Artificial Intelligence System for Predicting Prostate Cancer Lesions from Shear Wave Elastography Measurements
Abstract
:1. Introduction
2. Aim of the Study
3. Material and Methods
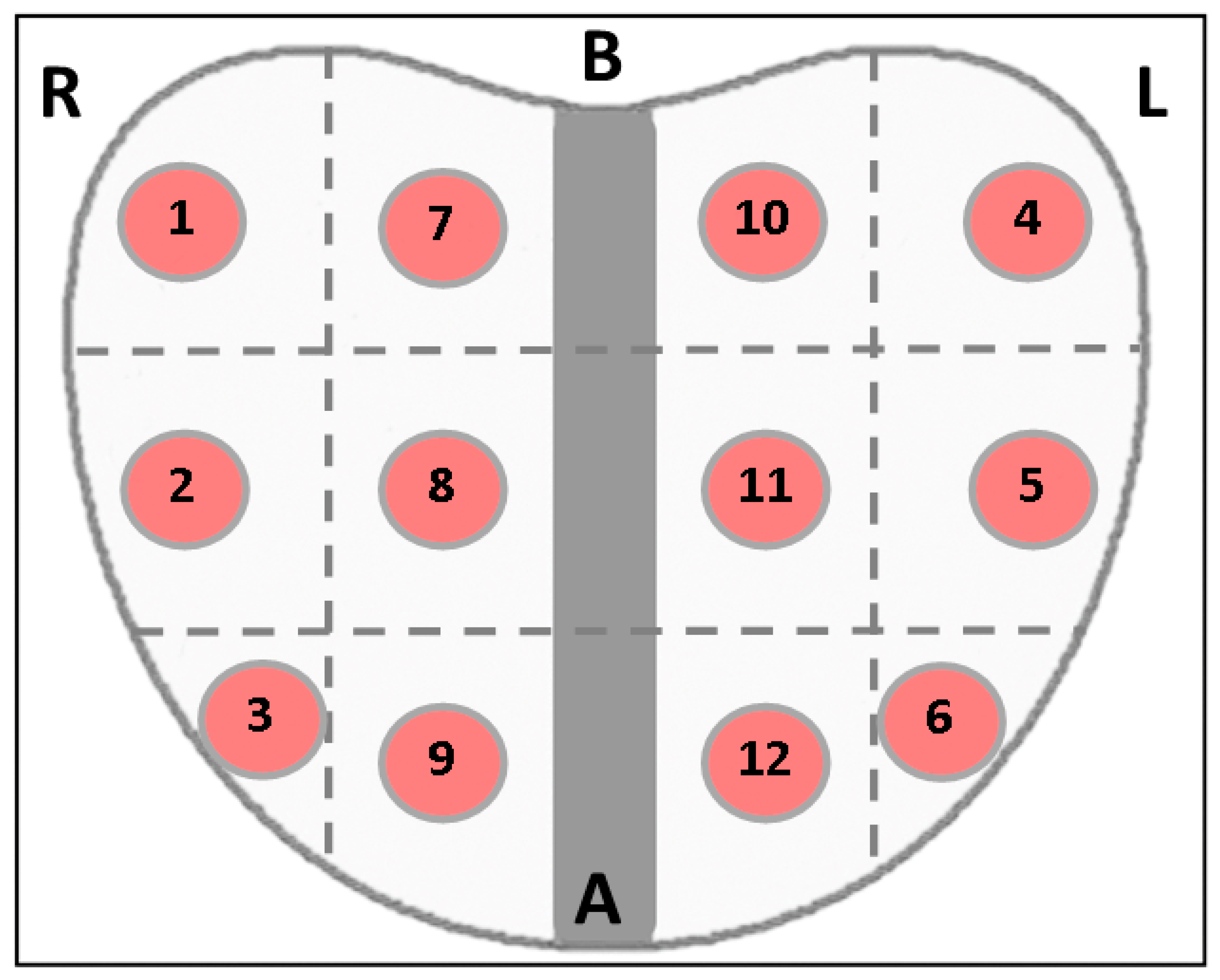
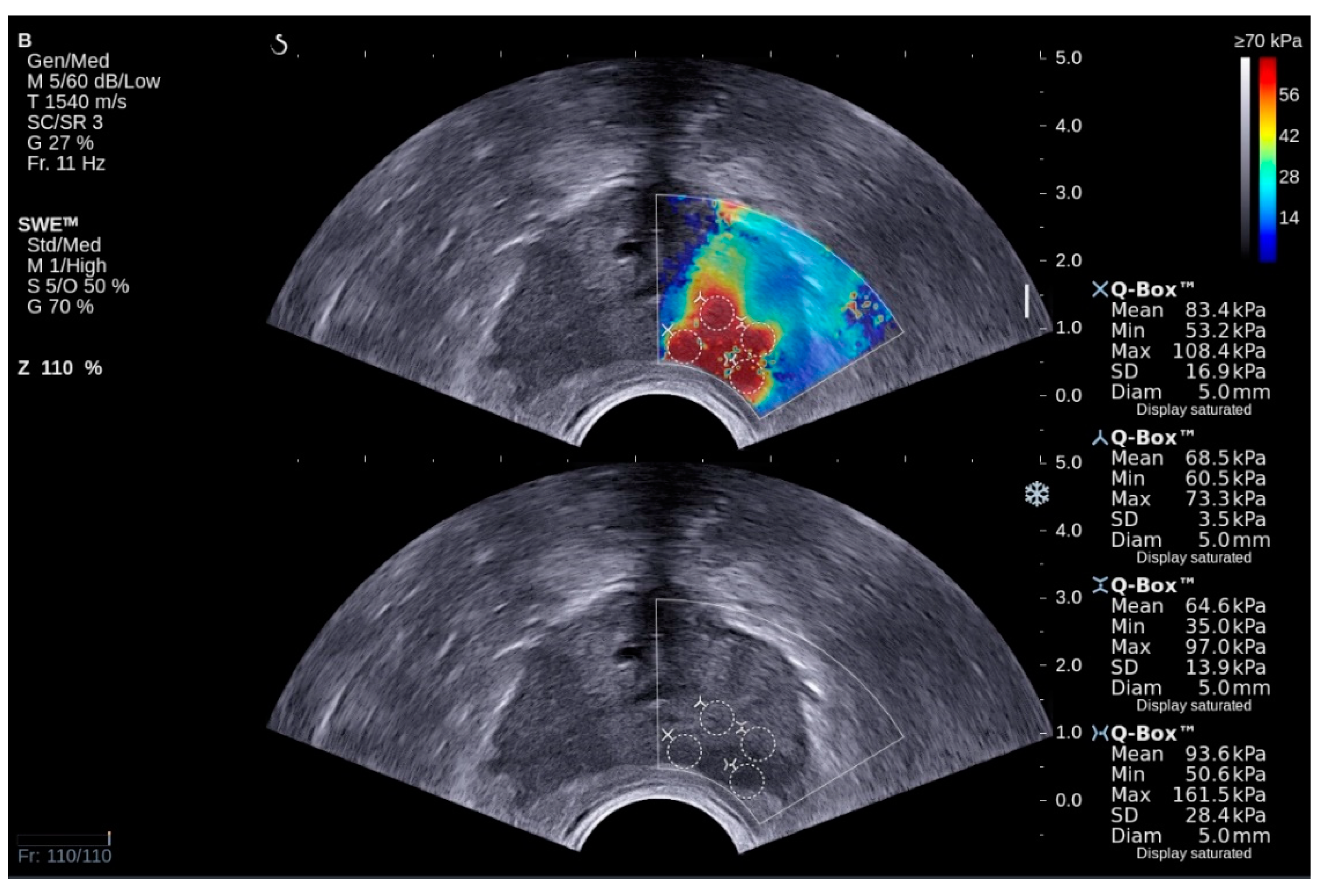
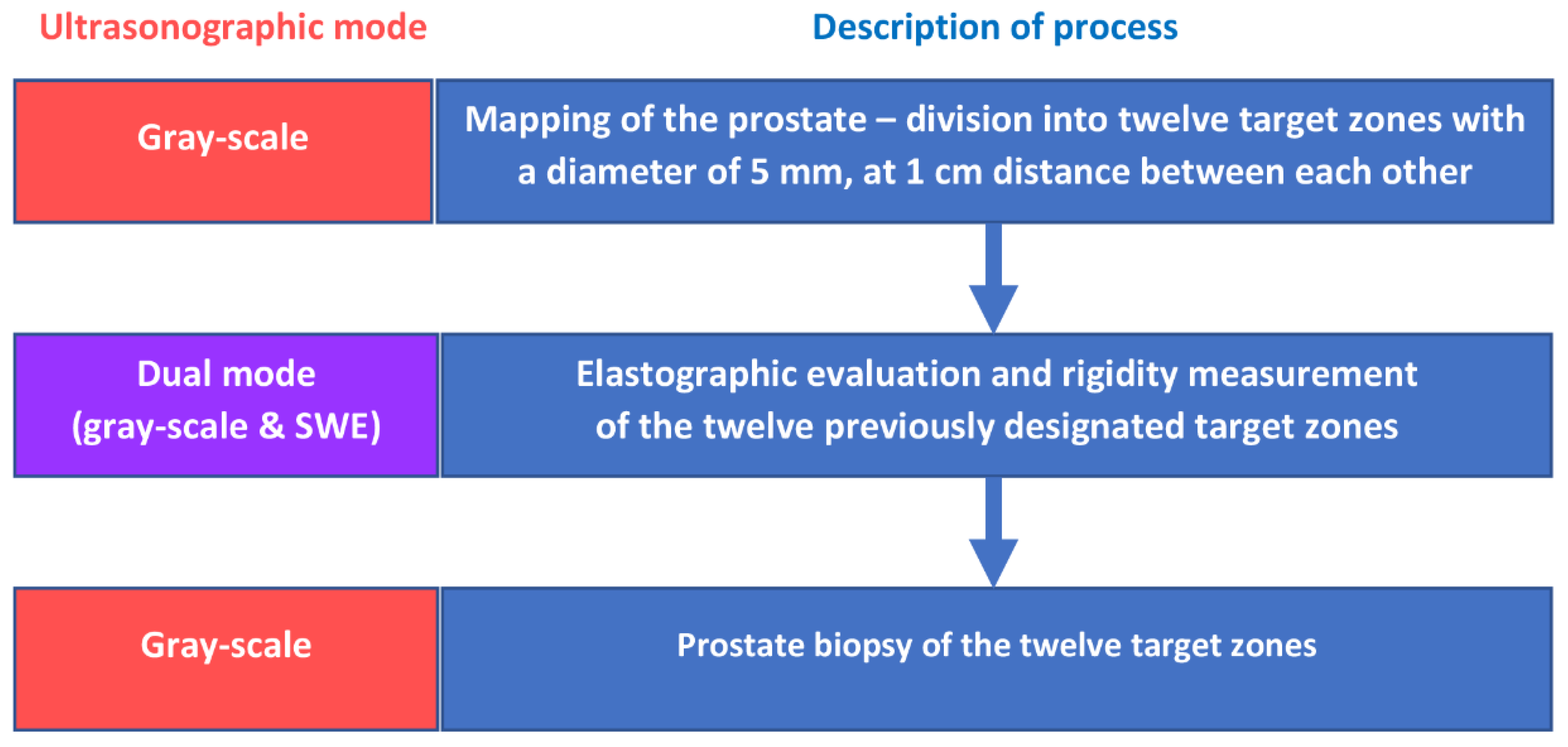
3.1. Methodology for Elastography
3.2. Methodology for Prostate Biopsy
3.3. Creation of the Dataset
3.4. Implementation of Machine Learning Techniques
3.5. Neural Network Classifier Implementation
3.6. Statistical Analysis
4. Results
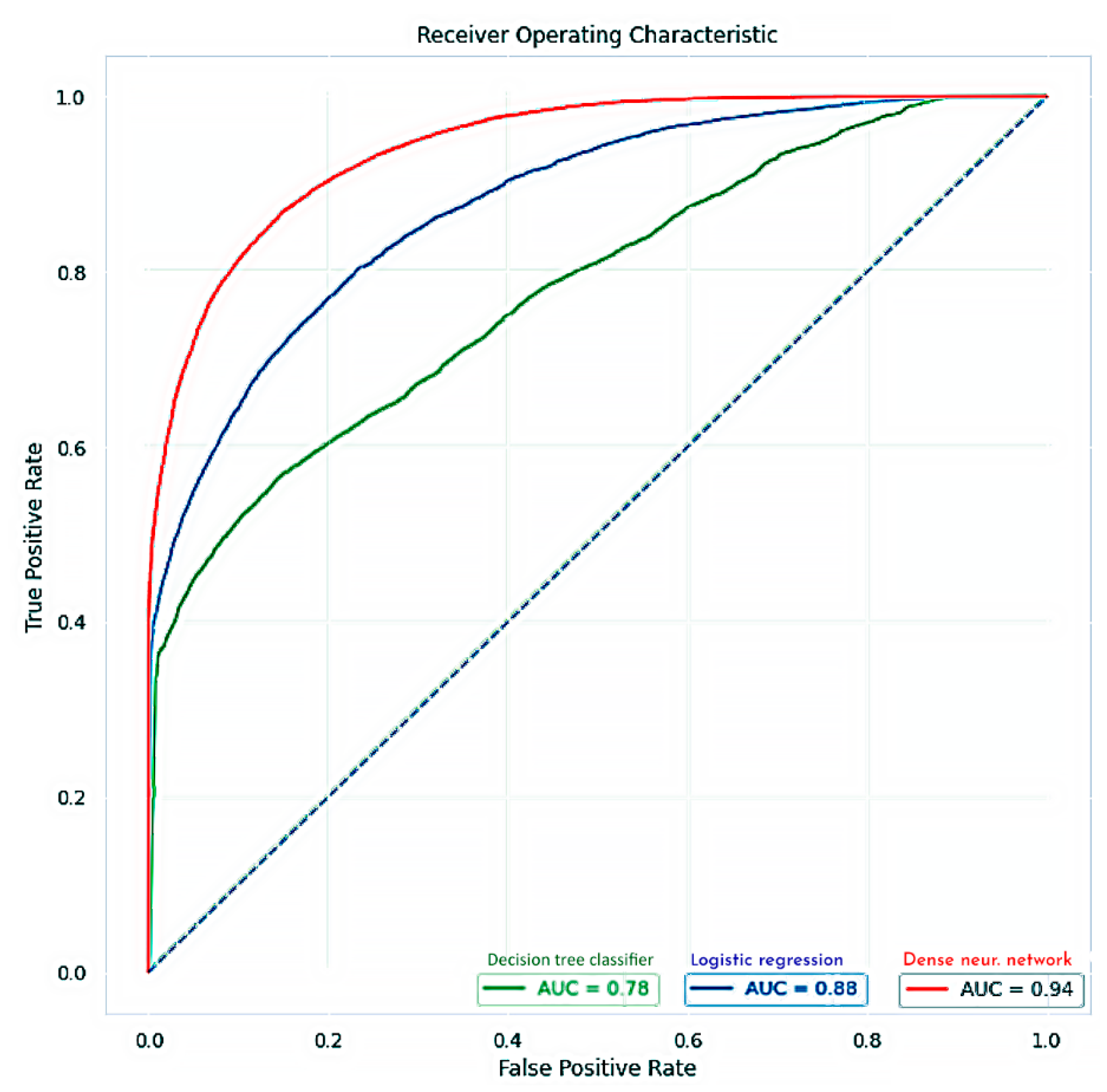
4.1. Re-Training of the Neural Network
4.2. Ensemble Learning Model
5. Discussion
6. Conclusions and Future Work
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Poppel, H.; Roobol, M.J.; Chapple, C.R.; Catto, J.W.; N’Dow, J.; Sønksen, J.; Stenzl, A.; Wirth, M. Prostate-specific Antigen Testing as Part of a Risk-Adapted Early Detection Strategy for Prostate Cancer: European Association of Urology Position and Recommendations for 2021. Eur. Urol. 2021, 80, 703–711. [Google Scholar] [CrossRef]
- Mazzone, E.; Stabile, A.; Pellegrino, F.; Basile, G.; Cignoli, D.; Cirulli, G.O.; Sorce, G.; Barletta, F.; Scuderi, S.; Bravi, C.A.; et al. Positive Predictive Value of Prostate Imaging Reporting and Data System Version 2 for the Detection of Clinically Significant Prostate Cancer: A Systematic Review and Meta-analysis. Eur. Urol. Oncol. 2021, 4, 697–713. [Google Scholar] [CrossRef]
- Itoh, A.; Ueno, E.; Tohno, E.; Kamma, H.; Takahashi, H.; Shiina, T.; Yamakawa, M.; Matsumura, T. Breast Disease: Clinical Application of US Elastography for Diagnosis. Radiology 2006, 239, 341–350. [Google Scholar] [CrossRef]
- Pallwein, L.; Mitterberger, M.; Struve, P.; Horninger, W.; Aigner, F.; Bartsch, G.; Gradl, J.; Schurich, M.; Pedross, F.; Frauscher, F. Comparison of sonoelastography guided biopsy with systematic biopsy: Impact on prostate cancer detection. Eur. Radiol. 2007, 17, 2278–2285. [Google Scholar] [CrossRef]
- König, K.; Scheipers, U.; Pesavento, A.; Lorenz, A.; Ermert, H.; Senge, T. Initial Experiences with Real-Time Elastography Guided Biopsies of the Prostate. J. Urol. 2005, 174, 115–117. [Google Scholar] [CrossRef]
- Barr, R.G.; Cosgrove, D.; Brock, M.; Cantisani, V.; Correas, J.M.; Postema, A.W.; Salomon, G.; Tsutsumi, M.; Xu, H.-X.; Dietrich, C.F. WFUMB Guidelines and Recommendations on the Clinical Use of Ultrasound Elastography: Part Prostate. Ultrasound Med. Biol. 2017, 43, 27–48. [Google Scholar] [CrossRef] [Green Version]
- Aigner, F.; Pallwein, L.; Junker, D.; Schäfer, G.; Mikuz, G.; Pedross, F.; Mitterberger, M.J.; Jaschke, W.; Halpern, E.J.; Frauscher, F. Value of Real-Time Elastography Targeted Biopsy for Prostate Cancer Detection in Men With Prostate Specific Antigen 1.25 ng/ml or Greater and 4.00 ng/ml or Less. J. Urol. 2010, 184, 913–917. [Google Scholar] [CrossRef]
- Giurgiu, C.R.; Manea, C.; Crişan, N.; Bungărdean, C.; Coman, I.; Dudea, S.M. Real-time sonoelastography in the diagnosis of prostate cancer. Med. Ultrason. 2011, 13, 5–9. [Google Scholar]
- Lorenz, A.; Ermert, H.; Sommerfeld, H.-J.; Garcia-Schürmann, M.; Senge, T.; Philippou, S. Ultraschall-Elastographie der Prostata. Ultrasound elastography of the prostate. A new technique for tumor detection. Ultraschall Med. 2000, 21, 8–15. [Google Scholar] [CrossRef] [Green Version]
- Kamoi, K.; Okihara, K.; Ochiai, A.; Ukimura, O.; Mizutani, Y.; Kawauchi, A.; Miki, T. The Utility of Transrectal Real-Time Elastography in the Diagnosis of Prostate Cancer. Ultrasound Med. Biol. 2008, 34, 1025–1032. [Google Scholar] [CrossRef] [PubMed]
- Tu, X.; Qiu, S.; Chang, T.; Jin, K.; Bao, Y.; Yang, L.; Wei, Q. The role of real-time elastography-targeted biopsy in the detection and diagnosis of prostate cancer. A systematic review and meta-analysis. Medicine 2018, 97, e0220. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.; Kim, S.Y.; Cho, J.Y.; Kim, S.H. Shear Wave Elastography for Detection of Prostate Cancer: A Preliminary Study. Korean J. Radiol. 2014, 15, 346–355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferraioli, G.; Barr, R.G.; Farrokh, A.; Radzina, M.; Cui, X.W.; Dong, Y.; Rocher, L.; Cantisani, V.; Polito, E.; D’Onofrio, M.; et al. How to perform shear wave elastography. Part II. Med. Ultrason. 2022, 24, 196. [Google Scholar] [CrossRef]
- Dudea, S.M.; Giurgiu, C.R.; Dumitriu, D.; Chiorean, A.; Ciurea, A.; Botar-Jid, C.; Coman, I. Value of ultrasound elastography in the diagnosis and management of prostate carcinoma. Med. Ultrason. 2011, 13, 45–53. [Google Scholar]
- Stenzl, A.; Sternberg, C.N.; Ghith, J.; Serfass, L.; Schijvenaars, B.J.; Sboner, A. Application of artificial intelligence to overcome clinical information overload in urological cancer. BJU Int. 2021; (ahead of print). [Google Scholar] [CrossRef]
- Mitchell, T. Machine Learning; McGraw-Hill: New York, NY, USA, 1997. [Google Scholar]
- Wang, H.; Zheng, B.; Yoon, S.W.; Ko, H.S. A support vector machine-based ensemble algorithm for breast cancer diagnosis. Eur. J. Oper. Res. 2017, 267, 687–699. [Google Scholar] [CrossRef]
- Lu, H.; Wang, H.; Yoon, S.W. A dynamic gradient boosting machine using genetic optimizer for practical breast cancer prognosis. Expert Syst. Appl. 2019, 116, 340–350. [Google Scholar] [CrossRef]
- Wang, Y.; Duggar, W.N.; Thomas, T.V.; Roberts, P.R.; Bian, L.; Wang, H. Extracapsular extension identification for head and neck cancer using multi-scale 3D deep neural network. In Proceedings of the 12th ACM Conference on Bioinformatics, Computational Biology, and Health Informatics, Gainesville, FL, USA, 1–4 August 2021; pp. 1–5. [Google Scholar] [CrossRef]
- Nagendran, M.; Chen, Y.; Lovejoy, C.A.; Gordon, A.; Komorowski, M.; Harvey, H.; Topol, E.J.; Ioannidis, J.P.A.; Collins, G.; Maruthappu, M. Artificial intelligence versus clinicians: Systematic review of design, reporting standards, and claims of deep learning studies. BMJ 2020, 368, m689. [Google Scholar] [CrossRef] [Green Version]
- Tătaru, O.; Vartolomei, M.; Rassweiler, J.; Virgil, O.; Lucarelli, G.; Porpiglia, F.; Amparore, D.; Manfredi, M.; Carrieri, G.; Falagario, U.; et al. Artificial Intelligence and Machine Learning in Prostate Cancer Patient Management—Current Trends and Future Perspectives. Diagnostics 2021, 11, 354. [Google Scholar] [CrossRef]
- Yan, R.; Chen, X.; Wang, P.; Onchis, D.M. Deep learning for fault diagnosis and prognosis in manufacturing systems. Comput. Ind. 2019, 110, 1–2. [Google Scholar] [CrossRef]
- Wang, Y.; Zamiela, C.; Thomas, T.V.; Duggar, W.N.; Roberts, P.R.; Bian, L.; Wang, H. 3D Texture Feature-Based Lymph Node Automated Detection in Head and Neck Cancer Analysis. In Proceedings of the 2020 IEEE International Conference on Bioinformatics and Biomedicine (BIBM), Seoul, Korea, 16–19 December 2020; pp. 2113–2119. [Google Scholar]
- Hameed, B.; Dhavileswarapu, A.S.; Raza, S.; Karimi, H.; Khanuja, H.; Shetty, D.; Ibrahim, S.; Shah, M.; Naik, N.; Paul, R.; et al. Artificial Intelligence and Its Impact on Urological Diseases and Management: A Comprehensive Review of the Literature. J. Clin. Med. 2021, 10, 1864. [Google Scholar] [CrossRef] [PubMed]
- Checcucci, E.; Autorino, R.; Cacciamani, G.E.; Amparore, D.; De Cillis, S.; Piana, A.; Piazzolla, P.; Vezzetti, E.; Fiori, C.; Veneziano, D.; et al. Uro-technology and SoMe Working Group of the Young Academic Urologists Working Party of the European Association of Urology. Artificial intelligence and neural networks in urology: Current clinical applications. Minerva Urol. E Nefrol. 2020, 72, 49–57. [Google Scholar] [CrossRef]
- Logistic Regression. Available online: https://scikit-learn.org/stable/modules/linear_model.html#logistic-regression (accessed on 19 July 2021).
- Naive Bayes. Available online: https://scikit-learn.org/stable/modules/naive_bayes.html (accessed on 19 July 2021).
- The Sequential Class. Available online: https://keras.io/api/models/sequential/ (accessed on 19 July 2021).
- TensorFlow. Available online: https://www.tensorflow.org/ (accessed on 19 July 2021).
- Keras. Available online: https://keras.io/ (accessed on 19 July 2021).
- Kingma, D.P.; Ba, J. Adam: A method for stochastic optimization. arXiv 2014, arXiv:1412.6980. [Google Scholar]
- Brown, C.D.; Davis, H.T. Receiver operating characteristics curves and related decision measures: A tutorial. Chemom. Intell. Lab. Syst. 2006, 80, 24–38. [Google Scholar] [CrossRef]
- Chawla, N.; Bowyer, K.; Hall, L.O.; Kegelmeyer, W.P. SMOTE: Synthetic minority over-sampling technique. J. Artif. Intel. Res. 2002, 16, 321–357. [Google Scholar] [CrossRef]
- Polikar, R. “Ensemble Learning” Ensemble Machine Learning; Springer: Boston, MA, USA, 2012; pp. 1–34. [Google Scholar]
- Rebuffi, S.-A.; Kolesnikov, A.; Sperl, G.; Lampert, C.H. iCaRL: Incremental Classifier and Representation Learning. In Proceedings of the 2017 IEEE Conference on Computer Vision and Pattern Recognition, Honolulu, HI, USA, 21–26 July 2017; pp. 5533–5542. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, A.; Thomas, S.; Oto, A. Prostate MR: Pitfalls and benign lesions. Abdom. Radiol. 2019, 45, 2154–2164. [Google Scholar] [CrossRef]
- Gold, S.A.; Hale, G.R.; Bloom, J.B.; Smith, C.P.; Rayn, K.N.; Valera, V.; Wood, B.J.; Choyke, P.L.; Turkbey, B.; Pinto, P.A. Follow-up of negative MRI-targeted prostate biopsies: When are we missing cancer? World J. Urol. 2018, 37, 235–241. [Google Scholar] [CrossRef]
- Kaneko, M.; Fukuda, N.; Nagano, H.; Yamada, K.; Yamada, K.; Konishi, E.; Sato, Y.; Ukimura, O. Artificial intelligence trained with integration of multiparametric MR-US imaging data and fusion biopsy trajectory-proven pathology data for 3D prediction of prostate cancer: A proof-of-concept study. Prostate 2022, 82, 793–803. [Google Scholar] [CrossRef]
- Correas, J.-M.; Tissier, A.-M.; Khairoune, A.; Khoury, G.; Eiss, D.; Hélénon, O. Ultrasound elastography of the prostate: State of the art. Diagn. Interv. Imaging 2013, 94, 551–560. [Google Scholar] [CrossRef] [Green Version]
- Woo, S.; Suh, C.H.; Kim, S.Y.; Cho, J.Y.; Kim, S.H. Shear-Wave Elastography for Detection of Prostate Cancer: A Systematic Review and Diagnostic Meta-Analysis. Am. J. Roentgenol. 2017, 209, 806–814. [Google Scholar] [CrossRef]
- Anbarasan, T.; Wei, C.; Bamber, J.; Barr, R.; Nabi, G. Characterisation of Prostate Lesions Using Transrectal Shear Wave Elastography (SWE) Ultrasound Imaging: A Systematic Review. Cancers 2021, 13, 122. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhao, X.; Shi, J.; Huang, Y. Value of shear wave elastography for diagnosis of primary prostate cancer: A systematic review and meta-analysis. Med. Ultrason. 2019, 21, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Mannaerts, C.K.; Wildeboer, R.R.; Remmers, S.; van Kollenburg, R.A.A.; Kajtazovic, A.; Hagemann, J.; Postema, A.W.; van Sloun, R.J.G.; Roobol, M.J.; Tilki, D.; et al. Multiparametric Ultrasound for Prostate Cancer Detection and Localization: Correlation of B-mode, Shear Wave Elastography and Contrast Enhanced Ultrasound with Radical Prostatectomy Specimens. J. Urol. 2019, 202, 1166–1173. [Google Scholar] [CrossRef] [PubMed]
- Local Surrogate (LIME). Available online: https://christophm.github.io/interpretable-ml-book/lime.html (accessed on 19 July 2021).
- Onchis, D.M.; Gillich, G.-R. Stable and explainable deep learning damage prediction for prismatic cantilever steel beam. Comput. Ind. 2021, 125, 103359. [Google Scholar] [CrossRef]
- Wei, C.; Zhang, Y.; Zhang, X.; Ageeli, W.; Szewczyk-Bieda, M.; Serhan, J.; Wilson, J.; Li, C.; Nabi, G. Prostate Cancer Gleason Score From Biopsy to Radical Surgery: Can Ultrasound Shear Wave Elastography and Multiparametric Magnetic Resonance Imaging Narrow the Gap? Front. Oncol. 2021, 11, 740724. [Google Scholar] [CrossRef] [PubMed]

| Layer (Type) | Output Shape | Parameter Number |
|---|---|---|
| dense_1 (Dense) | (None, 24) | 312 |
| dense_2 (Dense) | (None, 12) | 300 |
| dense_3 (Dense) | (None, 6) | 78 |
| dense_4 (Dense) | (None, 1) | 7 |
| No. | Mean Age | Mean PSA | % of Positive Cores | % of DRE Positive | |
|---|---|---|---|---|---|
| ISUP 1 | 90 | 63.21 | 10.915 | 34.2% | 16.7% |
| ISUP 2 | 14 | 61.07 | 13.586 | 41% | 28.6% |
| ISUP 3 | 61 | 64.83 | 17.776 | 47.3% | 50.8% |
| ISUP 4 | 31 | 60.64 | 23.127 | 59.1% | 80.6% |
| ISUP 5 | 27 | 71.88 | 46.383 | 68.8% | 85.2% |
| Classification Algorithm | Accuracy Score | Sensitivity | Specificity |
|---|---|---|---|
| Logistic regression | 0.8041 | 0.6163 | 0.9160 |
| Decision tree classifier | 0.6862 | 0.8490 | 0.4297 |
| Dense neural network | 0.8697 | 0.8550 | 0.8223 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Secasan, C.C.; Onchis, D.; Bardan, R.; Cumpanas, A.; Novacescu, D.; Botoca, C.; Dema, A.; Sporea, I. Artificial Intelligence System for Predicting Prostate Cancer Lesions from Shear Wave Elastography Measurements. Curr. Oncol. 2022, 29, 4212-4223. https://doi.org/10.3390/curroncol29060336
Secasan CC, Onchis D, Bardan R, Cumpanas A, Novacescu D, Botoca C, Dema A, Sporea I. Artificial Intelligence System for Predicting Prostate Cancer Lesions from Shear Wave Elastography Measurements. Current Oncology. 2022; 29(6):4212-4223. https://doi.org/10.3390/curroncol29060336
Chicago/Turabian StyleSecasan, Ciprian Cosmin, Darian Onchis, Razvan Bardan, Alin Cumpanas, Dorin Novacescu, Corina Botoca, Alis Dema, and Ioan Sporea. 2022. "Artificial Intelligence System for Predicting Prostate Cancer Lesions from Shear Wave Elastography Measurements" Current Oncology 29, no. 6: 4212-4223. https://doi.org/10.3390/curroncol29060336
APA StyleSecasan, C. C., Onchis, D., Bardan, R., Cumpanas, A., Novacescu, D., Botoca, C., Dema, A., & Sporea, I. (2022). Artificial Intelligence System for Predicting Prostate Cancer Lesions from Shear Wave Elastography Measurements. Current Oncology, 29(6), 4212-4223. https://doi.org/10.3390/curroncol29060336







