Newly Diagnosed Multifocal GBM: A Monocentric Experience and Literature Review
Abstract
Simple Summary
Abstract
1. Introduction
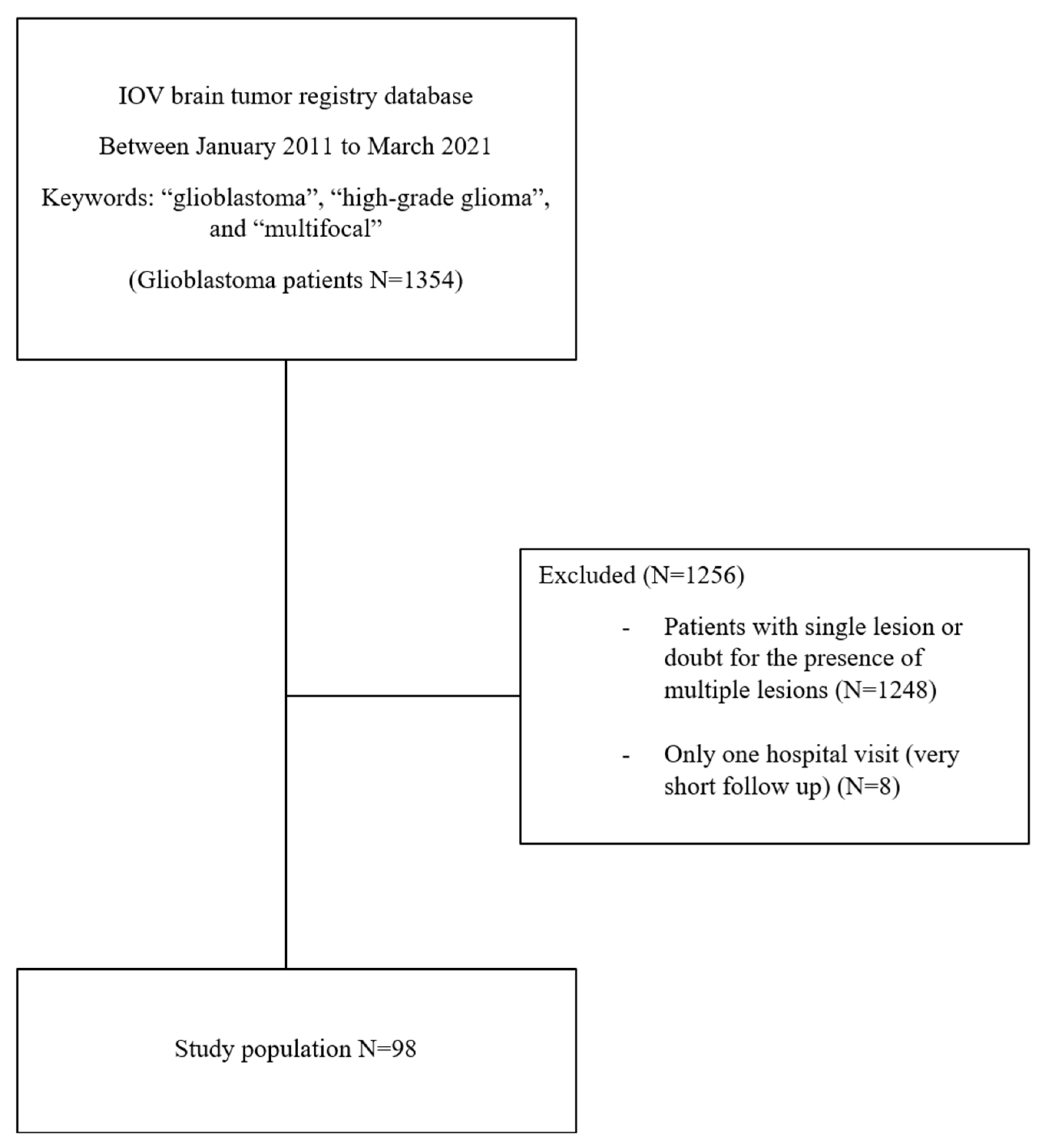
2. Materials and Methods
3. Results
3.1. Patient Characteristics
3.2. Response to Treatment
3.3. Progression and Survival
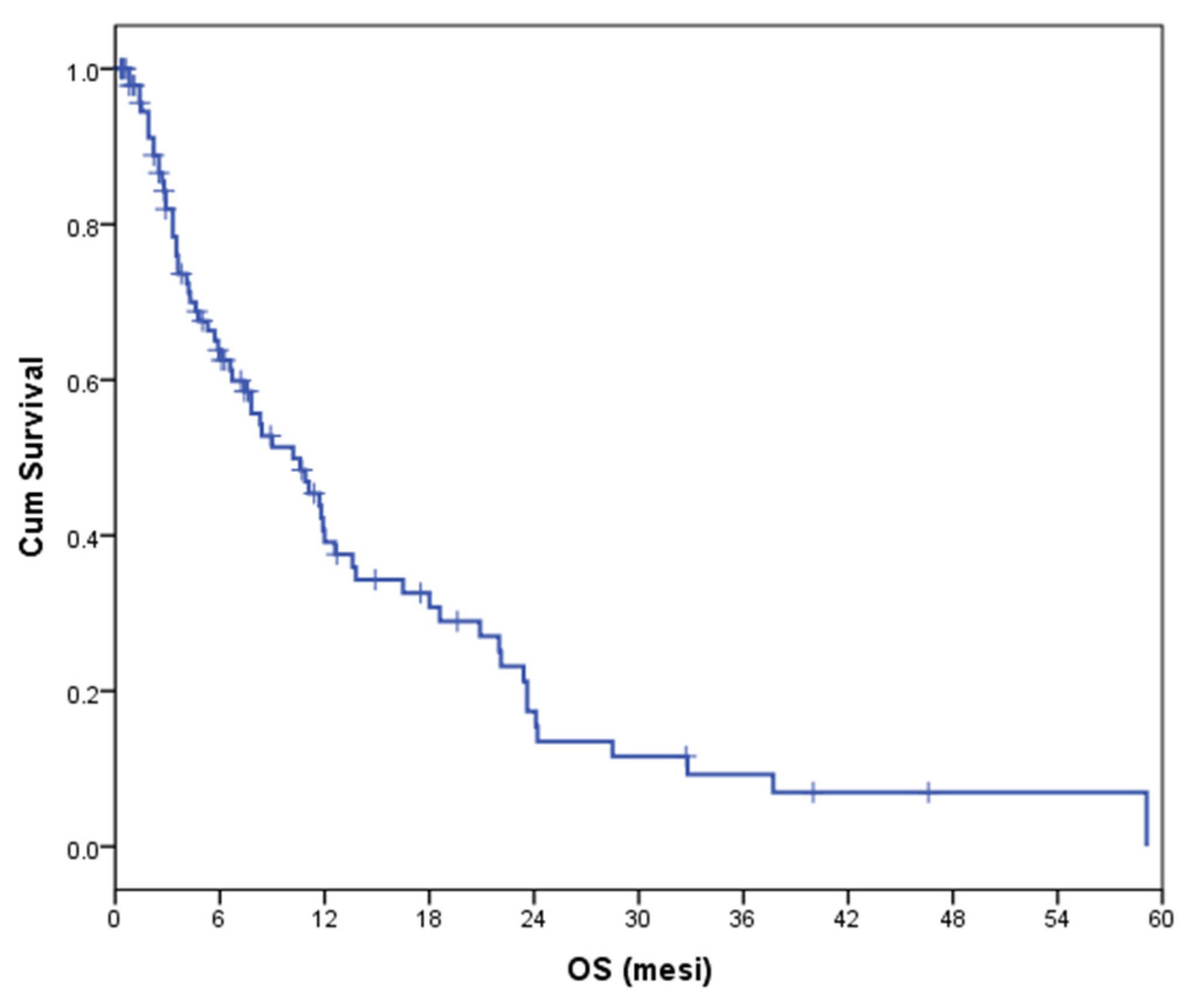
3.3.1. Overall Survival
3.3.2. Progression-Free Survival
3.4. Safety
4. Discussion
5. Conclusions
5.1. Strengths and Limitations
5.2. Clinical Implication and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ostrom, Q.T.; Patil, N.; Cioffi, G.; Waite, K.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2013–2017. Neuro-oncology 2020, 22, iv1–iv96. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Laws, J.E.R.; Parney, I.; Huang, W.; Anderson, J.F.A.; Morris, A.M.; Asher, A.; Lillehei, K.O.; Bernstein, M.; Brem, H.; Sloan, A.E.; et al. Survival following surgery and prognostic factors for recently diagnosed malignant glioma: Data from the Glioma Outcomes Project. J. Neurosurg. 2003, 99, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Hegi, M.E.; Diserens, A.C.; Gorlia, T.; Hamou, M.F.; De Tribolet, N.; Weller, M.; Stupp, R. MGMT Gene Silencing and Benefit from Temozolomide in Glioblastoma. N. Engl. J. Med. 2005, 352, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Salvati, M.; Caroli, E.; Orlando, E.R.; Frati, A.; Artizzu, S.; Ferrante, L. Multicentric glioma: Our experience in 25 patients and critical review of the literature. Neurosurg. Rev. 2003, 26, 275–279. [Google Scholar] [CrossRef]
- Dono, A.; Wang, E.; Lopez-Rivera, V.; Ramesh, A.V.; Tandon, N.; Ballester, L.Y.; Esquenazi, Y. Molecular characteristics and clinical features of multifocal glioblastoma. J. Neuro-Oncol. 2020, 148, 389–397. [Google Scholar] [CrossRef]
- Showalter, T.N.; Andrel, J.; Andrews, D.W.; Curran, W.J.; Daskalakis, C.; Werner-Wasik, M. Multifocal Glioblastoma Multiforme: Prognostic Factors and Patterns of Progression. Int. J. Radiat. Oncol. 2007, 69, 820–824. [Google Scholar] [CrossRef]
- Patil, C.G.; Yi, A.; Elramsisy, A.; Hu, J.; Mukherjee, D.; Irvin, D.K.; Yu, J.S.; Bannykh, S.I.; Black, K.L.; Nuño, M. Prognosis of patients with multifocal glioblastoma: A case-control study. J. Neurosurg. 2012, 117, 705–711. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, Y.; Li, W.; Wang, X.; Sawaya, R.; Lang, F.F.; Yung, W.K.A.; Chen, K.; Fuller, G.; Zhang, W. Genetic, epigenetic, and molecular landscapes of multifocal and multicentric glioblastoma. Acta Neuropathol. 2015, 130, 587–597. [Google Scholar] [CrossRef]
- Haque, W.; Thong, Y.; Verma, V.; Rostomily, R.; Butler, E.B.; Teh, B.S. Patterns of management and outcomes of unifocal versus multifocal glioblastoma. J. Clin. Neurosci. 2020, 74, 155–159. [Google Scholar] [CrossRef]
- Paulsson, A.K.; Holmes, J.A.; Peiffer, A.M.; Miller, L.D.; Liu, W.; Xu, J.; Hinson, W.H.; Lesser, G.J.; Laxton, A.W.; Tatter, S.B.; et al. Comparison of clinical outcomes and genomic characteristics of single focus and multifocal glioblastoma. J. Neuro-Oncol. 2014, 119, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Syed, M.; Liermann, J.; Verma, V.; Bernhardt, D.; Bougatf, N.; Paul, A.; Rieken, S.; Debus, J.; Adeberg, S. Survival and recurrence patterns of multifocal glioblastoma after radiation therapy. Cancer Manag. Res. 2018, 10, 4229–4235. [Google Scholar] [CrossRef] [PubMed]
- Kasper, J.; Hilbert, N.; Wende, T.; Fehrenbach, M.; Wilhelmy, F.; Jähne, K.; Frydrychowicz, C.; Hamerla, G.; Meixensberger, J.; Arlt, F. On the prognosis of multifocal glioblastoma: An evaluation incorporating volumetric MRI. Curr. Oncol. 2021, 28, 1437–1446. [Google Scholar] [CrossRef] [PubMed]
- Lasocki, A.; Gaillard, F.; Tacey, M.; Drummond, K.; Stuckey, S. Multifocal and multicentric glioblastoma: Improved characterisation with FLAIR imaging and prognostic implications. J. Clin. Neurosci. 2016, 31, 92–98. [Google Scholar] [CrossRef]
- Thomas, R.P.; Xu, L.W.; Lober, R.M.; Li, G.; Nagpal, S. The incidence and significance of multiple lesions in glioblastoma. J. Neuro-Oncol. 2013, 112, 91–97. [Google Scholar] [CrossRef]
- Batzdorf, U.; Malamud, N. The Problem of Multicentric Gliomas. J. Neurosurg. 1963, 20, 122–136. [Google Scholar] [CrossRef]
- Di, L.; Heath, R.N.; Shah, A.H.; Sanjurjo, A.D.; Eichberg, D.G.; Luther, E.M.; De La Fuente, M.I.; Komotar, R.J.; Ivan, M.E. Resection versus biopsy in the treatment of multifocal glioblastoma: A weighted survival analysis. J. Neuro-Oncol. 2020, 148, 155–164. [Google Scholar] [CrossRef]
- Lahmi, L.; Idbaih, A.; Del Campo, E.R.; Hoang-Xuan, K.; Mokhtari, K.; Sanson, M.; Canova, C.; Carpentier, A.; Jacob, J.; Maingon, P.; et al. Whole brain radiotherapy with concurrent temozolomide in multifocal and/or multicentric newly diagnosed glioblastoma. J. Clin. Neurosci. 2019, 68, 39–44. [Google Scholar] [CrossRef]
- Giannopoulos, S.; Kyritsis, A.P. Diagnosis and Management of Multifocal Gliomas. Oncology 2010, 79, 306–312. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Z.-X.; Huang, G.-H.; Xiang, Y.; Yang, L.; Pei, Y.-C.; Yang, W.; Lv, S.-Q. A systematic review of multifocal and multicentric glioblastoma. J. Clin. Neurosci. 2021, 83, 71–76. [Google Scholar] [CrossRef]
- Liu, Y.; Hao, S.; Yu, L.; Gao, Z. Long-term temozolomide might be an optimal choice for patient with multifocal glioblastoma, especially with deep-seated structure involvement: A case report and literature review. World J. Surg. Oncol. 2015, 13, 142. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A summary. Neuro-oncology 2021, 23, 1231–1251. [Google Scholar] [CrossRef]
- Kyritsis, A.P.; Levin, V.A.; Yung, W.A.; Leeds, N.E. Imaging patterns of multifocal gliomas. Eur. J. Radiol. 1993, 16, 163–170. [Google Scholar] [CrossRef]
- Weller, M.; van den Bent, M.; Preusser, M.; Le Rhun, E.; Tonn, J.C.; Minniti, G.; Bendszus, M.; Balana, C.; Chinot, O.; Dirven, L.; et al. EANO guidelines on the diagnosis and treatment of diffuse gliomas of adulthood. Nat. Rev. Clin. Oncol. 2020, 18, 170–186. [Google Scholar] [CrossRef] [PubMed]
- Ohgaki, H.; Kleihues, P. The Definition of Primary and Secondary Glioblastoma. Clin. Cancer Res. 2012, 19, 764–772. [Google Scholar] [CrossRef]
- Hassaneen, W.; Levine, N.B.; Suki, D.; Salaskar, A.L.; Lima, A.D.M.; McCutcheon, I.E.; Prabhu, S.S.; Lang, F.F.; Demonte, F.; Rao, G.; et al. Multiple craniotomies in the management of multifocal and multicentric glioblastoma. J. Neurosurg. 2011, 114, 576–584. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Reifenberger, G.; Von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef]
- Pirotte, B.; Goldman, S.; Massager, N.; David, P.; Wikler, D.; Vandesteene, A.; Salmon, I.; Brotchi, J.; Levivier, M. Comparison of 18F-FDG and 11C-methionine for PET-guided stereotactic brain biopsy of gliomas. J. Nucl. Med. 2004, 45, 1293–1298. [Google Scholar]
- Pirotte, B.J.; Levivier, M.; Goldman, S.; Massager, N.; Wikler, D.; Dewitte, O.; Bruneau, M.; Rorive, S.; David, P.; Brotchi, J. Positron emission tomography-guided volumetric resection of supratentorial high-grade gliomas. Neurosurgery 2009, 64, 471–481. [Google Scholar] [CrossRef]
- Pafundi, D.H.; Laack, N.N.; Youland, R.S.; Parney, I.; Lowe, V.J.; Giannini, C.; Kemp, B.J.; Grams, M.P.; Morris, J.M.; Hoover, J.M.; et al. Biopsy validation of 18F-DOPA PET and biodistribution in gliomas for neurosurgical planning and radiotherapy target delineation: Results of a prospective pilot study. Neuro-oncology 2013, 15, 1058–1067. [Google Scholar] [CrossRef]
- Crespo, I.; Vital, A.L.; Gonzalez-Tablas, M.; Patino, M.D.C.; Otero, A.; Lopes, M.C.; de Oliveira, C.; Domingues, P.; Orfao, A.; Tabernero, M.D. Molecular and Genomic Alterations in Glioblastoma Multiforme. Am. J. Pathol. 2015, 185, 1820–1833. [Google Scholar] [CrossRef] [PubMed]
- Rivera, A.L.; Pelloski, C.E.; Gilbert, M.R.; Colman, H.; De La Cruz, C.; Sulman, E.P.; Bekele, B.N.; Aldape, K.D. MGMT promoter methylation is predictive of response to radiotherapy and prognostic in the absence of adjuvant alkylating chemotherapy for glioblastoma. Neuro-oncology 2009, 12, 116–121. [Google Scholar] [CrossRef] [PubMed]
| Patient Characteristics (No. = 98) | ||
|---|---|---|
| Age—yr | ||
| Mean | 62 | |
| Median | 63 | |
| Range | 16–84 | |
| Gender—no. (%) | ||
| Male | 63 | (64.30%) |
| Female | 35 | (35.70%) |
| ECOG PS—no. (%) | ||
| 0–2 | 74 | (75.50%) |
| >2 | 15 | (15.30%) |
| Data missing | 9 | (9.20%) |
| Surgery—no. (%) | ||
| Yes | 73 | (74.50%) |
| No | 25 | (25.50%) |
| Extent of resection—no. out of 73 (%) | ||
| Biopsy | 26 | (35.60%) |
| Partial resection | 45 | (61.60%) |
| Gross total resection | 0 | (0%) |
| Not otherwise specified | 2 | (2.70%) |
| MGMT—no. out of 73 (%) | ||
| Data available | 59 | (80.80%) |
| Data missing | 14 | (19.20%) |
| MGMT status—no. out of 59 (%) | ||
| Methylated | 28 | (47.50%) |
| Unmethylated | 31 | (52.50%) |
| IDH—no. out of 73 (%) | ||
| Data available | 60 | (82.20%) |
| Data missing | 13 | (17.80%) |
| IDH 1–2 status—no. out of 60 (%) | ||
| Wild Type | 60 | (100%) |
| Mutated | 0 | (0.00%) |
| Active oncological treatment—no. (%) | ||
| Yes | 82 | (83.70%) |
| No | 16 | (16.30%) |
| Type of active oncological treatment—no. out of 82 (%) | ||
| RT + TMZ | 54 | (65.90%) |
| TMZ alone | 26 | (31.70%) |
| RT alone | 2 | (2.40%) |
| Cycles of temozolomide—no. | ||
| Median | 2 | |
| Range | 0–16 |
| Type of Response—No. (%) | Active Oncological Treatment (No. = 82) | RT + TMZ (No. = 54) | TMZ Alone (No. = 26) | RT Alone (No. = 2) |
|---|---|---|---|---|
| CR | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) |
| PR | 6 (7%) | 4 (7%) | 2 (8%) | 0 (0%) |
| SD | 38 (46%) | 30 (56%) | 8 (31%) | 0 (0%) |
| ORR (CR + PR) | 6 (7%) | 4 (7%) | 2 (8%) | 0 (0%) |
| DCR (CR + PR + SD) | 44 (54%) | 34 (63%) | 10 (38%) | 0 (0%) |
| PD | 38 (46%) | 20 (37%) | 16 (62%) | 2 (100%) |
| Total No. | No. of Events | Median OS | IC 95% | p Value | |
|---|---|---|---|---|---|
| Extent of resection | 0.028 | ||||
| PR | 45 | 31 | 13.8 | 6.0–21.6 | |
| Biopsy | 26 | 14 | 10.2 | 5.5–14.9 | |
| ECOG PS | 0.012 | ||||
| 0–2 | 74 | 49 | 11.7 | 9.9–13.5 | |
| >2 | 15 | 14 | 3.5 | 3.1–3.9 | |
| MGMT status | 0.138 | ||||
| Unmet | 31 | 18 | 12.6 | 10.0–15.2 | |
| Met | 28 | 19 | 20.9 | 6.4–35.4 | |
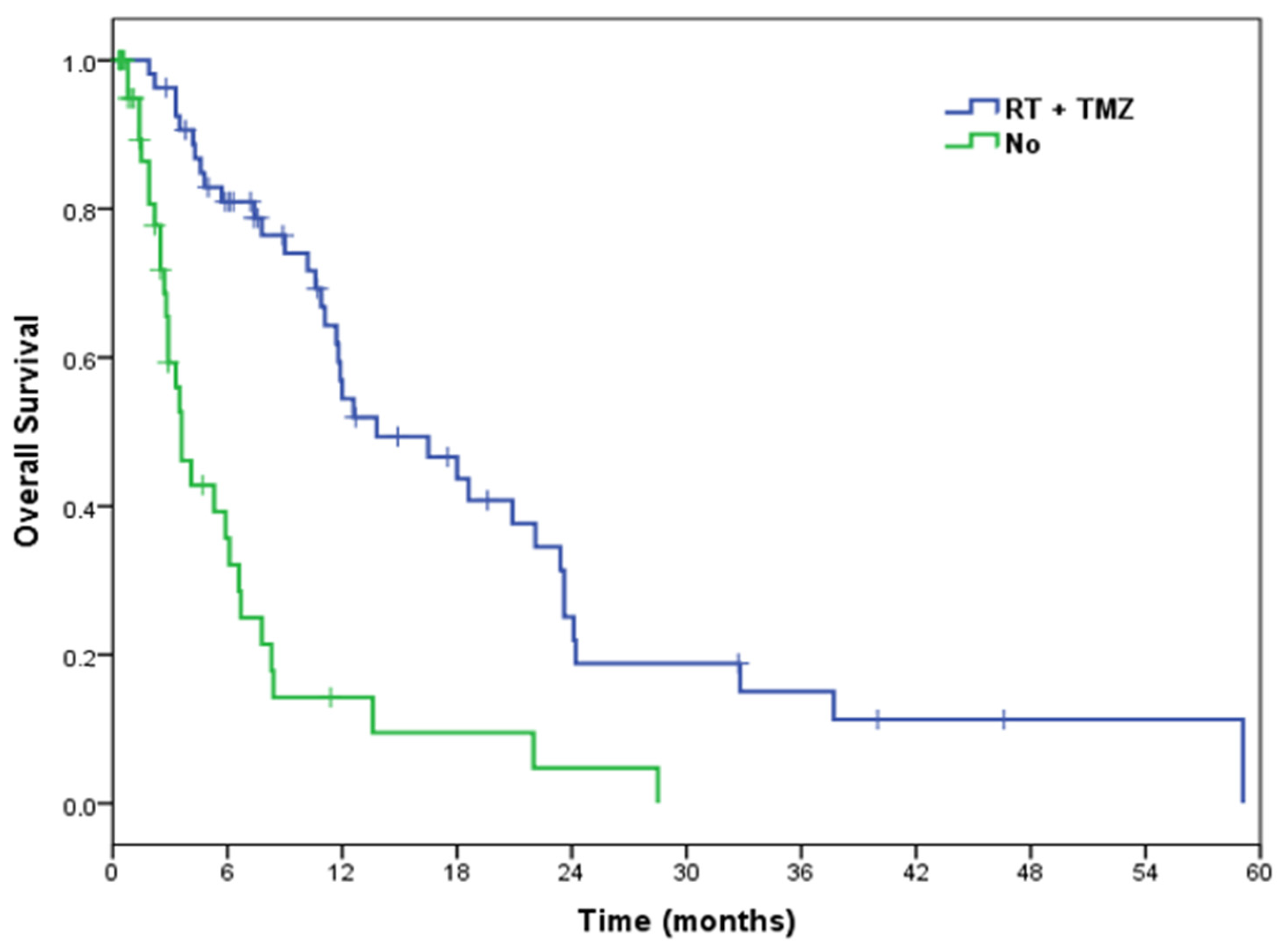
| Active oncological treatment | <0.001 | ||||
| Yes | 82 | 58 | 11.1 | 8.2–14.0 | |
| No | 16 | 8 | 1.9 | 0.7–3.1 | |
| RT + TMZ | <0.001 | ||||
| Yes | 54 | 36 | 13.8 | 6.9–20.7 | |
| No | 44 | 30 | 3.6 | 2.6–4.7 |
| B | Sig. | HR | IC 95% | |
|---|---|---|---|---|
| ECOG PS | 0.07 | 3 | 0.9–9.6 | |
| >2 vs. | 1.08 | |||
| 0–2 | 4 | |||
| MGMT | 0.075 | 2.1 | 0.9–5.0 | |
| Unmet vs. | 0.76 | |||
| Met | 2 | |||
| RT + TMZ | 0.014 | 3.1 | 1.3–7.7 | |
| No vs. | 1.13 | |||
| Yes | 6 |
| Authors | Incidence of mGBM (%) | No. of mGBM Patients |
|---|---|---|
| Salvati M. et al., 2003 [5] | 2 | 25 |
| Dono A et al., 2020 [6] | 9.9 | 39 |
| Showalter TN et al., 2007 [7] | 10 | 50 |
| Patil CG et al., 2012 [8] | 12.8 | 47 |
| Liu Q et al., 2015 [9] | 15.6 | 35 |
| Haque W et al., 2020 1 [10] | 17.2 | 7785 |
| Paulsson AK et al., 2014 [11] | 21 | 33 |
| Syed M et al., 2018 [12] | 24 | 63 |
| Kasper J et al. 2021 [13] | 29.5 | 54 |
| Lasocki A et al., 2016 [14] | 34 | 51 |
| Thomas RP et al., 2013 [15] | 35 | 67 |
| Present study | 7.2 | 98 |
| Authors | Median OS (Months) |
|---|---|
| Lasocki A et al., 2016 [14] | 6 |
| Patil CG et al., 2012 [8] | 6 |
| Liu Q et al., 2015 [9] | 6 |
| Showalter TN et al., 2007 [7] | 8.1 |
| Paulsson AK et al., 2014 [11] | 8.2 |
| Haque W et al., 2020 [10] | 8.3 |
| Lahmi L et al., 2019 [18] | 10 |
| Thomas RP et al., 2013 [15] | 10 |
| Syed M et al., 2018 [12] | 11.5 |
| Dono A et al., 2020 [6] | 13 |
| Present study | 10.2 |
| Authors | Predictive Factors | Explanations |
|---|---|---|
| Syed M et al., 2018 [12] | -RT + TMZ (OS univariate, OS multivariate) | NA |
| Haque W et al., 2020 [10] | -EOR (OS univariate) -RT + TMZ (OS univariate, OS multivariate) -MGMT status (OS univariate, OS multivariate) | EOR: gross total resection vs. subtotal resection vs. biopsy. RT + TMZ: analysis conducted on a subpopulation of biopsied mGBMs. MGMT status: analysis conducted on a population of both multifocal and unifocal glioblastomas. |
| Patil CG et al., 2012 [8] | -EOR (OS univariate) -KPS (OS univariate) | EOR: gross total or near gross total vs. partial resection or biopsy. KPS: ≥70 vs. <70. |
| Paulsson AK et al., 2014 [11] | -KPS (PFS univariate) | KPS: ≥80 vs. <80. |
| Di L et al., 2020 [17] | -EOR (OS univariate, PFS multivariate) | EOR: resection vs. biopsy. |
| Showalter TN et al., 2007 [7] | -KPS (PFS univariate PFS, PFS multivariate, OS univariate, OS multivariate) -EOR (PFS multivariate, OS univariate) -RT + TMZ (PFS multivariate, OS univariate) | KPS: ≥70 vs. <70 EOG: gross total resection vs. biopsy. |
| Thomas RP et al., 2013 [15] | -KPS (OS multivariate, PFS multivariate) -EOR (OS multivariate, PFS multivariate) -MGMT status (OS multivariate, PFS multivariate) | EOR: gross total resection vs. less extent of resection. |
| Kasper J et al., 2021 [13] | -EOR (OS univariate) -RT + TMZ (OS univariate, OS multivariate) | EOR: ratio of residual tumor volume (contrast-enhancing tumor volume assessed from subtraction sequences of T1 MR imaging with and without contrast) and initial tumor volume (sum of contrast-enhancing tumor volume and pre-operative necrosis volume) in percent. |
| Present study | -PS (OS univariate) -EOR (OS univariate, PFS univariate) -Active treatment (OS univariate, PFS univariate) -RT + TMZ (OS univariate, OS multivariate OS, PFS univariate) | PS: >2 vs. 0–2. EOR: partial resection vs. biopsy. Active treatment: RT + TMZ, TMZ alone, RT alone vs. palliative care. |
| Authors | Surgical Resection % | Explanation |
|---|---|---|
| Syed M et al., 2018 [12] | - 14% Total - 38% Subtotal - 48% Biopsy | NA |
| Dono A et al., 2020 [6] | Multifocal GBM: - 30% Gross total - 0% Near total - 50% Subtotal - 20% Biopsy Multicentric GBM: - 5% Gross total - 5% Near total - 80% Subtotal - 10% Biopsy | NA |
| Haque W et al., 2020 [10] | - 25.8% Gross total - 35.1% Subtotal - 34% Biopsy - 5.2% Surgery not otherwise specified | NA |
| Lahmi L et al., 2019 [18] | - 18% Partial - 82% Biopsy | NA |
| Patil CG et al., 2012 [8] | - 8.5% Gross total - 4.3% Near gross total - 29.8% Partial - 57.4% Biopsy | Gross-total: all of the enhancing tumor resected. Near gross total: >95% of the enhancing tumor resected. Partial: <95% of the enhancing tumor resected. Biopsy: procedure only. |
| Paulsson AK et al.l, 2014 [11] | Multifocal GBM: - 24% Gross total - 48% Subtotal - 28% Biopsy Multicentric GBM: - 12.5% Gross total - 25% Subtotal - 63.5% Biopsy | Gross total: all enhancing tumor removed. Subtotal: greater than 10% of the tumor mass debulked, but surgery was not a GTR. Biopsy: less than 10% of the tumor was debulked. |
| Di L et al., 2020 [17] | - 47.1% Resection: - 25% Resection ≥ 85% - 56.3% Resection < 85% - 18.7% Resection not otherwise specified - 52.9% Biopsy | Resection: removal of the largest contrast-enhancing lesion. |
| Showalter TN et al., 2007 [7] | - 12% Gross total - 66% Subtotal - 22% Biopsy | NA |
| Thomas RP et al., 2013 [15] | Multifocal - 26% Gross total - 28% Subtotal - 47% Biopsy Multicentric - 0% Gross total - 11% Subtotal - 89% Biopsy | NA |
| Liu Q et al., 2015 [9] | - 90% Resection - 10% Biopsy | NA |
| Present study | - 0% Gross total - 61.6% Partial - 35.5% Biopsy - 2.7% Surgery not otherwise specified | Gross total: removal of all enhancing areas on MRI. Partial: ranging from less than 100% of resection to more than 10% of enhancing areas on MRI. Biopsy: resection of less than 10% of the enhancing lesions (open biopsy, stereotactic biopsy). |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baro, V.; Cerretti, G.; Todoverto, M.; Della Puppa, A.; Chioffi, F.; Volpin, F.; Causin, F.; Busato, F.; Fiduccia, P.; Landi, A.; et al. Newly Diagnosed Multifocal GBM: A Monocentric Experience and Literature Review. Curr. Oncol. 2022, 29, 3472-3488. https://doi.org/10.3390/curroncol29050280
Baro V, Cerretti G, Todoverto M, Della Puppa A, Chioffi F, Volpin F, Causin F, Busato F, Fiduccia P, Landi A, et al. Newly Diagnosed Multifocal GBM: A Monocentric Experience and Literature Review. Current Oncology. 2022; 29(5):3472-3488. https://doi.org/10.3390/curroncol29050280
Chicago/Turabian StyleBaro, Valentina, Giulia Cerretti, Michela Todoverto, Alessandro Della Puppa, Franco Chioffi, Francesco Volpin, Francesco Causin, Fabio Busato, Pasquale Fiduccia, Andrea Landi, and et al. 2022. "Newly Diagnosed Multifocal GBM: A Monocentric Experience and Literature Review" Current Oncology 29, no. 5: 3472-3488. https://doi.org/10.3390/curroncol29050280
APA StyleBaro, V., Cerretti, G., Todoverto, M., Della Puppa, A., Chioffi, F., Volpin, F., Causin, F., Busato, F., Fiduccia, P., Landi, A., d’Avella, D., Zagonel, V., Denaro, L., & Lombardi, G. (2022). Newly Diagnosed Multifocal GBM: A Monocentric Experience and Literature Review. Current Oncology, 29(5), 3472-3488. https://doi.org/10.3390/curroncol29050280







