CRAFT—A Proposed Framework for Decentralized Clinical Trials Participation in Canada
Abstract
1. Introduction
2. Methods
3. Results
3.1. Results from the Literature Review
3.2. Two Case Studies of Remote Trial Participation
3.2.1. COSA Australasian Tele-Trial Model (COSA ATM)
3.2.2. Pediatric Oncology Group of Ontario (POGO) Satellite Program
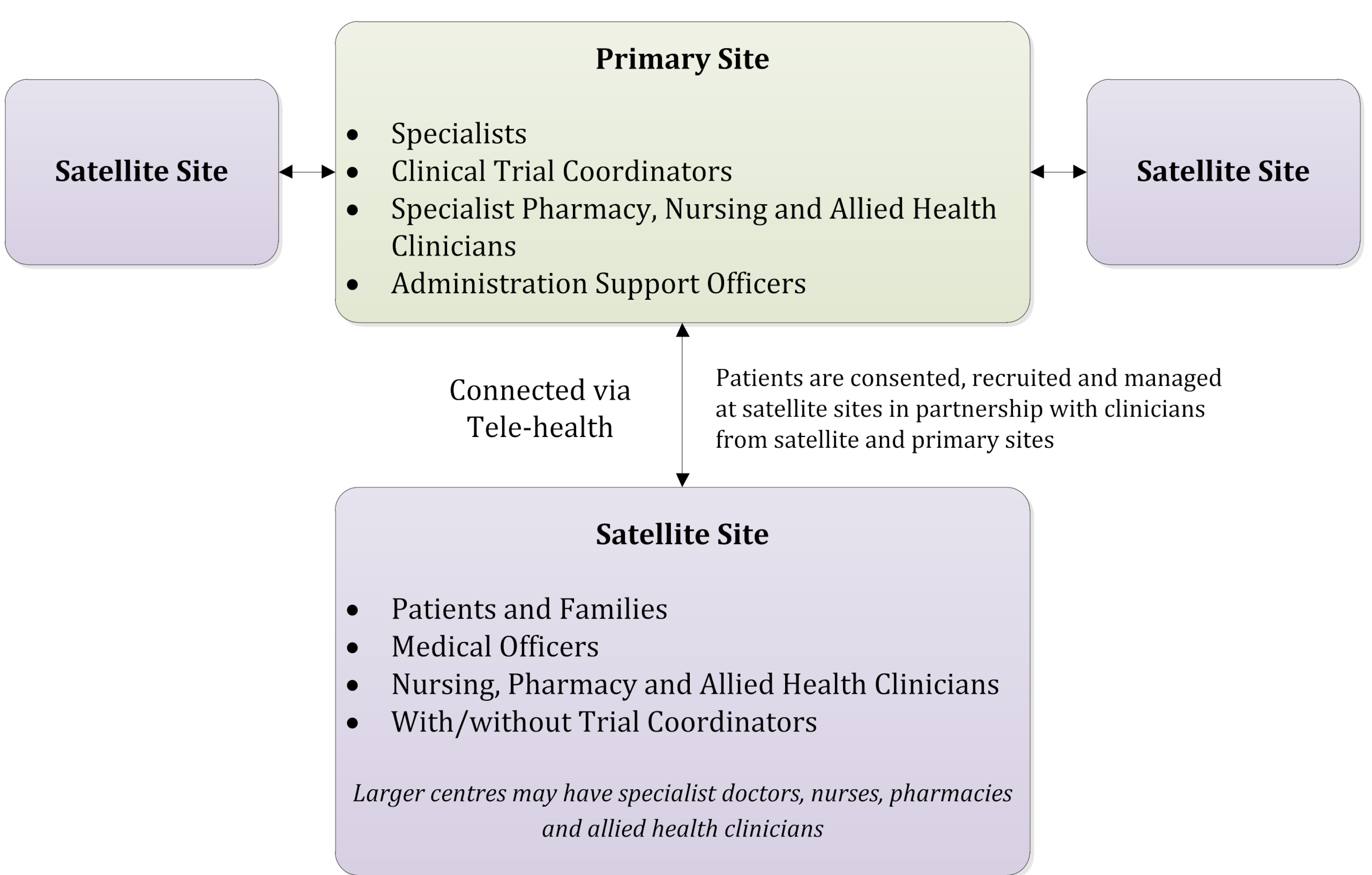
3.3. Recommended Model
3.4. Canadian Remote Access Framework for Clinical Trials (CRAFT) Recommendations
- Conduct pilot studies to evaluate the model and share findings. Pilot implementation projects should build upon existing regional networks of shared clinical care with personnel that are supportive of improving trial participation. Pilot clusters would extend care delivery to include trial delivery, leveraging existing regional patterns of care and telemedicine capacity across site networks. The cluster could begin by participating in a trial of interventions of lower risk and complexity, such as a trial assessing different standard of care treatments or supportive care measures. Such a trial experience would establish the cluster, and the processes to ensure trial oversight and conduct (e.g., site contracts, REB, training, delegation of responsibilities).
- Identify agreement terms among investigators, sub-investigators, healthcare providers (or their representatives), insurers and sponsors that can address research responsibilities, professional liability, and indemnity. Ideally, a core set of template documents could be developed to be used by those interested in implementing the model and framework.
- Identify feasible and cost-effective options for establishing linkages between centres that consider professional capacity, existing workflows, and scheduling requirements. Research activities should be distributed to efficiently and effectively ensure optimal engagement of personnel and resources.
- Continue to consult with Health Canada to ensure recognition and support of models of trial conduct within a cluster across regulations, guidance documents, review of clinical trial applications and site/sponsor inspections.
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Member | Affiliation(s) | Title(s) |
|---|---|---|
| Stephen Sundquist * (Chair) | 3CTN | Executive Director |
| Gerry Batist | Quebec-Clinical Research Organization in Cancer | Scientific Director |
| Kathy Brodeur-Robb * | C-17 | Executive Director |
| Janet Dancey * | 3CTN; Canadian Cancer Trials Group | Scientific Director; Executive Director |
| Kathryn Dyck | CancerCare Manitoba | Clinical Trials Manager |
| Bernie Eigl | BC Cancer | Provincial Director—Clinical Trials |
| David K. Lee | Health Canada—Health Products and Food Branch | Chief Regulatory Officer |
| Jacqueline Limoges * | Ontario Cancer Research Ethics Board | Chair |
| Jim Pankovich | Bold Therapeutics Inc | Executive Vice President, Clinical Development |
| Anna Sadura | Canadian Cancer Trials Group | Manager, Trial Management Group (retired) |
| Patrick Sullivan | Team Finn Foundation; 3CTN; Canadian Cancer Trials Group | Childhood Cancer Research Advocacy; Patient Representative; Research Advisor |
| Holly Longstaff * (Writing Committee only) | Provincial Health Services Authority | Director, Privacy and Access, PHSA Research and New Initiatives Research & Academic Services |
| *Writing Committee Member |
References
- Canadian Partnership Against Cancer. The 2018 Cancer System Performance Report; Canadian Partnership Against Cancer: Toronto, ON, Canada, 2018. [Google Scholar]
- Sinha, G. United Kingdom Becomes the Cancer Clinical Trials Recruitment Capital of the World. J. Natl. Cancer Inst. 2007, 99, 420–422. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Statistics Canada. Population Centre. 2017. Available online: https://www12.statcan.gc.ca/census-recensement/2016/ref/dict/geo049a-eng.cfm (accessed on 13 January 2020).
- Canadian Organization of Medical Physicists. Canadian Cancer Centres. Available online: https://www.comp-ocpm.ca/english/career-education/career-resources/canadian-cancer-centres.html (accessed on 14 February 2020).
- Unger, J.M.; Cook, E.; Tai, E.; Bleyer, A. The Role of Clinical Trial Participation in Cancer Research: Barriers, Evidence, and Strategies. Am. Soc. Clin. Oncol. Educ. Book 2016, 35, 185–198. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society Cancer Action Network. Barriers to Patient Enrollment in Therapeutic Clinical Trials for Cancer: A Landscape Report; American Cancer Society Cancer Action Network: Washington, DC, USA, 2018. [Google Scholar]
- Schroen, A.T.; Petroni, G.R.; Wang, H.; Gray, R.; Wang, X.F.; Cronin, W.; Sargent, D.; Benedetti, J.; Wickerham, D.L.; Djulbegovic, B.; et al. Preliminary evaluation of factors associated with premature trial closure and feasibility of accrual benchmarks in phase III oncology trials. Clin. Trials 2010, 7, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Government of Canada. TCPS 2 (2018)—Chapter 1: Ethics Framework. Government of Canada; 2019. Available online: https://ethics.gc.ca/eng/tcps2-eptc2_2018_chapter1-chapitre1.html#b (accessed on 8 January 2020).
- Government of Canada. Canada Health Act. In R.S.C.; Government of Canada; 1985. [Google Scholar]
- Health Canada. Management of Clinical Trials during the COVID-19 Pandemic: Notice to Clinical Trial Sponsors. 2021. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/announcements/management-clinical-trials-during-covid-19-pandemic.html (accessed on 27 May 2020).
- U.S. Food and Drug Administration. Conduct of Clinical Trials of Medical Products During the COVID-19 Public Health Emergency; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2020. [Google Scholar]
- European Medicines Agency. Guidance on the Management of Clinical Trials During the COVID-19 (Coronavirus) Pandemic; European Medicines Agency: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Queensland Health. Queensland Health Teletrials Pilot Analysis Report; Queensland Health: Brisbane, Australia, 2019. [Google Scholar]
- Clinical Trials Transformation Initiative. Decentralized Clinical Trials 2. 2018. Available online: https://www.ctti-clinicaltrials.org/sites/www.ctti-clinicaltrials.org/files/dct_recommendations_final.pdf (accessed on 10 February 2020).
- Unger, J.M.; Vaidya, R.; Hershman, D.L.; Minasian, L.M.; Fleury, M.E. Systematic Review and Meta-Analysis of the Magnitude of Structural, Clinical, and Physician and Patient Barriers to Cancer Clinical Trial Participation. J. Natl. Cancer Inst. 2019, 111, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Federation of Medical Regulatory Authorities of Canada. FMRAC Framework on Telemedicine. Available online: http://fmrac.ca/fmrac-framework-on-telemedicine/ (accessed on 1 October 2019).
- Humer, M.F.; Campling, B.G. The Role of Telemedicine in Providing Thoracic Oncology Care to Remote Areas of British Columbia. Curr. Oncol. Rep. 2017, 19, 52. [Google Scholar] [CrossRef] [PubMed]
- Basch, E.; Deal, A.M.; Dueck, A.C.; Scher, H.I.; Kris, M.G.; Hudis, C.; Schrag, D. Overall Survival Results of a Trial Assessing Patient-Reported Outcomes for Symptom Monitoring During Routine Cancer Treatment. JAMA 2017, 318, 197–198. [Google Scholar] [CrossRef] [PubMed]
- Sabesan, S.; Zalcberg, J. Telehealth models could be extended to conducting clinical trials-a teletrial approach. Eur. J. Cancer Care 2016, 27, e12587. [Google Scholar] [CrossRef] [PubMed]
- Alexander, S.; Greenberg, M.; Malkin, D.; Portwine, C.; Johnston, D.; Silva, M.; Zelcer, S.; Sonshine, S.; Manzo, J.; Bennett, C.; et al. Pediatric oncology clinical trial participation where the geography is vast: Development of a clinical research system for tertiary and satellite centers in Ontario, Canada. Pediatr. Blood Cancer 2017, 65, e26901. [Google Scholar] [CrossRef] [PubMed]
- Clinical Oncology Society of Australia. Australasian Tele-Trial Model: A National Guide for Implementation; Clinical Oncology Society of Australia: Sydney, Australia, 2016. [Google Scholar]
- Sabesan, S.; Zalcberg, J.; Underhill, C.; Zielinski, R.; Burbury, K.; Ansari, Z.; Rainey, N.; Collins, I.; Osbourne, R.; Sanmugarajah, J.; et al. Implementation of the Australasian Teletrial Model: Lessons from practice. Asia Pac. J. Clin. Oncol. 2019, 15 (Suppl. 8), 3–14. [Google Scholar]
- Clinical Oncology Society of Australia. Pilot Implementation of the Australasian Tele-Trial Model Project Completion Report; Clinical Oncology Society of Australia: Sydney, Australia, 2021. [Google Scholar]
- Hirsch, B.R.; Locke, S.; Abernethy, A.P. Experience of the National Cancer Institute Community Cancer Centers Program on Community-Based Cancer Clinical Trials Activity. J. Oncol. Pract. 2016, 12, e350–e358. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pediatric Oncology Group of Ontario. Satellite Manual. 1997. Available online: https://www.pogo.ca/satellite-manual/ (accessed on 1 August 2019).
- U.S. Food and Drug Administration. Advancing Oncology Decentralized Trials. 2021. Available online: https://www.fda.gov/about-fda/oncology-center-excellence/advancing-oncology-decentralized-trials (accessed on 27 May 2021).
- Clinical Trials and Translational Research Advisory Committee. Strategic Planning Working Group Report. 2020. Available online: https://deainfo.nci.nih.gov/advisory/ctac/1120/Loehrer1.pdf (accessed on 27 May 2021).
| Network | Description |
|---|---|
| Alberta’s Community Cancer Network | Comprised of two tertiary centres, four associate centres and 11 community cancer centres. Community cancer centres must satisfy eligibility criteria for safe and effective chemotherapy treatment and follow-up care. Provides treatment, psychosocial & palliative care, prevention and screening services. |
| The North East Regional Community Oncology Clinic Network (COCN) | Tele-oncology program operating out of Sudbury, Ontario serves a population of 600,000 spread over about 300,000 square kilometres. A regional network of fourteen regional satellite clinics offer imaging, chemotherapy and in one case, radiotherapy to 5,000 patient consults annually. |
| Atlantic Provinces Pediatric Hematology/Oncology Network (APPHON) | Platform for healthcare providers, patients and caregivers to access comprehensive health services and clinical care at regional centres located in Halifax, Nova Scotia and St. John’s, Newfoundland as well as education and research related to paediatric hematologic or oncologic disorders and supports for member organizations and groups in development of standards. |
| Trial | Sponsor |
|---|---|
| Abemaciclib Combined with Endocrine Therapy for the Adjuvant Treatment of HR+, HER2-, Node-Positive, High-Risk, Early Breast Cancer (monarchE) | Eli Lilly and Company |
| Targeted thromboprophylaxis in ambulatory patients receiving anticancer therapies (TARGET-TP) | Victorian Cancer Agency/Peter MacCallum Cancer Centre |
| Aspirin for Dukes C and High-Risk Dukes B Colorectal Cancers: An International, Multi-Centre, Double Blind, Randomised Placebo Controlled Phase III Trial (ASCOLT) | Australasian Gastro-Intestinal Trial Group |
| Framework Element (s) | Recommendations | ||
|---|---|---|---|
| 1 | Infrastructure, personnel and system development | 1.1 | Address human resources, equipment and facility requirements at satellite centres. |
| 1.2 | Develop contingency plans to assure patient participation can be supported throughout the course of the clinical trial and long-term follow-up. | ||
| 1.3 | Use a risk-based approach to identify protocol-specific training needs for satellite personnel that is based on the extent of delegated responsibilities and scope of practice. | ||
| 1.4 | Assess what aspects of core clinical trial competency training (ICH GCP E6(2), Ethical Conduct of Research Involving Humans (TCPS 2), may be required for remote activities. | ||
| 1.5 | Establish mentoring relationships with satellite personnel for professional trial competencies development. | ||
| 1.6 | Provide a decision guide for risk-based assessment with criteria for establishing satellite site suitability for a trial. | ||
| 1.7 | Provide templates for clinical trial budgets, agreements between the sponsor and primary site in a cluster as well as sub-agreements between the primary site and each satellite. | ||
| 1.8 | Provide tools (e.g., template checklists) to inform supervision plans and roles and responsibilities for satellite activities. | ||
| 2 | Costs and funding requirements | 2.1 | Invest in applied studies and evaluations that can demonstrate feasibility for a range of trial types, patient populations, distributed care models, trial cluster configurations, etc. |
| 2.2 | Provide financial support to primary sites to support initial costs to create infrastructure, systems, training and visits at satellite centres to set up the cluster. | ||
| 3 | Trial planning and conduct | 3.1 | Design clusters to be robust and flexible to allow the addition of satellite sites throughout the period a trial is open. |
| 3.2 | Leverage pre-existing telemedicine/care delivery practices with satellites, when feasible. | ||
| 3.3 | Engage clinicians and patients from rural and remote sites in trial design. | ||
| 3.4 | Consider protocol accommodations that allow for clinical trial conduct at satellite centre. | ||
| 3.5 | Adopt risk-based criteria to determine remote centres involvement in the trial. Such criteria may include complexity of trial design, product safety profile, or required protocol assessments. | ||
| 3.6 | Adopt a risk-based criteria to determine activities that can be delegated to a satellite site, required staffing complement, qualifications, equipment and facilities. | ||
| 4 | Health Canada regulatory guidelines and inspections | 4.1 | Update or interpret the Health Canada Food and Drug Regulations, Part C, Division 5 “Drugs for Clinical Trials Involving Human Subjects” to recognize the required elements of the proposed framework. Specifically, that:
|
| 4.2 | Health Canada reviews and inspections should recognize the trial cluster, delegation of Qualified Investigator responsibilities to satellite sites and assess regulatory compliance so as not to cause undue burden for the primary site or for satellite sites. | ||
| 5 | Ethics review | 5.1 | Recognize the primary site’s REB as the REB of record for the cluster so as not to introduce added steps or barriers to the ethics review process for satellite sites. |
| 6 | Patient privacy | 6.1 | Adopt the interpretation of the Personal Information Protection and Electronic Documents Act (PIPEDA) legislation and provincial privacy laws that recognizes healthcare professionals performing trial related activities as part of a circle of care may have access to personal health information. |
| 7 | Trial agreements, Indemnity and insurance | 7.1 | Trial sponsors should be willing to execute agreements with the primary site and extend terms of coverage for the scope of a primary site’s coordination of satellite centres. |
| 8 | Engagement, communications and advocacy | 8.1 | Develop dissemination and knowledge mobilization strategies to generating broader awareness and advocacy among sponsors, researchers, clinicians, patient communities, ethics boards and regulators that can be scaled and sustained over time. |
| 8.2 | Create a strategy for health policy advocacy to recognize and support clinical trials as standard of care. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sundquist, S.; Batist, G.; Brodeur-Robb, K.; Dyck, K.; Eigl, B.J.; Lee, D.K.; Limoges, J.; Longstaff, H.; Pankovich, J.; Sadura, A.; et al. CRAFT—A Proposed Framework for Decentralized Clinical Trials Participation in Canada. Curr. Oncol. 2021, 28, 3857-3865. https://doi.org/10.3390/curroncol28050329
Sundquist S, Batist G, Brodeur-Robb K, Dyck K, Eigl BJ, Lee DK, Limoges J, Longstaff H, Pankovich J, Sadura A, et al. CRAFT—A Proposed Framework for Decentralized Clinical Trials Participation in Canada. Current Oncology. 2021; 28(5):3857-3865. https://doi.org/10.3390/curroncol28050329
Chicago/Turabian StyleSundquist, Stephen, Gerald Batist, Kathy Brodeur-Robb, Kathryn Dyck, Bernhard J. Eigl, David K. Lee, Jaqueline Limoges, Holly Longstaff, Jim Pankovich, Anna Sadura, and et al. 2021. "CRAFT—A Proposed Framework for Decentralized Clinical Trials Participation in Canada" Current Oncology 28, no. 5: 3857-3865. https://doi.org/10.3390/curroncol28050329
APA StyleSundquist, S., Batist, G., Brodeur-Robb, K., Dyck, K., Eigl, B. J., Lee, D. K., Limoges, J., Longstaff, H., Pankovich, J., Sadura, A., Sullivan, P., & Dancey, J. E. (2021). CRAFT—A Proposed Framework for Decentralized Clinical Trials Participation in Canada. Current Oncology, 28(5), 3857-3865. https://doi.org/10.3390/curroncol28050329






