Virtual Care in Patients with Cancer: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Systematic Review Planning and Registration
2.2. Literature Search Method
2.3. Search Strategy and Study Selection
2.4. Data Extraction and Assessment of Risk of Bias
2.5. Synthesizing the Evidence
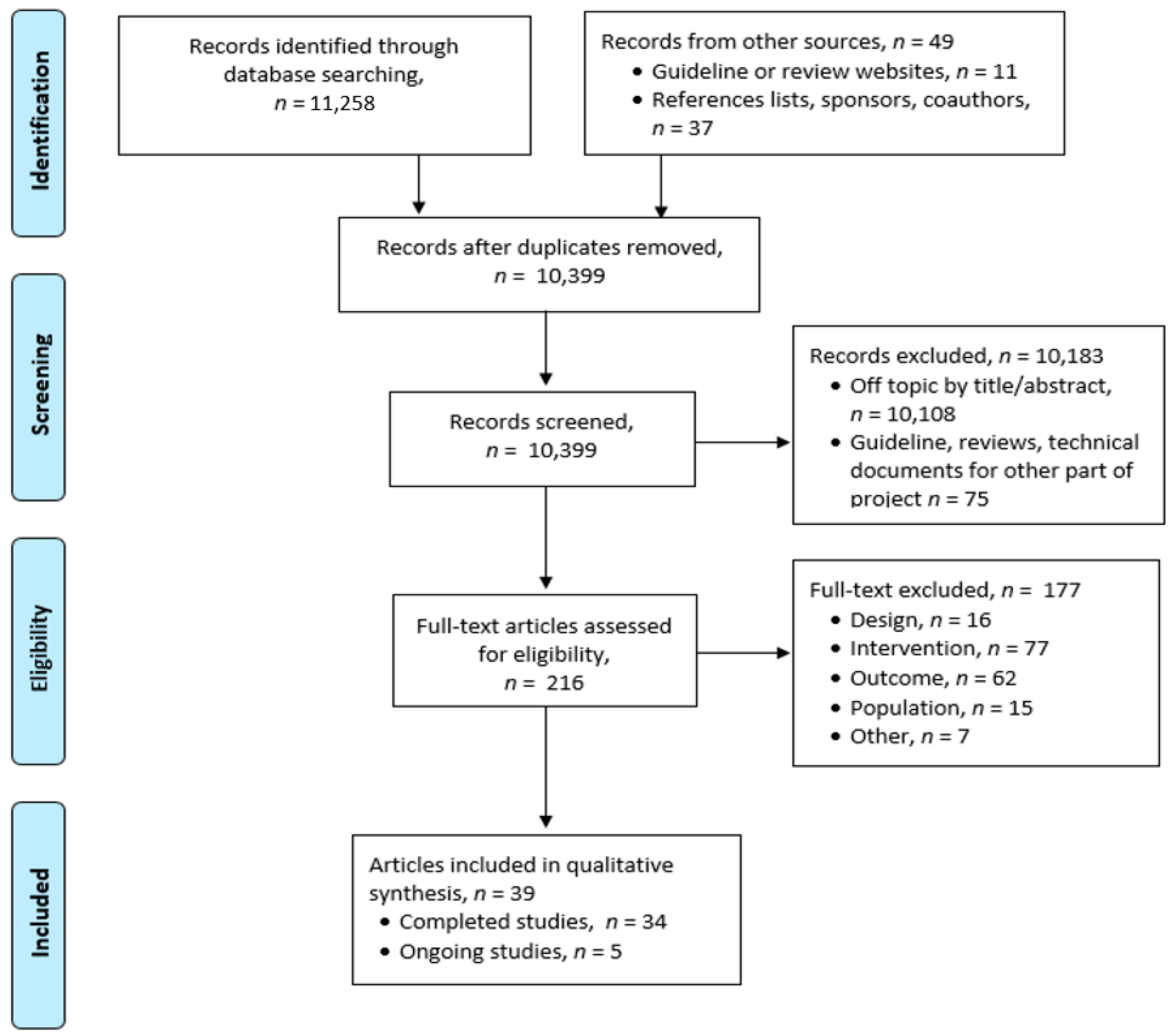
3. Results
3.1. Counselling Studies
3.2. Medical or Supportive Care
3.3. Ongoing, Unpublished, or Incomplete Studies
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions Version 6.1; Updated September 2020; Cochrane: London, UK, 2020; Available online: www.training.cochrane.org/handbook (accessed on 11 March 2021).
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [Green Version]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef] [Green Version]
- Schünemann, H.; Brożek, J.; Guyatt, G.; Oxman, A. (Eds.) Handbook for Grading the Quality of Evidence and the Strength of Recommendations Using the GRADE Approach (Updated October 2013). GRADE Working Group, 2013. Available online: https://gdt.gradepro.org/app/handbook/handbook.html (accessed on 11 March 2021).
- Lleras de Frutos, M.; Medina, J.C.; Vives, J.; Casellas-Grau, A.; Marzo, J.L.; Borras, J.M.; Ochoa-Arnedo, C. Video conference vs face-to-face group psychotherapy for distressed cancer survivors: A randomized controlled trial. Psychooncology 2020, 29, 1995–2003. [Google Scholar] [CrossRef] [PubMed]
- Watson, M.; White, C.; Lynch, A.; Mohammed, K. Telephone-delivered individual cognitive behavioural therapy for cancer patients: An equivalence randomised trial. Psychooncology 2017, 26, 301–308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harrigan, M.; Cartmel, B.; Loftfield, E.; Sanft, T.; Chagpar, A.B.; Zhou, Y.; Playdon, M.; Li, F.; Irwin, M.L. Randomized trial comparing telephone versus in-person weight loss counseling on body composition and circulating biomarkers in women treated for breast cancer: The Lifestyle, Exercise, and Nutrition (LEAN) Study. J. Clin. Oncol. 2016, 34, 669–676. [Google Scholar] [CrossRef] [PubMed]
- van der Lee, M.L.; Schellekens, M.P.J. Bridging the distance: Continuing psycho-oncological care via video-consults during the COVID-19 pandemic. Psychooncology 2020, 13, 13. [Google Scholar]
- Buchanan, A.H.; Datta, S.K.; Skinner, C.S.; Hollowell, G.P.; Beresford, H.F.; Freeland, T.; Rogers, B.; Boling, J.; Marcom, P.K.; Adams, M.B. Randomized trial of telegenetics vs. in-person cancer genetic counseling: Cost, patient satisfaction and attendance. J. Genet. Couns. 2015, 24, 961–970. [Google Scholar] [CrossRef] [Green Version]
- Datta, S.K.; Buchanan, A.H.; Hollowell, G.P.; Beresford, H.F.; Marcom, P.K.; Adams, M.B. Telemedicine vs in-person cancer genetic counseling: Measuring satisfaction and conducting economic analysis. Comp. Eff. Res. (Auckl.) 2011, 1, 43–50. [Google Scholar] [CrossRef]
- Steffen, L.E.; Du, R.; Gammon, A.; Kohlmann, W.K.; Lee, J.H.; Buys, S.S.; Stroup, A.M.; Campo, R.A.; Flores, K.G. Genetic testing in a population-based sample of breast and ovarian cancer survivors from the REACH randomized trial: Cost barriers and moderators of counseling mode. Cancer Epidemiol. Biomarkers Prev. 2017, 26, 1772–1780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kinney, A.Y.; Steffen, L.E.; Brumbach, B.H.; Kohlmann, W.; Du, R.; Lee, J.H.; Gammon, A.; Butler, K.; Buys, S.S.; Stroup, A.M.; et al. Randomized noninferiority trial of telephone delivery of BRCA1/2 genetic counseling compared with in-person counseling: 1-year follow-up. J. Clin. Oncol. 2016, 34, 2914–2924. [Google Scholar] [CrossRef] [Green Version]
- Chang, Y.; Near, A.M.; Butler, K.M.; Hoeffken, A.; Edwards, S.L.; Stroup, A.M.; Kohlmann, W.; Gammon, A.; Buys, S.S.; Schwartz, M.D.; et al. Economic evaluation alongside a clinical trial of telephone versus in-person genetic counseling for BRCA1/2 mutations in geographically underserved areas. J. Oncol. Pract. 2016, 12, e1–e13. [Google Scholar] [CrossRef] [Green Version]
- Jacobs, A.S.; Schwartz, M.D.; Valdimarsdottir, H.; Nusbaum, R.H.; Hooker, G.W.; DeMarco, T.A.; Heinzmann, J.E.; McKinnon, W.; McCormick, S.R.; Davis, C.; et al. Patient and genetic counselor perceptions of in-person versus telephone genetic counseling for hereditary breast/ovarian cancer. Fam. Cancer. 2016, 15, 529–539. [Google Scholar] [CrossRef] [Green Version]
- Peshkin, B.N.; Kelly, S.; Nusbaum, R.H.; Similuk, M.; DeMarco, T.A.; Hooker, G.W.; Valdimarsdottir, H.B.; Forman, A.D.; Joines, J.R.; Davis, C.; et al. Patient perceptions of telephone vs. in-person BRCA1/BRCA2 genetic counseling. J. Genet. Couns. 2016, 25, 472–482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwartz, M.D.; Valdimarsdottir, H.B.; Peshkin, B.N.; Mandelblatt, J.; Nusbaum, R.; Huang, A.-T.; Chang, Y.; Graves, K.; Isaacs, C.; Wood, M.; et al. Randomized noninferiority trial of telephone versus in-person genetic counseling for hereditary breast and ovarian cancer. J. Clin. Oncol. 2014, 32, 618–626. [Google Scholar] [CrossRef]
- Butrick, M.; Kelly, S.; Peshkin, B.N.; Luta, G.; Nusbaum, R.; Hooker, G.W.; Graves, K.; Feeley, L.; Isaacs, C.; Valdimarsdottir, H.B.; et al. Disparities in uptake of BRCA1/2 genetic testing in a randomized trial of telephone counseling. Genet Med. 2015, 17, 467–475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mette, L.A.; Saldivar, A.M.; Poullard, N.E.; Torres, I.C.; Seth, S.G.; Pollock, B.H.; Tomlinson, G.E. Reaching high-risk underserved individuals for cancer genetic counseling by video-teleconferencing. J. Community Support. Oncol. 2016, 14, 162–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solomons, N.M.; Lamb, A.E.; Lucas, F.L.; McDonald, E.F.; Miesfeldt, S. Examination of the patient-focused impact of cancer telegenetics among a rural population: Comparison with traditional in-person services. Telemed. J. e-Health 2018, 24, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Tutty, E.; Petelin, L.; McKinley, J.; Young, M.A.; Meiser, B.; Rasmussen, V.M.; Forbes Shepherd, R.; James, P.A.; Forrest, L.E. Evaluation of telephone genetic counselling to facilitate germline BRCA1/2 testing in women with high-grade serous ovarian cancer. Eur. J. Hum. Genet. 2019, 27, 1186–1196. [Google Scholar] [CrossRef]
- Kelleher, S.A.; Winger, J.G.; Dorfman, C.S.; Ingle, K.K.; Moskovich, A.A.; Abernethy, A.P.; Keefe, F.J.; Samsa, G.P.; Kimmick, G.G.; Somers, T.J. A behavioral cancer pain intervention: A randomized noninferiority trial comparing in-person with videoconference delivery. Psychooncology 2019, 28, 1671–1678. [Google Scholar] [CrossRef]
- Winger, J.G.; Nunez, C.; Kelleher, S.A.; Ingle, K.K.; Gandhi, V.; Keefe, F.J.; Somers, T.J. Predictors of intervention session completion in a randomized clinical trial of a behavioral cancer pain intervention. J. Pain Symptom Manag. 2020, 59, 1268–1277. [Google Scholar] [CrossRef] [PubMed]
- Walle, T. NCT MOBILE—Smartphone-Based Video Consultations in Medical Oncology, 2018. Available online: http://www.drks.de/DRKS00015788 (accessed on 29 October 2020).
- Walle, T.; Erdal, E.; Mühlsteffen, L.; Singh, H.M.; Gnutzmann, E.; Grün, B.; Hofmann, H.; Ivanova, A.; Köhler, B.; Korell, F.; et al. Completion rate and impact on physician-patient relationship of video consultations in medical oncology: A randomized controlled open label trial. ESMO Open 2020, 5, e000912. [Google Scholar] [CrossRef] [PubMed]
- Beaver, K.; Williamson, S.; Sutton, C.; Hollingworth, W.; Gardner, A.; Allton, B.; Abdel-Aty, M.; Blackwood, K.; Burns, S.; Curwen, D.; et al. Comparing hospital and telephone follow-up for patients treated for stage-I endometrial cancer (ENDCAT trial): A randomised, multicentre, non-inferiority trial. BJOG 2017, 124, 150–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dixon, P.; Beaver, K.; Williamson, S.; Sutton, C.; Martin-Hirsch, P.; Hollingworth, W. Cost-consequence analysis alongside a randomised controlled trial of hospital versus telephone follow-up after treatment for endometrial cancer. Appl. Health Econ. Health Policy 2018, 16, 415–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beaver, K.; Williamson, S.; Sutton, C.J.; Gardner, A.; Martin-Hirsch, P. Endometrial cancer patients’ preferences for follow-up after treatment: A cross-sectional survey. Eur. J. Oncol. Nurs. 2020, 45, 101722. [Google Scholar] [CrossRef]
- Viers, B.R.; Lightner, D.J.; Rivera, M.E.; Tollefson, M.K.; Boorjian, S.A.; Karnes, R.J.; Thompson, R.H.; O’Neil, D.A.; Hamilton, R.L.; Gardner, M.R.; et al. Efficiency, satisfaction, and costs for remote video visits following radical prostatectomy: A randomized controlled trial. Eur. Urol. 2015, 68, 729–735. [Google Scholar] [CrossRef]
- Chan, B.A.; Larkins, S.L.; Evans, R.; Watt, K.; Sabesan, S. Do teleoncology models of care enable safe delivery of chemotherapy in rural towns? Med. J. Aust. 2015, 203, 406–406.e6. [Google Scholar] [CrossRef]
- Hamilton, E.; Van Veldhuizen, E.; Brown, A.; Brennan, S.; Sabesan, S. Telehealth in radiation oncology at the Townsville Cancer Centre: Service evaluation and patient satisfaction. Clin. Transl. Radiat. Oncol. 2019, 15, 20–25. [Google Scholar] [CrossRef] [Green Version]
- Jue, J.S.; Spector, S.A.; Spector, S.A. Telemedicine broadening access to care for complex cases. J. Surg. Res. 2017, 220, 164–170. [Google Scholar] [CrossRef]
- Li, M.; Zhang, M.; Wang, H.; Pan, X.; Wu, W.; Zhang, Q.; Liu, Y.; Zhang, H. The efficacy of internet-based intervention on quality of life for patients with chronic post-surgical pain. Iran. J. Public Health 2016, 45, 1604–1609. [Google Scholar] [PubMed]
- Verma, R.; Treasure, P.; Hughes, R. Development and evaluation of radiographer led telephone follow up following radical radiotherapy to the prostate. A report of a Macmillan Cancer Support sponsored pilot project at Mount Vernon Hospital. Radiography 2015, 21, 16–24. [Google Scholar] [CrossRef]
- Patil, V.M.; Pande, N.; Chandrasekharan, A.M.C.; Tonse, R.; Krishnatry, R.; Goda, J.S.; Dsouza, H.; Vallathol, D.H.; Chakraborty, S. Shadow study: Randomized comparison of clinic with video follow-up in glioma undergoing adjuvant temozolomide therapy. CNS Oncol. 2018, 7, CNS14. [Google Scholar] [CrossRef] [Green Version]
- Smrke, A.; Younger, E.; Wilson, R.; Husson, O.; Farag, S.; Merry, E.; Macklin-Doherty, A.; Cojocaru, E.; Arthur, A.; Benson, C.; et al. Telemedicine during the COVID-19 pandemic: Impact on care for rare cancers. JCO Glob. Oncol. 2020, 6, 1046–1051. [Google Scholar] [CrossRef] [PubMed]
- Rodler, S.; Apfelbeck, M.; Schulz, G.B.; Ivanova, T.; Buchner, A.; Staehler, M.; Heinemann, V.; Stief, C.; Casuscelli, J. Telehealth in uro-oncology beyond the pandemic: Toll or lifesaver? Eur. Urol. Focus 2020, 10, 10. [Google Scholar]
- Layfield, E.; Triantafillou, V.; Prasad, A.; Deng, J.; Shanti, R.M.; Newman, J.G.; Rajasekaran, K. Telemedicine for head and neck ambulatory visits during COVID-19: Evaluating usability and patient satisfaction. Head Neck 2020, 42, 1681–1689. [Google Scholar] [CrossRef] [PubMed]
- Triantafillou, V.; Layfield, E.; Prasad, A.; Deng, J.; Shanti, R.M.; Newman, J.G.; Rajasekaran, K. Patient perceptions of head and neck ambulatory telemedicine visits: A qualitative study. Otolaryngol. Head Neck Surg. 2020, 164, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Chua, I.S.; Zachariah, F.; Dale, W.; Feliciano, J.; Hanson, L.; Blackhall, L.; Quest, T.; Curseen, K.; Grey, C.; Rhodes, R.; et al. Early integrated telehealth versus in-person palliative care for patients with advanced lung cancer: A study protocol. J. Palliat. Med. 2019, 22, 7–19. [Google Scholar] [CrossRef] [Green Version]
- National Library of Medicine (US). Telemedicine Nurse-Led Intervention for Rural Cancer Survivors. 2020. Available online: https://clinicaltrials.gov/show/NCT04267627 (accessed on 7 August 2020).
- Australian and New Zealand Clinical Trials Registry. Telehealth for Palliative Care Patients in Metropolitan and Rural Settings; NHMRC Clinical Trials Centre, University of Sydney: Sydney, Australia, 2018; Available online: https://anzctr.org.au/Trial/Registration/TrialReview.aspx?ACTRN=12618001007224 (accessed on 7 August 2020).
- National Library of Medicine (US). Supportive Care Delivered by Telemedicine to Cancer Patients at Home. 2019. Available online: https://clinicaltrials.gov/show/NCT04136340 (accessed on 7 August 2020).
- National Library of Medicine (US). Assessing the System for High-Intensity Evaluation during Radiotherapy during Changes in Response to COVID-19 (CORONA-SHIELD). 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04357574 (accessed on 10 November 2020).
- Luo, C.; Sanger, N.; Singhal, N.; Pattrick, K.; Shams, I.; Shahid, H.; Hoang, P.; Schmidt, J.; Lee, J.; Haber, S.; et al. A comparison of electronically-delivered and face to face cognitive behavioural therapies in depressive disorders: A systematic review and meta-analysis. EClinicalMedicine 2020, 24, 100442. [Google Scholar] [CrossRef]
- Osenbach, J.E.; O’Brien, K.M.; Mishkind, M.; Smolenski, D.J. Synchronous telehealth technologies in psychotherapy for depression: A meta-analysis. Depress Anxiety 2013, 30, 1058–1067. [Google Scholar] [CrossRef]
- Sloan, D.M.; Gallagher, M.W.; Feinstein, B.A.; Lee, D.J.; Pruneau, G.M. Efficacy of telehealth treatments for posttraumatic stress-related symptoms: A meta-analysis. Cogn. Behav. Ther. 2011, 40, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Berryhill, M.B.; Culmer, N.; Williams, N.; Halli-Tierney, A.; Betancourt, A.; Roberts, H.; King, M. Videoconferencing psychotherapy and depression: A systematic review. Telemed J. e-Health 2019, 25, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Varker, T.; Brand, R.M.; Ward, J.; Terhaag, S.; Phelps, A. Efficacy of synchronous telepsychology interventions for people with anxiety, depression, posttraumatic stress disorder, and adjustment disorder: A rapid evidence assessment. Psychol. Serv. 2019, 16, 621–635. [Google Scholar] [CrossRef] [PubMed]
- Drago, A.; Winding, T.N.; Antypa, N. Videoconferencing in psychiatry, a meta-analysis of assessment and treatment. Eur. Psychiatry 2020, 36, 29–37. [Google Scholar] [CrossRef] [PubMed]
| Author and Country | Patient Characteristics | Trial Type and Intervention | Sample Size (Patients per Group) | Outcome | Evidence Certainty |
|---|---|---|---|---|---|
| General counselling studies | |||||
| Lleras de Frutos et al., 2020 [5] | Adult women with cancer (81% breast cancer) and emotional distress after primary oncological treatment | Pragmatic RCT; Online videoconference vs. face-to-face group positive psychotherapy for cancer survivors; 12 weekly group sessions, online group had 11 sessions plus 1 in-person session | 269 of which 225 (108 vs. 117) randomized and 44 (16 vs. 28) selected their group | Measurement tools: HADS, PCL-C, PTGI; Emotional distress (anxiety and depression), post-traumatic stress symptoms improved in both groups, no significant difference between groups; Post-traumatic stress still above cut-off after treatment; No significant difference in attrition, integrity, or effectiveness after adjusting for baseline differences; Online counselling is not superior, both may be effective. | Very low |
| Watson et al., 2017 [6] UK | Cancer patients (except non-melanoma skin cancer) with high psychological needs for mental health and coping referred for psychological care | Equivalence RCT; Telephone-delivered cognitive behavioural therapy to face-to-face (treatment as usual) therapy; Median 4 sessions | 118 (60 vs. 58) randomized; 78 analyzed (43 vs. 35; including 5 vs. 6 who switched arms) | Measurement tools: HADS, MAC H/H, CLCC, CCQ, and additional post-therapy study-specific questionnaire; Both arms had significant improvement in anxiety, depression, HADS total score, cancer (p < 0.01); In-person but not telephone group had significant improvement in helpless/hopeless scale (p = 0.13 and p = 0.015); Stress and worry improved and were equivalent between groups. | Very low |
| Harrigan et al., 2016 [7] USA | Breast cancer survivors with BMI ≥ 25.0 kg/m2 diagnosed in the 5 years before enrollment with stage 0 to 3 breast cancer, who had completed chemotherapy and/or radiation therapy ≥3 months before enrollment | 3 Arm RCT Telephone vs. in-person weight-loss counselling (11 sessions over 6 months) vs. usual care (brochures and referred to survivorship clinic, which offers a 2-session weight management program and an in-person counselling session) | 100 (34 vs. 33 vs. 33) 6-month data: 24 vs. 30 vs. 31 12-month data: 15 vs. 22 vs. 19 | Weight change at 6 months: 4.8 kg (−5.4%), 5.6 kg (−6.4%), 1.7 kg (−2.0%); p = 0.46 telephone to in-person, p = 0.009 telephone to usual care; p = 0.001 in-person to usual care; Self-reported weight loss at 12 months was not significantly different between groups; Reduction in % body fat was significant for in-person (p = 0.05) but not telephone (p = 0.37) compared to usual care; Difference between telephone and in-person care was not significant (p = 0.35); Increased activity: 96 ± 154 min vs. 114 ± 130 min vs. 17 ± 110 min (p < 0.05); Change in number of steps per day: 948 vs. 1847 vs. −330 (p < 0.05). | Very low |
| Van der Lee et al., 2020 [8] Netherlands | Cancer out-patients or their family members | Single-arm study; Psycho-oncology counselling via video-consults instead of in-person; Survey of 34 psychologists and 2 psychiatrists giving video-consults | 239 surveys (209 patients, 30 therapists) | Patients reported being grateful for continued care, video-consults as more distant, harder for some to express feelings, missed travel time as time to prepare and process afterwards, missed having a place outside the home to leave their distress; Others found video from home as quieter and relaxed, with less stress due to travel and face-to-face contact; Therapists missed non-verbal communication and informal physical contact that normally helps release tension; Therapists had to work harder and felt more exhausted; All willing to continue video-consults if requested by patients; Preference for in-person sessions for more complex therapies. | Very low |
| Genetic counselling studies | |||||
| Buchanan et al., 2015 [9]; Datta et al., 2011 [10] USA | People referred to CGC and who preferred CGC locally instead of at the academic medical centre | RCT; Telegenetics vs. in-person; Same genetics counsellor for both groups; Not designed to test inferiority or equivalence of telegenetics vs. in-person counselling | 162 (81 vs. 81) randomized; 59 vs. 71 analyzed | Measurement tools: VSQ and GCSS 9GCSS0; Cost to health care system: $106 vs. $244/patient; Patient satisfaction was high and did not differ between groups; CGC attendance: 79% vs. 89%, p = 0.03; Lower computer comfort associated with lower attendance (p = 0.02); Of telegenetics group, 98% were comfortable with the system, but 32% would have preferred an in-person visit | Very low |
| Steffen et al., 2017 [11]; Kinney et al., 2016 [12]; Chang et al., 2016 [13] USA | Breast (91.5%) and ovarian cancer (8.1%) survivors at increased hereditary risk for BRCA1/2 mutations; history suggestive of HBOC meeting NCCN criteria for genetic counselling | RCT equivalency/ noninferiority trial; Telephone (TC) vs. in-person counselling (IPC) | 502 vs. 510 randomized; 464 vs. 437 eligible and received counselling; 402 vs. 379 analyzed [11]; 493 vs. 495 [12] | Groups did not differ at low levels of distress (27% vs. 30%) and risk (23.8% vs. 29.8%); At high distress uptake was 26.3% TC vs. 44.3% IPC (OR = 0.45, 95% CI = 0.27 to 0.76) and at high perceived risk uptake was 33.9% vs. 50.5% (OR = 0.50, 95% CI = 0.29 to 0.87); At 1-yr follow-up, TC was non-inferior to IPC for all psychosocial and informed decision-making outcomes (anxiety, cancer-specific distress, perceived personal control, decisional conflict); Telephone counselling cost: $120 (range $80−$200) vs. $270 (range $180−$400) per person. | Low |
| Jacobs et al., 2016 [14]; Peshkin et al., 2016 [15]; Schwartz et al., 2014 [16]; Butrick et al., 2015 [17] USA | Women with BRCA 1/2-associated hereditary breast/ovarian cancer | Non-inferiority RCT; Telephone genetic counselling (TC) vs. usual care (UC; in-person) by trained genetic counselor; Follow-up telephone interview approximately 2 weeks later to assess perception and satisfaction with pre-test counselling | 669 (335 vs. 334) randomized; 554 (272 vs. 282) completed baseline and 2-week follow-up interview; 479 (236 vs. 243) analyzed | Measurement tools: BCGCKS, DCS, IES, MCS, PCS; TC noninferior to UC on all outcomes (knowledge, decision conflict, cancer distress, perceived stress, genetic counseling satisfaction); No difference and TC non-inferior in pre-test and post-test survey for satisfaction (83.1% vs. 86.8% very satisfied, p = 0.22), knowledge, and perceived stress; UC group had more decisional conflict but within non-inferiority bounds; TC was more convenient (OR = 4.78, 95% CI = 3.32 to 6.89) but with lower perceived support (52.9% vs. 66%, p = 0.002; OR = 0.56, 95% CI = 0.40 to 0.80) and emotional recognition (55.5% vs. 68.8%, p = 0.001; OR = 0.53, 95% CI = 0.37 to 0.76); 80.9% of TC preferred TC or had no preference; 84.2% of UC preferred UC or had no preference (p = 0.3); TC group had less uptake of subsequent BRCA 1/2 testing (84.2% vs. 90.1%; logistic regression model OR = 1.65, 95% CI 1.00 to 2.72); Genetic counsellor scores did not differ overall (p = 0.910); Scores did not differ by group, but were lower for minorities. | Moderate |
| Mette et al., 2016 [18] USA | Underserved primarily Hispanic population (95%); high risk based on their personal and/or family medical histories and meeting NCCN guidelines for genetic counselling | Non-randomized study; Telemedicine or video-teleconferencing vs. in-person; In-person appointments about once a month at each center (otherwise it was by video) and patients had no input on type of visit | 353 surveys, 119 responses (56 vs. 63) | There were no differences between the two groups for satisfaction, comfort talking, feeling listened to, enough time, understanding information, finding information valuable, information helpful to make health decisions, or likelihood of recommending the program. | Very low |
| Solomons et al., 2018 [19] USA | New rural patients with personal or family history suggestive of HBOC susceptibility | Non-randomized study; Live-interactive videoconferencing from remote clinic vs. in-person; Groups matched by gender, race, health insurance status; Same counselor or oncologist for remote or in-person counselling; Questionnaire at pre-counselling, immediately after, 1 month after by mail, plus 4 weeks after test results for those undergoing genetic testing | 174 (106 vs. 68) 158 (90 vs. 68; 85% vs. 100%) returned pre- and post-counselling surveys; 65 (41 vs. 24; 46% vs. 35%) returned 1-month surveys | Measurement tools: 9 HBOC-related knowledge questions, PHQ-4, pre-validated survey; HBOC knowledge improved equally (evaluated only in patients with personal or family history of breast/ovarian cancer); Remote group had higher anxiety and depression pre-counselling; Decreased anxiety in both groups; Depression improved more in telegenetics group initially but was lower at 1 month in both groups; Telegenetics (remote) reduced transportation need and work absence; All patients satisfied with quality of care, 32% of remote patients noted preference for in-person care. | Very low |
| Tutty et al., 2019 [20] Australia | Women with high-grade serous ovarian cancer | Single-arm study; Telephone genetic counselling prior to testing and after testing if patient decides to be tested; Those with BRCA1/2 variant affecting function (n = 26, 9.2%) or significant family history requiring evaluation (e.g., Lynch syndrome) offered further in-person counselling | 284 counselled; 277 surveys; 107 responses (39%) | 40% had poor knowledge (<5/7 correct answers) for knowledge of hereditary breast and ovarian cancer syndromes; 97% satisfied with timing of telephone call and 94% satisfied with information provided; 17% would have preferred face-to-face counselling, 34% had no preference; Median per patient cost was AUD $91.52 telephone vs. $107.37 in-person. | Very low |
| Author and Country | Patient Characteristics | Trial Type and Intervention | Sample Size (Patients per Group) | Outcome | Evidence Certainty |
|---|---|---|---|---|---|
| Kelleher et al., 2019 [21]; Winger et al., 2020 [22] USA | Patients with breast, lung, prostate, or colorectal cancer | Non-inferiority RCT; Videoconference vs. in-person psychosocial pain management; 4 sessions; Videoconference group given tablet (iPad) with data plan (also given to patients in the in-person group if needed to access website); Both groups had access to study website for self-assessment and to indicate preferences for content of next session | 178 (89 vs. 89) randomized; 137 (75 vs. 62) post-treatment; 128 (70 vs. 58) at 3-month follow-up | Measurement tools: CSQ and individual questions items; Similar patient burden and acceptability in both groups, but better feasibility in videoconference group; Pain severity, pain interference, physical well-being, physical symptoms, psychological distress, and self-efficacy for pain management all improved post-treatment compared to baseline in both groups, with continued improvement at 3-months only in the videoconference group; Videoconferencing was non-inferior at post-treatment, and at 3 months post-treatment; Completion of all sessions predicted improvement in pain severity, pain interference, and pain self-efficacy; Videoconference group was more likely to complete all sessions (83% vs. 65%, p = 0.006); Patients near medical center, with early cancer or less comorbidity, were more likely to complete in-person sessions. | Low |
| Walle et al., 2018, 2020 [23,24] Germany | Solid tumours and systemic therapy needing follow-up visit at outpatient clinic in 2–14 days, follow-up for 6 months | RCT; Mobile telephone vs. in-person visit | 66 (33 vs. 33) randomized; 48 (22 vs. 26) evaluable questionnaires | Measurement tools: Questionnaire developed by research team and STAI-S for psychological morbidity; Patient satisfaction was greater with video call due to confidence in their physician (p = 0.006), efficiency (p = 0.003), and punctuality (p = 0.003), saving time (p < 0.0001) and cost (p < 0.0001); Physical exam in 2 vs. 8 visits (9% vs. 31%), prescriptions in 9% vs. 50% of patients, referrals to other professionals in 5% vs. 12% | Very low |
| Beaver et al., 2017 [25]; Dixon et al., 2018 [26]; Beaver et al., 2020 [27] England | Patients with hysterectomy for stage I endometrial cancer; Only 4% had RT | Non-inferiority RCT; Gynecology oncology nurse-led specialist TFU vs. traditional HFU, appointments every 3 or 4 months for 2 years post-treatment followed 6–monthly and annually up to 3–5 years; TFU was on same schedule as HFU; Questionnaires at baseline and immediately after appointments by mail | 259 (129 vs. 130) randomized; 217 (111 vs. 106) analyzed; 211 (105 vs. 106) responded | Psychological morbidity was non-inferior (33.0 ± 11.0 vs. 35.5 ± 13.0); Patient satisfaction with information: OR = 0.9, 95% CI 0.4 to 2.1, p = 0.83; Patient satisfaction with service: 9.2 ± 1.5 vs. 8.9±1.7, 95% CI −0.5 to 0.3, p = 0.58; Recurrence rate of 4%, 5 from each group, all symptomatic and presented as interval events reported to general practitioner or nurse specialist between scheduled appointments; Time from randomisation to diagnosis of recurrence: TFU median 307 days, range 48–662 days; HFU 172 days, range 99–436 days; Cost analysis: no difference at 6 or 12 months; TFU more likely to have appointments on time (p < 0.001) and thorough (p = 0.011) No statistically significant differences for being able to ask questions, having questions answered, feeling anxious prior to appointments, or feeling reassured; HFU more likely to be kept waiting (p = 0.001), and indicated nurse was less likely to be familiar with their case (p = 0.005); No significant difference in QoL. | Moderate |
| Viers et al., 2015 [28] USA | Patients with radical prostatectomy ≥90 days and undergoing surveillance, no active urologic concerns requiring physical examination as determined by pre-visit phone call | Equivalence RCT; Video visit (at home or work) vs. office visits by urologist for one visit; Urologists completed a 12-point questionnaire at the conclusion of each visit | 70 (34 vs. 36) randomized; 55 (28 vs. 27) completed the study | Measurement tools: Questionnaire with 7-point Likert scale (1 = strongly agree, 7 = strongly disagree); 100% of video patients and 96% of office visit patients agreed they would meet with their provider in the same setting again; When considering cost, 83% and 59% would choose remote encounter for subsequent visit; No difference in patient trust of the provider (1.0 vs. 1.0), perception of visit confidentiality (1.1 vs. 1.0), or ability to share sensitive/personal information (1.3 vs. 1.0); Similar perceived efficiency (2.1 vs. 1.4), quality of education provided (1.3 vs. 1.4), and overall satisfaction with the encounter (1.2 vs. 1.1); High level of urologist satisfaction in both groups, no difference in quality of medical history, therapeutic management, or perceived patient satisfaction; Different distribution of missed visits: video had 3 technical, 1 canceled, 2 medical, and office visits had 9 late or no show. | Very low |
| Chan et al., 2015 [29] Australia | Chemotherapy patients excluding those with RT or on clinical trials | Non-randomized study; Teleoncology (by general physicians, chemotherapy-proficient nurses, allied health professionals, pharmacist) vs. in-person; Both groups supervised by same medical oncologists | 89 vs. 117 | Serious adverse effects: palliative 5.4% vs. 15%, curative /adjuvant 2.9% vs. 3.6%; Grade 3/4 toxicity in palliative patients: neutropenia 21% vs. 23%, diarrhea 0% vs. 12%, neuropathy 8.8% vs. 0%, fatigue 0% vs. 1.8%, other 8.8% vs. 21%; Grade 3/4 toxicity in curative patients: neutropenia 34% vs. 13%, nausea and vomiting 0% vs. 3.3%, diarrhea 1.8% vs. 1.7%, neuropathy 0% vs. 3.3%, fatigue 0% vs. 6.7%, other 16% vs. 30%; Hospital admissions: palliative 36% vs. 43%, curative 15% vs. 27%; Dose intensity: palliative 97.4% vs. 98.2%, curative 84.4% vs. 88.1%. | Very low |
| Hamilton et al., 2019 [30] Australia | Patients deemed suitable by radiation oncologist or referring specialist; Both new and follow-up appointments eligible | Single-arm study; Tele-radiation oncology program | 311 charts were audited; Survey sent to subset of 231 patients; 106 responses | Survey response rate of 106/231 (46%); 55% preferred telehealth for future appointments, 1% face-to-face, 35% mixed (telehealth and face-to-face), 9% unknown; 80% or more strongly agreed (≥90% agreed or strongly agreed) they could hear the doctor clearly, felt privacy and confidentiality were respected, could ask questions easily, felt it was easy to establish rapport, and thought diagnosis and treatment options were adequately explained; 68% strongly agreed and 15% agreed that they felt reassured when there was a nurse or local doctor present. | Very low |
| Jue et al., 2017 [31] USA | Patients referred to surgical oncologist | Single-arm study; Visit via video from local centre with centralized surgical oncologist who directed oncology treatment, physical exam by nurse practitioner under video supervision of surgical oncologist; Only surgery itself (if needed) was done in-person by surgical oncologist; Single surgeon had medical oncology background | 296 (755 visits) | Reduction in patient travel distance by 80.7% (213,008 miles), saved system $155,627 as patients are normally reimbursed for travel expenses; 86% of patients believed care was more accessible; Average satisfaction scores 4.4/5 to 4.7/5 for most categories; 4.2/5 average for video being more cost and time efficient; 3.8/5 average preference for next visit to be in-person; 3.6/5 average score for believing they would have received better in-person. | Very low |
| Li et al., 2016 [32] China | Lung cancer patients with chronic post-surgical pain after surgery without postoperative complications | Non-randomized comparative study; Remote pain intervention (smartphone or internet) vs. conventional care (weekly in outpatient clinic) | 81 (41 vs. 40) | Measurement tools: SF-36 for QoL at 1 and 3 months after therapy; Similar QoL between groups, p > 0.05; Remote group had higher satisfaction (90.2% vs. 72.5%), p < 0.05. | Very low |
| >Verma et al., 2015 [33] UK | Men who had RT for localized low to medium risk prostate cancer | Single-arm study; Remote telephone follow-up by radiographer | 134 had first review at 6 months; 69 at 12 months; 9 at 24 months | 88/134 patients (66%) returned questionnaires at 6 months; 77% reported telephone follow-up as more convenient, 3% as not more convenient, 8% no preference; 9% would have preferred an in-person visit with a radiologist and 15% would have preferred an in-person visit with a doctor; For future follow-up, 76% preferred phone, 6% preferred in-person, 7% no preference. | Very low |
| Patil et al., 2018 [34] India | Adult patients with intermediate- to high-grade (grade II-IV) glioma on adjuvant TMZ for 2+ cycles | Single-arm study; Video (VF) vs. clinic follow-up (CF) in same patients; VF on day 24 from previous CF to determine if patients needed continuation of TMZ, supportive medications, imaging, molecular testing, and rehabilitation; In-person exam about 4 days later to determine agreement of decision; Two groups of 3 clinicians did assessment; Patients randomized as to which group did assessment | 65 | Concurrence in decision to administer TMZ was 100%; Patient satisfaction rate (somewhat or extremely) was 100% post-VF and 98.5% post-CF; Median cost $58.15 US vs. $131.23 US. | Very low |
| Smrke et al., 2020 [35] | Patients with sarcoma | Non-randomized comparative study; Telemedicine vs. face-to-face; Anonymous patient experience survey and clinician survey | 108 patient surveys returned (70 vs. 34); 18 clinician surveys | Patient satisfaction: 8.99/10 vs. 8.35/10; 80% indicated they would like at least some future appointments to be by telemedicine for reasons of travel time (42%), travel expense (20%), and convenience (30%); 48% would not want bad news over the phone; Of those who preferred face-to-face, 42% felt it was more reassuring and 20% felt the treatment plan was clearer; 78% clinicians found appointments shorter by phone; 89% felt it did not increase workload, but affected ability to perform exams sometimes (44%) or often (17%); Most clinicians favoured telemedicine for active surveillance or stable-dose oral medication. | Very low |
| Rodler et al., 2020 [36] Germany | Patients with advanced genitourinary cancer and systemic therapy switched to virtual treatment (phone or video) where possible due to COVID-19 pandemic; Visits limited to therapy application | Single-arm study; Survey of all patients during one week by email, phone, or in-person | 101 patients; 92 responded | Majority of patients valued continuation of therapy higher than COVID-19 prevention measures; 77.2% were unwilling to postpone a staging exam; 44.6% were afraid of progression (61.8% chemotherapy; 33.3% immunotherapy) and did not want to delay or interrupt treatment; Acceptance of virtual discussion of staging results and therapy decisions: median 8/10 (IQR 5–9); Referral to secondary care oncologists for therapy: median 2.5/10 (IQR 0–6.75); Preference for telehealth beyond the pandemic: median 4/10 overall (IQR 2–7; 3/10 for immunotherapy and 5/10 for chemotherapy); 62.6% preferred in-person care after the pandemic; High acceptance of external laboratory controls (60.9%) but lower acceptance for online visit management (48.9%), remote treatment planning (44.6%), and referral to secondary care oncologists (17.4%). | Very low |
| Layfield et al., 2020 [37]; Triantafillou et al., 2020 [38] USA | New (6%) or returning (94%) otolaryngology patients who previously had telemedicine visits; 73% malignant, 9% premalignant | Single-arm study; Video-based telemedicine visits | 100 qualitative comments from subset of 56 patients | Mean scores ± SD: Usefulness 6.10 ± 0.50; Ease of use 6.21 ± 0.13; Effectiveness 6.20 ± 0.60; Reliability 4.86 ± 0.84; Satisfaction 6.29 ± 0.32; Concerns were related to limitation on physical exams and lack of touch; 29% required technical assistance from family or caregiver; 32% expressed relief they could receive care during the pandemic; 25% indicated telemedicine was more convenient. | Very low |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, S.; Fletcher, G.G.; Yao, X.; Sussman, J. Virtual Care in Patients with Cancer: A Systematic Review. Curr. Oncol. 2021, 28, 3488-3506. https://doi.org/10.3390/curroncol28050301
Singh S, Fletcher GG, Yao X, Sussman J. Virtual Care in Patients with Cancer: A Systematic Review. Current Oncology. 2021; 28(5):3488-3506. https://doi.org/10.3390/curroncol28050301
Chicago/Turabian StyleSingh, Simron, Glenn G. Fletcher, Xiaomei Yao, and Jonathan Sussman. 2021. "Virtual Care in Patients with Cancer: A Systematic Review" Current Oncology 28, no. 5: 3488-3506. https://doi.org/10.3390/curroncol28050301
APA StyleSingh, S., Fletcher, G. G., Yao, X., & Sussman, J. (2021). Virtual Care in Patients with Cancer: A Systematic Review. Current Oncology, 28(5), 3488-3506. https://doi.org/10.3390/curroncol28050301





