The Challenge of Return to Work after Breast Cancer: The Role of Family Situation, CANTO Cohort
Abstract
:1. Introduction
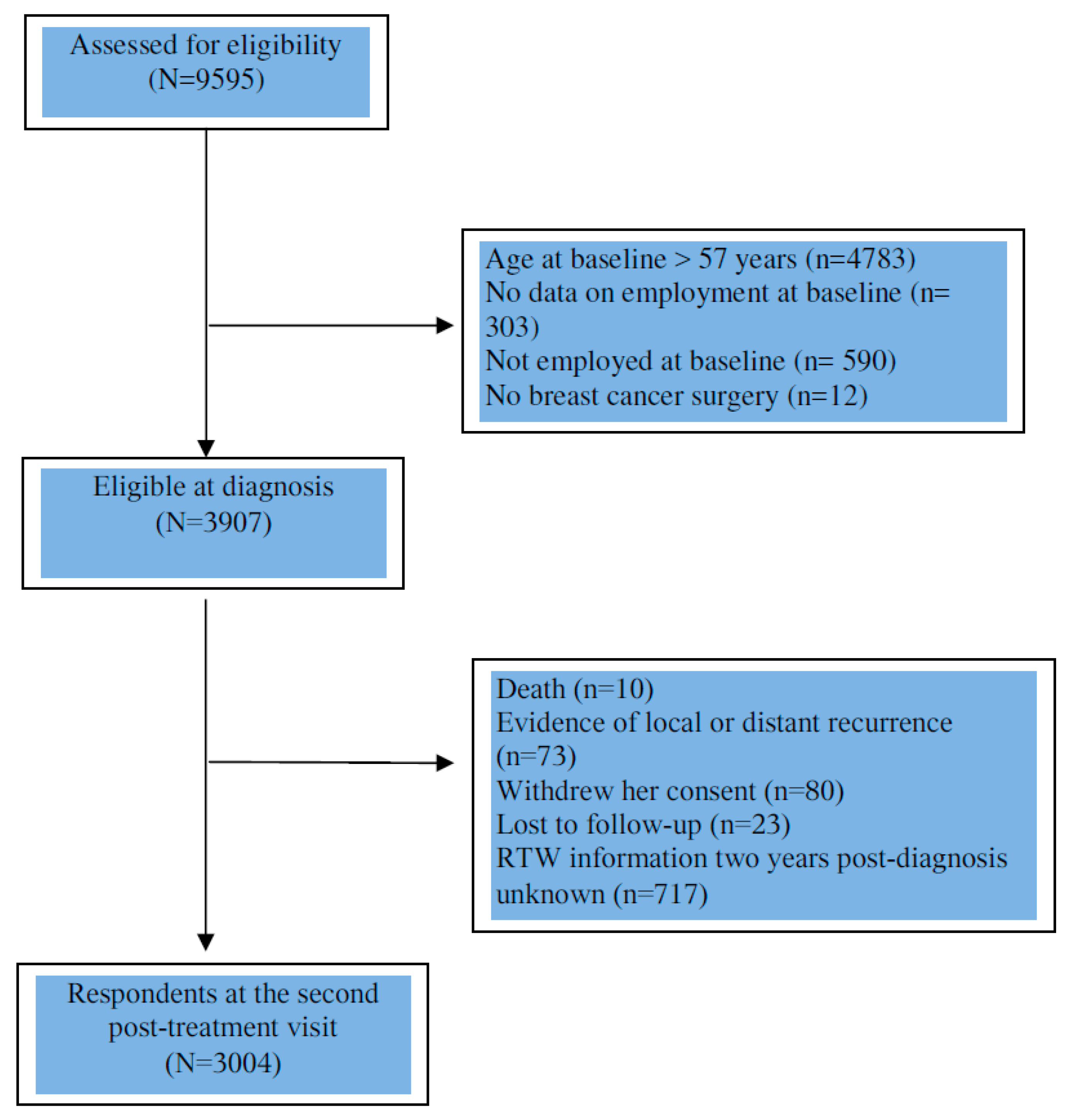
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Société Canadienne du Cancer. Statistics on Breast Cancer. Available online: https://www.cancer.ca:443/fr-ca/cancer-information/cancer-type/breast/statistics/?region=on (accessed on 15 December 2019).
- Di Cosimo, S.; Appierto, V.; Pizzamiglio, S.; Silvestri, M.; Baselga, J.; Piccart, M.; Huober, J.; Izquierdo, M.; de la Pena, L.; Hilbers, F.S.; et al. Early Modulation of Circulating MicroRNAs Levels in HER2-Positive Breast Cancer Patients Treated with Trastuzumab-Based Neoadjuvant Therapy. Int. J. Mol. Sci. 2020, 21, 1386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Von Minckwitz, G.; Procter, M.; de Azambuja, E.; Zardavas, D.; Benyunes, M.; Viale, G.; Suter, T.; Arahmani, A.; Rouchet, N.; Clark, E.; et al. Adjuvant Pertuzumab and Trastuzumab in Early HER2-Positive Breast Cancer. N. Eng. J. Med. 2017, 377, 122–131. [Google Scholar] [CrossRef]
- Santé Public France. National Estimates of Cancer Incidence and Mortality in Metropolitan France between 1990 and 2018—Volume 1: Solid Tumors: Study Based on the Cancer Registers of the Francim Network. Available online: https://www.santepubliquefrance.fr/maladies-et-traumatismes/cancers/cancer-du-sein/documents/rapport-synthese/estimations-nationales-de-l-incidence-et-de-la-mortalite-par-cancer-en-france-metropolitaine-entre-1990-et-2018-volume-1-tumeurs-solides-etud (accessed on 7 January 2020).
- Butow, P.; Laidsaar-Powell, R.; Konings, S.; Lim, C.Y.S.; Koczwara, B. Return to work after a cancer diagnosis: A meta-review of reviews and a meta-synthesis of recent qualitative studies. J. Cancer Surviv. 2020, 14, 114–134. [Google Scholar] [CrossRef]
- Cocchiara, R.; Sciarra, I.; D’Egidio, V.; Sestili, C.; Mancino, M.; Backhaus, I.; Mannocci, A.; De Luca, A.; Frusone, F.; Di Bella, O.; et al. Returning to work after breast cancer: A systematic review of reviews. Work 2018, 61, 463–476. [Google Scholar] [CrossRef]
- Ganz, P.A. Current issues in cancer rehabilitation. Cancer 1990, 65, 742–751. [Google Scholar] [CrossRef]
- Azzani, M.; Roslani, A.C.; Su, T.T. The perceived cancer-related financial hardship among patients and their families: A systematic review. Support. Care Cancer 2014, 23, 889–898. [Google Scholar] [CrossRef]
- Duijts, S.F.A.; Van Egmond, M.P.; Gits, M.; Van Der Beek, A.J.; Bleiker, E.M. Cancer survivors’ perspectives and experiences regarding behavioral determinants of return to work and continuation of work. Disabil. Rehabil. 2016, 39, 2164–2172. [Google Scholar] [CrossRef]
- Waddell, G.; Burton, A.K. Is Work Good for Your Health and Well-Being? The Stationery Office: London, UK, 2006. [Google Scholar]
- Dumas, A.; Luis, I.V.; Bovagnet, T.; El Mouhebb, M.; Di Meglio, A.; Pinto, S.; Charles, C.; Dauchy, S.; Delaloge, S.; Arveux, P.; et al. Impact of Breast Cancer Treatment on Employment: Results of a Multicenter Prospective Cohort Study (CANTO). J. Clin. Oncol. 2020, 38, 734–743. [Google Scholar] [CrossRef]
- Ekenga, C.C.; Pérez, M.; Margenthaler, J.A.; Jeffe, D.B. Early-stage breast cancer and employment participation after 2 years of follow-up: A com-parison with age-matched controls. Cancer 2018, 124, 2026. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; Kang, H.S.; Lee, K.S.; Lee, E.S. Three-Year Prospective Cohort Study of Factors Associated with Return to Work after Breast Cancer Diagnosis. J. Occup. Rehabil. 2016, 27, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.E.; Scherer, S.; Wiskemann, J.; Steindorf, K. Return to work after breast cancer: The role of treatment-related side effects and potential impact on quality of life. Eur. J. Cancer Care 2019, 28, e13051. [Google Scholar] [CrossRef]
- Mehnert, A.; De Boer, A.; Feuerstein, M. Employment challenges for cancer survivors. Cancer 2013, 119, 2151–2159. [Google Scholar] [CrossRef]
- Porro, B.; Durand, M.-J.; Petit, A.; Bertin, M.; Roquelaure, Y. Return to work of breast cancer survivors: Toward an integrative and transactional conceptual model. J. Cancer Surviv. 2021, 1–14. [Google Scholar] [CrossRef]
- Salonen, P.; Rantanen, A.; Kellokumpu-Lehtinen, P.-L.; Huhtala, H.; Kaunonen, M. The quality of life and social support in significant others of pa-tients with breast cancer—A longitudinal study. Eur. J. Cancer Care 2014, 23, 274–283. [Google Scholar] [CrossRef]
- Zissimopoulos, J.M.; Karney, B.; Rauer, A.J. Marriage and economic well-being at older ages. Rev. Econ. Househ. 2013, 13, 1–35. [Google Scholar] [CrossRef]
- Suwankhong, D.; Liamputtong, P. Social Support and Women Living with Breast Cancer in the South of Thailand. J. Nurs. Sch. 2015, 48, 39–47. [Google Scholar] [CrossRef]
- Fantoni, S.Q.; Peugniez, C.; Duhamel, A.; Skrzypczak, J.; Frimat, P.; Leroyer, A. Factors Related to Return to Work by Women with Breast Cancer in Northern France. J. Occup. Rehabil. 2009, 20, 49–58. [Google Scholar] [CrossRef]
- Wang, L.; Hong, B.Y.; Kennedy, S.A.; Chang, Y.; Hong, C.J.; Craigie, S.; Kwon, H.Y.; Romerosa, B.; Couban, R.J.; Reid, S.; et al. Predictors of Unemployment after Breast Cancer Surgery: A Systematic Review and Meta-Analysis of Observational Studies. J. Clin. Oncol. 2018, 36, 1868–1879. [Google Scholar] [CrossRef] [PubMed]
- Heuser, C.; Halbach, S.; Kowalski, C.; Enders, A.; Pfaff, H.; Ernstmann, N. Sociodemographic and disease-related determinants of return to work among women with breast cancer: A German longitudinal cohort study. BMC Health Serv. Res. 2018, 18, 1000. [Google Scholar] [CrossRef]
- Johnsson, A.; Fornander, T.; Olsson, M.; Nystedt, M.; Johansson, H.; Rutqvist, L.E. Factors associated with return to work after breast cancer treatment. Acta Oncol. 2007, 46, 90–96. [Google Scholar] [CrossRef] [Green Version]
- Nilsson, M.; Olsson, M.; Wennman-Larsen, A.; Petersson, L.-M.; Alexanderson, K. Return to work after breast cancer: Women’s experiences of encounters with different stakeholders. Eur. J. Oncol. Nurs. 2011, 15, 267–274. [Google Scholar] [CrossRef]
- De Boer, A.G.; Torp, S.; Popa, A.; Horsboel, T.; Zadnik, V.; Rottenberg, Y.; Bardi, E.; Bultmann, U.; Sharp, L. eLong-term work retention after treatment for cancer: A systematic review and meta-analysis. J. Cancer Surviv. 2020, 14, 135–150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landeiro, L.C.G.; Gagliato, D.M.; Fêde, A.B.; Fraile, N.M.; Lopez, R.M.; da Fonseca, L.G.; Testa, L.; Hoff, P.M.; Mano, M.S. Return to work after breast cancer diagnosis: An obser-vational prospective study in Brazil. Cancer 2018, 124, 4700–4710. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carlsen, K.; Jensen, A.J.; Rugulies, R.; Christensen, J.; Bidstrup, P.E.; Johansen, C.; Madsen, I.E.H.; Dalton, S.O. Self-reported work ability in long-term breast cancer survi-vors. A population-based questionnaire study in Denmark. Acta Oncol. 2013, 52, 423–429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- INSEE. Nuptiality—Demographic Report 2019. Available online: https://www.insee.fr/fr/statistiques/1892240?sommaire=1912926 (accessed on 11 August 2020).
- Bureau UC. Historical Marital Status Tables. The United States Census Bureau. Available online: https://www.census.gov/data/tables/time-series/demo/families/marital.html (accessed on 14 September 2020).
- CDC. Child Development: Middle Childhood (6–8 Years Old)|CDC Centers for Disease Control and Prevention. 2021. Available online: https://www.cdc.gov/ncbddd/childdevelopment/positiveparenting/middle.html (accessed on 28 July 2021).
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE); Version 4.0.; US Department of Health and Human Services: Washington, DC, USA, 2009; p. 196.
- Giesinger, J.M.; Kuijpers, W.; Young, T.; Tomaszewski, K.; Friend, E.; Zabernigg, A.; Holzner, B.; Aaronson, N.K. Thresholds for clinical importance for four key domains of the EORTC QLQ-C30: Physical functioning, emotional functioning, fatigue and pain. Health Qual. Life Outcomes 2016, 14, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Zigmond, A.S.; Snaith, R.P. The Hospital Anxiety and Depression Scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [Green Version]
- Bouknight, R.R.; Bradley, C.J.; Luo, Z. Correlates of Return to Work for Breast Cancer Survivors. J. Clin. Oncol. 2006, 24, 345–353. [Google Scholar] [CrossRef] [Green Version]
- Domínguez-Folgueras, M.; Lesnard, L. Family and social changement. L’Ann. Sociol. 2018, 68, 295–314. [Google Scholar]
- Eurostat. Conciliation between Work and Family Life in the EU25 in 2003. Available online: https://ec.europa.eu/eurostat/web/products-euro-indicators/-/3-12042005-ap (accessed on 14 March 2011).
- Tamminga, S.J.; de Boer, A.G.E.M.; Verbeek, J.H.A.M.; Frings-Dresen, M.H.W. Breast cancer survivors’ views of factors that influence the return-to-work process—A qualitative study. Scand. J. Work Environ. Health 2012, 38, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Liamputtong, P.; Suwankhong, D. Breast cancer diagnosis: Biographical disruption, emotional experiences and strategic management in Thai women with breast cancer. Sociol. Health Illn. 2015, 37, 1086–1101. [Google Scholar] [CrossRef] [PubMed]
- Trusson, D.; Pilnick, A.; Roy, S. A new normal? Women’s experiences of biographical disruption and liminality following treatment for early stage breast cancer. Soc. Sci. Med. 2016, 151, 121–129. [Google Scholar] [CrossRef]
| N (%) † | % RTW | |
|---|---|---|
| Entire study population (N = 3004) | ||
| Living with a partner | ||
| Yes | 2305 (78.5) | 79.5 |
| No | 632 (21.5) | 79.9 |
| Missing | 67 | |
| Household structure | ||
| Single woman with no economically dependent children | 284 (9.7) | 80.3 |
| Partnered woman with no economically dependent children | 646 (22.2) | 76.5 |
| Single mother | 333 (11.4) | 79.9 |
| Partnered woman with economically dependent children | 1653 (56.7) | 80.7 |
| Missing | 88 | |
| Women living with a partner (N = 2305) | ||
| Marital status | ||
| Married | 1675 (73.0) | 79.0 |
| Not married | 618 (27.0) | 81.1 |
| Missing | 12 | |
| Number of economically dependent children | ||
| 0 | 646 (28.1) | 76.5 |
| 1 | 524 (22.8) | 79.0 |
| 2 | 833 (36.2) | 82.4 |
| 3 or more | 296 (12.9) | 79.1 |
| Missing | 6 | |
| Support by the partner | ||
| Very strong | 1930 (84.7) | 79.4 |
| A little, not enough, not at all | 348 (15.3) | 80.5 |
| Missing | 27 | |
| All Women | Analyses by Age Group | Analyses by Monthly Household Income | |||
|---|---|---|---|---|---|
| N = 2347 | <50 Years of Age N = 1412 | ≥50 Years of Age N = 935 | <2500 Euros N = 673 | ≥2500 Euros N = 1674 | |
| OR † [95% CI] | OR † [95% CI] | OR † [95% CI] | OR † [95% CI] | OR † [95% CI] | |
| Living with a partner | |||||
| No | 1 | 1 | 1 | 1 | 1 |
| Yes | 0.63 [0.47–0.86] | 0.71 [0.47–1.08] | 0.55 [0.35–0.88] | 0.68 [0.47–1.00] | 0.56 [0.32–0.99] |
| Household structure | |||||
| Single woman with no economically dependent children | 1.62 [1.07–2.47] | 1.49 [0.82–2.73] | 1.77 [0.96–3.25] | 1.7 [0.98–2.84] | 2.23 [0.88–5.65] |
| Partnered woman with no economically dependent children | 0.90 [0.68–1.20] | 1.04 [0.65–1.66] | 0.84 [0.57–1.24] | 1.22 [0.67–2.21] | 0.81 [0.58–1.12] |
| Single mother with economically dependent children | 1.45 [0.98–2.13] | 1.38 [0.85–2.25] | 1.47 [0.76–2.86] | 1.48 [0.90–2.45] | 1.37 [0.68–2,76] |
| Partnered woman with economically dependent children | 1 | 1 | 1 | 1 | 1 |
| All Partnered Women | Analyses by Age Group | Analyses by Monthly Household Income | |||
|---|---|---|---|---|---|
| N = 1817 | <50 Years of Age N = 1100 | ≥50 Years of Age N = 717 | <2500 Euros N = 314 | ≥2500 Euros N = 1503 | |
| OR † [95% CI] | OR † [95% CI] | OR † [95% CI] | OR † [95% CI] | OR † [95% CI] | |
| Marital status | |||||
| Not married | 1 | 1 | 1 | 1 | 1 |
| Married | 0.79 [0.59- 1.06] | 0.92 [0.64–1.33] | 0.57 [0.34- 0.95] | 0.63 [0.34–1.18] | 0.88 [0.63–1.23] |
| Number of economically dependent children | |||||
| 0 | 1 | 1 | 1 | 1 | 1 |
| 1 | 1.02 [0.72–1.44] | 0.76 [0.44–1.34] | 1.30 [0.80–2.09] | 1.03 [0.46–2.28] | 1.02 [0.69–1.51] |
| 2 | 1.25 [0.88–1.77] | 1.11 [0.66–1.9] | 1.17 [0.69–2.00] | 0.65 [0.28–1,48] | 1.42 [0.96–2.10] |
| 3 or more | 0.9 [0.64–1.55] | 0.79 [0.44–1.42] | 1.34 [0.48–3.76] | 0.28 [0.10–0,80] | 1.33 [0.79–2.23] |
| Perceived support by the partner | |||||
| A little, not enough, not at all | 1 | 1 | 1 | 1 | 1 |
| Verystrong | 0.83 [0.58–1.19] | 0.72 [0.43–1.21] | 1.05 [0.62–1.76] | 0.52 [0.24–1.11] | 0.97 [0.64–1.48] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caumette, E.; Vaz-Luis, I.; Pinto, S.; Havas, J.; Bovagnet, T.; Ruiz de Azua, G.; Di Meglio, A.; Martin, A.-L.; Everhard, S.; Cottu, P.; et al. The Challenge of Return to Work after Breast Cancer: The Role of Family Situation, CANTO Cohort. Curr. Oncol. 2021, 28, 3866-3875. https://doi.org/10.3390/curroncol28050330
Caumette E, Vaz-Luis I, Pinto S, Havas J, Bovagnet T, Ruiz de Azua G, Di Meglio A, Martin A-L, Everhard S, Cottu P, et al. The Challenge of Return to Work after Breast Cancer: The Role of Family Situation, CANTO Cohort. Current Oncology. 2021; 28(5):3866-3875. https://doi.org/10.3390/curroncol28050330
Chicago/Turabian StyleCaumette, Elsa, Inès Vaz-Luis, Sandrine Pinto, Julie Havas, Thomas Bovagnet, Garazi Ruiz de Azua, Antonio Di Meglio, Anne-Laure Martin, Sibille Everhard, Paul Cottu, and et al. 2021. "The Challenge of Return to Work after Breast Cancer: The Role of Family Situation, CANTO Cohort" Current Oncology 28, no. 5: 3866-3875. https://doi.org/10.3390/curroncol28050330
APA StyleCaumette, E., Vaz-Luis, I., Pinto, S., Havas, J., Bovagnet, T., Ruiz de Azua, G., Di Meglio, A., Martin, A.-L., Everhard, S., Cottu, P., Vanlemmens, L., Jouannaud, C., Lerebours, F., Dumas, A., & Menvielle, G. (2021). The Challenge of Return to Work after Breast Cancer: The Role of Family Situation, CANTO Cohort. Current Oncology, 28(5), 3866-3875. https://doi.org/10.3390/curroncol28050330





