection: A meta-analysis. Scand. J. Gastroenterol. 2018, 53, 734–740. [Google Scholar] [CrossRef]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrel, L.D.; Liu, Y.C.; Torbenson, M.S.; Unalp-Arida, A.; et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef]
- Caviglia, G.P.; Armandi, A.; Rosso, C.; Gaia, S.; Aneli, S.; Rolle, E.; Abate, M.L.; Olivero, A.; Nicolosi, A.; Guariglia, M.; et al. Biomarkers of Oncogenesis, Adipose Tissue Dysfunction and Systemic Inflammation for the Detection of Hepatocellular Carcinoma in Patients with Nonalcoholic Fatty Liver Disease. Cancers 2021, 13, 2305. [Google Scholar] [CrossRef]
- Caviglia, G.P.; Touscoz, G.A.; Smedile, A.; Pellicano, R. Noninvasive assessment of liver fibrosis: Key messages for clinicians. Pol. Arch. Med. Wewn. 2014, 124, 329–335. [Google Scholar] [CrossRef] [Green Version]
- Gaia, S.; Campion, D.; Evangelista, A.; Spandre, M.; Cosso, L.; Brunello, F.; Ciccone, G.; Bugianesi, E.; Rizzetto, M. Non-invasive score system for fibrosis in chronic hepatitis: Proposal for a model based on biochemical, FibroScan and ultrasound data. Liver Int. 2015, 35, 2027–2035. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Bru, C.; Bruix, J. Prognosis of hepatocellular carcinoma: The BCLC staging classification. Semin. Liver Dis. 1999, 19, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Caviglia, G.P.; Abate, M.L.; Gaia, S.; Petrini, E.; Bosco, C.; Olivero, A.; Rosso, C.; Ciancio, A.; Pellicano, R.; Saracco, G.M.; et al. Risk of hepatocellular carcinoma in HBV cirrhotic patients assessed by the combination of miR-122, AFP and PIVKA-II. Panminerva Med. 2017, 59, 283–289. [Google Scholar] [CrossRef]
- Caviglia, G.P.; Troshina, Y.; Garro, E.; Gesualdo, M.; Aneli, S.; Birolo, G.; Pittaluga, F.; Cavallo, R.; Saracco, G.M.; Ciancio, A. Usefulness of a Hepatitis B Surface Antigen-Based Model for the Prediction of Functional Cure in Patients with Chronic Hepatitis B Virus Infection Treated with Nucleos(t)ide Analogues: A Real-World Study. J. Clin. Med. 2021, 10, 3308. [Google Scholar] [CrossRef] [PubMed]
- Kanwal, F.; Shubrook, J.H.; Younossi, Z.; Natarajan, Y.; Bugianesi, E.; Rinella, M.E.; Harrison, S.A.; Mantzoros, C.; Pfotenhauer, K.; Klein, S.; et al. Preparing for the NASH Epidemic: A Call to Action. Gastroenterology 2021, 161, 1030–1042.e8. [Google Scholar] [CrossRef] [PubMed]
- Saracco, G.M.; Evangelista, A.; Fagoonee, S.; Ciccone, G.; Bugianesi, E.; Caviglia, G.P.; Abate, M.L.; Rizzetto, M.; Pellicano, R.; Smedile, A. Etiology of chronic liver diseases in the Northwest of Italy, 1998 through 2014. World J. Gastroenterol. 2016, 22, 8187–8193. [Google Scholar] [CrossRef]
- Ribaldone, D.G.; Caviglia, G.P.; Gaia, S.; Rolle, E.; Risso, A.; Campion, D.; Brunocilla, P.R.; Saracco, G.M.; Carucci, P. Effect of COVID-19 Pandemic on Hepatocellular Carcinoma Diagnosis: Results from a Tertiary Care Center in North-West Italy. Curr. Oncol. 2022, 29, 1422–1429. [Google Scholar] [CrossRef] [PubMed]
- Singal, A.G.; El-Serag, H.B. Rational HCC screening approaches for patients with NAFLD. J. Hepatol. 2022, 76, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Henry, L. Epidemiology of non-alcoholic fatty liver disease and hepatocellular carcinoma. JHEP Rep. 2021, 3, 100305. [Google Scholar] [CrossRef] [PubMed]
- Ricco, G.; Cosma, C.; Bedogni, G.; Biasiolo, A.; Guarino, M.; Pontisso, P.; Morisco, F.; Oliveri, F.; Cavallone, D.; Bonino, F.; et al. Modeling the time-related fluctuations of AFP and PIVKA-II serum levels in patients with cirrhosis undergoing surveillance for hepatocellular carcinoma. Cancer Biomark. 2020, 29, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Loglio, A.; Iavarone, M.; Facchetti, F.; Di Paolo, D.; Perbellini, R.; Lunghi, G.; Ceriotti, F.; Galli, C.; Sandri, M.T.; Viganò, M.; et al. The combination of PIVKA-II and AFP improves the detection accuracy for HCC in HBV caucasian cirrhotics on long-term oral therapy. Liver Int. 2020, 40, 1987–1996. [Google Scholar] [CrossRef] [PubMed]
- Caviglia, G.P.; Ciruolo, M.; Abate, M.L.; Carucci, P.; Rolle, E.; Rosso, C.; Olivero, A.; Troshina, G.; Risso, A.; Nicolosi, A.; et al. Alpha-Fetoprotein, Protein Induced by Vitamin K Absence or Antagonist II and Glypican-3 for the Detection and Prediction of Hepatocellular Carcinoma in Patients with Cirrhosis of Viral Etiology. Cancers 2020, 12, 3218. [Google Scholar] [CrossRef]
- Degasperi, E.; Perbellini, R.; D’Ambrosio, R.; Uceda Renteria, S.C.; Ceriotti, F.; Perego, A.; Orsini, C.; Borghi, M.; Iavarone, M.; Bruccoleri, M.; et al. Prothrombin induced by vitamin K absence or antagonist-II and alpha foetoprotein to predict development of hepatocellular carcinoma in Caucasian patients with hepatitis C-related cirrhosis treated with direct-acting antiviral agents. Aliment. Pharmacol. Ther. 2022, 55, 350–359. [Google Scholar] [CrossRef] [PubMed]
| Variables | Cirrhosis | HCC | p Value |
|---|---|---|---|
| Patients, n | 58 | 62 | |
| Age (years), median (IQR) | 63 (57–69) | 66 (62–70) | 0.016 |
| Gender (M/F) | 29/29 | 49/13 | 0.001 |
| BMI (kg/m2), median (IQR) | 31.0 (27.1–34.0) | 29.8 (26.0–32.0) | 0.043 |
| Obesity (BMI ≥ 30.0 kg/m2), n (%) | 34 (59%) | 30 (48%) | 0.278 |
| T2DM, n (%) | 40 (69%) | 36 (58%) | 0.257 |
| Dyslipidemia, n (%) * | 38 (66%) | 36 (58%) | 0.455 |
| Arterial hypertension, n (%) | 42 (72%) | 42 (68%) | 0.691 |
| Child-Pugh score, n (%) | <0.001 | ||
| A | 57 (98%) | 46 (74%) | |
| B | 1 (2%) | 13 (21%) | |
| C | 0 | 3 (5%) | |
| ALT (U/L), median (IQR) | 41 (25–64) | 34 (25–45) | 0.056 |
| AST (U/L), median (IQR) | 43 (31–55) | 39 (33–56) | 0.727 |
| Platelets (×109/L) | 183 (138–223) | 113 (81–151) | <0.001 |
| Albumin (g/dL), median (IQR) | 4.2 (4.0–4.4) | 3.5 (3.0–4.3) | <0.001 |
| Total bilirubin (mg/dL), median (IQR) | 0.7 (0.5–1.1) | 1.2 (0.7–1.7) | 0.001 |
| INR, median (IQR) | 1.09 (1.04–1.16) | 1.25 (1.11–1.41) | <0.001 |
| Fasting glucose (mg/dL), median (IQR) | 116 (97–139) | 120 (95–143) | 0.936 |
| Total cholesterol (mg/dL), median (IQR) | 177 (153–202) | 164 (131–185) | 0.078 |
| HDL-cholesterol (mg/dL), median (IQR) | 47 (39–63) | 45 (35–54) | 0.290 |
| Triglycerides (mg/dL), median (IQR) | 127 (101–175) | 104 (79–136) | 0.009 |
| BCLC staging, n (%) | |||
| 0 | 7 (11%) | ||
| A | 31 (50%) | ||
| B | 14 (23%) | ||
| C | 7 (11%) | ||
| D | 3 (5%) | ||
| HCC nodules, n (%) | |||
| 1 | 28 (45%) | ||
| 2 | 12 (19%) | ||
| 3 | 8 (13%) | ||
| >3 | 14 (23%) | ||
| Size of major nodule (mm), median (IQR) | 27 (20–42) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caviglia, G.P.; Nicolosi, A.; Abate, M.L.; Carucci, P.; Rosso, C.; Rolle, E.; Armandi, A.; Aneli, S.; Olivero, A.; Risso, A.; et al. Liver Cancer-Specific Isoform of Serine Protease Inhibitor Kazal for the Detection of Hepatocellular Carcinoma: Results from a Pilot Study in Patients with Dysmetabolic Liver Disease. Curr. Oncol. 2022, 29, 5457-5465. https://doi.org/10.3390/curroncol29080431
Caviglia GP, Nicolosi A, Abate ML, Carucci P, Rosso C, Rolle E, Armandi A, Aneli S, Olivero A, Risso A, et al. Liver Cancer-Specific Isoform of Serine Protease Inhibitor Kazal for the Detection of Hepatocellular Carcinoma: Results from a Pilot Study in Patients with Dysmetabolic Liver Disease. Current Oncology. 2022; 29(8):5457-5465. https://doi.org/10.3390/curroncol29080431
Chicago/Turabian StyleCaviglia, Gian Paolo, Aurora Nicolosi, Maria Lorena Abate, Patrizia Carucci, Chiara Rosso, Emanuela Rolle, Angelo Armandi, Serena Aneli, Antonella Olivero, Alessandra Risso, and et al. 2022. "Liver Cancer-Specific Isoform of Serine Protease Inhibitor Kazal for the Detection of Hepatocellular Carcinoma: Results from a Pilot Study in Patients with Dysmetabolic Liver Disease" Current Oncology 29, no. 8: 5457-5465. https://doi.org/10.3390/curroncol29080431
APA StyleCaviglia, G. P., Nicolosi, A., Abate, M. L., Carucci, P., Rosso, C., Rolle, E., Armandi, A., Aneli, S., Olivero, A., Risso, A., Ribaldone, D. G., Fermer, C., Saracco, G. M., Gaia, S., & Bugianesi, E. (2022). Liver Cancer-Specific Isoform of Serine Protease Inhibitor Kazal for the Detection of Hepatocellular Carcinoma: Results from a Pilot Study in Patients with Dysmetabolic Liver Disease. Current Oncology, 29(8), 5457-5465. https://doi.org/10.3390/curroncol29080431








