Prognostic and Predictive Relevance of Tumor-Infiltrating Lymphocytes in Squamous Cell Head–Neck Cancer Patients Treated with Radical Radiotherapy/Chemo-Radiotherapy
Abstract
:1. Introduction
2. Materials and Methods
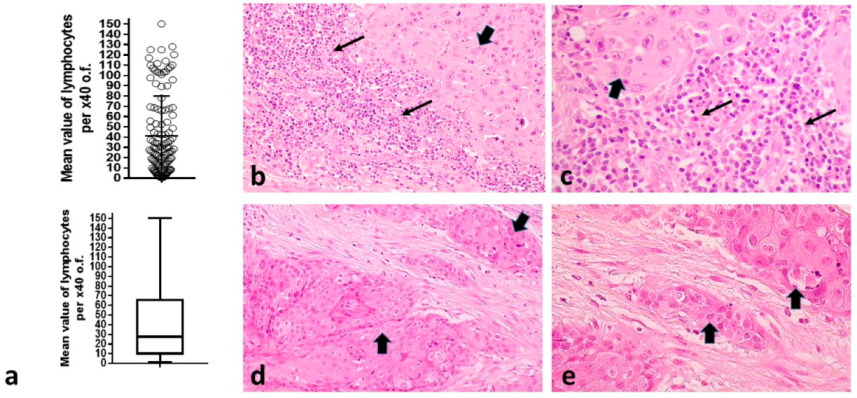
2.1. Assessment of TILs
2.2. Assessment of Hypoxia-Related Markers
2.3. Statistical Analysis
3. Results
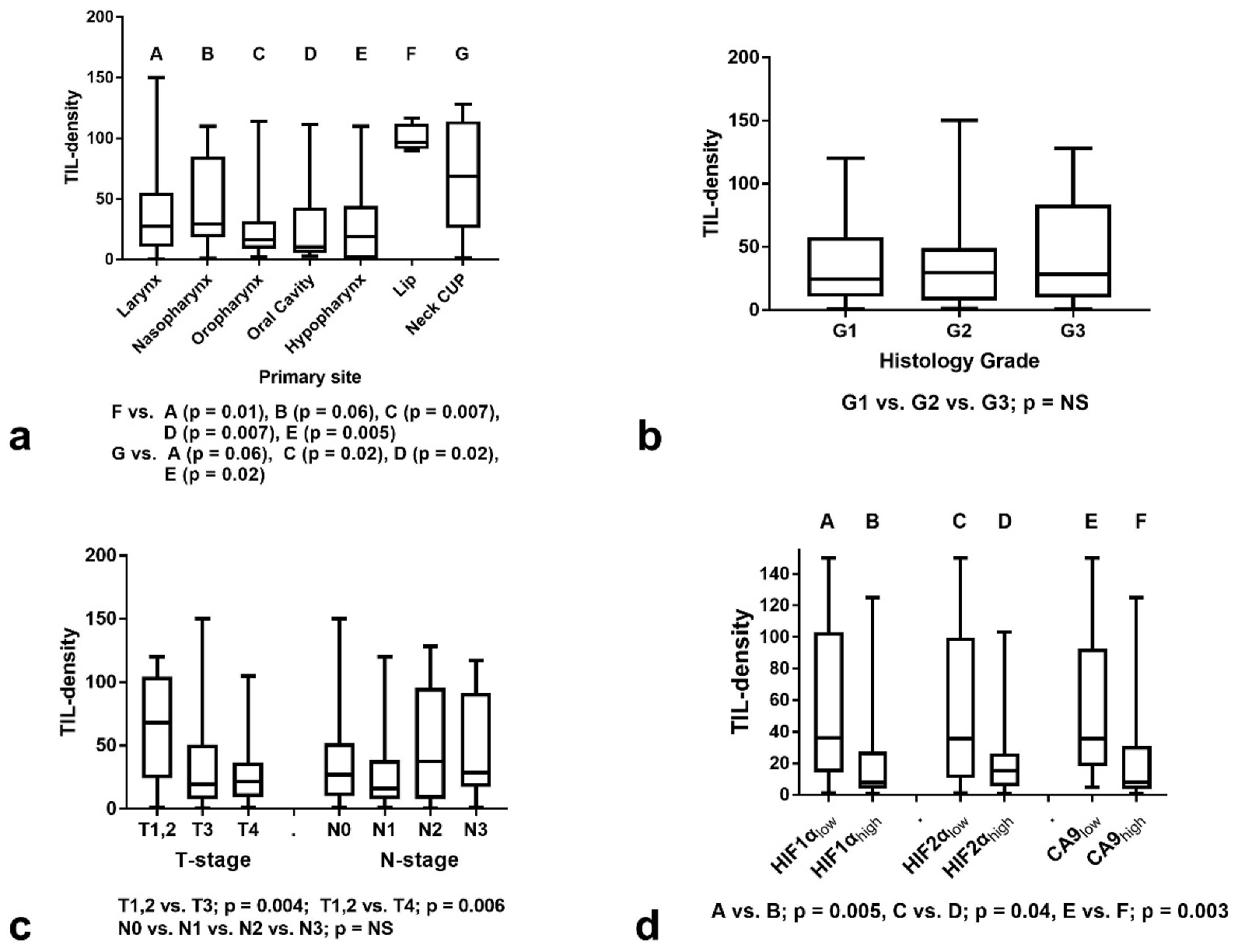
3.1. TILs and Histopathological Variables
3.2. TILs and Hypoxia Markers
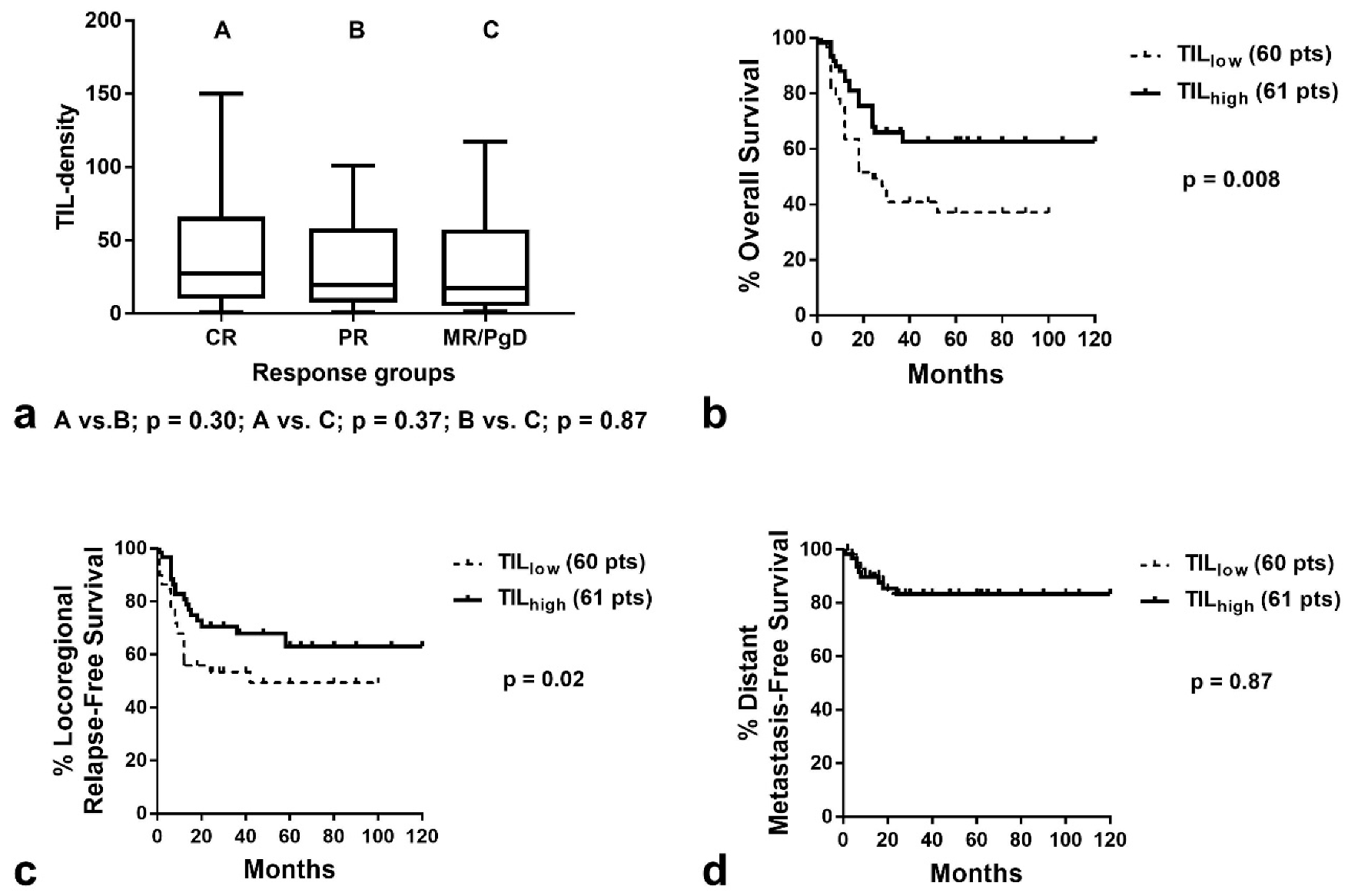
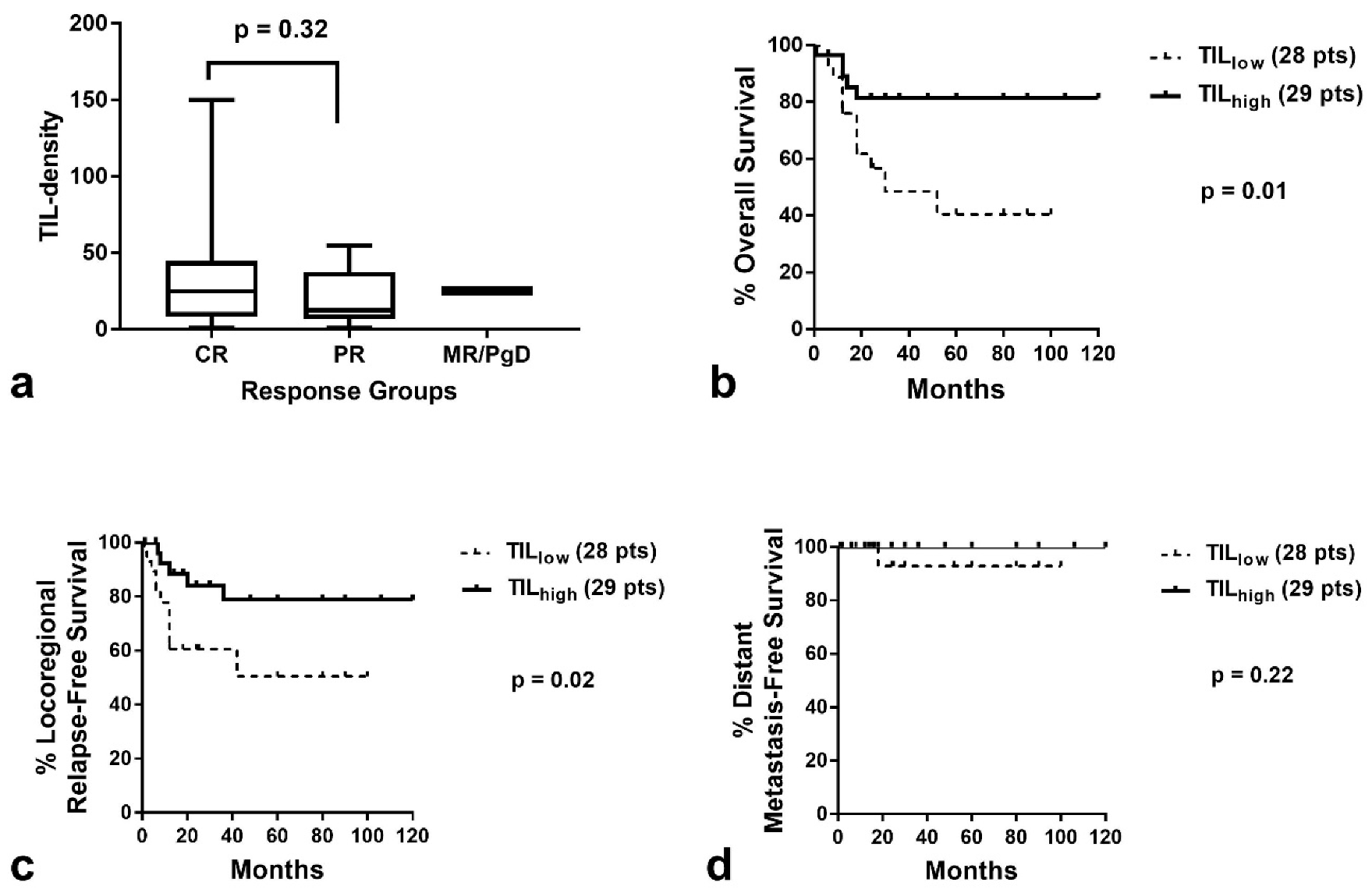
3.3. TILs and Response to Therapy
3.4. TILs and Patient Prognosis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shaw, R.; Beasley, N. Aetiology and Risk Factors for Head and Neck Cancer: United Kingdom National Multidisciplinary Guidelines. J. Laryngol. Otol. 2016, 130, S9–S12. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network. Available online: https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf (accessed on 15 May 2022).
- Keam, B.; Machiels, J.P.; Kim, H.R.; Licitra, L.; Golusinski, W.; Gregoire, V.; Lee, Y.G.; Belka, C.; Guo, Y.; Rajappa, S.J.; et al. Pan-Asian adaptation of the EHNS-ESMO-ESTRO Clinical Practice Guidelines for the diagnosis, treatment and follow-up of patients with squamous cell carcinoma of the head and neck. ESMO Open 2021, 6, 100309. [Google Scholar] [CrossRef] [PubMed]
- Tiwana, M.S.; Wu, J.; Hay, J.; Wong, F.; Cheung, W.; Olson, R.A. 25 year survival outcomes for squamous cell carcinomas of the head and neck: Population-based outcomes from a Canadian province. Oral Oncol. 2014, 50, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Birchall, M.; Bailey, D.; King, P. South West Cancer Intelligence Service Head and Neck Tumour Panel. Effect of process standards on survival of patients with head and neck cancer in the south and west of England. Br. J. Cancer 2004, 91, 1477–1481. [Google Scholar] [CrossRef] [PubMed]
- Fasano, M.; Corte, C.M.D.; Liello, R.D.; Viscardi, G.; Sparano, F.; Iacovino, M.L.; Paragliola, F.; Piccolo, A.; Napolitano, S.; Martini, G.; et al. Immunotherapy for head and neck cancer: Present and future. Crit. Rev. Oncol./Hematol. 2022, 174, 103679. [Google Scholar] [CrossRef]
- Zhang, F.; Manna, S.; Pop, L.M.; Chen, Z.J.; Fu, Y.X.; Hannan, R. Type I Interferon Response in Radiation-Induced Anti-Tumor Immunity. Semin. Radiat. Oncol. 2020, 30, 129–138. [Google Scholar] [CrossRef]
- Pevzner, A.M.; Tsyganov, M.M.; Ibragimova, M.K.; Litvyakov, N.V. Abscopal effect in the radio and immunotherapy. Radiat. Oncol. J. 2021, 39, 247–253. [Google Scholar] [CrossRef]
- Koukourakis, M.I.; Giatromanolaki, A. Tumor microenvironment, immune response and post-radiotherapy tumor clearance. Clin. Transl. Oncol. 2020, 22, 2196–2205. [Google Scholar] [CrossRef]
- Koukourakis, I.M.; Zygogianni, A.; Kouloulias, V.; Kyrgias, G.; Panteliadou, M.; Nanos, C.; Abatzoglou, I.; Koukourakis, M.I. Is Locally Advanced Head-Neck Cancer One More Candidate for Accelerated Hypofractionation? Anticancer Res. 2021, 41, 467–475. [Google Scholar] [CrossRef]
- Koukourakis, M.I.; Giatromanolaki, A.; Sivridis, E.; Simopoulos, C.; Turley, H.; Talks, K.; Gatter, K.C.; Harris, A.L. Hypoxia-inducible factor (HIF1A and HIF2A), angiogenesis, and chemo-radiotherapy outcome of squamous cell head-and-neck cancer. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 1192–1202. [Google Scholar] [CrossRef]
- Koukourakis, M.I.; Giatromanolaki, A.; Danielidis, V.; Sivridis, E. Hypoxia inducible factor (HIf1alpha and HIF2alpha) and carbonic anhydrase 9 (CA9) expression and response of head-neck cancer to hypofractionated and accelerated radiotherapy. Int. J. Radiat. Biol. 2008, 84, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Ager, A.; Watson, H.A.; Wehenkel, S.C.; Mohammed, R.N. Homing to solid cancers: A vascular checkpoint in adoptive cell therapy using CAR T-cells. Biochem. Soc. Trans. 2016, 44, 377–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calcinotto, A.; Filipazzi, P.; Grioni, M.; Iero, M.; De Milito, A.; Ricupito, A.; Cova, A.; Canese, R.; Jachetti, E.; Rossetti, M.; et al. Modulation of microenvironment acidity reverses anergy in human and murine tumor-infiltrating T lymphocytes. Cancer Res. 2012, 72, 2746–2756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Severin, T.; Muller, B.; Giese, G.; Uhl, B.; Wolf, B.; Hauschildt, S.; Kreutz, W. pH-dependent LAK cell cytotoxicity. Tumor Biol. 1994, 15, 304–310. [Google Scholar] [CrossRef]
- Giatromanolaki, A.; Kouroupi, M.; Pouliliou, S.; Mitrakas, A.; Hasan, F.; Pappa, A.; Koukourakis, M.I. Ectonucleotidase CD73 and CD39 expression in non-small cell lung cancer relates to hypoxia and immunosuppressive pathways. Life Sci. 2020, 259, 118389. [Google Scholar] [CrossRef]
- van Baren, N.; Van den Eynde, B.J. Tryptophan-degrading enzymes in tumoral immune resistance. Front. Immunol. 2015, 6, 34. [Google Scholar] [CrossRef] [Green Version]
- Vijayan, D.; Young, A.; Teng, M.W.L.; Smyth, M.J. Targeting immunosuppressive adenosine in cancer. Nat. Rev. Cancer 2017, 17, 709–724. [Google Scholar] [CrossRef]
- de Ruiter, E.J.; Ooft, M.L.; Devriese, L.A.; Willems, S.M. The prognostic role of tumor infiltrating T-lymphocytes in squamous cell carcinoma of the head and neck: A systematic review and meta-analysis. Oncoimmunology 2017, 6, e1356148. [Google Scholar] [CrossRef] [Green Version]
- Ruffini, E.; Asioli, S.; Filosso, P.L.; Lyberis, P.; Bruna, M.C.; Macrì, L.; Daniele, L.; Oliaro, A. Clinical significance of tumor-infiltrating lymphocytes in lung neoplasms. Ann. Thorac. Surg. 2009, 87, 365–372. [Google Scholar] [CrossRef]
- Adams, S.; Gray, R.J.; Demaria, S.; Goldstein, L.; Perez, E.A.; Shulman, L.N.; Martino, S.; Wang, M.; Jones, V.E.; Saphner, T.J.; et al. Prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancers from two phase III randomized adjuvant breast cancer trials: ECOG 2197 and ECOG 1199. J. Clin. Oncol. 2014, 32, 2959–2966. [Google Scholar] [CrossRef]
- Giatromanolaki, A.; Gkegka, A.G.; Pouliliou, S.; Biziota, E.; Kakolyris, S.; Koukourakis, M. Hypoxia and anaerobic metabolism relate with immunologically cold breast cancer and poor prognosis. Breast Cancer Res. Treat. 2022, 194, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Hendry, S.; Salgado, R.; Gevaert, T.; Russell, P.A.; John, T.; Thapa, B.; Christie, M.; van de Vijver, K.; Estrada, M.V.; Gonzalez-Ericsson, P.I.; et al. Assessing Tumor-Infiltrating Lymphocytes in Solid Tumors: A Practical Review for Pathologists and Proposal for a Standardized Method from the International Immuno-Oncology Biomarkers Working Group: Part 2: TILs in Melanoma, Gastrointestinal Tract Carcinomas, Non-Small Cell Lung Carcinoma and Mesothelioma, Endometrial and Ovarian Carcinomas, Squamous Cell Carcinoma of the Head and Neck, Genitourinary Carcinomas, and Primary Brain Tumors. Adv. Anat. Pathol. 2017, 24, 311–335. [Google Scholar] [CrossRef] [PubMed]
- De Keukeleire, S.J.; Vermassen, T.; De Meulenaere, A.; Deron, P.; Huvenne, W.; Duprez, F.; Creytens, D.; Van Dorpe, J.; Rottey, S.; Ferdinande, L. Tumour infiltrating lymphocytes in oropharyngeal carcinoma: Prognostic value and evaluation of a standardised method. Pathology 2021, 53, 836–843. [Google Scholar] [CrossRef] [PubMed]
- De Keukeleire, S.J.; Vermassen, T.; Deron, P.; Huvenne, W.; Duprez, F.; Creytens, D.; Van Dorpe, J.; Ferdinande, L.; Rottey, S. Concordance, Correlation, and Clinical Impact of Standardized PD-L1 and TIL Scoring in SCCHN. Cancers 2022, 14, 2431. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.Y.; Park, H.E.; Kim, J.H.; Wen, X.; Jeong, S.; Cho, N.Y.; Gwon, H.G.; Kim, K.; Lee, H.S.; Jeong, S.Y.; et al. Whole-Slide Image Analysis Reveals Quantitative Landscape of Tumor-Immune Microenvironment in Colorectal Cancers. Clin. Cancer Res. 2020, 26, 870–881. [Google Scholar] [CrossRef]
- Ou, D.; Adam, J.; Garberis, I.; Blanchard, P.; Nguyen, F.; Levy, A.; Casiraghi, O.; Gorphe, P.; Breuskin, I.; Janot, F.; et al. Clinical relevance of tumor infiltrating lymphocytes, PD-L1 expression and correlation with HPV/p16 in head and neck cancer treated with bio- or chemo-radiotherapy. Oncoimmunology 2017, 6, e1341030. [Google Scholar] [CrossRef] [Green Version]
- Balermpas, P.; Rödel, F.; Rödel, C.; Krause, M.; Linge, A.; Lohaus, F.; Baumann, M.; Tinhofer, I.; Budach, V.; Gkika, E.; et al. CD8+ tumour-infiltrating lymphocytes in relation to HPV status and clinical outcome in patients with head and neck cancer after postoperative chemo-radiotherapy: A multicentre study of the German cancer consortium radiation oncology group (DKTK-ROG). Int. J. Cancer 2016, 138, 171–181. [Google Scholar] [CrossRef]
- Fiedler, M.; Weber, F.; Hautmann, M.G.; Bohr, C.; Reichert, T.E.; Ettl, T. Infiltrating immune cells are associated with radiosensitivity and favorable survival in head and neck cancer treated with definitive radiotherapy. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 129, 612–620. [Google Scholar] [CrossRef]
- Balermpas, P.; Michel, Y.; Wagenblast, J.; Seitz, O.; Weiss, C.; Rödel, F.; Rödel, C.; Fokas, E. Tumour-infiltrating lymphocytes predict response to definitive chemo-radiotherapy in head and neck cancer. Br. J. Cancer 2014, 110, 501–509. [Google Scholar] [CrossRef]
- Suzuki, H.; Kawasaki, Y.; Miura, M.; Hatakeyama, H.; Shina, K.; Suzuki, S.; Yamada, T.; Suzuki, M.; Ito, A.; Omori, Y. Tumor Infiltrating Lymphocytes Are Prognostic Factors and Can Be Markers of Sensitivity to Chemoradiotherapy in Head and Neck Squamous Cell Carcinoma. Asian Pac. J. Cancer Prev. 2022, 23, 1271–1278. [Google Scholar] [CrossRef]
- Distel, L.V.; Fickenscher, R.; Dietel, K.; Hung, A.; Iro, H.; Zenk, J.; Nkenke, E.; Büttner, M.; Niedobitek, G.; Grabenbauer, G.G. Tumour infiltrating lymphocytes in squamous cell carcinoma of the oro- and hypopharynx: Prognostic impact may depend on type of treatment and stage of disease. Oral Oncol. 2009, 45, e167–e174. [Google Scholar] [CrossRef] [PubMed]
- Spector, M.E.; Bellile, E.; Amlani, L.; Zarins, K.; Smith, J.; Brenner, J.C.; Rozek, L.; Nguyen, A.; Thomas, D.; McHugh, J.B.; et al. Prognostic Value of Tumor-Infiltrating Lymphocytes in Head and Neck Squamous Cell Carcinoma. JAMA Otolaryngol. Head Neck Surg. 2019, 145, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Giatromanolaki, A.; Koukourakis, I.M.; Balaska, K.; Mitrakas, A.G.; Harris, A.L.; Koukourakis, M.I. Programmed death-1 receptor (PD-1) and PD-ligand-1 (PD-L1) expression in non-small cell lung cancer and the immune-suppressive effect of anaerobic glycolysis. Med. Oncol. 2019, 36, 76. [Google Scholar] [CrossRef] [PubMed]
- Luo, F.; Lu, F.T.; Cao, J.X.; Ma, W.J.; Xia, Z.F.; Zhan, J.H.; Zeng, K.M.; Huang, Y.; Zhao, H.Y.; Zhang, L. HIF-1α inhibition promotes the efficacy of immune checkpoint blockade in the treatment of non-small cell lung cancer. Cancer Lett. 2022, 531, 39–56. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakash, P.; Ai, M.; Liu, A.; Budhani, P.; Bartkowiak, T.; Sheng, J.; Ager, C.; Nicholas, C.; Jaiswal, A.R.; Sun, Y.; et al. Targeted hypoxia reduction restores T cell infiltration and sensitizes prostate cancer to immunotherapy. J. Clin. Investig. 2018, 128, 5137–5149. [Google Scholar] [CrossRef] [PubMed]
- Chafe, S.C.; McDonald, P.C.; Saberi, S.; Nemirovsky, O.; Venkateswaran, G.; Burugu, S.; Gao, D.; Delaidelli, A.; Kyle, A.H.; Baker, J.H.E.; et al. Targeting Hypoxia-Induced Carbonic Anhydrase IX Enhances Immune-Checkpoint Blockade Locally and Systemically. Cancer Immunol. Res. 2019, 7, 1064–1078. [Google Scholar] [CrossRef] [Green Version]
| No Patients | 121 |
| Age | |
| Range | 35–86 |
| Median | 66 |
| Performance Status | |
| 0 | 99 (81.8) |
| 1 | 22 (18.2) |
| Sex | |
| Male | 108 (89.2) |
| Female | 13 (10.8) |
| Primary Tumor Location | |
| Larynx | 57 (47.1) |
| Hypopharynx | 9 (7.4) |
| Oropharynx | 12 (9.9) |
| Oral cavity | 15 (12.4) |
| Nasopharynx | 13 (10.7) |
| Neck CUP * | 9 (7.5) |
| Parotid | 2 (1.7) |
| Lower Lip | 4 (3.3) |
| Differentiation | |
| Grade 1 | 38 (31.5) |
| Grade 2 | 30 (24.9) |
| Grade 3 | 53 (43.6) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koukourakis, I.M.; Gkegka, A.G.; Xanthopoulou, E.; Nanos, C.; Giatromanolaki, A.; Koukourakis, M.I. Prognostic and Predictive Relevance of Tumor-Infiltrating Lymphocytes in Squamous Cell Head–Neck Cancer Patients Treated with Radical Radiotherapy/Chemo-Radiotherapy. Curr. Oncol. 2022, 29, 4274-4284. https://doi.org/10.3390/curroncol29060342
Koukourakis IM, Gkegka AG, Xanthopoulou E, Nanos C, Giatromanolaki A, Koukourakis MI. Prognostic and Predictive Relevance of Tumor-Infiltrating Lymphocytes in Squamous Cell Head–Neck Cancer Patients Treated with Radical Radiotherapy/Chemo-Radiotherapy. Current Oncology. 2022; 29(6):4274-4284. https://doi.org/10.3390/curroncol29060342
Chicago/Turabian StyleKoukourakis, Ioannis M., Anastasia G. Gkegka, Erasmia Xanthopoulou, Christos Nanos, Alexandra Giatromanolaki, and Michael I. Koukourakis. 2022. "Prognostic and Predictive Relevance of Tumor-Infiltrating Lymphocytes in Squamous Cell Head–Neck Cancer Patients Treated with Radical Radiotherapy/Chemo-Radiotherapy" Current Oncology 29, no. 6: 4274-4284. https://doi.org/10.3390/curroncol29060342
APA StyleKoukourakis, I. M., Gkegka, A. G., Xanthopoulou, E., Nanos, C., Giatromanolaki, A., & Koukourakis, M. I. (2022). Prognostic and Predictive Relevance of Tumor-Infiltrating Lymphocytes in Squamous Cell Head–Neck Cancer Patients Treated with Radical Radiotherapy/Chemo-Radiotherapy. Current Oncology, 29(6), 4274-4284. https://doi.org/10.3390/curroncol29060342






