What Has Changed in the Management of Uterine Serous Carcinomas? Two Decades of Experience
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Statistical Analysis
3. Results
3.1. Patients’ Characteristics
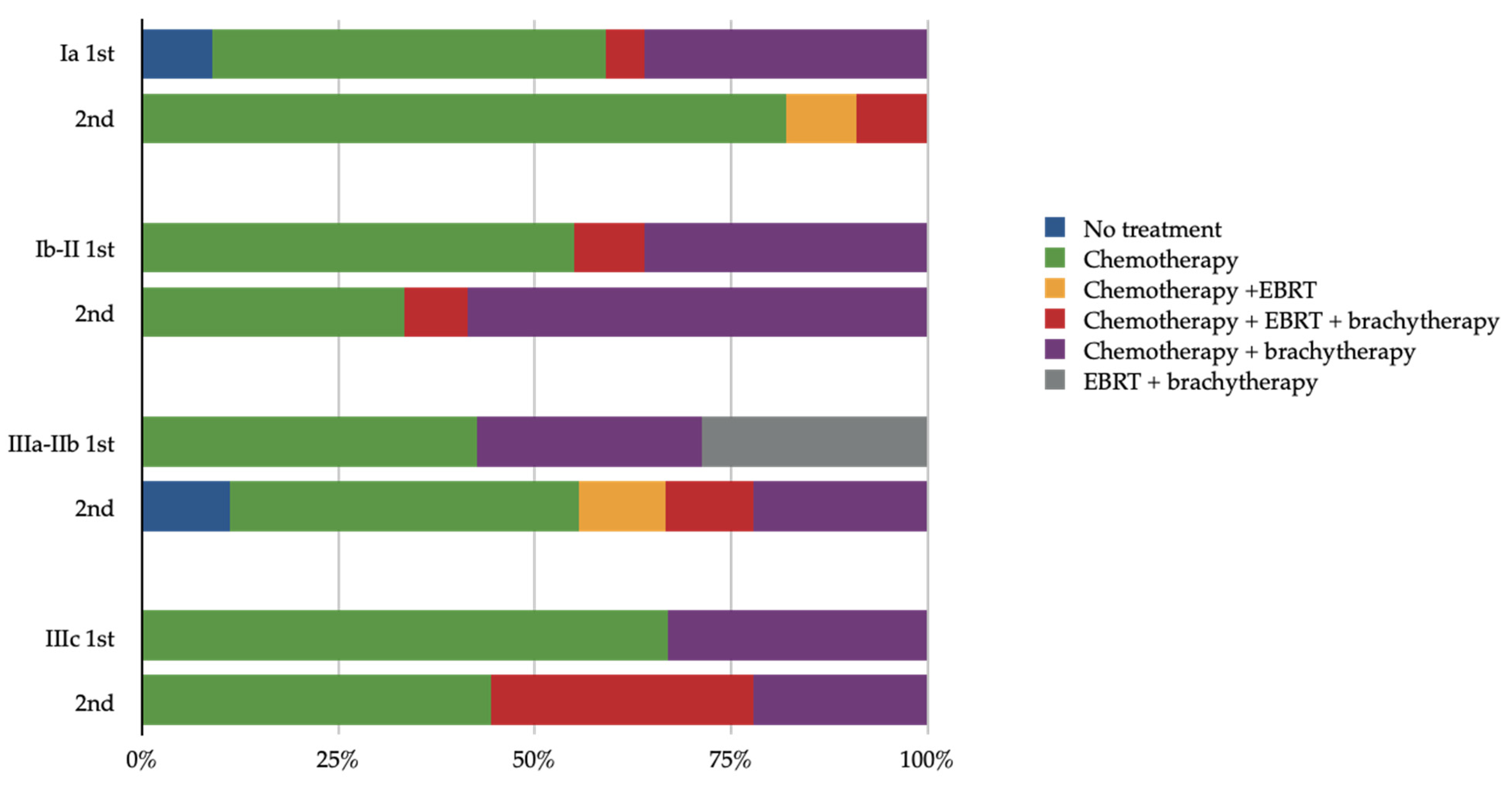
3.2. Adjuvant Treatment
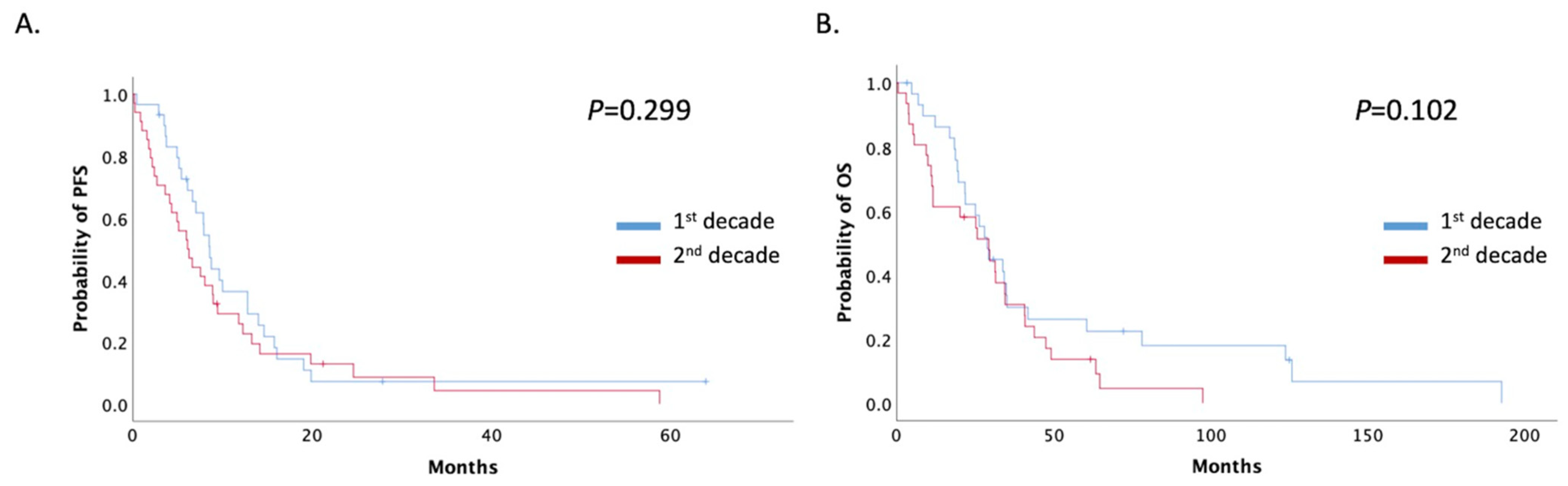
3.3. First-Line Treatment
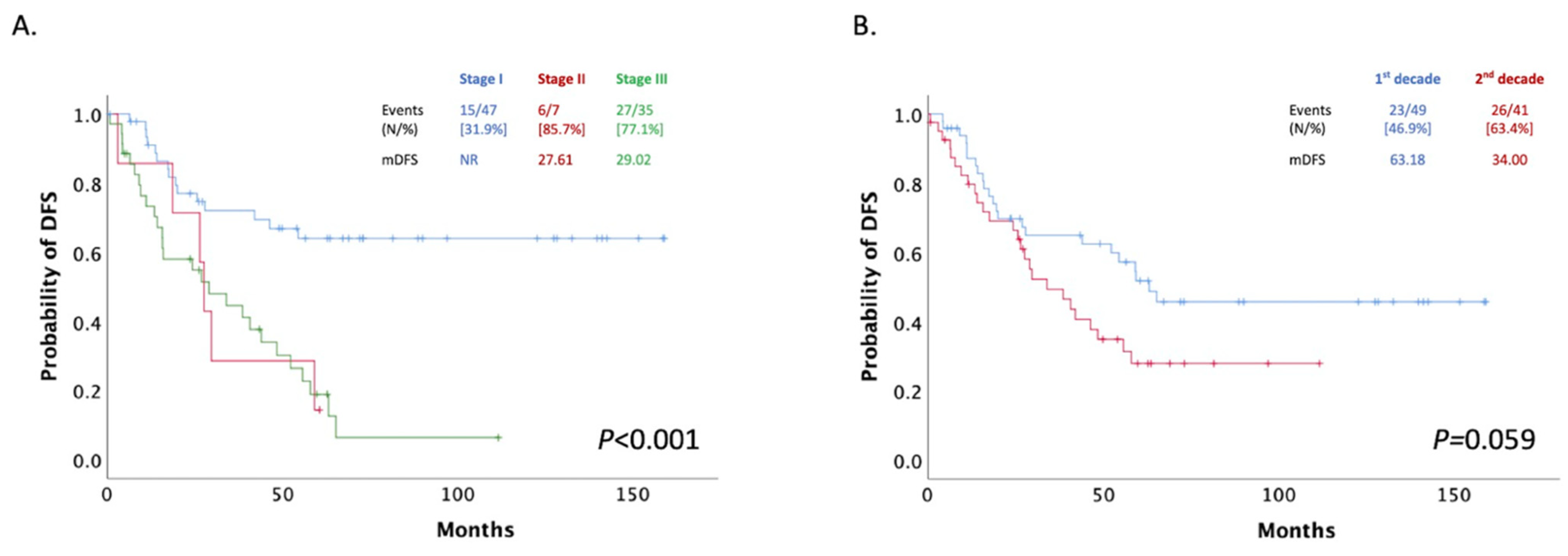
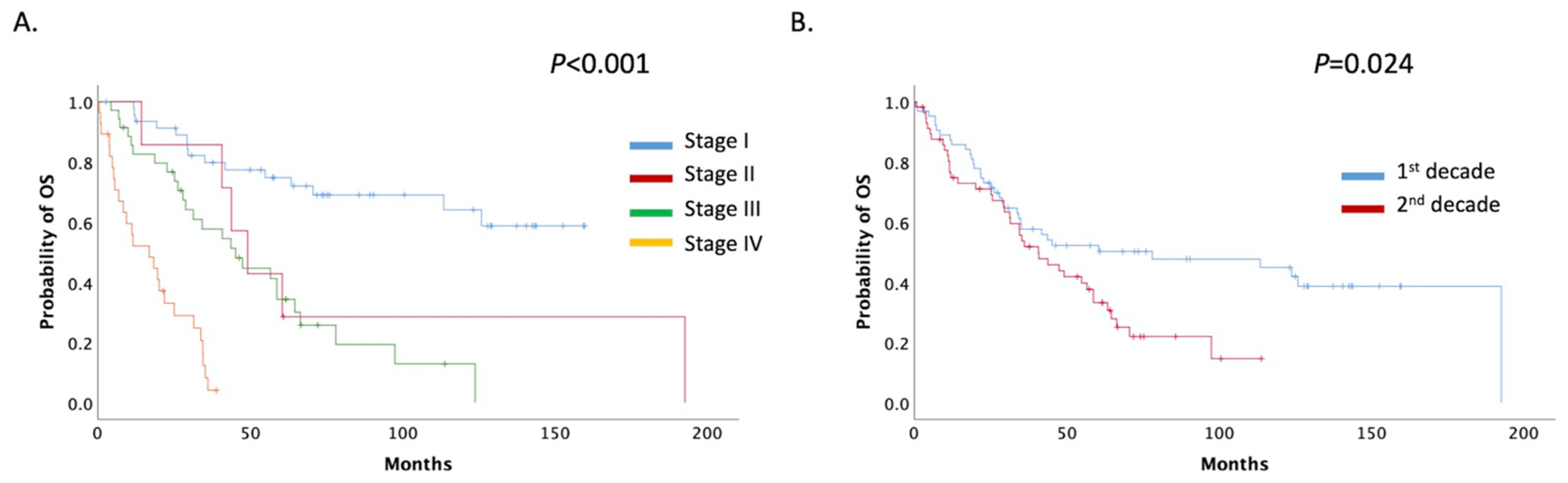
3.4. Overall Survival
3.5. Multivariate Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Epidemiology of Endometrial Cancer Consortium (E2C2). Available online: https://epi.grants.cancer.gov/eecc/ (accessed on 10 October 2019).
- Dedes, K.J.; Wetterskog, D.; Ashworth, A.; Kaye, S.B.; Reis-Filho, J.S. Emerging therapeutic targets in endometrial cancer. Nat. Rev. Clin. Oncol. 2011, 8, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Gatius, S.; Matias-Guiu, X. Practical issues in the diagnosis of serous carcinoma of the endometrium. Mod. Pathol. 2016, 29, S45–S58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scarfone, G.; Secomandi, R.; Parazzini, F.; Viganò, R.; Mangili, G.; Frigerio, L.; Villa, A.; Tateo, S.; Ricci, E.; Bolis, G. Clear cell and papillary serous endometrial carcinomas: Survival in a series of 128 cases. Arch. Gynecol. Obstet. 2013, 287, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Tang, Y.H.; Chiang, Y.C.; Wang, K.L.; Fu, H.C.; Ke, Y.M.; Lau, H.Y.; Hsu, K.F.; Wu, C.H.; Cheng, W.F. Impact of management on the prognosis of pure uterine papillary serous cancer—A Taiwanese Gynecologic Oncology Group (TGOG) study. Gynecol. Oncol. 2014, 133, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Kosary, C. Cancer of the Corpus Uteri. In SEER Survival Monograph: Cancer Survival among Adults: U.S. SEER Program, 1988–2001, Patient and Tumor Characteristics; NCI, SEER Program, National Cancer Institute: Bethesda, MD, USA, 2007. [Google Scholar]
- DeLair, D.F.; Burke, K.A.; Selenica, P.; Lim, R.S.; Scott, S.N.; Middha, S.; Mohanty, A.S.; Cheng, D.T.; Berger, M.F.; Soslow, R.A.; et al. The genetic landscape of endometrial clear cell carcinomas. J. Pathol. 2017, 243, 230–241. [Google Scholar] [CrossRef]
- Huh, W.K.; Powell, M.; Leath, C.A., 3rd; Straughn, J.M., Jr.; Cohn, D.E.; Gold, M.A.; Falkner, C.A.; Carey, D.E.; Herzog, T.; Fowler, J.M.; et al. Uterine papillary serous carcinoma: Comparisons of outcomes in surgical Stage I patients with and without adjuvant therapy. Gynecol. Oncol. 2003, 91, 470–475. [Google Scholar] [CrossRef]
- Wilczyński, M.; Danielska, J.; Wilczyński, J. An update of the classical Bokhman’s dualistic model of endometrial cancer. Prz. Menopauzalny 2016, 15, 63–68. [Google Scholar] [CrossRef]
- Kandoth, C.; McLellan, M.D.; Vandin, F.; Ye, K.; Niu, B.; Lu, C.; Xie, M.; Zhang, Q.; McMichael, J.F.; Wyczalkowski, M.A.; et al. Mutational landscape and significance across 12 major cancer types. Nature 2013, 502, 333–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bell, D.W.; Ellenson, L.H. Molecular Genetics of Endometrial Carcinoma. Annu. Rev. Pathol. 2019, 14, 339–367. [Google Scholar] [CrossRef]
- de Boer, S.M.; Powell, M.E.; Mileshkin, L.; Katsaros, D.; Bessette, P.; Haie-Meder, C.; Ottevanger, P.B.; Ledermann, J.A.; Khaw, P.; Colombo, A.; et al. Adjuvant chemoradiotherapy versus radiotherapy alone for women with high-risk endometrial cancer (PORTEC-3): Final results of an international, open-label, multicentre, randomised, phase 3 trial. Lancet Oncol. 2018, 19, 295–309. [Google Scholar] [CrossRef] [Green Version]
- Santoro, A.; Angelico, G.; Travaglino, A.; Inzani, F.; Arciuolo, D.; Valente, M.; D’Alessandris, N.; Scaglione, G.; Fiorentino, V.; Raffone, A.; et al. New Pathological and Clinical Insights in Endometrial Cancer in View of the Updated ESGO/ESTRO/ESP Guidelines. Cancers 2021, 13, 2623. [Google Scholar] [CrossRef] [PubMed]
- Concin, N.; Matias-Guiu, X.; Vergote, I.; Cibula, D.; Mirza, M.R.; Marnitz, S.; Ledermann, J.; Bosse, T.; Chargari, C.; Fagotti, A.; et al. ESGO/ESTRO/ESP guidelines for the management of patients with endometrial carcinoma. Int. J. Gynecol. Cancer 2021, 31, 12–39. [Google Scholar] [CrossRef] [PubMed]
- Fader, A.N.; Roque, D.M.; Siegel, E.; Buza, N.; Hui, P.; Abdelghany, O.; Chambers, S.; Secord, A.A.; Havrilesky, L.; O’Malley, D.M.; et al. Randomized Phase II Trial of Carboplatin-Paclitaxel Compared with Carboplatin-Paclitaxel-Trastuzumab in Advanced (Stage III–IV) or Recurrent Uterine Serous Carcinomas that Overexpress Her2/Neu (NCT01367002): Updated Overall Survival Analysis. Clin. Cancer Res. 2020, 26, 3928–3935. [Google Scholar] [CrossRef] [PubMed]
- Makker, V.; Taylor, M.H.; Aghajanian, C.; Oaknin, A.; Mier, J.; Cohn, A.L.; Romeo, M.; Bratos, R.; Brose, M.S.; DiSimone, C.; et al. Lenvatinib Plus Pembrolizumab in Patients with Advanced Endometrial Cancer. J. Clin. Oncol. 2020, 38, 2981–2992. [Google Scholar] [CrossRef]
- Testing the Addition of the Immunotherapy Drug, Pembrolizumab, to the Usual Radiation Treatment for Newly Diagnosed Early Stage High Intermediate Risk Endometrial Cancer. Identifier: NCT04214067. Available online: https://clinicaltrials.gov/ct2/show/NCT04214067 (accessed on 5 May 2021).
- van den Heerik, A.; Horeweg, N.; Nout, R.A.; Lutgens, L.; van der Steen-Banasik, E.M.; Westerveld, G.H.; van den Berg, H.A.; Slot, A.; Koppe, F.L.A.; Kommoss, S.; et al. PORTEC-4a: International randomized trial of molecular profile-based adjuvant treatment for women with high-intermediate risk endometrial cancer. Int. J. Gynecol. Cancer 2020, 30, 2002–2007. [Google Scholar] [CrossRef]
- Testing the Addition of the Immunotherapy Drug Pembrolizumab to the Usual Chemotherapy Treatment (Paclitaxel and Carboplatin) in Stage III–IV or Recurrent Endometrial Cancer. Identifier: NCT03914612. Available online: https://clinicaltrials.gov/ct2/show/NCT03914612 (accessed on 5 May 2021).
- A Study to Evaluate Dostarlimab Plus Carboplatin-Paclitaxel Versus Placebo Plus Carboplatin-Paclitaxel in Participants with Recurrent or Primary Advanced Endometrial Cancer (RUBY). Identifier: NCT03981796. Available online: https://clinicaltrials.gov/ct2/show/NCT03981796 (accessed on 5 May 2021).
- Durvalumab with or without Olaparib as Maintenance Therapy after First-Line Treatment of Advanced and Recurrent Endometrial Cancer (DUO-E) Identifier: NCT04269200. Available online: https://clinicaltrials.gov/ct2/show/NCT04269200 (accessed on 5 May 2021).
- Pembrolizumab (MK-3475) Plus Lenvatinib (E7080/MK-7902) Versus Chemotherapy for Endometrial Carcinoma (ENGOT-en9/MK-7902-001) (LEAP-001). Identifier: NCT03884101. Available online: https://clinicaltrials.gov/ct2/show/NCT03884101 (accessed on 5 May 2021).
- Maintenance with Selinexor/Placebo after Combination Chemotherapy in Participants with Endometrial Cancer [SIENDO] (ENGOT-EN5). Identifier: NCT03555422. Available online: https://clinicaltrials.gov/ct2/show/NCT03555422 (accessed on 5 May 2021).
- Study of Pembrolizumab (MK-3475) in Combination with Adjuvant Chemotherapy with or without Radiotherapy in Participants with Newly Diagnosed Endometrial Cancer after Surgery with Curative Intent (MK-3475-B21/KEYNOTE-B21/ENGOT-en11/GOG-3053). Identifier: NCT04634877. Available online: https://clinicaltrials.gov/ct2/show/NCT04634877 (accessed on 5 May 2021).
- Oken, M.M.; Creech, R.H.; Tormey, D.C.; Horton, J.; Davis, T.E.; McFadden, E.T.; Carbone, P.P. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 1982, 5, 649–655. [Google Scholar] [CrossRef]
- Altman, A.D.; Ferguson, S.E.; Atenafu, E.G.; Kobel, M.; McAlpine, J.N.; Panzarella, T.; Lau, S.; Gien, L.T.; Gilks, B.; Clarke, B.; et al. Canadian high risk endometrial cancer (CHREC) consortium: Analyzing the clinical behavior of high risk endometrial cancers. Gynecol. Oncol. 2015, 139, 268–274. [Google Scholar] [CrossRef]
- Surveillance, Epidemiology, and End Results Program. Cancer Stat Facts: Uterine Cancer. Available online: http://seer.cancer.gov/statfacts/html/corp.html (accessed on 7 January 2021).
- Slomovitz, B.M.; Burke, T.W.; Eifel, P.J.; Ramondetta, L.M.; Silva, E.G.; Jhingran, A.; Oh, J.C.; Atkinson, E.N.; Broaddus, R.R.; Gershenson, D.M.; et al. Uterine papillary serous carcinoma (UPSC): A single institution review of 129 cases. Gynecol. Oncol. 2003, 91, 463–469. [Google Scholar] [CrossRef]
- Felix, A.S.; Stone, R.A.; Bowser, R.; Chivukula, M.; Edwards, R.P.; Weissfeld, J.L.; Linkov, F. Comparison of survival outcomes between patients with malignant mixed mullerian tumors and high-grade endometrioid, clear cell, and papillary serous endometrial cancers. Int. J. Gynecol. Cancer 2011, 21, 877–884. [Google Scholar] [CrossRef] [Green Version]
- Park, J.Y.; Nam, J.H.; Kim, Y.T.; Kim, Y.M.; Kim, J.H.; Kim, D.Y.; Sohn, I.; Lee, S.W.; Sung, C.O.; Kim, K.R. Poor prognosis of uterine serous carcinoma compared with grade 3 endometrioid carcinoma in early stage patients. Virchows Arch. 2013, 462, 289–296. [Google Scholar] [CrossRef]
- Kaban, A.; Topuz, S.; Sözen, H.; Minareci, Y.; Salihoğlu, Y. Clinicopathologic and survival results in serous endometrium carcinoma and subgroup analysis for mixed serous and pure serous histology. J. Turk. Ger. Gynecol. Assoc. 2018, 19, 23–28. [Google Scholar] [CrossRef] [Green Version]
- Mori, S.; Gotoh, O.; Kiyotani, K.; Low, S.K. Genomic alterations in gynecological malignancies: Histotype-associated driver mutations, molecular subtyping schemes, and tumorigenic mechanisms. J. Hum. Genet. 2021, 66, 853–868. [Google Scholar] [CrossRef] [PubMed]
- Roelofsen, T.; van Ham, M.A.; van Tilburg, J.M.W.; Zomer, S.F.; Bol, M.; Massuger, L.F.; Bulten, J. Pure compared with mixed serous endometrial carcinoma: Two different entities? Obstet. Gynecol. 2012, 120, 1371–1381. [Google Scholar] [CrossRef] [PubMed]
- Thomakos, N.; Dimopoulou, S.; Sotiropoulou, M.; Machairiotis, N.; Pandraklakis, A.; Haidopoulos, D.; Liontos, M.; Bamias, A.; Rodolakis, A. How do different histologic components of mixed endometrial carcinomas affect prognosis? Does it really matter? Horm. Mol. Biol. Clin. Investig. 2020, 1, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Colombo, N.; Creutzberg, C.; Amant, F.; Bosse, T.; González-Martín, A.; Ledermann, J.; Marth, C.; Nout, R.; Querleu, D.; Mirza, M.R.; et al. ESMO-ESGO-ESTRO Consensus Conference on Endometrial Cancer: Diagnosis, treatment and follow-up. Ann. Oncol. 2016, 27, 16–41. [Google Scholar] [CrossRef]
- Casarin, J.; Bogani, G.; Piovano, E.; Falcone, F.; Ferrari, F.; Odicino, F.; Puppo, A.; Bonfiglio, F.; Donadello, N.; Pinelli, C.; et al. Survival implication of lymphadenectomy in patients surgically treated for apparent early-stage uterine serous carcinoma. J. Gynecol. Oncol. 2020, 31, e64. [Google Scholar] [CrossRef]
- Venigalla, S.; Chowdhry, A.K.; Shalowitz, D.I. Survival implications of staging lymphadenectomy for non-endometrioid endometrial cancers. Gynecol. Oncol. 2018, 149, 531–538. [Google Scholar] [CrossRef]
- Zapardiel, I.; Blancafort, C.; Cibula, D.; Jaunarena, I.; Gorostidi, M.; Gil-Moreno, A.; De Santiago, J. Utility and Actual Use of European and Spanish Guidelines on the Management of Endometrial Cancer Among Gynecologic Oncologists in Spain. Int. J. Gynecol. Cancer 2017, 27, 1293–1297. [Google Scholar] [CrossRef]
- Amant, F.; Mirza, M.R.; Koskas, M.; Creutzberg, C.L. Cancer of the corpus uteri. Int. J. Gynaecol. Obstet. 2018, 143 (Suppl. 2), 37–50. [Google Scholar] [CrossRef] [Green Version]
- de Boer, S.M.; Powell, M.E.; Mileshkin, L.; Katsaros, D.; Bessette, P.; Haie-Meder, C.; Ottevanger, P.B.; Ledermann, J.A.; Khaw, P.; D’Amico, R.; et al. Adjuvant chemoradiotherapy versus radiotherapy alone in women with high-risk endometrial cancer (PORTEC-3): Patterns of recurrence and post-hoc survival analysis of a randomised phase 3 trial. Lancet Oncol. 2019, 20, 1273–1285. [Google Scholar] [CrossRef] [Green Version]
- Nasioudis, D.; Roy, A.G.; Ko, E.M.; Cory, L.; Giuntoli Ii, R.L.; Haggerty, A.F.; Kim, S.H.; Morgan, M.A.; Latif, N.A. Adjuvant treatment for patients with FIGO stage I uterine serous carcinoma confined to the endometrium. Int. J. Gynecol. Cancer 2020, 30, 1089–1094. [Google Scholar] [CrossRef]
- Tétreault-Laflamme, A.; Nguyen-Huynh, T.V.; Carrier, J.F.; Samouëlian, V.; Sauthier, P.; Beauchemin, M.C.; Barkati, M. Adjuvant Chemotherapy and Vaginal Vault Brachytherapy with or without Pelvic Radiotherapy for Stage 1 Papillary Serous or Clear Cell Endometrial Cancer. Int. J. Gynecol. Cancer 2016, 26, 301–306. [Google Scholar] [CrossRef]
- Van Weelden, W.J.; Reijnen, C.; Eggink, F.A.; Boll, D.; Ottevanger, P.B.; van den Berg, H.A.; van der Aa, M.A.; Pijnenborg, J.M.A. Impact of different adjuvant treatment approaches on survival in stage III endometrial cancer: A population-based study. Eur. J. Cancer 2020, 133, 104–111. [Google Scholar] [CrossRef]
- Jamieson, A.; Bosse, T.; McAlpine, J.N. The emerging role of molecular pathology in directing the systemic treatment of endometrial cancer. Ther. Adv. Med. Oncol. 2021, 13, 17588359211035959. [Google Scholar] [CrossRef]
- Roelofsen, T.; van Ham, M.A.; de Hullu, J.A.; Massuger, L.F. Clinical management of uterine papillary serous carcinoma. Expert Rev. Anticancer Ther. 2011, 11, 71–81. [Google Scholar] [CrossRef]
- McGunigal, M.; Liu, J.; Kalir, T.; Chadha, M.; Gupta, V. Survival Differences among Uterine Papillary Serous, Clear Cell and Grade 3 Endometrioid Adenocarcinoma Endometrial Cancers: A National Cancer Database Analysis. Int. J. Gynecol. Cancer 2017, 27, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Mueller, J.J.; Pedra Nobre, S.; Braxton, K.; Alektiar, K.M.; Leitao, M.M., Jr.; Aghajanian, C.; Ellenson, L.H.; Abu-Rustum, N.R. Incidence of pelvic lymph node metastasis using modern FIGO staging and sentinel lymph node mapping with ultrastaging in surgically staged patients with endometrioid and serous endometrial carcinoma. Gynecol. Oncol. 2020, 157, 619–623. [Google Scholar] [CrossRef] [PubMed]
- Boruta, D.M., 2nd; Gehrig, P.A.; Fader, A.N.; Olawaiye, A.B. Management of women with uterine papillary serous cancer: A Society of Gynecologic Oncology (SGO) review. Gynecol. Oncol. 2009, 115, 142–153. [Google Scholar] [CrossRef] [PubMed]
- Baekelandt, M.M.; Castiglione, M. Endometrial carcinoma: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann. Oncol. 2008, 19 (Suppl. 2), ii19–ii20. [Google Scholar] [CrossRef] [PubMed]
- Fader, A.N.; Boruta, D.; Olawaiye, A.B.; Gehrig, P.A. Uterine papillary serous carcinoma: Epidemiology, pathogenesis and management. Curr. Opin. Obstet. Gynecol. 2010, 22, 21–29. [Google Scholar] [CrossRef]
- Morice, P.; Leary, A.; Creutzberg, C.; Abu-Rustum, N.; Darai, E. Endometrial cancer. Lancet 2016, 387, 1094–1108. [Google Scholar] [CrossRef]
- Li, Y.; Bian, Y.; Wang, K.; Wan, X.P. POLE mutations improve the prognosis of endometrial cancer via regulating cellular metabolism through AMF/AMFR signal transduction. BMC Med. Genet. 2019, 20, 202. [Google Scholar] [CrossRef]
- Buza, N.; Roque, D.M.; Santin, A.D. HER2/neu in Endometrial Cancer: A Promising Therapeutic Target With Diagnostic Challenges. Arch. Pathol. Lab. Med. 2014, 138, 343–350. [Google Scholar] [CrossRef] [Green Version]
- Mentrikoski, M.J.; Stoler, M.H. HER2 immunohistochemistry significantly overestimates HER2 amplification in uterine papillary serous carcinomas. Am. J. Surg. Pathol. 2014, 38, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Smith, C.L.; Cheetham, G.; Dodd, T.J.; Davy, M.L. Serous carcinoma of the uterus-determination of HER-2/neu status using immunohistochemistry, chromogenic in situ hybridization, and quantitative polymerase chain reaction techniques: Its significance and clinical correlation. Int. J. Gynecol. Cancer 2008, 18, 1344–1351. [Google Scholar] [CrossRef] [PubMed]
- Slomovitz, B.M.; Broaddus, R.R.; Burke, T.W.; Sneige, N.; Soliman, P.T.; Wu, W.; Sun, C.C.; Munsell, M.F.; Gershenson, D.M.; Lu, K.H. Her-2/neu overexpression and amplification in uterine papillary serous carcinoma. J. Clin. Oncol. 2004, 22, 3126–3132. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Peng, H.; Qi, X.; Wu, M.; Zhao, X. Targeted therapies in gynecological cancers: A comprehensive review of clinical evidence. Signal Transduct. Target. Ther. 2020, 5, 137. [Google Scholar] [CrossRef]
- Post, C.C.B.; Westermann, A.M.; Bosse, T.; Creutzberg, C.L.; Kroep, J.R. PARP and PD-1/PD-L1 checkpoint inhibition in recurrent or metastatic endometrial cancer. Crit. Rev. Oncol. Hematol. 2020, 152, 102973. [Google Scholar] [CrossRef] [PubMed]
- Sacks, D.; Baxter, B.; Campbell, B.C.V.; Carpenter, J.S.; Cognard, C.; Dippel, D.; Eesa, M.; Fischer, U.; Hausegger, K.; Hirsch, J.A.; et al. Multisociety Consensus Quality Improvement Revised Consensus Statement for Endovascular Therapy of Acute Ischemic Stroke. Int. J. Stroke 2018, 13, 612–632. [Google Scholar] [CrossRef] [Green Version]
- Westin, S.N.; Moore, K.N.; Nieuwenhuysen, E.V.; Oza, A.M.; Mileshkin, L.R.; Okamoto, A.; Suzuki, A.; Meyer, K.; Barker, L.; Rhee, J.; et al. DUO-E/GOG-3041/ENGOT-EN10: A randomized phase III trial of first-line carboplatin (carb) and paclitaxel (pac) in combination with durvalumab (durva), followed by maintenance durva with or without olaparib (ola), in patients (pts) with newly diagnosed (nd) advanced or recurrent endometrial cancer (EC). J. Clin. Oncol. 2020, 38, TPS6108. [Google Scholar] [CrossRef]
- Vergote, I.; Perez-Fidalgo, J.A.; Hamilton, E.P.; Gorp, T.V.; Valabrega, G.; Laenen, A.; Oza, A.M.; Levy, T.; Cibula, D.; Sehouli, J.; et al. SIENDO/ENGOT-EN5/GOG-3055: A randomized phase 3 trial of maintenance selinexor versus placebo after combination platinum-based chemotherapy in advanced or recurrent endometrial cancer. J. Clin. Oncol. 2021, 39, TPS5610. [Google Scholar] [CrossRef]
| Patient Characteristics | All Patients | 1st Decade | 2nd Decade | p-Value |
|---|---|---|---|---|
| Age (median, 95% Cl) | 66.49 (64.9–67.8) | 65.9 (63.8–68.1) | 66.7 (64.4–68.9) | 0.777 |
| n(%) | n(%) | n(%) | ||
| Stage | 0.368 | |||
| IA | 33 (27.3%) | 22 (34.4%) | 11 (19.3%) | |
| IB-II | 23 (19.0%) | 11 (17.2%) | 12 (21.1%) | |
| III | 34 (28.1%) | 16 (25.0%) | 18 (31.6%) | |
| IV | 28 (23.1%) | 13 (20.3%) | 15 (26.3%) | |
| Missing | 3 (4.7%) | 2 (3.1%) | 1 (1.8%) | |
| Lymph node dissection | 0.129 | |||
| No | 50 (41.3%) | 23 (35.9%) | 27 (47.4%) | |
| Yes | 63 (52.1%) | 38 (59.4%) | 25 (43.9%) | |
| Missing | 2 (1.6%) | 1 (1.6%) | 1 (1.7%) | |
| Not applicable | 6 (5%) | 2 (3.1%) | 4 (7%) | |
| Omentum excision | 0.610 | |||
| No | 30 (24.8%) | 15 (23.4%) | 15 (26.3%) | |
| Yes | 83 (68.6%) | 46 (71.8%) | 37 (64.9%) | |
| Missing | 2 (1.6%) | 1 (1.6%) | 1 (1.7%) | |
| Not applicable | 6 (5%) | 2 (3.1%) | 4 (7%) | |
| Type of adjuvant therapy | 0.745 | |||
| Chemotherapy | 47 (38.8%) | 26 (40.6%) | 21 (36.8%) | |
| Radiotherapy | 2 (1.7%) | 2 (3.1%) | 0 (0%) | |
| Chemotherapy and radiotherapy | 38 (31.4%) | 19 (29.7%) | 19 (33.3%) | |
| Not applicable | 34 (28.1%) | 17 (26.6%) | 17 (29.8%) |
| Patients’ Characteristics | HR | 95% CI | p-Value |
|---|---|---|---|
| Age | 0.771 | ||
| <65 years | 1 | ||
| >65 years | 1.132 | 0.492–2.607 | |
| Histology | 0.925 | ||
| Only serous | 1 | ||
| Mixed | 0.962 | 0.428–2.162 | |
| Stage | 0.027 | ||
| I | 1 | ||
| II | 4.147 | 0.814–21.125 | |
| III | 2.716 | 1.238–5.959 | |
| Type of adjuvant | 0.268 | ||
| Chemotherapy | 1 | ||
| Chemotherapy plus radiotherapy | 0.626 | 0.273–1.435 | |
| Lymph node dissection | 0.179 | ||
| No | 1 | ||
| Yes | 0.509 | 0.190–1.363 | |
| Omentectomy | 0.563 | ||
| No | 1 | ||
| Yes | 0.775 | 0.326–1.839 | |
| Decade | 0.717 | ||
| 1st | 1 | ||
| 2nd | 1.161 | 0.517–2.611 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liontos, M.; Svarna, A.; Theofanakis, C.; Fiste, O.; Andrikopoulou, A.; Kaparelou, M.; Koutsoukos, K.; Thomakos, N.; Haidopoulos, D.; Rodolakis, A.; et al. What Has Changed in the Management of Uterine Serous Carcinomas? Two Decades of Experience. Curr. Oncol. 2021, 28, 4862-4873. https://doi.org/10.3390/curroncol28060410
Liontos M, Svarna A, Theofanakis C, Fiste O, Andrikopoulou A, Kaparelou M, Koutsoukos K, Thomakos N, Haidopoulos D, Rodolakis A, et al. What Has Changed in the Management of Uterine Serous Carcinomas? Two Decades of Experience. Current Oncology. 2021; 28(6):4862-4873. https://doi.org/10.3390/curroncol28060410
Chicago/Turabian StyleLiontos, Michalis, Anna Svarna, Charalampos Theofanakis, Oraianthi Fiste, Angeliki Andrikopoulou, Maria Kaparelou, Konstantinos Koutsoukos, Nikolaos Thomakos, Dimitrios Haidopoulos, Alexandros Rodolakis, and et al. 2021. "What Has Changed in the Management of Uterine Serous Carcinomas? Two Decades of Experience" Current Oncology 28, no. 6: 4862-4873. https://doi.org/10.3390/curroncol28060410
APA StyleLiontos, M., Svarna, A., Theofanakis, C., Fiste, O., Andrikopoulou, A., Kaparelou, M., Koutsoukos, K., Thomakos, N., Haidopoulos, D., Rodolakis, A., Dimopoulos, M. A., & Zagouri, F. (2021). What Has Changed in the Management of Uterine Serous Carcinomas? Two Decades of Experience. Current Oncology, 28(6), 4862-4873. https://doi.org/10.3390/curroncol28060410






