Clinical Study of Sentinel Lymph Node Detection Using Photodynamic Eye for Abdominal Radical Trachelectomy
Abstract
:1. Introduction
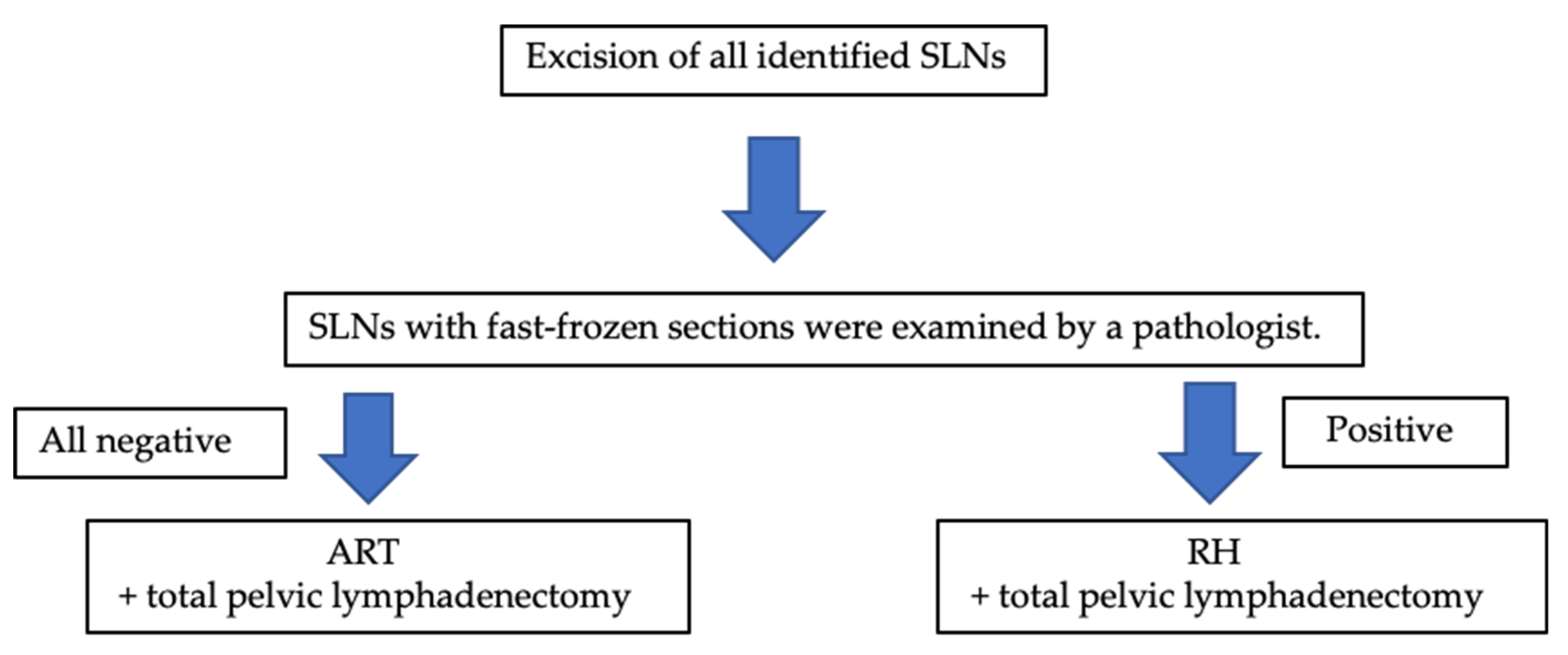
2. Materials and Methods
3. Results
3.1. Patients
3.2. Operation and Complications
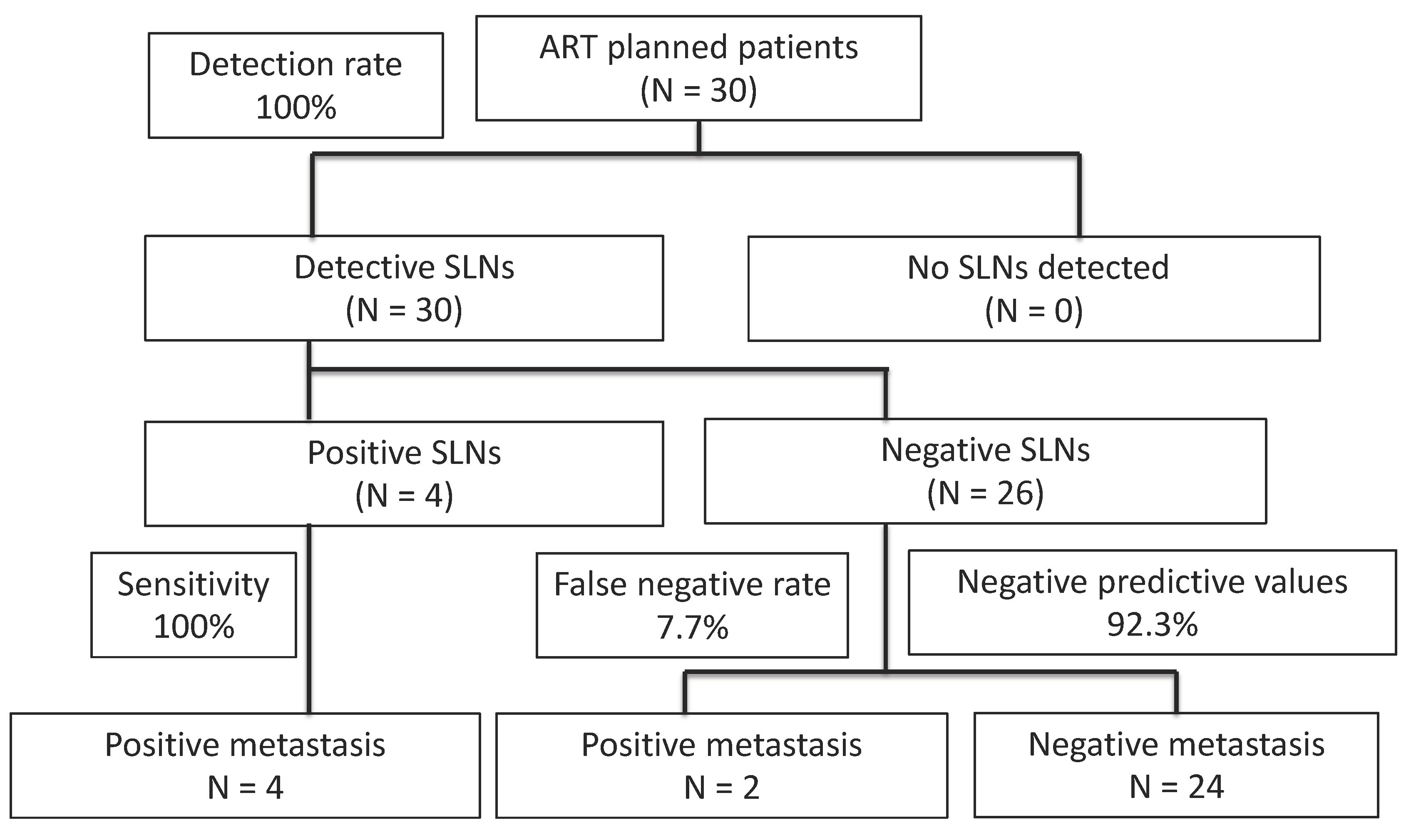
3.3. SLN Detection
3.4. Adjuvant Chemotherapy
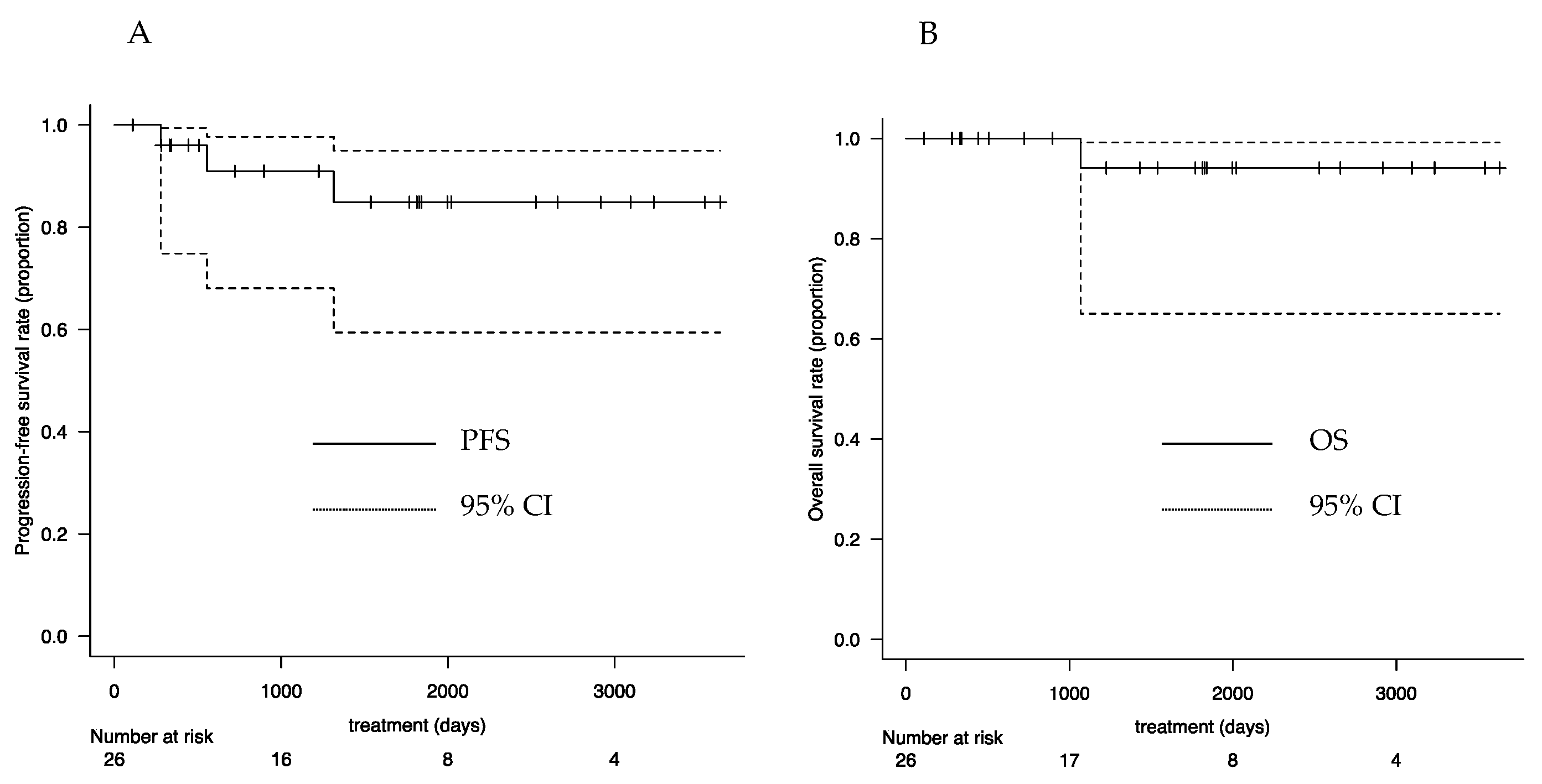
3.5. Follow-Up Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [Green Version]
- International Agency for Research on Cancer. Cancer Today. Available online: https://gco.iarc.fr/today/home (accessed on 7 September 2021).
- Lei, J.; Ploner, A.; Elfstrom, K.M.; Wang, J.; Roth, A.; Fang, F.; Sundstrom, K.; Dillner, J.; Sparen, P. HPV vaccination and the risk of invasive cervical cancer. N. Engl. J. Med. 2020, 383, 1340–1348. [Google Scholar] [CrossRef]
- Simms, K.T.; Steinberg, J.; Caruana, M.; Smith, M.A.; Lew, J.B.; Soerjomataram, I.; Castle, P.E.; Bray, F.; Canfell, K. Impact of scaled up human papillomavirus vaccination and cervical screening and the potential for global elimination of cervical cancer in 181 countries, 2020–2099: A modelling study. Lancet Oncol. 2019, 20, 394–407. [Google Scholar] [CrossRef]
- WHO. WHO EB Recommends the Adoption of the Strategy for Elimination of Cervical Cancer. Available online: https://www.who.int/news/item/05-02-2020-who-eb-recommends-the-adoption-of-the-strategy-for-elimination-of-cervical-cancer (accessed on 7 September 2021).
- Gilmour, S.; Kanda, M.; Kusumi, E.; Tanimoto, T.; Kami, M.; Shibuya, K. HPV vaccination programme in Japan. Lancet 2013, 382, 768. [Google Scholar] [CrossRef]
- Simms, K.T.; Hanley, S.J.B.; Smith, M.A.; Keane, A.; Canfell, K. Impact of HPV vaccine hesitancy on cervical cancer in Japan: A modelling study. Lancet Public Health 2020, 5, e223–e234. [Google Scholar] [CrossRef]
- NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Cervical Cancer. Available online: https://www.nccn.org/professionals/physician_gls/pdf/cervical.pdf (accessed on 20 August 2019).
- Diaz, J.P.; Sonoda, Y.; Leitao, M.M.; Zivanovic, O.; Brown, C.L.; Chi, D.S.; Barakat, R.R.; Abu-Rustum, N.R. Oncologic outcome of fertility-sparing radical trachelectomy versus radical hysterectomy for stage IB1 cervical carcinoma. Gynecol. Oncol. 2008, 111, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Bentivegna, E.; Gouy, S.; Maulard, A.; Chargari, C.; Leary, A.; Morice, P. Oncological outcomes after fertility-sparing surgery for cervical cancer: A systematic review. Lancet Oncol. 2016, 17, e240–e253. [Google Scholar] [CrossRef]
- Marchiole, P.; Tigaud, J.D.; Costantini, S.; Mammoliti, S.; Buenerd, A.; Moran, E.; Mathevet, P. Neoadjuvant chemotherapy and vaginal radical trachelectomy for fertility-sparing treatment in women affected by cervical cancer (FIGO stage IB-IIA1). Gynecol. Oncol. 2011, 122, 484–490. [Google Scholar] [CrossRef]
- Deng, X.; Zhang, Y.; Li, D.; Zhang, X.; Guo, H.; Wang, F.; Sheng, X. Abdominal radical trachelectomy guided by sentinel lymph node biopsy for stage IB1 cervical cancer with tumors >2 cm. Oncotarget 2016, 8, 3422–3429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zusterzeel, P.L.M.; Aarts, J.W.M.; Pol, F.J.M.; Ottevanger, P.B.; van Ham, M. Neoadjuvant chemotherapy followed by vaginal radical trachelectomy as fertility-preserving treatment for patients with FIGO 2018 stage 1B2 cervical cancer. Oncologist 2020, 25, e1051–e1059. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, O.K.; Chen, J.; Dodson, M.; Lee, C.M.; Gaffney, D.K. Prognostic significance of histology and positive lymph node involvement following radical hysterectomy in carcinoma of the cervix. Am. J. Clin. Oncol. 2009, 32, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Biewenga, P.; van der Velden, J.; Mol, B.W.; Stalpers, L.J.; Schilthuis, M.S.; van der Steeg, J.W.; Burger, M.P.; Buist, M.R. Prognostic model for survival in patients with early stage cervical cancer. Cancer 2011, 117, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Marin, F.; Plesca, M.; Bordea, C.I.; Voinea, S.C.; Burlanescu, I.; Ichim, E.; Jianu, C.G.; Nicolaescu, R.R.; Teodosie, M.P.; Maher, K.; et al. Postoperative surgical complications of lymphadenohysterocolpectomy. J. Med. Life 2014, 7, 60–66. [Google Scholar]
- Benedetti-Panici, P.; Maneschi, F.; Scambia, G.; Greggi, S.; Cutillo, G.; D’Andrea, G.; Rabitti, C.; Coronetta, F.; Capelli, A.; Mancuso, S. Lymphatic spread of cervical cancer: An anatomical and pathological study based on 225 radical hysterectomies with systematic pelvic and aortic lymphadenectomy. Gynecol. Oncol. 1996, 62, 19–24. [Google Scholar] [CrossRef]
- Ansari, M.; Rad, M.A.; Hassanzadeh, M.; Gholami, H.; Yousefi, Z.; Dabbagh, V.R.; Sadeghi, R. Sentinel node biopsy in endometrial cancer: Systematic review and meta-analysis of the literature. Eur. J. Gynaecol. Oncol. 2013, 34, 387–401. [Google Scholar]
- Hassanzade, M.; Attaran, M.; Treglia, G.; Yousefi, Z.; Sadeghi, R. Lymphatic mapping and sentinel node biopsy in squamous cell carcinoma of the vulva: Systematic review and meta-analysis of the literature. Gynecol. Oncol. 2013, 130, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Lécuru, F.; Mathevet, P.; Querleu, D.; Leblanc, E.; Morice, P.; Darai, E.; Marret, H.; Magaud, L.; Gillaizeau, F.; Chatellier, G.; et al. Bilateral negative sentinel nodes accurately predict absence of lymph node metastasis in early cervical cancer: Results of the SENTICOL study. J. Clin. Oncol. 2011, 29, 1686–1691. [Google Scholar] [CrossRef]
- Zaal, A.; Zweemer, R.P.; Zikan, M.; Dusek, L.; Querleu, D.; Lecuru, F.; Bats, A.S.; Jach, R.; Sevcik, L.; Graf, P.K.; et al. Pelvic lymphadenectomy improves survival in patients with cervical cancer with low-volume disease in the sentinel node: A retrospective multicenter cohort study. Int. J. Gynecol. Cancer 2014, 24, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Du, X.-L.; Sheng, X.-D.; Jiang, T.; Li, Q.-S.; Yu, H.; Pan, C.-X.; Lu, C.-H.; Wang, C.; Song, Q.-Q. Sentinel lymph node biopsy as guidance for radical trachelectomy in young patients with early stage cervical cancer. BMC Cancer 2011, 11, 157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yahata, H.; Kobayashi, H.; Sonoda, K.; Kodama, K.; Yagi, H.; Yasunaga, M.; Ohgami, T.; Onoyama, I.; Kaneki, E.; Okugawa, K.B.; et al. Prognostic outcome and complications of sentinel lymph node navigation surgery for early-stage cervical cancer. Int. J. Clin. Oncol. 2018, 23, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Fang, F.; Li, Y.F. Sentinel-lymph-node procedures in early stage cervical cancer: A systematic review and meta-analysis. Med. Oncol. 2015, 32, 385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kadkhodayan, S.; Hasanzadeh, M.; Treglia, G.; Azad, A.; Yousefi, Z.; Zarifmahmoudi, L.; Sadeghi, R. Sentinel node biopsy for lymph nodal staging of uterine cervix cancer: A systematic review and meta-analysis of the pertinent literature. Eur. J. Surg. Oncol. 2015, 41, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Li, Z.; Wu, H.; Yu, J. Sentinel lymph node biopsy in cervical cancer: A meta-analysis. Mol. Clin. Oncol. 2013, 1, 1025–1030. [Google Scholar] [CrossRef]
- Ross, G.; Shoaib, T.; Scott, J.; Soutar, D.S.; Gray, H.; Mackie, R. The learning curve for sentinel node biopsy in malignant melanoma. Br. J. Plast. Surg. 2002, 55, 298–301. [Google Scholar] [CrossRef]
- Pareja, R.; Rendon, G.J.; Vasquez, M.; Echeverri, L.; Sanz-Lomana, C.M.; Ramirez, P.T. Immediate radical trachelectomy versus neoadjuvant chemotherapy followed by conservative surgery for patients with stage IB1 cervical cancer with tumors 2 cm or larger: A literature review and analysis of oncological and obstetrical outcomes. Gynecol. Oncol. 2015, 137, 574–580. [Google Scholar] [CrossRef]
- Plante, M.; Lau, S.; Brydon, L.; Swenerton, K.; LeBlanc, R.; Roy, M. Neoadjuvant chemotherapy followed by vaginal radical trachelectomy in bulky stage IB1 cervical cancer: Case report. Gynecol. Oncol. 2006, 101, 367–370. [Google Scholar] [CrossRef]
- Liu, H.; Peng, Z.L.; Lou, J.Y.; Ping, W. Pregnancy after neoadjuvant chemotherapy followed by pelvic lymphadenectomy and radical trachelectomy in bulky stage IB1 cervical cancer: A case report. Aust. N. Z. J. Obstet. Gynaecol. 2008, 48, 517–518. [Google Scholar] [CrossRef]
- Lanowska, M.; Mangler, M.; Speiser, D.; Bockholdt, C.; Schneider, A.; Köhler, C.; Vasiljeva, J.; Al-Hakeem, M.; Vercellino, G.F. Radical vaginal trachelectomy after laparoscopic staging and neoadjuvant chemotherapy in women with early-stage cervical cancer over 2 cm: Oncologic, fertility, and neonatal outcome in a series of 20 patients. Int. J. Gynecol. Cancer 2014, 24, 586–593. [Google Scholar] [CrossRef]
- Tanaka, T.; Terai, Y.; Ashihara, K.; Tsunetoh, S.; Akagi, H.; Yamada, T.; Ohmichi, M. The detection of sentinel lymph nodes in laparoscopic surgery for uterine cervical cancer using 99m-technetium-tin colloid, indocyanine green, and blue dye. J. Gynecol. Oncol. 2017, 28, e13. [Google Scholar] [CrossRef] [Green Version]
- Sethi, D.; Sen, R.; Parshad, S.; Khetarpal, S.; Garg, M.; Sen, J. Histopathologic changes following neoadjuvant chemotherapy in various malignancies. Int. J. Appl. Basic Med. Res 2012, 2, 111–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leijte, J.A.; van der Ploeg, I.M.; Valdes Olmos, R.A.; Nieweg, O.E.; Horenblas, S. Visualization of tumor blockage and rerouting of lymphatic drainage in penile cancer patients by use of SPECT/CT. J. Nucl. Med. 2009, 50, 364–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruscito, I.; Gasparri, M.L.; Braicu, E.I.; Bellati, F.; Raio, L.; Sehouli, J.; Mueller, M.D.; Panici, P.B.; Papadia, A. Sentinel node mapping in cervical and endometrial cancer: Indocyanine green versus other conventional dyes—A meta-analysis. Ann. Surg. Oncol. 2016, 23, 3749–3756. [Google Scholar] [CrossRef]
- Jewell, E.L.; Huang, J.J.; Abu-Rustum, N.R.; Gardner, G.J.; Brown, C.L.; Sonoda, Y.; Barakat, R.R.; Levine, D.A.; Leitao, M.M., Jr. Detection of sentinel lymph nodes in minimally invasive surgery using indocyanine green and near-infrared fluorescence imaging for uterine and cervical malignancies. Gynecol. Oncol. 2014, 133, 274–277. [Google Scholar] [CrossRef] [Green Version]
- Diab, Y. Sentinel lymph nodes mapping in cervical cancer a comprehensive review. Int. J. Gynecol. Cancer 2017, 27, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Polom, K.; Murawa, D.; Rho, Y.S.; Nowaczyk, P.; Hunerbein, M.; Murawa, P. Current trends and emerging future of indocyanine green usage in surgery and oncology: A literature review. Cancer 2011, 117, 4812–4822. [Google Scholar] [CrossRef]
- Bizzarri, N.; Pedone Anchora, L.; Zannoni, G.F.; Santoro, A.; Valente, M.; Inzani, F.; Gallotta, V.; Conte, C.; Chiantera, V.; Fanfani, F.F.; et al. Role of one-step nucleic acid amplification (OSNA) to detect sentinel lymph node low-volume metastasis in early-stage cervical cancer. Int. J. Gynecol. Cancer 2020, 30, 364–371. [Google Scholar] [CrossRef]
- Santoro, A.; Angelico, G.; Inzani, F.; Arciuolo, D.; Spadola, S.; Valente, M.; D’Alessandris, N.; Piermattei, A.; Fiorentino, V.; Clanfrini, F.; et al. Standard ultrastaging compared to one-step nucleic acid amplification (OSNA) for the detection of sentinel lymph node metastases in early stage cervical cancer. Int. J. Gynecol. Cancer 2020, 30, 1871–1877. [Google Scholar] [CrossRef]
- Marchiole, P.; Buenerd, A.; Benchaib, M.; Nezhat, K.; Dargent, D.; Mathevet, P. Clinical significance of lympho vascular space involvement and lymph node micrometastases in early-stage cervical cancer: A retrospective case-control surgico-pathological study. Gynecol. Oncol. 2005, 97, 727–732. [Google Scholar] [CrossRef]
- Cibula, D.; Abu-Rustum, N.R.; Dusek, L.; Zikan, M.; Zaal, A.; Sevcik, L.; Kenter, G.G.; Querleu, D.; Jach, R.; Bats, A.S.D.; et al. Prognostic significance of low volume sentinel lymph node disease in early-stage cervical cancer. Gynecol. Oncol. 2012, 124, 496–501. [Google Scholar] [CrossRef]
- Guani, B.; Dorez, M.; Magaud, L.; Buenerd, A.; Lecuru, F.; Mathevet, P. Impact of micrometastasis or isolated tumor cells on recurrence and survival in patients with early cervical cancer: SENTICOL Trial. Int. J. Gynecol. Cancer 2019, 29, 447–452. [Google Scholar] [CrossRef]
- Pareja, R.; Rendon, G.J.; Sanz-Lomana, C.M.; Monzon, O.; Ramirez, P.T. Surgical, oncological, and obstetrical outcomes after abdominal radical trachelectomy—A systematic literature review. Gynecol. Oncol. 2013, 131, 77–82. [Google Scholar] [CrossRef]
- Okugawa, K.; Yahata, H.; Sonoda, K.; Ohgami, T.; Yasunaga, M.; Kaneki, E.; Kato, K. Safety evaluation of abdominal trachelectomy in patients with cervical tumors ≥2 cm: A single-institution, retrospective analysis. J. Gynecol. Oncol. 2020, 31, e41. [Google Scholar] [CrossRef]
- Seki, T.; Tanabe, H.; Nagata, C.; Suzuki, J.; Suzuki, K.; Takano, H.; Isonishi, S.; Ochiai, K.; Takakura, S.; Okamoto, A. Adjuvant therapy after radical surgery for stage IB-IIB cervical adenocarcinoma with risk factors. Jpn. J. Clin. Oncol. 2017, 47, 32–38. [Google Scholar] [CrossRef] [Green Version]
- Ronco, G.; Dillner, J.; Elfstrom, K.M.; Tunesi, S.; Snijders, P.J.F.; Arbyn, M.; Kitchener, H.; Segnan, N.; Gilham, C.; Giorgi-Rossi, P.; et al. Efficacy of HPV-based screening for prevention of invasive cervical cancer: Follow-up of four European randomised controlled trials. Lancet 2014, 383, 524–532. [Google Scholar] [CrossRef]
- Li, X.; Xia, L.; Li, J.; Chen, X.; Ju, X.; Wu, X. Reproductive and obstetric outcomes after abdominal radical trachelectomy (ART) for patients with early-stage cervical cancers in Fudan, China. Gynecol. Oncol. 2020, 157, 418–422. [Google Scholar] [CrossRef]
- Kim, C.H.; Abu-Rustum, N.R.; Chi, D.S.; Gardner, G.J.; Leitao, M.M., Jr.; Carter, J.; Barakat, R.R.; Sonoda, Y. Reproductive outcomes of patients undergoing radical trachelectomy for early-stage cervical cancer. Gynecol. Oncol. 2012, 125, 585–588. [Google Scholar] [CrossRef]
| Age (Years) | 34 (23–40) |
|---|---|
| FIGO 2009 stage | |
| IA1 | 2 (7) |
| IA2 | 1 (3) |
| IB1 | 25 (83) |
| IB2 | 2 (7) |
| Histology (%) | |
| Squamous cell carcinoma | 19 (63) |
| Adenocarcinoma | 6 (20) |
| Adenosquamous carcinoma | 2 (7) |
| Mucinous adenocarcinoma | 2 (7) |
| Small cell adenocarcinoma | 1 (3) |
| Tumor size (cm) | |
| <2 | 15 (50) |
| ≥2 | 15 (50) |
| Lymphovascular space invasion | |
| + | 5 (17) |
| − | 25 (83) |
| Neoadjuvant chemotherapy | |
| + | 4 (13) |
| − | 26 (87) |
| HPV vaccine | |
| + | 0 (0) |
| − | 30 (100) |
| Operative Time (Min) | 489 ± 69 (371–623) |
|---|---|
| Blood loss (g) | 1740 ± 824 (849–4200) |
| No. pelvic lymph nodes | |
| Right side | 11 ± 5 |
| Left side | 11 ± 6 |
| Total n | 622 |
| Adjuvant chemotherapy | |
| Paclitaxel and carboplatin | 9 (35) |
| Nedaplatin + doxifluridine | 3 (11) |
| Docetaxel + cisplatin | 1 (4) |
| No adjuvant therapy | 13 (50) |
| SLN Detection | 30/30 (100) |
|---|---|
| Total number of SLNs | 350 |
| Localization of SLNs (region) | |
| Common iliac | 90 (25.7) |
| External iliac | 98 (28) |
| Internal iliac | 22 (6.3) |
| Obturator | 122 (34.9) |
| Parametrial | 4 (1.1) |
| Sacral | 3 (0.9) |
| Suprainguinal | 11 (3.1) |
| Patient | No. Total SLNs | No. Positive LNs | Location (No. Positive LNs) | Treatment |
|---|---|---|---|---|
| 1 | 10 | 1 | Left obturator (1/1) | ART |
| 2 | 7 | 1 | Right obturator (1/1) | ART |
| 3 | 10 | 3 | Right common iliac (1/3) | RH |
| Right external iliac (1/3) | ||||
| Left external iliac (1/3) | ||||
| 4 | 8 | 4 | Left common iliac (2/4) | RH |
| Left internal iliac (1/4) | ||||
| Left obturator (1/4) | ||||
| 5 | 6 | 2 | Left suprainguinal (2/2) | RH |
| 6 | 5 | 1 | Right obturator (1/1) | RH |
| Age (Years) | Stage | Histology | Tumor Size (cm) | Lymphovascular Space Invasion | Stromal Invasion | Adjuvant Chemotherapy | Progression-Free Survival (Months) | Outcome |
|---|---|---|---|---|---|---|---|---|
| 36 | IB1 | SCC | 3.7 | Positive | Deep third | 6 cycles of TC | 42 | No evidence of disease |
| 30 | IB1 | Adeno | 1 | Positive | Superficial third | None | 18 | Death |
| Age (Years) | Stage | Histology | Tumor Size (before NAC) (cm) | Conization | NAC |
|---|---|---|---|---|---|
| 32 | IB1 | Adenocarcinoma | 2.4 | − | + |
| 25 | IB1 | Small cell carcinoma | 3.6 | − | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harano, N.; Sakamoto, M.; Fukushima, S.; Iwai, S.; Koike, Y.; Horikawa, S.; Suzuki, K.; Narui, C.; Matsuoka, K.; Ozeki, R.; et al. Clinical Study of Sentinel Lymph Node Detection Using Photodynamic Eye for Abdominal Radical Trachelectomy. Curr. Oncol. 2021, 28, 4709-4720. https://doi.org/10.3390/curroncol28060397
Harano N, Sakamoto M, Fukushima S, Iwai S, Koike Y, Horikawa S, Suzuki K, Narui C, Matsuoka K, Ozeki R, et al. Clinical Study of Sentinel Lymph Node Detection Using Photodynamic Eye for Abdominal Radical Trachelectomy. Current Oncology. 2021; 28(6):4709-4720. https://doi.org/10.3390/curroncol28060397
Chicago/Turabian StyleHarano, Naomi, Masaru Sakamoto, Souta Fukushima, Shinnosuke Iwai, Yuki Koike, Shingo Horikawa, Kayo Suzuki, Chikage Narui, Kazuko Matsuoka, Rinko Ozeki, and et al. 2021. "Clinical Study of Sentinel Lymph Node Detection Using Photodynamic Eye for Abdominal Radical Trachelectomy" Current Oncology 28, no. 6: 4709-4720. https://doi.org/10.3390/curroncol28060397
APA StyleHarano, N., Sakamoto, M., Fukushima, S., Iwai, S., Koike, Y., Horikawa, S., Suzuki, K., Narui, C., Matsuoka, K., Ozeki, R., Iwaya, K., Umayahara, K., Tanaka, T., & Okamoto, A. (2021). Clinical Study of Sentinel Lymph Node Detection Using Photodynamic Eye for Abdominal Radical Trachelectomy. Current Oncology, 28(6), 4709-4720. https://doi.org/10.3390/curroncol28060397







