Colorectal Cancer and Onset of Anxiety and Depression: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Literature Search Strategy
2.2. Study Screening and Eligibility Criteria
2.3. Data Extraction and Quality Assessment
2.4. Synthesis and Analysis
3. Results
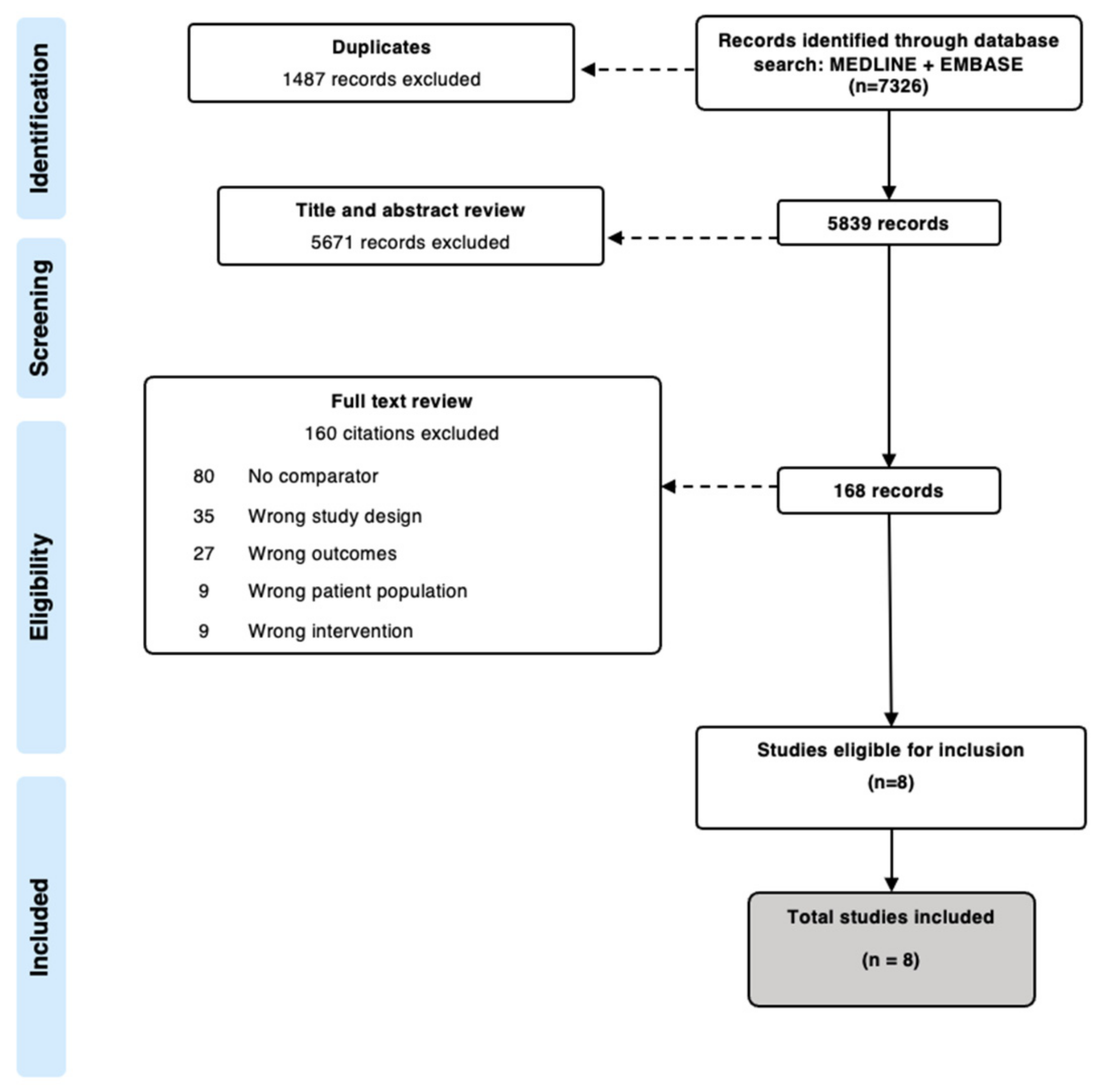
3.1. Search Results
3.2. Study Characteristics
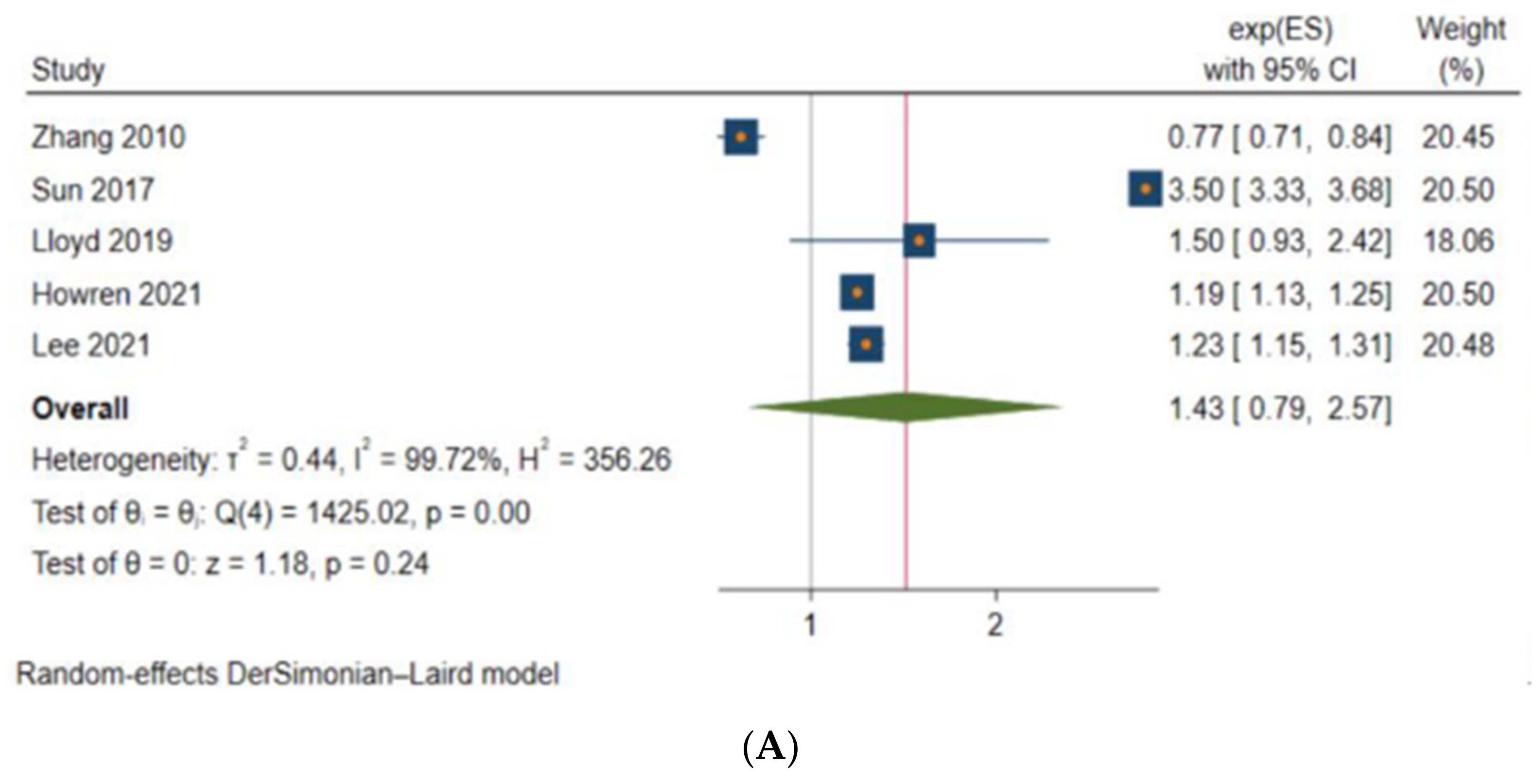
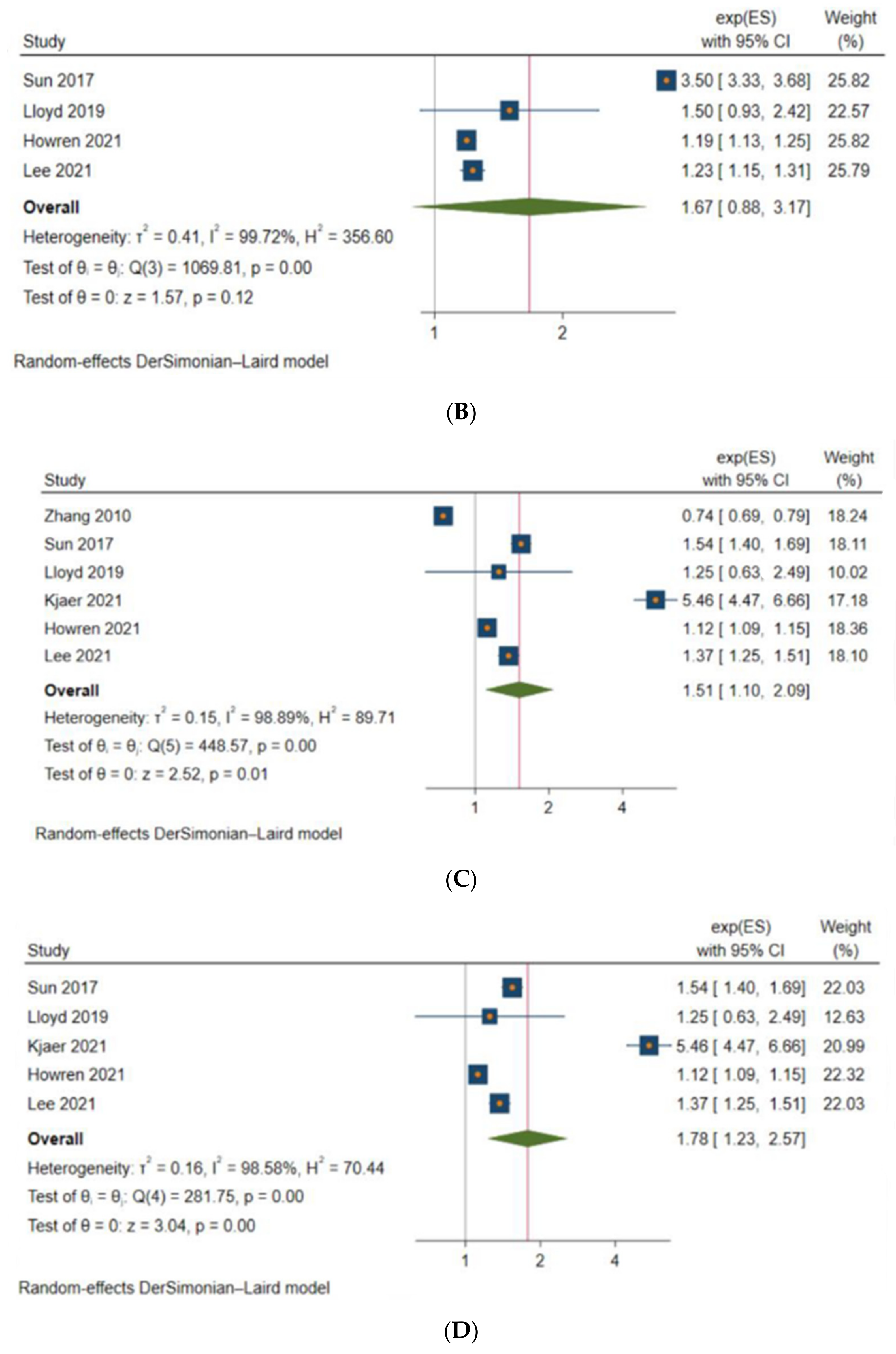
3.3. Anxiety and Depression among Patients with CRC
3.4. Predictors of Anxiety and Depression
3.5. Impacts of Anxiety and Depression
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Xi, Y.; Xu, P. Global colorectal cancer burden in 2020 and projections to 2040. Transl. Oncol. 2021, 14, 101174. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Jiang, T.; Zhang, L.; Zhou, F.; Ge, F. A Reconfigurable Convolutional Neural Network-Accelerated Coprocessor Based on RISC-V Instruction Set. Electronics 2020, 9, 1005. [Google Scholar] [CrossRef]
- Denlinger, C.S.; Barsevick, A.M. The challenges of colorectal cancer survivorship. J. Natl. Compr. Cancer Netw. 2009, 7, 883–893, quiz 894. [Google Scholar] [CrossRef] [PubMed]
- Marventano, S.; Forjaz, M.; Grosso, G.; Mistretta, A.; Giorgianni, G.; Platania, A.; Gangi, S.; Basile, F.; Biondi, A. Health related quality of life in colorectal cancer patients: State of the art. BMC Surg. 2013, 13 (Suppl. 2), S15. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.N.; Huang, M.L.; Kao, C.H. Prevalence of Depression and Anxiety in Colorectal Cancer Patients: A Literature Review. Int. J. Environ. Res. Public Health 2019, 16, 411. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, S.; Baraghoshi, D.; Tao, R.; Garrido-Laguna, I.; Gilcrease, G.W., 3rd; Whisenant, J.; Weis, J.R.; Scaife, C.; Pickron, T.B.; Huang, L.C.; et al. Mental Health Disorders are More Common in Colorectal Cancer Survivors and Associated With Decreased Overall Survival. Am. J. Clin. Oncol. 2019, 42, 355–362. [Google Scholar] [CrossRef]
- Kjaer, T.K.; Moustsen-Helms, I.R.; Albieri, V.; Larsen, S.B.; Degett, T.H.; Tjonneland, A.; Johansen, C.; Kjaer, S.K.; Gogenur, I.; Dalton, S.O. Risk of Pharmacological or Hospital Treatment for Depression in Patients with Colorectal Cancer-Associations with Pre-Cancer Lifestyle, Comorbidity and Clinical Factors. Cancers 2021, 13, 1979. [Google Scholar] [CrossRef]
- Howren, A.; Sayre, E.C.; Cheng, V.; Oveisi, N.; McTaggart-Cowan, H.; Peacock, S.; De Vera, M.A. Risk of Anxiety and Depression after Diagnosis of Young-Onset Colorectal Cancer: A Population-Based Cohort Study. Curr. Oncol. 2022, 29, 3072–3081. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Covidence Systematic Review Software. Available online: www.covidence.org (accessed on 30 June 2022).
- Stata Statistical Software: Release 17; StataCorp LLC: College Station, TX, USA, 2021.
- Otten, D.; Tibubos, A.N.; Schomerus, G.; Brahler, E.; Binder, H.; Kruse, J.; Ladwig, K.H.; Wild, P.S.; Grabe, H.J.; Beutel, M.E. Similarities and Differences of Mental Health in Women and Men: A Systematic Review of Findings in Three Large German Cohorts. Front. Public Health 2021, 9, 553071. [Google Scholar] [CrossRef]
- Eaton, N.R.; Keyes, K.M.; Krueger, R.F.; Balsis, S.; Skodol, A.E.; Markon, K.E.; Grant, B.F.; Hasin, D.S. An invariant dimensional liability model of gender differences in mental disorder prevalence: Evidence from a national sample. J. Abnorm. Psychol. 2012, 121, 282–288. [Google Scholar] [CrossRef]
- Wells, G. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Ott. Ott. Hosp. Res. Inst. 2014, 2, 1–12. [Google Scholar]
- McPheeters, M.L.; Kripalani, S.; Peterson, N.B.; Idowu, R.T.; Jerome, R.N.; Potter, S.A.; Andrews, J.C. Closing the quality gap: Revisiting the state of the science (vol. 3: Quality improvement interventions to address health disparities). Evid. Rep. Technol. Assess. 2012, 208, 1–475. [Google Scholar]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2022. [Google Scholar]
- Zhang, A.Y.; Cooper, G.S. Recognition of Depression and Anxiety among Elderly Colorectal Cancer Patients. Nurs. Res. Pract. 2010, 2010, 693961. [Google Scholar] [CrossRef]
- Sun, L.M.; Liang, J.A.; Lin, C.L.; Sun, S.; Kao, C.H. Risk of mood disorders in patients with colorectal cancer. J. Affect. Disord. 2017, 218, 59–65. [Google Scholar] [CrossRef]
- Mols, F.; Schoormans, D.; de Hingh, I.; Oerlemans, S.; Husson, O. Symptoms of anxiety and depression among colorectal cancer survivors from the population-based, longitudinal PROFILES Registry: Prevalence, predictors, and impact on quality of life. Cancer 2018, 124, 2621–2628. [Google Scholar] [CrossRef]
- Lee, M.J.; Huang, C.W.; Lee, C.P.; Kuo, T.Y.; Fang, Y.H.; Chin-Hung Chen, V.; Yang, Y.H. Investigation of anxiety and depressive disorders and psychiatric medication use before and after cancer diagnosis. Psychooncology 2021, 30, 919–927. [Google Scholar] [CrossRef]
- Weissman, S.; Ghoneim, S.; Sanayei, A.; Said, H.; Rosenblatt, S.; Mohammed, A.; Jafri, S.; Atoot, A.; Feuerstein, J.D. New-onset depression after colorectal cancer diagnosis: A population- based longitudinal study. Int. J. Color. Dis. 2021, 36, 2599–2602. [Google Scholar] [CrossRef]
- King, M.T. The interpretation of scores from the EORTC quality of life questionnaire QLQ-C30. Qual. Life Res. 1996, 5, 555–567. [Google Scholar] [CrossRef]
- Cocks, K.; King, M.T.; Velikova, G.; Martyn St-James, M.; Fayers, P.M.; Brown, J.M. Evidence-based guidelines for determination of sample size and interpretation of the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30. J. Clin. Oncol. 2011, 29, 89–96. [Google Scholar] [CrossRef]
- Niedzwiedz, C.L.; Knifton, L.; Robb, K.A.; Katikireddi, S.V.; Smith, D.J. Depression and anxiety among people living with and beyond cancer: A growing clinical and research priority. BMC Cancer 2019, 19, 943. [Google Scholar] [CrossRef] [PubMed]
- Naser, A.Y.; Hameed, A.N.; Mustafa, N.; Alwafi, H.; Dahmash, E.Z.; Alyami, H.S.; Khalil, H. Depression and Anxiety in Patients With Cancer: A Cross-Sectional Study. Front Psychol. 2021, 12, 585534. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.R. Depression in cancer patients: Pathogenesis, implications and treatment (Review). Oncol. Lett. 2015, 9, 1509–1514. [Google Scholar] [CrossRef] [PubMed]
- Renna, M.E.; Shrout, M.R.; Madison, A.A.; Alfano, C.M.; Povoski, S.P.; Lipari, A.M.; Carson, W.E., 3rd; Malarkey, W.B.; Kiecolt-Glaser, J.K. Depression and anxiety in colorectal cancer patients: Ties to pain, fatigue, and inflammation. Psychooncology 2022, 31, 1536–1544. [Google Scholar] [CrossRef] [PubMed]
- Trudel-Fitzgerald, C.; Tworoger, S.S.; Zhang, X.; Giovannucci, E.L.; Meyerhardt, J.A.; Kubzansky, L.D. Anxiety, Depression, and Colorectal Cancer Survival: Results from Two Prospective Cohorts. J Clin Med. 2020, 9, 3174. [Google Scholar] [CrossRef]
- Reese, J.B.; Handorf, E.; Haythornthwaite, J.A. Sexual quality of life, body image distress, and psychosocial outcomes in colorectal cancer: A longitudinal study. Support Care Cancer 2018, 26, 3431–3440. [Google Scholar] [CrossRef]
- Krendl, A.C. Countries and Cultural Differences in the Stigma of Mental Illness: The East–West Divide. J. Cross-Cult. Psychol. 2020, 51, 149–167. [Google Scholar] [CrossRef]
- Uddin, M.N.; Bhar, S.; Islam, F.M.A. An assessment of awareness of mental health conditions and its association with socio-demographic characteristics: A cross-sectional study in a rural district in Bangladesh. BMC Health Serv. Res. 2019, 19, 562. [Google Scholar] [CrossRef]
- Pelletier, L.; O’Donnell, S.; Dykxhoorn, J.; McRae, L.; Patten, S.B. Under-diagnosis of mood disorders in Canada. Epidemiol Psychiatr. Sci. 2017, 26, 414–423. [Google Scholar] [CrossRef]
- Brenner, D.R.; Heer, E.; Sutherland, R.L.; Ruan, Y.; Tinmouth, J.; Heitman, S.J.; Hilsden, R.J. National Trends in Colorectal Cancer Incidence Among Older and Younger Adults in Canada. JAMA Netw. Open 2019, 2, e198090. [Google Scholar] [CrossRef]
- Patel, P.; De, P. Trends in colorectal cancer incidence and related lifestyle risk factors in 15-49-year-olds in Canada, 1969-2010. Cancer Epidemiol. 2016, 42, 90–100. [Google Scholar] [CrossRef]
- Howren, A.; Sayre, E.C.; Loree, J.M.; Gill, S.; Brown, C.J.; Raval, M.J.; Farooq, A.; De Vera, M.A. Trends in the Incidence of Young-Onset Colorectal Cancer With a Focus on Years Approaching Screening Age: A Population-Based Longitudinal Study. J. Natl. Cancer Inst. 2021, 113, 863–868. [Google Scholar] [CrossRef]
- Moore, K.J.; Sussman, D.A.; Koru-Sengul, T. Age-Specific Risk Factors for Advanced Stage Colorectal Cancer, 1981-2013. Prev. Chronic Dis. 2018, 15, E106. [Google Scholar] [CrossRef]
- Patten, S.B.; Williams, J.V.; Lavorato, D.H.; Wang, J.L.; McDonald, K.; Bulloch, A.G. Descriptive epidemiology of major depressive disorder in Canada in 2012. Can. J. Psychiatry 2015, 60, 23–30. [Google Scholar] [CrossRef]
- Wang, J.; Williams, J.; Lavorato, D.; Schmitz, N.; Dewa, C.; Patten, S.B. The incidence of major depression in Canada: The National Population Health Survey. J. Affect. Disord. 2010, 123, 158–163. [Google Scholar] [CrossRef]
- Altemus, M.; Sarvaiya, N.; Neill Epperson, C. Sex differences in anxiety and depression clinical perspectives. Front. Neuroendocr. 2014, 35, 320–330. [Google Scholar] [CrossRef]
- Shi, P.; Yang, A.; Zhao, Q.; Chen, Z.; Ren, X.; Dai, Q. A Hypothesis of Gender Differences in Self-Reporting Symptom of Depression: Implications to Solve Under-Diagnosis and Under-Treatment of Depression in Males. Front. Psychiatry 2021, 12, 589687. [Google Scholar] [CrossRef]
- CMHA. Recommendations for Preventing and Managing Co-Existing Chronic Physical Conditions and Mental Illnesses; CMHA: Singapore, 2008. [Google Scholar]
- Buneviciene, I.; Bunevicius, R.; Bagdonas, S.; Bunevicius, A. The impact of pre-existing conditions and perceived health status on mental health during the COVID-19 pandemic. J. Public Health 2022, 44, e88–e95. [Google Scholar] [CrossRef]
- Vodermaier, A.; Linden, W.; MacKenzie, R.; Greig, D.; Marshall, C. Disease stage predicts post-diagnosis anxiety and depression only in some types of cancer. Br. J. Cancer 2011, 105, 1814–1817. [Google Scholar] [CrossRef]
- Chang, C.K.; Hayes, R.D.; Broadbent, M.T.; Hotopf, M.; Davies, E.; Moller, H.; Stewart, R. A cohort study on mental disorders, stage of cancer at diagnosis and subsequent survival. BMJ Open 2014, 4, e004295. [Google Scholar] [CrossRef]
- Wang, Y.; Duan, Z.; Ma, Z.; Mao, Y.; Li, X.; Wilson, A.; Qin, H.; Ou, J.; Peng, K.; Zhou, F.; et al. Epidemiology of mental health problems among patients with cancer during COVID-19 pandemic. Transl. Psychiatry 2020, 10, 263. [Google Scholar] [CrossRef]
- Shumye, S.; Belayneh, Z.; Mengistu, N. Health related quality of life and its correlates among people with depression attending outpatient department in Ethiopia: A cross sectional study. Health Qual. Life Outcomes 2019, 17, 169. [Google Scholar] [CrossRef]
- Tsunoda, A.; Nakao, K.; Hiratsuka, K.; Yasuda, N.; Shibusawa, M.; Kusano, M. Anxiety, depression and quality of life in colorectal cancer patients. Int. J. Clin. Oncol. 2005, 10, 411–417. [Google Scholar] [CrossRef]
| Author, Year | Country | Study Design | Data Source | Follow-Up Timeline | Sample Size | Sex (% Males) | CRC Information | Quality Assessment | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| CRC | Non-CRC | Age at Diagnosis | Cancer Site | Cancer Stage | |||||||
| Zhang [17], 2010 | US | Cohort | SEER a Medicare | 1998–2002 | 56,182 | 265,382 | CRC: 47 | 65–74: 42.7% 75–84: 41% ≥85: 16.3% | Colon, rectum | 0–IV | 4 |
| Non-CRC: 37 | |||||||||||
| Sun [18], 2017 | Taiwan | Cohort | NHIRD b | 2000–2010 | 27,242 | 10,8046 | CRC: 61 | 64.2 (13.5) | Colon, rectum | Not reported | 8 |
| Non-CRC: 61 | |||||||||||
| Mols [19], 2018 | Netherlands | Cohort | Netherlands Cancer Registry | 2000–2009 | 2625 | 315 | CRC: 55 | 69.4 (9.5) | Not reported | I–IV | 5 |
| Non-CRC: 55 | |||||||||||
| Lloyd [6], 2019 | US | Cohort | Utah Cancer Registry | 1997–2013 | 8961 | 35,897 | CRC: 52 | <40: 5.10% 40–49: 8.50% 50–59: 20.9% 60–69: 24.0% 70–79: 23.7% 80+: 17.8% | Colon, rectum | I–IV | 8 |
| Non-CRC: 51 | |||||||||||
| Kjaer [7], 2021 | Denmark | Cohort | Danish Cancer Registry | 2001–2016 | 1324 | 6620 | CRC: 58 | 71.3 (5.98) c | Colon, rectum | I–IV | 8 |
| Non-CRC: 58 | |||||||||||
| Lee [20], 2021 | Taiwan | Cohort | NHIRD b | 2000–2011 | 30,391 | 30,391 | CRC: 51 | 0–20: 0.3% 21–40: 8.6% 41–60: 42.6% 61–80: 42.1% >80: 6.5% | Not reported | Not reported | 7 |
| Non-CRC: 51 | |||||||||||
| Weissman [21], 2021 | US | Cohort | IBM Explorys Database | 1999–2021 | 46,710 | N/A | CRC: 68 | 18–65: 42% >65: 57% | Not reported | I–III | 6 |
| Non-CRC: N/A | |||||||||||
| Howren [8], 2022 | Canada | Cohort | Population Data BC, BC Cancer Registry | 1985–2017 | 54.634 | 546,340 | CRC: 53.5 | 67.6 (11.9) | Colon, rectum | 0–IV | 8 |
| Non-CRC: 53.5 | |||||||||||
| Predictors | Anxiety Estimate (95% Confidence Interval) | Depression Estimate (95% Confidence Interval) |
|---|---|---|
| Clinical characteristics | ||
| Comorbidities | ||
| a Mols [19], 2018 | 2 vs. 0: OR: 2.86 (1.75–4.55) 2 vs. 1: OR: 2.56 (1.69–4.00) | 2 vs. 0: OR: 4.76 (2.70–8.33) 2 vs. 1: OR: 2.94 (1.89–4.76) |
| b Lloyd [6], 2019 | 0–2 years post-diagnosis 1 + vs. 0: (a)HR: 1.10 (0.99–1.22) >2–5 years post-diagnosis 1 + vs. 0: (a)HR: 1.23 (1.01–1.49) >5 years post-diagnosis 1 + vs. 0: (a)HR: 1.67 (1.39–2.00) | 0–2 years post-diagnosis 1 + vs. 0: (a)HR: 1.53 (1.31–1.79) >2–5 years post-diagnosis 1 + vs. 0: (a)HR: 1.12 (0.88–1.44) >5 years post-diagnosis 1 + vs. 0: (a)HR: 1.51 (1.19–1.91) |
| c Kjaer [7], 2021 | 1–2 vs. 0: (a)HR: 1.74 (1.24–2.43) 3 + vs. 0: (a)HR: 2.74 (1.84–4.09) | |
| Cancer stage | ||
| Mols [19], 2018 | IV vs. I: OR: 1.14 (0.35–3.70) IV vs. II: OR: 0.98 (0.31–3.13) IV vs. III: OR: 0.90 (0.30–2.70) | IV vs. I: OR: 2.38 (0.87–6.25) IV vs. II: OR: 2.33 (0.88–6.25) IV vs. III: OR: 1.96 (0.78–5.00) |
| Lloyd [6], 2019 | 0–2 years post-dx II vs. I: (a)HR: 1.31 (1.13–1.51) III vs. I: (a)HR: 1.34 (1.17–1.53) IV vs. I: (a)HR: 1.71 (1.48–1.99) >2–5 years post-dx II vs. I: (a)HR: 1.15 (0.90–1.48) III vs. I: (a)HR: 1.38 (1.09–1.74) IV vs. I: (a)HR: 2.63 (1.84–3.74) >5 years post-dx II vs. I: (a)HR: 0.99 (0.81–1.21) III vs. I: (a)HR: 0.86 (0.69–1.07) IV vs. I: (a)HR: 1.03 (0.58–1.85) | 0–2 years post-diagnosis II vs. I: (a)HR: 1.38 (1.11–1.72) III vs. I: (a)HR: 1.35 (1.10–1.67) IV vs. I: (a)HR: 1.68 (1.32–2.13) >2–5 years post-diagnosis II vs. I: (a)HR: 1.02 (0.74–1.41) III vs. I: (a)HR: 1.39 (1.03–1.87) IV vs. I: (a)HR: 2.59 (1.68–3.98) >5 years post-diagnosis II vs. I: (a)HR: 1.03 (0.79–1.35) III vs. I: (a)HR: 1.08 (0.81–1.43) IV vs. I: (a)HR: 0.92 (0.43–1.98) |
| Kjaer [7], 2021 | II vs. I: (a)HR: 0.88 (0.56–1.38) III vs. I: (a)HR: 1.10 (0.70–1.73) IV vs. I: (a)HR: 3.07 (1.95–4.83) | |
| Cancer site | ||
| Proximal colon | ||
| Lloyd [6], 2019 | 0–2 years post-dx (a)HR: 1.10 (0.86–1.14) >2–5 years post-dx: (a)HR: 1.76 (1.12–2.76) >5 years post-dx: (a)HR: 0.79 (0.48–1.30) | 0–2 years post-dx: (a)HR: 1.45 (1.03–2.06) >2–5 years post-dx: (a)HR: 2.18 (1.24–3.84) >5 years post-dx: (a)HR: 0.83 (0.44–1.56) |
| Rectum | ||
| Lloyd [6], 2019 | 0–2 years post-dx: (a)HR: 1.08 (0.93–1.27) >2–5 years post-dx: (a)HR: 1.43 (1.04–1.96) >5 years post-dx: (a)HR: 0.91 (0.7–1.18) | 0–2 years post-dx: (a)HR: 1.09 (0.85–1.39) >2–5 years post-dx: (a)HR: 1.52 (0.99–2.34) >5 years post-dx: (a)HR: 0.91 (0.64–1.29) |
| Kjaer [7], 2021 | (a)HR: 0.90 (0.65–1.23) | |
| Cancer treatments | ||
| Radiotherapy | ||
| Sun [18], 2017 | (a)HR: 0.77 (0.69–0.86) | (a)HR: 0.83 (0.66–1.03) |
| Mols [19], 2018 | YES vs. NO: OR: 1.03 (0.70–1.52) | YES vs. NO: OR: 1.23 (0.83–1.85) |
| Lloyd [6], 2019 | 0–2 years post-dx: (a)HR: 2.48 (1.03–5.98) | 0–2 years post-dx: (a)HR: 2.74 (0.68–11.02) |
| Kjaer [7], 2021 | (a)HR: 2.76 (1.82–4.19) | |
| Chemotherapy | ||
| Sun [18], 2017 | (a)HR: 0.78 (0.71–0.85) | (a)HR: 1.00 (0.83–1.20) |
| Mols [19], 2018 | YES vs. NO: OR: 0.96 (0.58–1.59) | YES vs. NO: OR: 0.98 (0.58–1.67) |
| Lloyd [6], 2019 | 0–2 years post-dx (a)HR: 2.28 (1.70–3.06) >2–5 years post-dx (a)HR: 4.36 (1.08–17.63) | 0–2 years post-dx (a)HR: 2.04 (1.27–3.29) >2–5 years post-dx (a)HR: 6.58 (2.41–17.94) |
| Kjaer [7], 2021 | (a)HR: 0.98 (0.67–1.42) | |
| Colostomy | ||
| Sun [18], 2017 | (a)HR: 0.67 (0.52–0.87) | (a)HR: 0.71 (0.42–1.21) |
| Lloyd [6], 2019 | 0–2 years post-dx (a)HR: 1.80 (1.48–2.18) >2–5 years post-dx (a)HR: 1.54 (0.99–2.39) >5 years post-dx (a)HR: 1.55 (0.98–2.46) | 0–2 years post-dx (a)HR: 1.99 (1.52–2.61) >2–5 years post-dx (a)HR: 2.23 (1.46–3.40) >5 years post-dx (a)HR: 1.19 (0.65–2.17) |
| Kjaer [7], 2021 | (a)HR: 1.61 (0.88–2.96) | |
| Sociodemographic characteristics | ||
| Age | ||
| Mols [19], 2018 | Old vs. Young: OR: 1.01 (0.99–1.03) | Old vs. Young: OR: 1.01 (1.00–1.03) |
| Lloyd [6], 2019 | 0–2 years post-diagnosis ≥65 vs. <65 years: (a)HR: 1.05 (0.95–1.17) >2–5 years post-diagnosis ≥65 vs. <65 years: (a)HR: 1.42 (1.17–1.72) >5 years post-diagnosis ≥65 vs. <65 years: (a)HR: 2.23 (1.87–2.67) | 0–2 years post-diagnosis ≥65 vs. <65 years: (a)HR: 0.94 (0.80–1.11) >2–5 years post-diagnosis ≥65 vs. <65 years: (a)HR: 0.98 (0.77–1.25) >5 years post-diagnosis ≥65 vs. <65 years: (a)HR: 2.16 (1.81–2.57) |
| Howren [8], 2022 | <50 vs. ≥50 years: (a)HR: 1.17 (0.79–1.75) Male (<50 vs. ≥50 years): (a)HR: 0.93 (0.47–1.83) Female (<50 vs. ≥50 years): (a)HR: 1.34 (0.81–2.22) | <50 vs. ≥50 years: (a)HR: 1.56 (1.13–2.14) Male (<50 vs. ≥50 years): (a)HR: 1.60 (1.02–2.52) Female (<50 vs. ≥50 years): (a)HR: 1.49 (0.93–2.37) |
| Sex | ||
| Mols [19], 2018 | Female vs. Male: OR: 1.92 (1.33–2.78) | Female vs. Male: OR: 0.97 (0.66–1.41) |
| Lloyd [6], 2019 | 0–2 years post-dx Female vs. Male: HR: 0.79 (0.88–0.71) >2–5 years post-dx Female vs. Male: HR: 0.99 (0.82–1.20) >5 years post-dx Female vs. Male: HR: 0.95 (0.80–1.13) | 0–2 years post-dx Female vs. Male: HR: 1.38 (1.18–1.62) >2–5 years post-dx Female vs. Male: HR: 1.48 (1.16–1.88) >5 years post-dx Female vs. Male: HR: 1.15 (0.92–1.44) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, V.; Oveisi, N.; McTaggart-Cowan, H.; Loree, J.M.; Murphy, R.A.; De Vera, M.A. Colorectal Cancer and Onset of Anxiety and Depression: A Systematic Review and Meta-Analysis. Curr. Oncol. 2022, 29, 8751-8766. https://doi.org/10.3390/curroncol29110689
Cheng V, Oveisi N, McTaggart-Cowan H, Loree JM, Murphy RA, De Vera MA. Colorectal Cancer and Onset of Anxiety and Depression: A Systematic Review and Meta-Analysis. Current Oncology. 2022; 29(11):8751-8766. https://doi.org/10.3390/curroncol29110689
Chicago/Turabian StyleCheng, Vicki, Niki Oveisi, Helen McTaggart-Cowan, Jonathan M. Loree, Rachel A. Murphy, and Mary A. De Vera. 2022. "Colorectal Cancer and Onset of Anxiety and Depression: A Systematic Review and Meta-Analysis" Current Oncology 29, no. 11: 8751-8766. https://doi.org/10.3390/curroncol29110689
APA StyleCheng, V., Oveisi, N., McTaggart-Cowan, H., Loree, J. M., Murphy, R. A., & De Vera, M. A. (2022). Colorectal Cancer and Onset of Anxiety and Depression: A Systematic Review and Meta-Analysis. Current Oncology, 29(11), 8751-8766. https://doi.org/10.3390/curroncol29110689






