Early-Age-Onset Colorectal Cancer in Canada: Evidence, Issues and Calls to Action
Abstract
:1. Introduction
1.1. Symposium Objectives
- To summarize evidence and educate on the issues related to EAO-CRC;
- To prioritize responses to address issues related to EAO-CRC in Canada; and
- To influence practice change amongst primary care physicians or providers to support the early diagnosis of EAO-CRC.
1.2. Symposium Organization
1.3. Participants
1.4. Agenda
2. Evidence Review
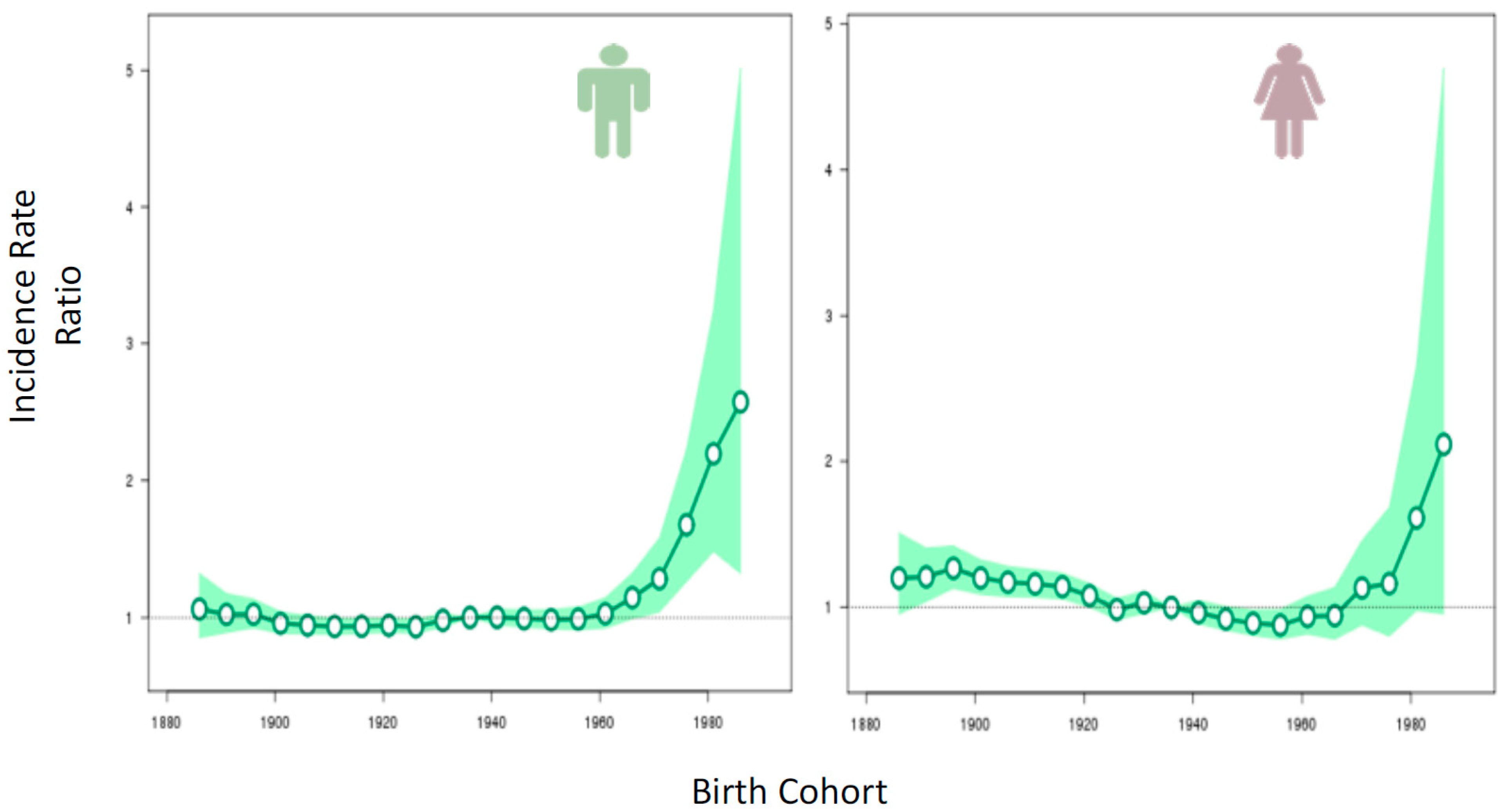
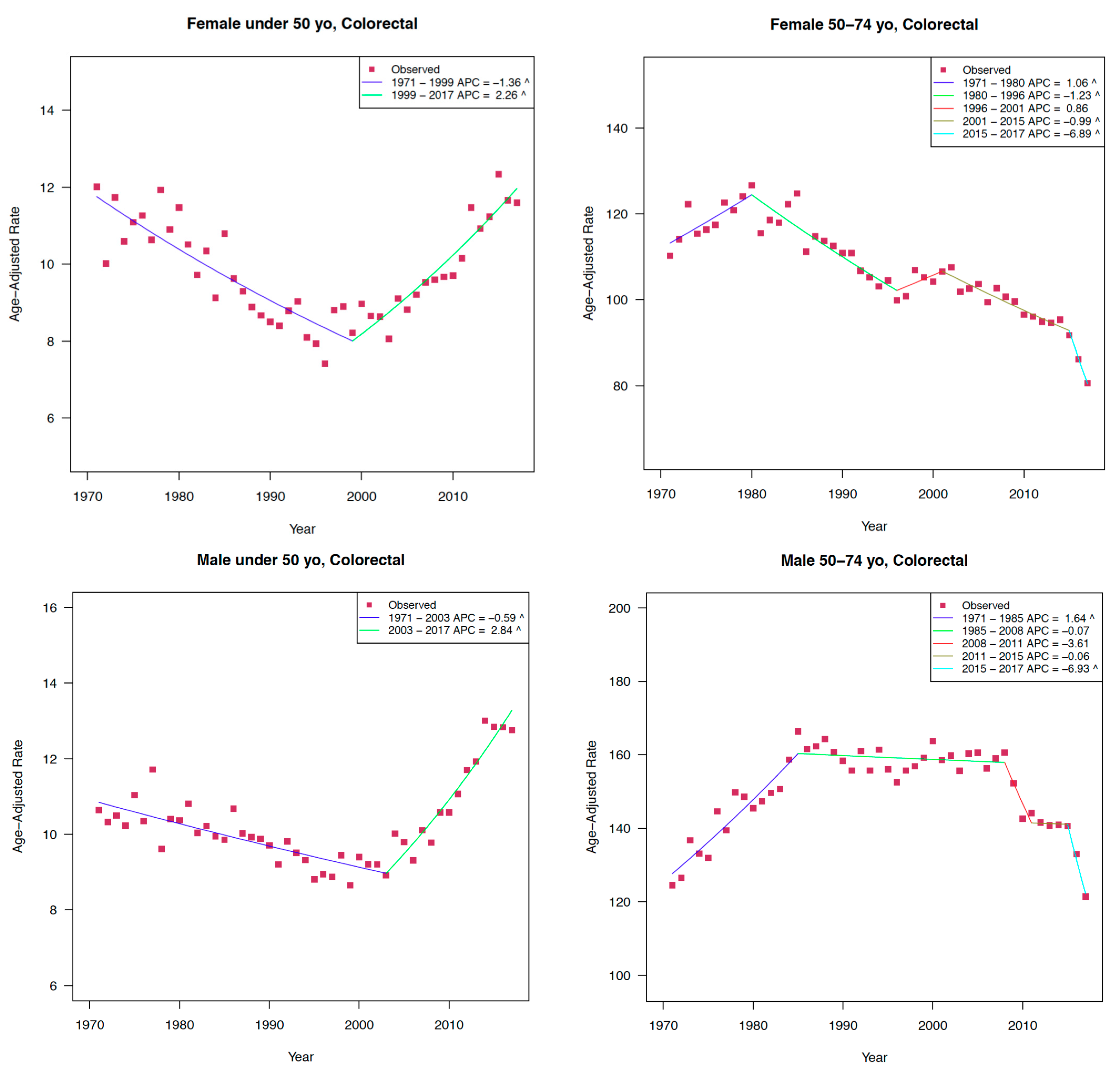
2.1. Incidence
2.2. Tumour Characteristics
2.3. Genetics and Genomics
- More microsatellite instability (MSI-high);
- Fewer BRAF mutations and mutations in MAPK gene pathways;
- More chromosomal instability;
- More copy number alterations;
- Greater long interspersed nuclear element-1 (LINE-1) DNA hypomethylation;
- Less DNA hypermethylation in promoter regions;
- More MSI/immune or canonical APC/B-catenin subtypes;
- Less metabolic or mesenchymal subtypes.
2.4. Primary Prevention: Risk Factor Identification
- Lynch syndrome, also called hereditary nonpolyposis colorectal cancer (HNPCC);
- Familial adenomatous polyposis (FAP);
- Attenuated familial adenomatous polyposis (AFAP), a subtype of FAP;
- Gardner syndrome, a subtype of FAP;
- Juvenile polyposis syndrome (JPS);
- Muir–Torre syndrome, a subtype of Lynch syndrome;
- MYH-associated polyposis (MAP);
- Peutz–Jeghers syndrome (PJS);
- Turcot syndrome, a subtype of FAP and Lynch syndrome.
- Moderate evidence was found for male sex, obesity and alcohol consumption;
- Suggestive evidence was found for Caucasian ethnicity and cigarette smoking;
- Emerging evidence was found for the role of other risk factors including:
- o
- Comorbidities: hyperlipidemia, diabetes, ulcerative colitis, hypertension, metabolic syndrome, and chronic kidney disease;
- o
- Lifestyle factors: sedentary behaviour, processed meat, insufficient fruit and vegetable consumption, and insufficient fish consumption;
- o
- Micronutrients/medications: aspirin use, B-carotene, vitamin C, vitamin E, and folate all potentially lower risk;
- o
- Occupational exposure to organic dust.
- Examine associations of potential risk factors with the development of EAO-CRC;
- Compare the magnitude of risk associated with different exposures between early and later-onset CRC (60+);
- Identify combinations of risk factors that are associated with the highest risk of developing EAO-CRC.
2.5. Secondary Prevention: Early Detection
2.6. Tertiary Prevention: Reducing Metastasis and Recurrence
- Have more lymph nodes examined;
- Are more likely to receive adjuvant chemotherapy in early stages;
- Are more likely to receive radiotherapy in all stages;
- Are more likely to receive multiagent therapies and more lines of therapy in stage IV disease.
- Mental health (mood, anxiety, depression, coping challenges, fear of recurrence);
- Fatigue;
- Family and relationship concerns (children, fertility, sexual health, altered family roles, psychological functioning of spouse/partner);
- Employment/school (financial pressures, impact on careers);
- Personal considerations (aesthetic outcome, altered body image);
- Lifestyle (diet, exercise);
- Physical health (anemia, bowel habits, stoma, menopause, perioperative care).
3. Issues and Actions
- Increased awareness of EAO-CRC;
- Promotion of earlier detection in younger populations;
- Need to improve the primary care health system in Canada such that every citizen has access to a primary care provider that they can see at least once per year;
- Continuation of research to identify unique risk factor profiles, tumour characteristics and treatment models that can inform tailored EAO-CRC management approaches.
3.1. Improve Awareness of Risk Factors and Symptoms
3.2. Expand Organized Screening Programs
3.3. Invest in Research
- A detailed understanding of the trends in histology, topography, initial stage at diagnosis, and mortality among EAO-CRC;
- Detailed analysis of cost-effectiveness outlining the impacts on current screening programs with potential harms and benefits;
- A comprehensive understanding of risk factor profiles that may lead to meaningful recommendations for screening decisions within the 40–49 age group in the absence of wide-spread screening programs;
- An evaluation of the effectiveness of current and novel screening tests or biomarkers specifically in the 40–49 age group.
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Canadian Cancer Statistics Advisory Committee in collaboration with the Canadian Cancer Society, Statistics Canada and the Public Health Agency of Canada. Canadian Cancer Statistics 2021; Canadian Cancer Society: Toronto, ON, Canada, 2021; Available online: Cancer.ca/Canadian-Cancer-Statistics-2021-EN (accessed on 8 December 2021).
- O’Sullivan, D.E.; Sutherland, R.L.; Town, S.; Chow, K.; Fan, J.; Forbes, N.; Heitman, S.J.; Hilsden, R.J.; Brenner, D.R. Risk Factors for Early-Onset Colorectal Cancer: A Systematic Review and Meta-analysis. Clin. Gastroenterol. Hepatol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.-H.; Wu, K.; Ng, K.; Zauber, A.G.; Nguyen, L.; Song, M.; He, X.; Fuchs, C.S.; Ogino, S.; Willett, W.C.; et al. Association of obesity with risk of early-onset colorectal cancer among women. JAMA Oncol. 2019, 5, 37–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saad El Din, K.; Loree, J.M.; Sayre, E.C.; Gill, S.; Brown, C.J.; Dau, H.; De Vera, M.A. Trends in the epidemiology of young-onset colorectal cancer: A worldwide systematic review. BMC Cancer 2020, 20, 288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Araghi, M.; Soerjomataram, I.; Bardot, A.; Ferlay, J.; Cabasag, C.J.; Morrison, D.S.; De, P.; Tervonen, H.; Walsh, P.M.; Bucher, O.; et al. Changes in colorectal cancer incidence in seven high-income countries: A population-based study. Lancet Hepatol. Gastroenterol. 2019, 4, 511–518. [Google Scholar] [CrossRef]
- Brenner, D.R.; Heer, E.; Sutherland, R.L.; Ruan, Y.; Tinmouth, J.; Heitman, S.J.; Hilsden, R.J. National trends in colorectal cancer incidence among older and younger adults in Canada. JAMA Netw. Open 2019, 2, e198090. [Google Scholar] [CrossRef] [Green Version]
- Hofseth, L.J.; Hebert, J.R.; Chanda, A.; Chen, H.; Love, B.L.; Pena, M.M.; Murphy, E.A.; Sajish, M.; Sheth, A.; Buckhaults, P.J.; et al. Early-onset colorectal cancer: Initial clues and current views. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 352–364. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fedewa, S.A.; Ahnen, D.J.; Meester, R.G.S.; Barzi, A.; Jemal, A. Colorectal cancer statistics, 2017. CA Cancer J. Clin. 2017, 67, 177–193. [Google Scholar] [CrossRef]
- Gausman, V.; Dornblaser, D.; Anand, S.; Hayes, R.B.; O’Connell, K.; Du, M.; Liang, P.S. Risk Factors Associated With Early-Onset Colorectal Cancer. Clin. Gastroenterol. Hepatol. 2020, 18, 2752–2759. [Google Scholar] [CrossRef]
- Lee, S.E.; Jo, H.B.; Kwack, W.G.; Jeong, Y.J.; Yoon, Y.-J.; Kang, H.W. Characteristics of and risk factors for colorectal neoplasms in young adults in a screening population. World J. Gastroenterol. 2016, 22, 2981–2992. [Google Scholar] [CrossRef]
- Kim, N.H.; Jung, Y.S.; Yang, H.J.; Park, S.-K.; Park, J.H.; Park, D.I.; Sohn, C.I. Prevalence of and risk factors for colorectal neoplasia in asymptomatic young adults (20–39 years old). Clin. Gastroenterol. Hepatol. 2019, 17, 115–122. [Google Scholar] [CrossRef] [Green Version]
- Decker, K.M.; Lambert, P.; Bravo, J.; Demers, A.; Singh, H. Time Trends in Colorectal Cancer Incidence Rates by Income and Age at Diagnosis in Canada from 1992 to 2016. JAMA Netw. Open 2021, 4, e2117556. [Google Scholar] [CrossRef] [PubMed]
- Mauri, G.; Sartore-Bianchi, A.; Russo, A.; Marsoni, S.; Bardelli, A.; Siena, S. Early-onset colorectal cancer in young individuals. Mol. Oncol. 2019, 13, 109–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdelsattar, Z.M.; Wong, S.L.; Regenbogen, S.E.; Jomaa, D.M.; Hardiman, K.M.; Hendren, S. Colorectal cancer outcomes and treatment patterns in patients too young for average-risk screening. Cancer 2016, 122, 929–934. [Google Scholar] [CrossRef] [Green Version]
- Patel, S.G.; Murphy, C.C.; Lieu, C.H.; Hampel, H. Early age onset colorectal cancer. Adv. Cancer Res. 2021, 151, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Colorectal Cancer Alliance. “Never Too Young” Survey Report; Colorectal Cancer Alliance: Washington, DC, USA, 2019; Available online: https://www.ccalliance.org/about/never-too-young/survey (accessed on 11 October 2021).
- O’Sullivan, D.E.; Hilsden, R.J.; Ruanc, Y.; Forbes, N.; Heitman, S.J.; Brenner, D.R. The incidence of young-onset colorectal cancer in Canada continues to increase. Cancer Epidemiol. 2020, 69, 101828. [Google Scholar] [CrossRef]
- Loomans-Kropp, H.A.; Umar, A. Increasing Incidence of Colorectal Cancer in Young Adults. J. Cancer Epidemiol. 2019, 2019, 9841295. [Google Scholar] [CrossRef] [Green Version]
- Sanford, N.N.; Dharwadkar, P.; Murphy, C.C. Early-onset colorectal cancer: More than one side to the story. Colorect Cancer 2020, 9, CRC28. [Google Scholar] [CrossRef]
- Akimoto, N.; Ugai, T.; Zhong, R.; Hamada, T.; Fujiyoshi, K.; Giannakis, M.; Wu, K.; Cao, Y.; Ng, K.; Oginoet, S. Rising incidence of early-onset colorectal cancer—A call to action. Nat. Rev. Clin. Oncol. 2021, 18, 230–243. [Google Scholar] [CrossRef]
- Roos, V.H.; Mangas-Sanjuan, C.; Rodriguez-Girondo, M.; Medina-Prado, L.; Steyerberg, E.W.; Bossuyt, P.M.M.; Dekker, E.; Jover, R.; van Leerdam, M.E. Effects of family history on relative and absolute risks for colorectal cancer: A systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2019, 17, 2657–2667.e9. [Google Scholar] [CrossRef] [Green Version]
- Carroll, J.C.; Permaul, J.A.; Semotiuk, K.; Yung, E.M.; Blaine, S.; Dicks, E.; Warner, E.; Rothenmund, H.; Esplen, M.J.; Moineddin, R.; et al. Hereditary colorectal cancer screening: A 10-year longitudinal cohort study following an educational intervention. Prev. Med. Rep. 2020, 20, 101189. [Google Scholar] [CrossRef]
- Ellison, L.F. Progress in net cancer survival in Canada over 20 years. Health Rep. 2018, 29, 10–18. [Google Scholar] [PubMed]
- Canadian Partnership against Cancer. Canadian Strategy for Cancer Control. 2019–2029; Canadian Partnership against Cancer: Toronto, ON, Canada, 2019; Available online: https://www.partnershipagainstcancer.ca/cancer-strategy/ (accessed on 11 October 2021).
- Austin, H.; Henley, S.J.; King, J.; Richardson, L.C.; Eheman, C. Changes in colorectal cancer incidence rates in young and older adults in the United States: What does it tell us about screening. Cancer Causes Control 2014, 25, 191–201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandhu, G.S.; Anders, R.; Blatchford, P.; Walde, A.; Leal, A.; King, G.; Leong, S.; Davis, S.L.; Purcell, W.T.; Goodman, K.A.; et al. High incidence of prolonged rectal bleeding and advanced stage cancer in early-onset colorectal cancer patients. Color. Cancer 2020, 9, CRC31. [Google Scholar] [CrossRef]
- Scott, R.B.; Rangel, L.E.; Osler, T.M.; Hyman, N.H. Rectal cancer in patients under the age of 50 years: The delayed diagnosis. Am. J. Surg. 2016, 211, 1014–1018. [Google Scholar] [CrossRef] [PubMed]
- Lowery, J.T.; Ahnen, D.J.; Schroy, P.C., III; Hampel, H.; Baxter, N.; Boland, C.R.; Burt, R.W.; Butterly, L.; Doerr, M.; Doroshenk, M.; et al. Understanding the Contribution of Family History to Colorectal Cancer Risk and Its Clinical Implications: A State-of-the-Science Review. Cancer 2016, 122, 2633–2645. [Google Scholar] [CrossRef]
- US Preventive Services Task Force. Screening for Colorectal Cancer. US Preventive Services Task Force Recommendation Statement. JAMA 2021, 325, 1965–1977. [Google Scholar] [CrossRef]
- O’Sullivan, D.E.; Cheung, W.Y.; Boyne, D.J.; Jarada, T.; Hilsden, R.J.; Brenner, D.R. Treatment patterns and survival outcomes of early-onset colorectal cancer patients in Alberta: A population-based study. 2022; Unpublished. [Google Scholar]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Colon Cancer (Version 2.2020). Available online: https://www.nccn.org/ (accessed on 5 April 2020).
- Van Cutsem, E.; Cervantes, A.; Adam, R.; Sobrero, A.; van Krieken, J.H.; Aderka, D.; Aguilar, E.A.; Bardelli, A.; Benson, A.; Bodoky, G.; et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann. Oncol. 2016, 27, 1386–1422. [Google Scholar] [CrossRef]
- Fakih, M.; Sandhu, J.; Wang, C.; Kim, J.; Chen, Y.-J.; Lai, L.; Melstrom, K.; Kaiser, A. Evaluation of Comparative Surveillance Strategies of Circulating Tumor DNA, Imaging, and Carcinoembryonic Antigen Levels in Patients with Resected Colorectal Cancer. AMA Netw. Open 2022, 5, e221093. [Google Scholar] [CrossRef]
- Canadian Task Force on Preventive Health Care. Recommendations on screening for colorectal cancer in primary care. Can. Med. Assoc. J. 2016, 188, 340–348. [Google Scholar] [CrossRef] [Green Version]
- Brenner, D.R.; O’Sullivan, D.E.; Hilsden, R.J. Implications of the United States recommendations for early-age-at-onset colorectal cancer screening in Canada. Prev. Med. 2021, 155, 106923. [Google Scholar] [CrossRef]
| Session | Speakers |
|---|---|
| Welcome and Introduction | Ms. Filomena Servidio-Italiano, CEO and President, CCRAN Dr. Christopher Lieu, Associate Director for Clinical Research, University of Colorado |
| The Patient’s Lived Experience: Young Adult Patient Panel Discussion | Facilitator: Ms. Dawn Richards, PhD, President, Five02 Labs and Global Patient Advocate Panel: Mr. Andrew Hare, Colorectal Cancer Survivor Ms. Ana Bettencourt, Colorectal Cancer Patient Ms. Armina Ligaya, Colorectal Cancer Survivor Mr. Bill McGinley, Colorectal Cancer Patient |
| The Impetus for Change in the U.S. | Ms. Kim Newcomer, Never Too Young Program Manager, Colorectal Cancer Alliance Ms. Andrea Dwyer, University of Colorado Cancer Center & Fight Colorectal Cancer |
| Exploring the Landscape of Ongoing Research Initiatives in EAO-CRC | Dr. Darren Brenner, Molecular Epidemiologist, University of Calgary |
| Colorectal Cancer: Why Family History Matters | Dr. June Carroll, Family Physician, Sinai Health System, University of Toronto |
| Treatment Pathways: Management of Early and Later Stage Disease | Dr. Shady Ashamalla, Head, Colorectal Cancer Surgical Oncology, Sunnybrook Health Sciences Centre, Toronto Dr. Petra Wildgoose, Family Physician, Sunnybrook Health Sciences Centre, Toronto Dr. Sharlene Gill, Medical Oncologist, BC Cancer Agency, Vancouver |
| The Case for Early Detection | Dr. Clarence Wong, Interim Section Chief, Gastroenterology, Alberta Health Services, Edmonton |
| Summary and Wrap Up | Ms. Filomena Servidio-Italiano |
| Action | Outcomes | Who |
| Increase awareness of EAO-CRC | General public and primary care practitioners know the risk factors and symptoms of CRC in people > 50 | Primary care practitioners Oncologists Patient support groups Health educators |
| Promote earlier detection in younger populations | CRC screening programs include ages 45+ | Provincial health ministries |
| Increase number of family physicians in Canada | All Canadians can see a family physician at least once per year | Provincial and federal governments Medical associations and colleges |
| Support research to identify unique risk factor profiles, tumour characteristics and treatment models | Risk-stratified approaches to managing EAO-CRC | Medical researchers Government and non-government funders |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Vera, M.A.; Gill, S.; Ashamalla, S.; Schiller, D.; Brenner, D.R.; Wong, C.; Wildgoose, P.; Esplen, M.J.; Lieu, C.; Fitzpatrick, R.; et al. Early-Age-Onset Colorectal Cancer in Canada: Evidence, Issues and Calls to Action. Curr. Oncol. 2022, 29, 3149-3159. https://doi.org/10.3390/curroncol29050256
De Vera MA, Gill S, Ashamalla S, Schiller D, Brenner DR, Wong C, Wildgoose P, Esplen MJ, Lieu C, Fitzpatrick R, et al. Early-Age-Onset Colorectal Cancer in Canada: Evidence, Issues and Calls to Action. Current Oncology. 2022; 29(5):3149-3159. https://doi.org/10.3390/curroncol29050256
Chicago/Turabian StyleDe Vera, Mary A., Sharlene Gill, Shady Ashamalla, Dan Schiller, Darren R. Brenner, Clarence Wong, Petra Wildgoose, Mary Jane Esplen, Christopher Lieu, Roslyn Fitzpatrick, and et al. 2022. "Early-Age-Onset Colorectal Cancer in Canada: Evidence, Issues and Calls to Action" Current Oncology 29, no. 5: 3149-3159. https://doi.org/10.3390/curroncol29050256
APA StyleDe Vera, M. A., Gill, S., Ashamalla, S., Schiller, D., Brenner, D. R., Wong, C., Wildgoose, P., Esplen, M. J., Lieu, C., Fitzpatrick, R., O’Sullivan, D. E., & Servidio-Italiano, F. (2022). Early-Age-Onset Colorectal Cancer in Canada: Evidence, Issues and Calls to Action. Current Oncology, 29(5), 3149-3159. https://doi.org/10.3390/curroncol29050256






