Current Overview of Treatment for Metastatic Bone Disease
Abstract
1. Introduction
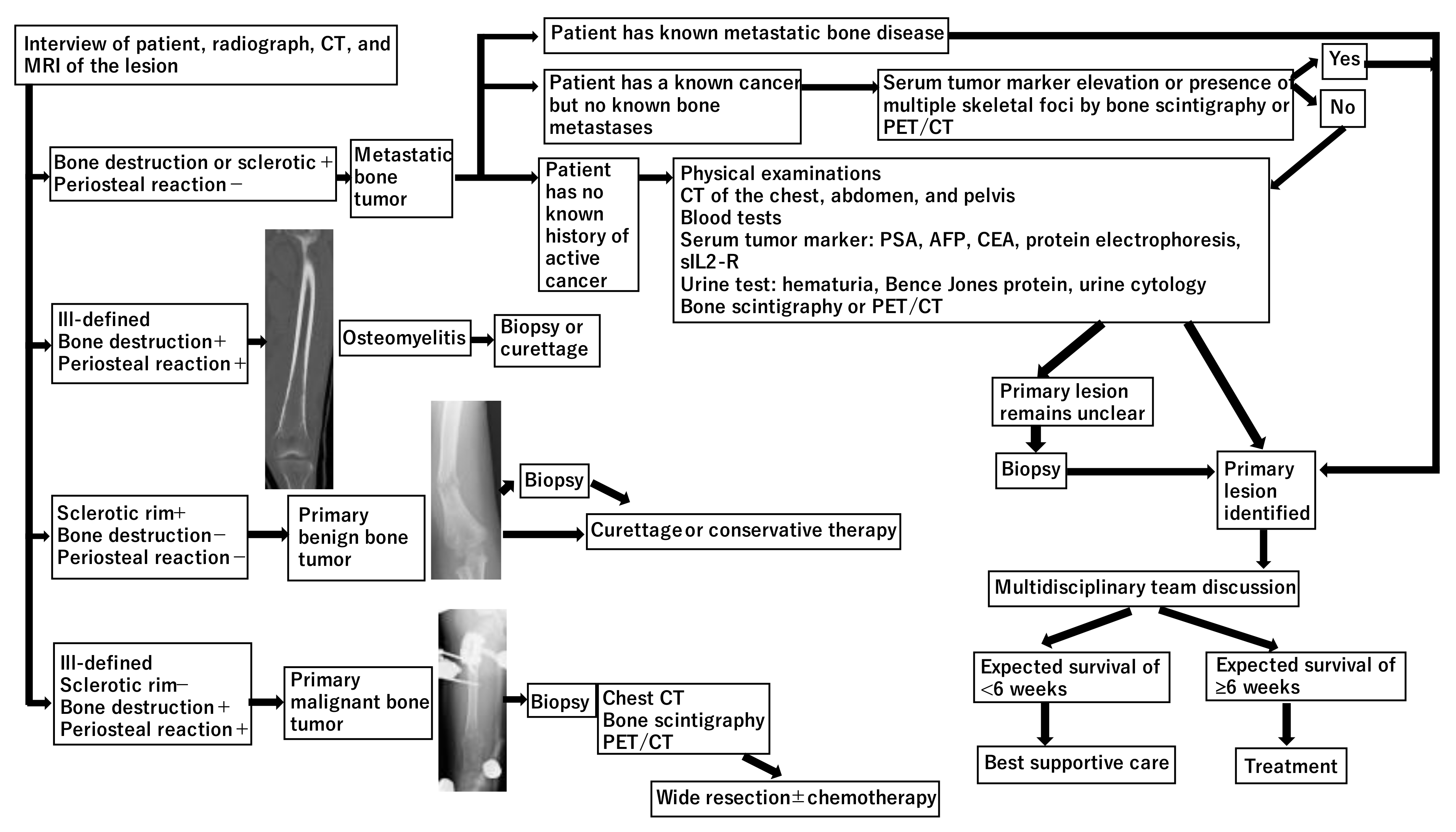
2. Diagnosis
3. Prognosis
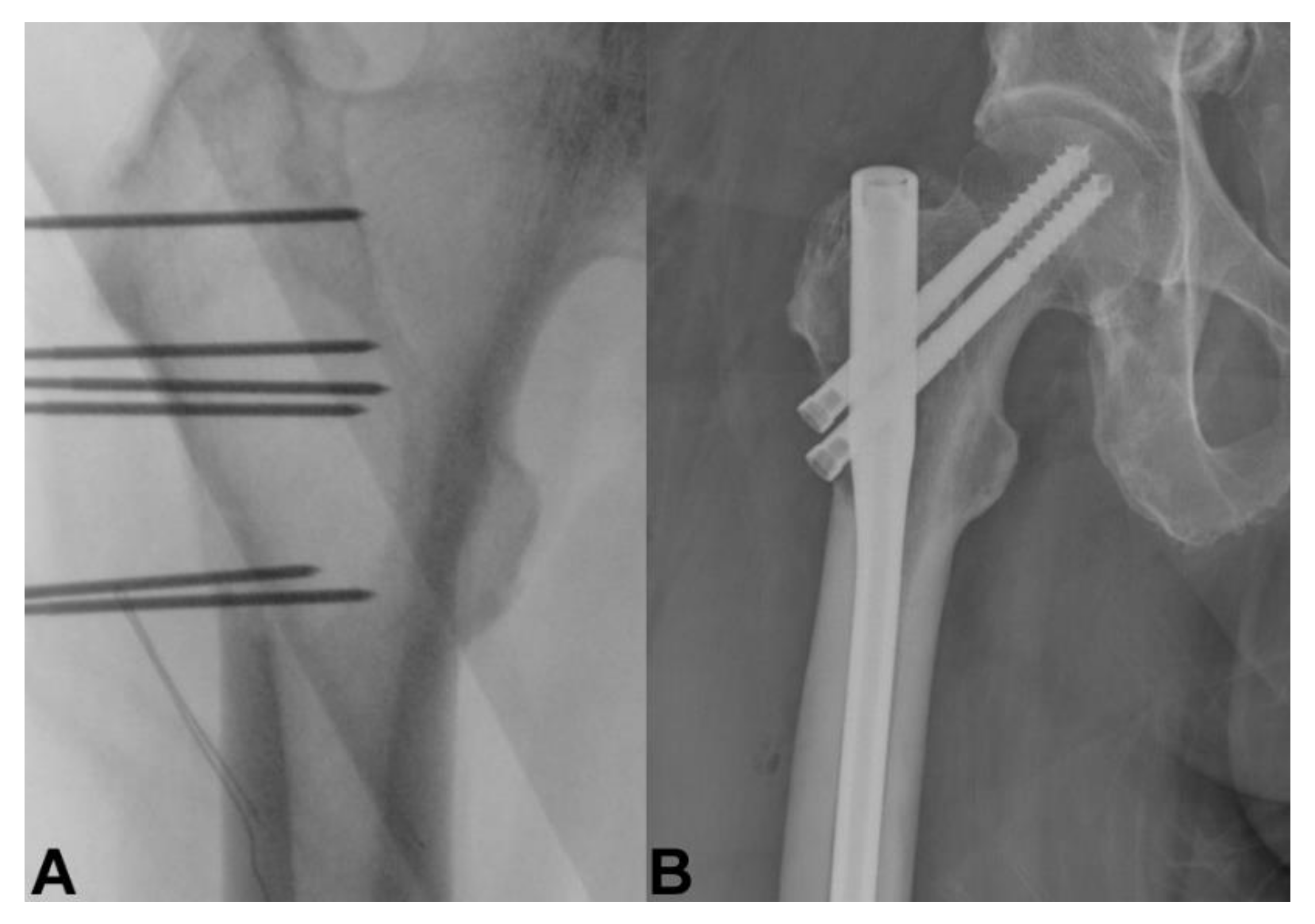
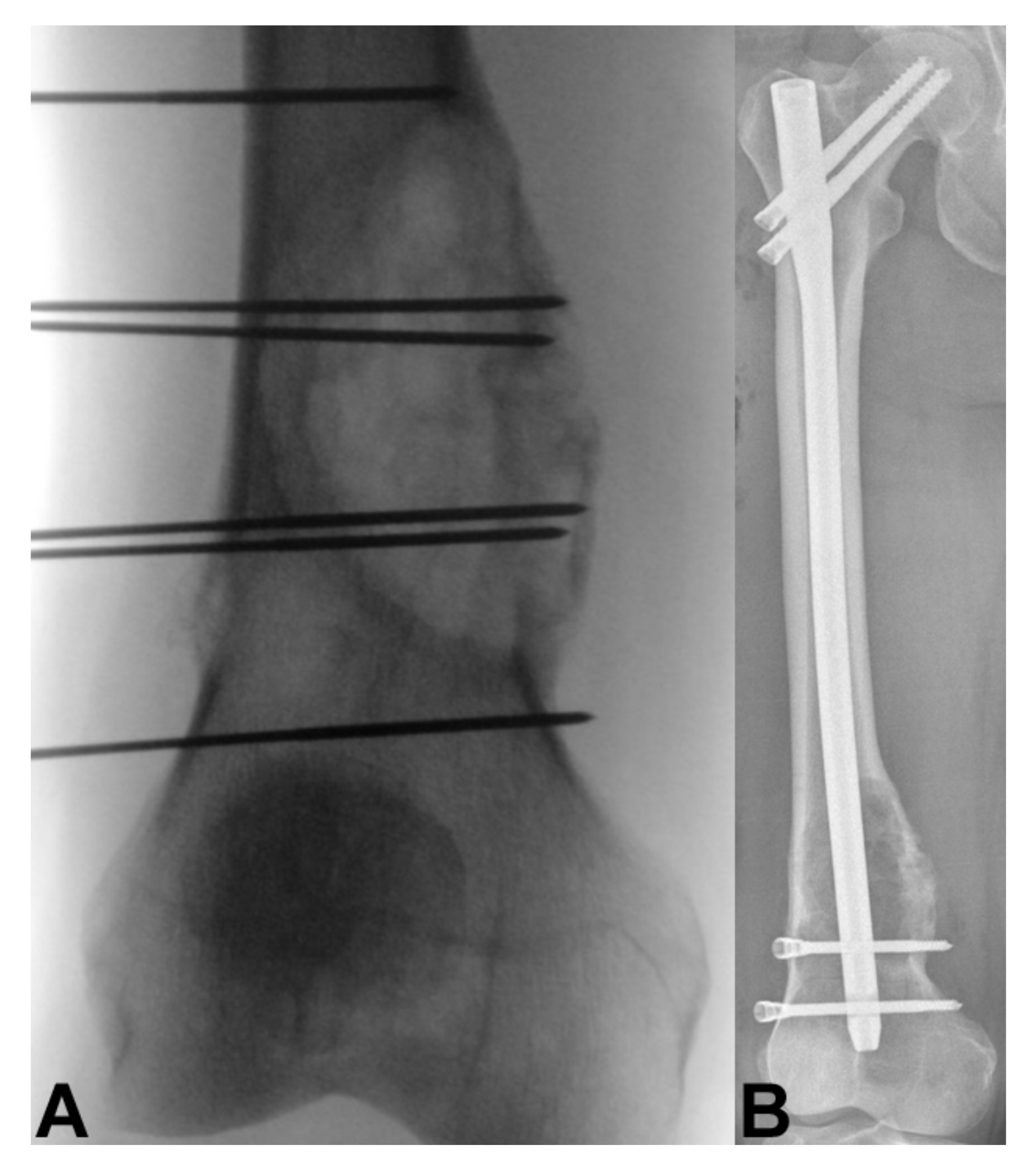
4. Bone Metastasis of the Extremities
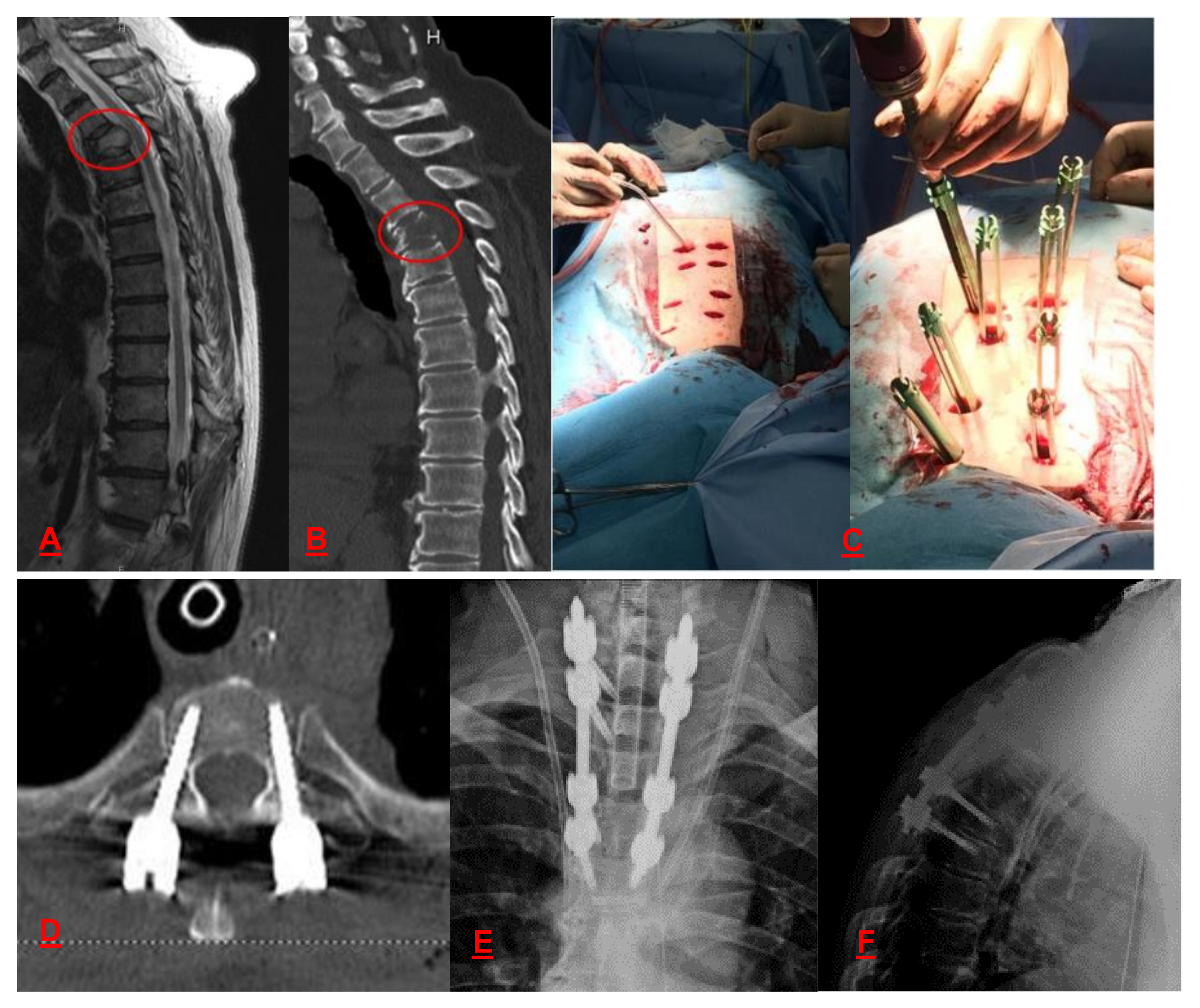
5. Bone Metastasis of the Spine
6. Bone Metastasis of the PELVIS
7. Radiotherapy
8. Bisphosphonates and Denosumab
9. Embolization
10. Radiofrequency Thermal ABLATION (RFA)
11. Electrochemotherapy
12. High-Intensity Focused Ultrasound (HIFU)
13. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sousa, S.; Clézardin, P. Bone-targeted therapies in cancer-induced bone disease. Calcif. Tissue Int. 2018, 102, 227–250. [Google Scholar] [CrossRef]
- Li, S.; Peng, Y.; Weinhandl, E.D.; Blaes, A.H.; Cetin, K.; Chia, V.M.; Stryker, S.; Pinzone, J.J.; Acquavella, J.F.; Arneson, T.J. Estimated number of prevalent cases of metastatic bone disease in the US adult population. Clin. Epidemiol. 2012, 4, 87–93. [Google Scholar] [CrossRef]
- Bollen, L.; Dijkstra, S.P.D.; Bartels, R.H.M.A.; de Graeff, A.; Poelma, D.L.H.; Brouwer, T.; Algra, P.R.; Kuijlen, J.M.A.; Minnema, M.C.; Nijboer, C.; et al. Clinical management of spinal metastases—The Dutch national guideline. Eur. J. Cancer 2018, 104, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Anract, P.; Biau, D.; Boudou-Rouquette, P. Metastatic fractures of long limb bones. Orthop. Traumatol. Surg. Res. 2017, 103, S41–S51. [Google Scholar] [CrossRef]
- Soldatos, T.; Chalian, M.; Attar, S.; McCarthy, E.F.; Carrino, J.A.; Fayad, L.M. Imaging differentiation of pathologic fractures caused by primary and secondary bone tumors. Eur. J. Radiol. 2013, 82, e36–e42. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, Y.; Gu, Y.; Hou, Y.; Chen, Z. The need for bone biopsies in the diagnosis of new bone lesions in patients with a known primary malignancy: A comparative review of 117 biopsy cases. J. Bone Oncol. 2019, 14, 100213. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, A.J.; Le, H.V.; Marjoua, Y.; Leonard, D.A.; Belmont, P.J.; Bono, C.M.; Harris, M.B. Assessing the utility of a clinical prediction score regarding 30-day morbidity and mortality following metastatic spinal surgery: The New England Spinal Metastasis Score (NESMS). Spine J. 2016, 16, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Willeumier, J.J.; van der Linden, Y.M.; van der Wal, C.W.P.G.; Jutte, P.C.; van der Velden, J.M.; Smolle, M.A.; van der Zwaal, P.; Koper, P.; Bakri, L.; de Pree, I.; et al. An easy-to-use prognostic model for survival estimation for patients with symptomatic long bone metastases. J. Bone Jt. Surg. Am. 2018, 100, 196–204. [Google Scholar] [CrossRef]
- Piccioli, A.; Spinelli, M.S.; Forsberg, J.A.; Wedin, R.; Healey, J.H.; Ippolito, V.; Daolio, P.A.; Ruggieri, P.; Maccauro, G.; Gasbarrini, A.; et al. How do we estimate survival? External validation of a tool for survival estimation in patients with metastatic bone disease-decision analysis and comparison of three international patient populations. BMC Cancer 2015, 15, 424. [Google Scholar] [CrossRef]
- Forsberg, J.A.; Wedin, R.; Bauer, H.C.F.; Hansen, B.H.; Laitinen, M.; Trovik, C.S.; Keller, J.Ø.; Boland, P.J.; Healey, J.H. External validation of the Bayesian Estimated Tools for Survival (BETS) models in patients with surgically treated skeletal metastases. BMC Cancer 2012, 12, 493. [Google Scholar] [CrossRef]
- Ogura, K.; Gokita, T.; Shinoda, Y.; Kawano, H.; Takagi, T.; Ae, K.; Kawai, A.; Wedin, R.; Forsberg, J.A. Can a multivariate model for survival estimation in skeletal metastases (PATHFx) be externally validated using Japanese patients? Clin. Orthop. Relat. Res. 2017, 475, 2263–2270. [Google Scholar] [CrossRef]
- Forsberg, J.A.; Wedin, R.; Boland, P.J.; Healey, J.H. Can we estimate short- and intermediate-term survival in patients undergoing surgery for metastatic bone disease? Clin. Orthop. Relat. Res. 2017, 475, 1252–1261. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.B.; Wedin, R.; Fabbri, N.; Boland, P.; Healey, J.; Forsberg, J.A. External validation of PATHFx Version 3.0 in patients treated surgically and nonsurgically for symptomatic skeletal metastases. Clin. Orthop. Relat. Res. 2020, 478, 808–818. [Google Scholar] [CrossRef]
- Meares, C.; Badran, A.; Dewar, D. Prediction of survival after surgical management of femoral metastatic bone disease—A comparison of prognostic models. J. Bone Oncol. 2019, 15, 100225. [Google Scholar] [CrossRef] [PubMed]
- Katagiri, H.; Okada, R.; Takagi, T.; Takahashi, M.; Murata, H.; Harada, H.; Nishimura, T.; Asakura, H.; Ogawa, H. New prognostic factors and scoring system for patients with skeletal metastasis. Cancer Med. 2014, 3, 1359–1367. [Google Scholar] [CrossRef]
- Ratasvuori, M.; Wedin, R.; Keller, J.; Nottrott, M.; Zaikova, O.; Bergh, P.; Kalen, A.; Nilsson, J.; Jonsson, H.; Laitinen, M. Insight opinion to surgically treated metastatic bone disease: Scandinavian Sarcoma Group Skeletal Metastasis Registry report of 1195 operated skeletal metastasis. Surg. Oncol. 2013, 22, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Janssen, S.J.; van der Heijden, A.S.; van Dijke, M.; Ready, J.E.; Raskin, K.A.; Ferrone, M.L.; Hornicek, F.J.; Schwab, J.H. 2015 Marshall Urist Young Investigator Award: Prognostication in patients with long bone metastases: Does a boosting algorithm improve survival estimates? Clin. Orthop. Relat. Res. 2015, 473, 3112–3121. [Google Scholar] [CrossRef]
- Sørensen, M.S.; Gerds, T.A.; Hindsø, K.; Petersen, M.M. External validation and optimization of the SPRING Model for prediction of survival after surgical treatment of bone metastases of the extremities. Clin. Orthop. Relat. Res. 2018, 476, 1591–1599. [Google Scholar] [CrossRef]
- Thio, Q.C.B.S.; Karhade, A.V.; Bindels, B.J.J.; Ogink, P.T.; Bramer, J.A.M.; Ferrone, M.L.; Calderón, S.L.; Raskin, K.A.; Schwab, J.H. Development and internal validation of machine learning algorithms for preoperative survival prediction of extremity metastatic disease. Clin. Orthop. Relat. Res. 2020, 478, 322–333. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, J.A.; Eberhardt, J.; Boland, P.J.; Wedin, R.; Healey, J.H. Estimating survival in patients with operable skeletal metastases: An application of a bayesian belief network. PLoS ONE 2011, 6, e19956. [Google Scholar] [CrossRef]
- Overmann, A.L.; Clark, D.M.; Tsagkozis, P.; Wedin, R.; Forsberg, J.A. Validation of PATHFx 2.0: An Open-Source Tool for Estimating Survival in Patients Undergoing Pathologic Fracture Fixation. J. Orthop. Res. 2020, 38, 2149–2156. [Google Scholar] [CrossRef]
- Karhade, A.V.; Thio, Q.C.B.S.; Ogink, P.T.; Shah, A.A.; Bono, C.M.; Oh, K.S.; Saylor, P.J.; Schoenfeld, A.J.; Shin, J.H.; Harris, M.B.; et al. Development of Machine Learning Algorithms for Prediction of 30-Day Mortality After Surgery for Spinal Metastasis. Neurosurgery 2019, 85, E83–E91. [Google Scholar] [CrossRef] [PubMed]
- Karhade, A.V.; Thio, Q.C.B.S.; Ogink, P.T.; Bono, C.M.; Ferrone, M.L.; Oh, K.S.; Saylor, P.J.; Schoenfeld, A.J.; Shin, J.H.; Harris, M.B.; et al. Predicting 90-Day and 1-Year Mortality in Spinal Metastatic Disease: Development and Internal Validation. Neurosurgery 2019, 85, E671–E681. [Google Scholar] [CrossRef]
- Errani, C.; Cosentino, M.; Ciani, G.; Ferra, L.; Alfaro, P.A.; Bordini, B.; Donati, D.M. C-reactive protein and tumour diagnosis predict survival in patients treated surgically for long bone metastases. Int. Orthop. 2021, 45, 1337–1346. [Google Scholar] [CrossRef] [PubMed]
- Karhade, A.V.; Thio, Q.C.B.S.; Kuverji, M.; Ogink, P.T.; Ferrone, M.L.; Schwab, J.H. Prognostic value of serum alkaline phosphatase in spinal metastatic disease. Br. J. Cancer 2019, 120, 640–646. [Google Scholar] [CrossRef]
- Kim, J.H.; Seo, S.W.; Chung, C.H. What factors are associated with early mortality in patients undergoing femur surgery for metastatic lung cancer? Clin. Orthop. Relat. Res. 2018, 476, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Sarahrudi, K.; Hora, K.; Heinz, T.; Millington, S.; Vécsei, V. Treatment results of pathological fractures of the long bones: A retrospective analysis of 88 patients. Int. Orthop. 2006, 30, 519–524. [Google Scholar] [CrossRef]
- Errani, C.; Mavrogenis, A.F.; Cevolani, L.; Spinelli, S.; Piccioli, A.; Maccauro, G.; Baldini, N.; Donati, D. Treatment for long bone metastases based on a systematic literature review. Eur. J. Orthop. Surg. Traumatol. 2017, 27, 205–211. [Google Scholar] [CrossRef]
- Ruatta, F.; Derosa, L.; Escudier, B.; Colomba, E.; Guida, A.; Baciarello, G.; Loriot, Y.; Fizazi, K.; Albiges, L. Prognosis of renal cell carcinoma with bone metastases: Experience from a large cancer centre. Eur. J. Cancer 2019, 107, 79–85. [Google Scholar] [CrossRef]
- Ruggieri, P.; Mavrogenis, A.F.; Casadei, R.; Errani, C.; Angelini, A.; Calabrò, T.; Pala, E.; Mercuri, M. Protocol of surgical treatment of long bone pathological fractures. Injury 2010, 41, 1161–1167. [Google Scholar] [CrossRef]
- Wedin, R.; Bauer, H.C.F. Surgical treatment of skeletal metastatic lesions of the proximal femur: Endoprosthesis or reconstruction nail? J. Bone Jt. Surg. Br. 2005, 87, 1653–1657. [Google Scholar] [CrossRef]
- Mirels, H. Metastatic disease in long bones. A proposed scoring system for diagnosing impending pathologic fractures. Clin. Orthop. Relat. Res. 1989, 249, 256–264. [Google Scholar] [CrossRef]
- Howard, E.L.; Shepherd, K.L.; Cribb, G.; Cool, P. The validity of the Mirels score for predicting impending pathological fractures of the lower limb. Bone Jt. J. 2018, 100-B, 1100–1105. [Google Scholar] [CrossRef]
- Van der Linden, Y.M.; Dijkstra, P.D.S.; Kroon, H.M.; Lok, J.J.; Noordijk, E.M.; Leer, J.W.H.; Marijnen, C.A.M. Comparative analysis of risk factors for pathological fracture with femoral metastases. J. Bone Jt. Surg. Br. 2004, 86, 566–573. [Google Scholar] [CrossRef]
- Shinoda, Y.; Sawada, R.; Ishibashi, Y.; Akiyama, T.; Zhang, L.; Hirai, T.; Oka, H.; Ohki, T.; Ikegami, M.; Okajima, K.; et al. Prediction of pathological fracture in patients with lower limb bone metastasis using computed tomography imaging. Clin. Exp. Metastasis 2020, 37, 607–616. [Google Scholar] [CrossRef]
- Philipp, T.C.; Mikula, J.D.; Doung, Y.-C.; Gundle, K.R. Is there an association between prophylactic femur stabilization and survival in patients with metastatic bone disease? Clin. Orthop. Relat. Res. 2020, 478, 540–546. [Google Scholar] [CrossRef]
- Ward, W.G.; Holsenbeck, S.; Dorey, F.J.; Spang, J.; Howe, D. Metastatic disease of the femur: Surgical treatment. Clin. Orthop. Relat. Res. 2003, 415, S230–S244. [Google Scholar] [CrossRef]
- McLynn, R.P.; Ondeck, N.T.; Grauer, J.N.; Lindskog, D.M. What is the adverse event profile after prophylactic treatment of femoral shaft or distal femur metastases? Clin. Orthop. Relat. Res. 2018, 476, 2381–2388. [Google Scholar] [CrossRef] [PubMed]
- Bindels, B.J.J.; Thio, Q.C.B.S.; Raskin, K.A.; Ferrone, M.L.; Lozano Calderón, S.A.; Schwab, J.H. Thirty-day postoperative complications after surgery for metastatic long bone disease are associated with higher mortality at 1 year. Clin. Orthop. Relat. Res. 2020, 478, 306–318. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Katagiri, H.; Murata, H.; Wasa, J.; Miyagi, M.; Honda, Y.; Takahashi, M. Surgery for femoral metastases. Bone Jt. J. 2020, 102-B, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Katagiri, H.; Takahashi, M.; Wakai, K.; Sugiura, H.; Kataoka, T.; Nakanishi, K. Prognostic factors and a scoring system for patients with skeletal metastasis. J. Bone Jt. Surg. Br. 2005, 87, 698–703. [Google Scholar] [CrossRef]
- Erol, B.; Saglam, F. Are cemented endoprosthetic reconstructions superior to uncemented endoprostheses in terms of postoperative outcomes and complications in patients with extremity-located bone metastasis scheduled for adjuvant radiotherapy? J. Arthroplast. 2021, 36, 1160–1167. [Google Scholar] [CrossRef]
- Gainor, B.J.; Buchert, P. Fracture healing in metastatic bone disease. Clin. Orthop. Relat. Res. 1983, 178, 297–302. [Google Scholar] [CrossRef]
- Capanna, R.; Campanacci, D.A. The treatment of metastases in the appendicular skeleton. J. Bone Jt. Surg. Br. 2001, 83, 471–481. [Google Scholar] [CrossRef]
- Willeumier, J.J.; van der Linden, Y.M.; Dijkstra, P.D.S. Lack of clinical evidence for postoperative radiotherapy after surgical fixation of impending or actual pathologic fractures in the long bones in patients with cancer; a systematic review. Radiother. Oncol. 2016, 121, 138–142. [Google Scholar] [CrossRef]
- Flynn, C.J.; Danjoux, C.; Wong, J.; Christakis, M.; Rubenstein, J.; Yee, A.; Yip, D.; Chow, E. Two cases of acrometastasis to the hands and review of the literature. Curr. Oncol. 2008, 15, 51–58. [Google Scholar] [CrossRef]
- Healey, J.H.; Turnbull, A.D.; Miedema, B.; Lane, J.M. Acrometastases. A study of twenty-nine patients with osseous involvement of the hands and feet. J. Bone Jt. Surg. Am. 1986, 68, 743–746. [Google Scholar] [CrossRef]
- Kerin, R. Metastatic tumors of the hand. A review of the literature. J. Bone Jt. Surg. Am. 1983, 65, 1331–1335. [Google Scholar] [CrossRef]
- Mavrogenis, A.F.; Mimidis, G.; Kokkalis, Z.T.; Karampi, E.-S.; Karampela, I.; Papagelopoulos, P.J.; Armaganidis, A. Acrometastases. Eur. J. Orthop. Surg. Traumatol. 2014, 24, 279–283. [Google Scholar] [CrossRef]
- Osterhouse, M.D.; Guebert, G.M. Bilateral acrometastasis secondary to breast cancer. J. Manip. Physiol. Ther. 2004, 27, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Shannon, F.J.; Antonescu, C.R.; Athanasian, E.A. Metastatic thymic carcinoma in a digit: A case report. J. Hand Surg. Am. 2000, 25, 1169–1172. [Google Scholar] [CrossRef] [PubMed]
- Baran, R.; Guillot, P.; Tosti, A. Metastasis from carcinoma of the bronchus to the distal aspect of two digits. Br. J. Dermatol. 1998, 138, 708. [Google Scholar] [CrossRef] [PubMed]
- Khokhar, N.; Lee, J.D. Phalangeal metastasis: First clinical sign of bronchogenic carcinoma. South. Med. J. 1983, 76, 927. [Google Scholar] [CrossRef]
- Bevan, D.A.; Ehrlich, G.E.; Gupta, V.P. Metastatic carcinoma simulating gout. JAMA 1977, 237, 2746–2747. [Google Scholar] [CrossRef]
- De Andrade, C.H.; Tsai, P.W.C.; Takagaki, T.Y. Thumb metastasis from small cell lung cancer treated with radiation. Rev. Hosp. Clin. Fac. Med. Sao Paulo 2002, 57, 283–286. [Google Scholar] [CrossRef]
- Mulvey, R.B. Peripheral bone metastases. Am. J. Roentgenol. Radium Ther. Nucl. Med. 1964, 91, 155–160. [Google Scholar]
- Okada, H.; Qing, J.; Ohnishi, T.; Watanabe, S. Metastasis of gastric carcinoma to a finger. Br. J. Dermatol. 1999, 140, 776–777. [Google Scholar]
- Plotkine, E.; Coscas, R.; Guigui, P. Digital metastasis of nasopharyngeal carcinoma: A surgical trap. Case report and literature review. Chir. Main 2008, 27, 187–190. [Google Scholar] [CrossRef]
- Igoumenou, V.G.; Mavrogenis, A.F.; Angelini, A.; Baracco, R.; Benzakour, A.; Benzakour, T.; Bork, M.; Vazifehdan, F.; Nena, U.; Ruggieri, P. Complications of spine surgery for metastasis. Eur. J. Orthop. Surg. Traumatol. 2020, 30, 37–56. [Google Scholar] [CrossRef] [PubMed]
- Quraishi, N.A.; Gokaslan, Z.L.; Boriani, S. The surgical management of metastatic epidural compression of the spinal cord. J. Bone Jt. Surg. Br. 2010, 92, 1054–1060. [Google Scholar] [CrossRef] [PubMed]
- Vazifehdan, F.; Karantzoulis, V.G.; Igoumenou, V.G. Surgical treatment for metastases of the cervical spine. Eur. J. Orthop. Surg. Traumatol. 2017, 27, 763–775. [Google Scholar] [CrossRef] [PubMed]
- Mavrogenis, A.F.; Pneumaticos, S.; Sapkas, G.S.; Papagelopoulos, P.J. Metastatic epidural spinal cord compression. Orthopedics 2009, 32, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Constans, J.P.; de Divitiis, E.; Donzelli, R.; Spaziante, R.; Meder, J.F.; Haye, C. Spinal metastases with neurological manifestations. Review of 600 cases. J. Neurosurg. 1983, 59, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Perrin, R.G.; Laxton, A.W. Metastatic Spine Disease: Epidemiology, pathophysiology, and evaluation of patients. Neurosurg. Clin. N. Am. 2004, 15, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Salvo, N.; Christakis, M.; Rubenstein, J.; de Sa, E.; Napolskikh, J.; Sinclair, E.; Ford, M.; Goh, P.; Chow, E. The role of plain radiographs in management of bone metastases. J. Palliat. Med. 2009, 12, 195–198. [Google Scholar] [CrossRef]
- Hamaoka, T.; Madewell, J.E.; Podoloff, D.A.; Hortobagyi, G.N.; Ueno, N.T. Bone imaging in metastatic breast cancer. J. Clin. Oncol. 2004, 22, 2942–2953. [Google Scholar] [CrossRef]
- Yang, H.-L.; Liu, T.; Wang, X.-M.; Xu, Y.; Deng, S.-M. Diagnosis of bone metastases: A meta-analysis comparing 18FDG PET, CT, MRI and bone scintigraphy. Eur. Radiol. 2011, 21, 2604–2617. [Google Scholar] [CrossRef]
- Jehn, C.F.; Diel, I.J.; Overkamp, F.; Kurth, A.; Schaefer, R.; Miller, K.; Lüftner, D. Management of metastatic bone disease algorithms for diagnostics and treatment. Anticancer Res. 2016, 36, 2631–2637. [Google Scholar]
- Peterson, J.J.; Kransdorf, M.J.; O’Connor, M.I. Diagnosis of occult bone metastases: Positron emission tomography. Clin. Orthop. Relat. Res. 2003, 415, S120–S128. [Google Scholar] [CrossRef]
- Chakraborty, D.; Bhattacharya, A.; Mete, U.K.; Mittal, B.R. Comparison of 18F fluoride PET/CT and 99mTc-MDP bone scan in the detection of skeletal metastases in urinary bladder carcinoma. Clin. Nucl. Med. 2013, 38, 616–621. [Google Scholar] [CrossRef]
- Duo, J.; Han, X.; Zhang, L.; Wang, G.; Ma, Y.; Yang, Y. Comparison of FDG PET/CT and gadolinium-enhanced MRI for the detection of bone metastases in patients with cancer: A meta-analysis. Clin. Nucl. Med. 2013, 38, 343–348. [Google Scholar] [CrossRef]
- Beheshti, M.; Vali, R.; Waldenberger, P.; Fitz, F.; Nader, M.; Hammer, J.; Loidl, W.; Pirich, C.; Fogelman, I.; Langsteger, W. The use of F-18 choline PET in the assessment of bone metastases in prostate cancer: Correlation with morphological changes on CT. Mol. Imaging Biol. 2010, 12, 98–107. [Google Scholar] [CrossRef]
- Shen, G.; Deng, H.; Hu, S.; Jia, Z. Comparison of choline-PET/CT, MRI, SPECT, and bone scintigraphy in the diagnosis of bone metastases in patients with prostate cancer: A meta-analysis. Skelet. Radiol. 2014, 43, 1503–1513. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, K.; Suenaga, Y.; Ueno, Y.; Kanda, T.; Maeda, T.; Makihara, N.; Ebina, Y.; Yamada, H.; Takahashi, S.; Sugimura, K. Value of fusion of PET and MRI in the detection of intra-pelvic recurrence of gynecological tumor: Comparison with 18F-FDG contrast-enhanced PET/CT and pelvic MRI. Ann. Nucl. Med. 2014, 28, 25–32. [Google Scholar] [CrossRef]
- Catalano, O.A.; Rosen, B.R.; Sahani, D.V.; Hahn, P.F.; Guimaraes, A.R.; Vangel, M.G.; Nicolai, E.; Soricelli, A.; Salvatore, M. Clinical impact of PET/MR imaging in patients with cancer undergoing same-day PET/CT: Initial experience in 134 patients—A hypothesis-generating exploratory study. Radiology 2013, 269, 857–869. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, S.; Mavrogenis, A.F.; Langevelde, K.V.; Vucht, N.V.; Kido, A.; Errani, C. Imaging of spinal bone tumors: Principles and practice. Curr. Med. Imaging 2021. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C.G.; DiPaola, C.P.; Ryken, T.C.; Bilsky, M.H.; Shaffrey, C.I.; Berven, S.H.; Harrop, J.S.; Fehlings, M.G.; Boriani, S.; Chou, D.; et al. A novel classification system for spinal instability in neoplastic disease: An evidence-based approach and expert consensus from the spine oncology study group. Spine 2010, 35, E1221–E1229. [Google Scholar] [CrossRef]
- Avrahami, E.; Tadmor, R.; Dally, O.; Hadar, H. Early MR demonstration of spinal metastases in patients with normal radiographs and CT and radionuclide bone scans. J. Comput. Assist. Tomogr. 1989, 13, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Algra, P.R.; Bloem, J.L.; Tissing, H.; Falke, T.H.; Arndt, J.W.; Verboom, L.J. Detection of vertebral metastases: Comparison between MR Imaging and bone scintigraphy. Radiographics 1991, 11, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Smoker, W.R.; Godersky, J.C.; Knutzon, R.K.; Keyes, W.D.; Norman, D.; Bergman, W. The role of MR imaging in evaluating metastatic spinal disease. AJR Am. J. Roentgenol. 1987, 149, 1241–1248. [Google Scholar] [CrossRef]
- Algra, P.R.; Heimans, J.J.; Valk, J.; Nauta, J.J.; Lachniet, M.; Van Kooten, B. Do metastases in vertebrae begin in the body or the pedicles? Imaging study in 45 patients. AJR Am. J. Roentgenol. 1992, 158, 1275–1279. [Google Scholar] [CrossRef]
- Kim, J.K.; Learch, T.J.; Colletti, P.M.; Lee, J.W.; Tran, S.D.; Terk, M.R. Diagnosis of vertebral metastasis, epidural metastasis, and malignant spinal cord compression: Are T(1)-weighted sagittal images sufficient? Magn. Reson. Imaging 2000, 18, 819–824. [Google Scholar] [CrossRef]
- Bilsky, M.H.; Laufer, I.; Fourney, D.R.; Groff, M.; Schmidt, M.H.; Varga, P.P.; Vrionis, F.D.; Yamada, Y.; Gerszten, P.C.; Kuklo, T.R. Reliability analysis of the epidural spinal cord compression scale. J. Neurosurg. Spine 2010, 13, 324–328. [Google Scholar] [CrossRef]
- Rades, D.; Rudat, V.; Veninga, T.; Stalpers, L.J.A.; Basic, H.; Karstens, J.H.; Hoskin, P.J.; Schild, S.E. A score predicting posttreatment ambulatory status in patients irradiated for metastatic spinal cord compression. Int. J. Radiat. Oncol. Biol. Phys. 2008, 72, 905–908. [Google Scholar] [CrossRef] [PubMed]
- Rades, D.; Douglas, S.; Huttenlocher, S.; Rudat, V.; Veninga, T.; Stalpers, L.J.A.; Basic, H.; Karstens, J.H.; Hoskin, P.J.; Adamietz, I.A.; et al. Validation of a score predicting post-treatment ambulatory status after radiotherapy for metastatic spinal cord compression. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 1503–1506. [Google Scholar] [CrossRef]
- Laufer, I.; Rubin, D.G.; Lis, E.; Cox, B.W.; Stubblefield, M.D.; Yamada, Y.; Bilsky, M.H. The NOMS Framework: Approach to the treatment of spinal metastatic tumors. Oncologist 2013, 18, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Matamalas, A.; Valverde, C.; Benavente, S.; Casas-Gomila, L.; Romagosa, C.; González-Tartière, P.; Pellisé, F. Team approach: Metastatic disease of the spine. JBJS Rev. 2018, 6, e6. [Google Scholar] [CrossRef] [PubMed]
- Patchell, R.A.; Tibbs, P.A.; Regine, W.F.; Payne, R.; Saris, S.; Kryscio, R.J.; Mohiuddin, M.; Young, B. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: A randomised trial. Lancet 2005, 366, 643–648. [Google Scholar] [CrossRef]
- Rades, D.; Huttenlocher, S.; Dunst, J.; Bajrovic, A.; Karstens, J.H.; Rudat, V.; Schild, S.E. Matched pair analysis comparing surgery followed by radiotherapy and radiotherapy alone for metastatic spinal cord compression. J. Clin. Oncol. 2010, 28, 3597–3604. [Google Scholar] [CrossRef]
- Tokuhashi, Y.; Matsuzaki, H.; Oda, H.; Oshima, M.; Ryu, J. A Revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine 2005, 30, 2186–2191. [Google Scholar] [CrossRef]
- Quraishi, N.A.; Arealis, G.; Salem, K.M.I.; Purushothamdas, S.; Edwards, K.L.; Boszczyk, B.M. The surgical management of metastatic spinal tumors based on an Epidural Spinal Cord Compression (ESCC) scale. Spine J. 2015, 15, 1738–1743. [Google Scholar] [CrossRef] [PubMed]
- Spratt, D.E.; Beeler, W.H.; de Moraes, F.Y.; Rhines, L.D.; Gemmete, J.J.; Chaudhary, N.; Shultz, D.B.; Smith, S.R.; Berlin, A.; Dahele, M.; et al. An integrated multidisciplinary algorithm for the management of spinal metastases: An International Spine Oncology Consortium report. Lancet Oncol. 2017, 18, e720–e730. [Google Scholar] [CrossRef]
- Thibault, I.; Al-Omair, A.; Masucci, G.L.; Masson-Côté, L.; Lochray, F.; Korol, R.; Cheng, L.; Xu, W.; Yee, A.; Fehlings, M.G.; et al. Spine stereotactic body radiotherapy for renal cell cancer spinal metastases: Analysis of outcomes and risk of vertebral compression fracture. J. Neurosurg. Spine 2014, 21, 711–718. [Google Scholar] [CrossRef]
- Sohn, S.; Chung, C.K.; Sohn, M.J.; Chang, U.-K.; Kim, S.H.; Kim, J.; Park, E. Stereotactic radiosurgery compared with external radiation therapy as a primary treatment in spine metastasis from renal cell carcinoma: A multicenter, matched-pair study. J. Neurooncol. 2014, 119, 121–128. [Google Scholar] [CrossRef]
- Folkert, M.R.; Bilsky, M.H.; Tom, A.K.; Oh, J.H.; Alektiar, K.M.; Laufer, I.; Tap, W.D.; Yamada, Y. Outcomes and toxicity for hypofractionated and single-fraction image-guided stereotactic radiosurgery for sarcomas metastasizing to the spine. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 1085–1091. [Google Scholar] [CrossRef]
- Weber, M.H.; Burch, S.; Buckley, J.; Schmidt, M.H.; Fehlings, M.G.; Vrionis, F.D.; Fisher, C.G. Instability and impending instability of the thoracolumbar spine in patients with spinal metastases: A systematic review. Int. J. Oncol. 2011, 38, 5–12. [Google Scholar]
- Fourney, D.R.; Frangou, E.M.; Ryken, T.C.; Dipaola, C.P.; Shaffrey, C.I.; Berven, S.H.; Bilsky, M.H.; Harrop, J.S.; Fehlings, M.G.; Boriani, S.; et al. Spinal Instability Neoplastic Score: An analysis of reliability and validity from the Spine Oncology Study Group. J. Clin. Oncol. 2011, 29, 3072–3077. [Google Scholar] [CrossRef]
- Leone, A.; Cianfoni, A.; Zecchi, V.; Cortese, M.C.; Rumi, N.; Colosimo, C. Instability and impending instability in patients with vertebral metastatic disease. Skelet. Radiol. 2019, 48, 195–207. [Google Scholar] [CrossRef]
- Park, J.-S.; Park, S.-J.; Lee, C.-S. Incidence and prognosis of patients with spinal metastasis as the initial manifestation of malignancy: Analysis of 338 patients undergoing surgical treatment. Bone Jt. J. 2019, 101-B, 1379–1384. [Google Scholar] [CrossRef]
- Klimo, P.; Thompson, C.J.; Kestle, J.R.W.; Schmidt, M.H. A meta-analysis of surgery versus conventional radiotherapy for the treatment of metastatic spinal epidural disease. Neuro Oncol. 2005, 7, 64–76. [Google Scholar] [CrossRef]
- Lee, C.-H.; Kwon, J.-W.; Lee, J.; Hyun, S.-J.; Kim, K.-J.; Jahng, T.-A.; Kim, H.-J. Direct decompressive surgery followed by radiotherapy versus radiotherapy alone for metastatic epidural spinal cord compression: A meta-analysis. Spine 2014, 39, E587–E592. [Google Scholar] [CrossRef]
- Rao, J.; Tiruchelvarayan, R.; Lee, L. Palliative surgery for cervical spine metastasis. Singap. Med. J. 2014, 55, 569–573. [Google Scholar] [CrossRef]
- Paulino Pereira, N.R.; Ogink, P.T.; Groot, O.Q.; Ferrone, M.L.; Hornicek, F.J.; van Dijk, C.N.; Bramer, J.A.M.; Schwab, J.H. Complications and reoperations after surgery for 647 patients with spine metastatic disease. Spine J. 2019, 19, 144–156. [Google Scholar] [CrossRef] [PubMed]
- Fourney, D.R.; Schomer, D.F.; Nader, R.; Chlan-Fourney, J.; Suki, D.; Ahrar, K.; Rhines, L.D.; Gokaslan, Z.L. Percutaneous vertebroplasty and kyphoplasty for painful vertebral body fractures in cancer patients. J. Neurosurg. 2003, 98, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Itagaki, M.W.; Talenfeld, A.D.; Kwan, S.W.; Brunner, J.W.M.; Mortell, K.E.; Brunner, M.C. Percutaneous vertebroplasty and kyphoplasty for pathologic vertebral fractures in the medicare population: Safer and less expensive than open surgery. J. Vasc. Interv. Radiol. 2012, 23, 1423–1429. [Google Scholar] [CrossRef]
- Ishida, Y.; Shigematsu, H.; Tsukamoto, S.; Morimoto, Y.; Iwata, E.; Okuda, A.; Kawasaki, S.; Tanaka, M.; Fujii, H.; Tanaka, Y.; et al. Impairment-driven cancer rehabilitation in patients with neoplastic spinal cord compression using minimally invasive spine stabilization. World J. Surg. Oncol. 2020, 18, 187. [Google Scholar] [CrossRef] [PubMed]
- Burström, G.; Persson, O.; Edström, E.; Elmi-Terander, A. Augmented Reality Navigation in Spine Surgery: A Systematic Review. Acta Neurochir. 2021, 163, 843–852. [Google Scholar] [CrossRef]
- Jud, L.; Fotouhi, J.; Andronic, O.; Aichmair, A.; Osgood, G.; Navab, N.; Farshad, M. Applicability of Augmented Reality in Orthopedic Surgery—A Systematic Review. BMC Musculoskelet. Disord. 2020, 21, 103. [Google Scholar] [CrossRef] [PubMed]
- Abrahm, J.L.; Banffy, M.B.; Harris, M.B. Spinal cord compression in patients with advanced metastatic cancer: “All I care about is walking and living my life”. JAMA 2008, 299, 937–946. [Google Scholar] [CrossRef]
- Ross, J.R.; Saunders, Y.; Edmonds, P.M.; Patel, S.; Wonderling, D.; Normand, C.; Broadley, K. A systematic review of the role of bisphosphonates in metastatic disease. Health Technol. Assess. 2004, 8, 1–176. [Google Scholar] [CrossRef]
- Facchini, G.; Di Tullio, P.; Battaglia, M.; Bartalena, T.; Tetta, C.; Errani, C.; Mavrogenis, A.F.; Rossi, G. Palliative embolization for metastases of the spine. Eur. J. Orthop. Surg. Traumatol. 2016, 26, 247–252. [Google Scholar] [CrossRef]
- Selvaggi, G.; Scagliotti, G.V. Management of bone metastases in cancer: A Review. Crit. Rev. Oncol. Hematol. 2005, 56, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Bickels, J.; Wittig, J.C.; Kollender, Y.; Kellar-Graney, K.; Malawer, M.M.; Meller, I. Sciatic nerve resection: Is that truly an indication for amputation? Clin. Orthop. Relat. Res. 2002, 399, 201–204. [Google Scholar] [CrossRef]
- Damron, T.A.; Sim, F.H. Surgical treatment for metastatic disease of the pelvis and the proximal end of the femur. Instr. Course Lect. 2000, 49, 461–470. [Google Scholar]
- Mankin, H.J.; Hornicek, F.J.; Temple, H.T.; Gebhardt, M.C. Malignant tumors of the pelvis: An outcome study. Clin. Orthop. Relat. Res. 2004, 425, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Marco, R.A.; Sheth, D.S.; Boland, P.J.; Wunder, J.S.; Siegel, J.A.; Healey, J.H. Functional and oncological outcome of acetabular reconstruction for the treatment of metastatic disease. J. Bone Jt. Surg. Am. 2000, 82, 642–651. [Google Scholar] [CrossRef]
- Patterson, F.R.; Peabody, T.D. Operative management of metastases to the pelvis and acetabulum. Orthop. Clin. N. Am. 2000, 31, 623–631. [Google Scholar] [CrossRef]
- Yasko, A.W.; Rutledge, J.; Lewis, V.O.; Lin, P.P. Disease- and recurrence-free survival after surgical resection of solitary bone metastases of the pelvis. Clin. Orthop. Relat. Res. 2007, 459, 128–132. [Google Scholar] [CrossRef]
- Müller, D.A.; Capanna, R. The surgical treatment of pelvic bone metastases. Adv. Orthop. 2015, 2015, 525363. [Google Scholar] [CrossRef]
- Harrington, K.D. Orthopaedic management of extremity and pelvic lesions. Clin. Orthop. Relat. Res. 1995, 312, 136–147. [Google Scholar]
- Rowell, P.; Lowe, M.; Sommerville, S.; Dickinson, I. Is an Acetabular cage and cement fixation sufficiently durable for the treatment of destructive acetabular metastases? Clin. Orthop. Relat. Res. 2019, 477, 1459–1465. [Google Scholar] [CrossRef]
- Tillman, R.; Tsuda, Y.; Puthiya Veettil, M.; Young, P.S.; Sree, D.; Fujiwara, T.; Abudu, A. The long-term outcomes of modified harrington procedure using antegrade pins for periacetabular metastasis and haematological diseases. Bone Jt. J. 2019, 101-B, 1557–1562. [Google Scholar] [CrossRef] [PubMed]
- Houdek, M.T.; Ferguson, P.C.; Abdel, M.P.; Griffin, A.M.; Hevesi, M.; Perry, K.I.; Rose, P.S.; Wunder, J.S.; Lewallen, D.G. Comparison of porous tantalum acetabular implants and harrington reconstruction for metastatic disease of the acetabulum. J. Bone Jt. Surg. Am. 2020, 102, 1239–1247. [Google Scholar] [CrossRef]
- Ruggieri, P.; Mavrogenis, A.F.; Angelini, A.; Mercuri, M. Metastases of the pelvis: Does resection improve survival? Orthopedics 2011, 34, e236–e244. [Google Scholar] [CrossRef]
- Cotten, A.; Deprez, X.; Migaud, H.; Chabanne, B.; Duquesnoy, B.; Chastanet, P. Malignant acetabular osteolyses: Percutaneous injection of acrylic bone cement. Radiology 1995, 197, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Butala, N.B.; Etheridge, B.S.; Siegel, H.J.; Lemons, J.E.; Eberhardt, A.W. A biomechanical study of periacetabular defects and cement filling. J. Biomech. Eng. 2007, 129, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Maccauro, G.; Liuzza, F.; Scaramuzzo, L.; Milani, A.; Muratori, F.; Rossi, B.; Waide, V.; Logroscino, G.; Logroscino, C.A.; Maffulli, N. Percutaneous acetabuloplasty for metastatic acetabular lesions. BMC Musculoskelet. Disord. 2008, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Kelekis, A.; Lovblad, K.O.; Mehdizade, A.; Somon, T.; Yilmaz, H.; Wetzel, S.G.; Seium, Y.; Dietrich, P.-Y.; Rufenacht, D.A.; Martin, J.-B. Pelvic osteoplasty in osteolytic metastases: Technical approach under fluoroscopic guidance and early clinical results. J. Vasc. Interv. Radiol. 2005, 16, 81–88. [Google Scholar] [CrossRef]
- Marcy, P.Y.; Palussière, J.; Descamps, B.; Magné, N.; Bondiau, P.Y.; Ciais, C.; Bruneton, J.N. Percutaneous cementoplasty for pelvic bone metastasis. Support. Care Cancer 2000, 8, 500–503. [Google Scholar] [CrossRef]
- Tucci, M.; Stucci, S.; Savonarola, A.; Ciavarella, S.; Cafforio, P.; Dammacco, F.; Silvestris, F. Immature dendritic cells in multiple myeloma are prone to osteoclast-like differentiation through interleukin-17A stimulation. Br. J. Haematol. 2013, 161, 821–831. [Google Scholar] [CrossRef]
- Culleton, S.; Kwok, S.; Chow, E. Radiotherapy for pain. Clin. Oncol. 2011, 23, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Sahgal, A.; Roberge, D.; Schellenberg, D.; Purdie, T.G.; Swaminath, A.; Pantarotto, J.; Filion, E.; Gabos, Z.; Butler, J.; Letourneau, D.; et al. The Canadian Association of Radiation Oncology scope of practice guidelines for lung, liver and spine stereotactic body radiotherapy. Clin. Oncol. 2012, 24, 629–639. [Google Scholar] [CrossRef] [PubMed]
- Sze, W.M.; Shelley, M.D.; Held, I.; Wilt, T.J.; Mason, M.D. Palliation of metastatic bone pain: Single fraction versus multifraction radiotherapy—A systematic review of randomised trials. Clin. Oncol. 2003, 15, 345–352. [Google Scholar] [CrossRef]
- Mavrogenis, A.F.; Angelini, A.; Vottis, C.; Pala, E.; Calabrò, T.; Papagelopoulos, P.J.; Ruggieri, P. Modern Palliative treatments for metastatic bone disease: Awareness of advantages, disadvantages, and guidance. Clin. J. Pain 2016, 32, 337–350. [Google Scholar] [CrossRef]
- Chow, E.; Zeng, L.; Salvo, N.; Dennis, K.; Tsao, M.; Lutz, S. Update on the systematic review of palliative radiotherapy trials for bone metastases. Clin. Oncol. 2012, 24, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Shiloh, R.; Krishnan, M. Radiation for treatment of painful bone metastases. Hematol. Oncol. Clin. N. Am. 2018, 32, 459–468. [Google Scholar] [CrossRef]
- Finlay, I.G.; Mason, M.D.; Shelley, M. Radioisotopes for the palliation of metastatic bone cancer: A systematic review. Lancet Oncol. 2005, 6, 392–400. [Google Scholar] [CrossRef]
- Hird, A.; Chow, E.; Zhang, L.; Wong, R.; Wu, J.; Sinclair, E.; Danjoux, C.; Tsao, M.; Barnes, E.; Loblaw, A. Determining the incidence of pain flare following palliative radiotherapy for symptomatic bone metastases: Results from three Canadian cancer centers. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 193–197. [Google Scholar] [CrossRef]
- Husain, Z.A.; Sahgal, A.; De Salles, A.; Funaro, M.; Glover, J.; Hayashi, M.; Hiraoka, M.; Levivier, M.; Ma, L.; Martínez-Alvarez, R.; et al. Stereotactic body radiotherapy for de novo spinal metastases: Systematic review. J. Neurosurg. Spine 2017, 27, 295–302. [Google Scholar] [CrossRef]
- Jawad, M.S.; Fahim, D.K.; Gerszten, P.C.; Flickinger, J.C.; Sahgal, A.; Grills, I.S.; Sheehan, J.; Kersh, R.; Shin, J.; Oh, K.; et al. Vertebral compression fractures after stereotactic body radiation therapy: A large, multi-institutional, multinational evaluation. J. Neurosurg. Spine 2016, 24, 928–936. [Google Scholar] [CrossRef]
- Huisman, M.; van den Bosch, M.A.A.J.; Wijlemans, J.W.; van Vulpen, M.; van der Linden, Y.M.; Verkooijen, H.M. Effectiveness of reirradiation for painful bone metastases: A systematic review and meta-analysis. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, 8–14. [Google Scholar] [CrossRef]
- Myrehaug, S.; Sahgal, A.; Hayashi, M.; Levivier, M.; Ma, L.; Martinez, R.; Paddick, I.; Régis, J.; Ryu, S.; Slotman, B.; et al. Reirradiation spine stereotactic body radiation therapy for spinal metastases: Systematic Review. J. Neurosurg. Spine 2017, 27, 428–435. [Google Scholar] [CrossRef]
- Macedo, F.; Ladeira, K.; Pinho, F.; Saraiva, N.; Bonito, N.; Pinto, L.; Goncalves, F. Bone metastases: An overview. Oncol. Rev. 2017, 11, 321. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.; Nilsson, S.; Heinrich, D.; Helle, S.I.; O’Sullivan, J.M.; Fosså, S.D.; Chodacki, A.; Wiechno, P.; Logue, J.; Seke, M.; et al. Alpha emitter Radium-223 and survival in metastatic prostate cancer. N. Engl. J. Med. 2013, 369, 213–223. [Google Scholar] [CrossRef]
- Terrisse, S.; Karamouza, E.; Parker, C.C.; Sartor, A.O.; James, N.D.; Pirrie, S.; Collette, L.; Tombal, B.F.; Chahoud, J.; Smeland, S.; et al. Overall survival in men with bone metastases from castration-resistant prostate cancer treated with bone-targeting radioisotopes: A meta-analysis of individual patient data from randomized clinical trials. JAMA Oncol. 2020, 6, 206–216. [Google Scholar] [CrossRef]
- Manafi-Farid, R.; Masoumi, F.; Divband, G.; Saidi, B.; Ataeinia, B.; Hertel, F.; Schweighofer-Zwink, G.; Morgenroth, A.; Beheshti, M. Targeted palliative radionuclide therapy for metastatic bone pain. J. Clin. Med. 2020, 9, 2622. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.; Body, J.J.; Aapro, M.; Hadji, P.; Herrstedt, J.; ESMO Guidelines Working Group. Bone health in cancer patients: ESMO Clinical Practice Guidelines. Ann. Oncol. 2014, 25 (Suppl. 3), iii124–iii137. [Google Scholar] [CrossRef]
- Hutton, B.; Addison, C.L.; Campbell, K.; Fergusson, D.; Mazarello, S.; Clemons, M. A systematic review of dosing frequency with bone-targeted agents for patients with bone metastases from breast cancer. J. Bone Oncol. 2013, 2, 123–131. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hortobagyi, G.N.; Van Poznak, C.; Harker, W.G.; Gradishar, W.J.; Chew, H.; Dakhil, S.R.; Haley, B.B.; Sauter, N.; Mohanlal, R.; Zheng, M.; et al. Continued treatment effect of zoledronic acid dosing every 12 vs 4 weeks in women with breast cancer metastatic to bone: The OPTIMIZE-2 Randomized Clinical Trial. JAMA Oncol. 2017, 3, 906–912. [Google Scholar] [CrossRef]
- Himelstein, A.L.; Foster, J.C.; Khatcheressian, J.L.; Roberts, J.D.; Seisler, D.K.; Novotny, P.J.; Qin, R.; Go, R.S.; Grubbs, S.S.; O’Connor, T.; et al. Effect of longer-interval vs standard dosing of zoledronic acid on skeletal events in patients with bone metastases: A randomized clinical trial. JAMA 2017, 317, 48–58. [Google Scholar] [CrossRef]
- Tsourdi, E.; Langdahl, B.; Cohen-Solal, M.; Aubry-Rozier, B.; Eriksen, E.F.; Guañabens, N.; Obermayer-Pietsch, B.; Ralston, S.H.; Eastell, R.; Zillikens, M.C. Discontinuation of denosumab therapy for osteoporosis: A systematic review and position statement by ECTS. Bone 2017, 105, 11–17. [Google Scholar] [CrossRef]
- Lipton, A.; Fizazi, K.; Stopeck, A.T.; Henry, D.H.; Smith, M.R.; Shore, N.; Martin, M.; Vadhan-Raj, S.; Brown, J.E.; Richardson, G.E.; et al. Effect of denosumab versus zoledronic acid in preventing skeletal-related events in patients with bone metastases by baseline characteristics. Eur. J. Cancer 2016, 53, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Pu, F. Safety of denosumab versus zoledronic acid in patients with bone metastases: A meta-analysis of randomized controlled trials. Oncol. Res. Treat. 2016, 39, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Forauer, A.R.; Kent, E.; Cwikiel, W.; Esper, P.; Redman, B. Selective palliative transcatheter embolization of bony metastases from renal cell carcinoma. Acta Oncol. 2007, 46, 1012–1018. [Google Scholar] [CrossRef] [PubMed]
- Hansch, A.; Neumann, R.; Pfeil, A.; Marintchev, I.; Pfleiderer, S.; Gajda, M.; Kaiser, W.-A. Embolization of an unusual metastatic site of hepatocellular carcinoma in the humerus. World J. Gastroenterol. 2009, 15, 2280–2282. [Google Scholar] [CrossRef] [PubMed]
- De Vries, M.M.; Persoon, A.C.M.; Jager, P.L.; Gravendeel, J.; Plukker, J.T.M.; Sluiter, W.J.; Links, T.P. Embolization therapy of bone metastases from epithelial thyroid carcinoma: Effect on symptoms and serum thyroglobulin. Thyroid 2008, 18, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Eustatia-Rutten, C.F.A.; Romijn, J.A.; Guijt, M.J.; Vielvoye, G.J.; van den Berg, R.; Corssmit, E.P.M.; Pereira, A.M.; Smit, J.W.A. Outcome of palliative embolization of bone metastases in differentiated thyroid carcinoma. J. Clin. Endocrinol. Metab. 2003, 88, 3184–3189. [Google Scholar] [CrossRef] [PubMed]
- Smit, J.W.; Vielvoye, G.J.; Goslings, B.M. Embolization for vertebral metastases of follicular thyroid carcinoma. J. Clin. Endocrinol. Metab. 2000, 85, 989–994. [Google Scholar] [CrossRef]
- Van Tol, K.M.; Hew, J.M.; Jager, P.L.; Vermey, A.; Dullaart, R.P.; Links, T.P. Embolization in combination with radioiodine therapy for bone metastases from differentiated thyroid carcinoma. Clin. Endocrinol. 2000, 52, 653–659. [Google Scholar] [CrossRef]
- Rossi, G.; Mavrogenis, A.F.; Casadei, R.; Bianchi, G.; Romagnoli, C.; Rimondi, E.; Ruggieri, P. Embolisation of bone metastases from renal cancer. Radiol. Med. 2013, 118, 291–302. [Google Scholar] [CrossRef]
- Rossi, G.; Mavrogenis, A.F.; Rimondi, E.; Braccaioli, L.; Calabrò, T.; Ruggieri, P. Selective embolization with n-butyl cyanoacrylate for metastatic bone disease. J. Vasc. Interv. Radiol. 2011, 22, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Barton, P.P.; Waneck, R.E.; Karnel, F.J.; Ritschl, P.; Kramer, J.; Lechner, G.L. Embolization of bone metastases. J. Vasc. Interv. Radiol. 1996, 7, 81–88. [Google Scholar] [CrossRef]
- Kato, S.; Hozumi, T.; Takaki, Y.; Yamakawa, K.; Goto, T.; Kondo, T. Optimal schedule of preoperative embolization for spinal metastasis surgery. Spine 2013, 38, 1964–1969. [Google Scholar] [CrossRef]
- Kim, W.; Han, I.; Jae, H.J.; Kang, S.; Lee, S.A.; Kim, J.S.; Kim, H.-S. Preoperative embolization for bone metastasis from hepatocellular carcinoma. Orthopedics 2015, 38, e99–e105. [Google Scholar] [CrossRef] [PubMed]
- Luksanapruksa, P.; Buchowski, J.M.; Tongsai, S.; Singhatanadgige, W.; Jennings, J.W. Systematic review and meta-analysis of effectiveness of preoperative embolization in surgery for metastatic spine disease. J. Neurointerv. Surg. 2018, 10, 596–601. [Google Scholar] [CrossRef]
- Pazionis, T.J.C.; Papanastassiou, I.D.; Maybody, M.; Healey, J.H. Embolization of hypervascular bone metastases reduces intraoperative blood loss: A case-control study. Clin. Orthop. Relat. Res. 2014, 472, 3179–3187. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Lang, E.V. Bone metastases from renal cell carcinoma: Preoperative embolization. J. Vasc. Interv. Radiol. 1998, 9, 263–269. [Google Scholar] [CrossRef]
- Kickuth, R.; Waldherr, C.; Hoppe, H.; Bonel, H.M.; Ludwig, K.; Beck, M.; Triller, J. Interventional management of hypervascular osseous metastasis: Role of embolotherapy before orthopedic tumor resection and bone stabilization. AJR Am. J. Roentgenol. 2008, 191, W240–W247. [Google Scholar] [CrossRef]
- Rosenthal, D.I. Radiofrequency treatment. Orthop. Clin. N. Am. 2006, 37, 475–484. [Google Scholar] [CrossRef]
- Rosenthal, D.I.; Alexander, A.; Rosenberg, A.E.; Springfield, D. Ablation of osteoid osteomas with a percutaneously placed electrode: A new procedure. Radiology 1992, 183, 29–33. [Google Scholar] [CrossRef]
- Lane, M.D.; Le, H.B.Q.; Lee, S.; Young, C.; Heran, M.K.S.; Badii, M.; Clarkson, P.W.; Munk, P.L. Combination radiofrequency ablation and cementoplasty for palliative treatment of painful neoplastic bone metastasis: Experience with 53 treated lesions in 36 patients. Skelet. Radiol. 2011, 40, 25–32. [Google Scholar] [CrossRef]
- Mendel, E.; Bourekas, E.; Gerszten, P.; Golan, J.D. Percutaneous techniques in the treatment of spine tumors: What are the diagnostic and therapeutic indications and outcomes? Spine 2009, 34, S93–S100. [Google Scholar] [CrossRef]
- Goetz, M.P.; Callstrom, M.R.; Charboneau, J.W.; Farrell, M.A.; Maus, T.P.; Welch, T.J.; Wong, G.Y.; Sloan, J.A.; Novotny, P.J.; Petersen, I.A.; et al. Percutaneous image-guided radiofrequency ablation of painful metastases involving bone: A multicenter study. J. Clin. Oncol. 2004, 22, 300–306. [Google Scholar] [CrossRef]
- Wallace, A.N.; Robinson, C.G.; Meyer, J.; Tran, N.D.; Gangi, A.; Callstrom, M.R.; Chao, S.T.; Van Tine, B.A.; Morris, J.M.; Bruel, B.M.; et al. The Metastatic Spine Disease Multidisciplinary Working Group Algorithms. Oncologist 2015, 20, 1205–1215. [Google Scholar] [CrossRef]
- Luigi Cazzato, R.; Auloge, P.; De Marini, P.; Rousseau, C.; Chiang, J.B.; Koch, G.; Caudrelier, J.; Rao, P.; Garnon, J.; Gangi, A. Percutaneous image-guided ablation of bone metastases: Local tumor control in oligometastatic patients. Int. J. Hyperth. 2018, 35, 493–499. [Google Scholar] [CrossRef]
- Belehradek, M.; Domenge, C.; Luboinski, B.; Orlowski, S.; Belehradek, J.; Mir, L.M. Electrochemotherapy, a new antitumor treatment. First clinical phase I-II trial. Cancer 1993, 72, 3694–3700. [Google Scholar] [CrossRef]
- Horiuchi, A.; Nikaido, T.; Mitsushita, J.; Toki, T.; Konishi, I.; Fujii, S. Enhancement of antitumor effect of bleomycin by low-voltage in vivo electroporation: A study of human uterine leiomyosarcomas in nude mice. Int. J. Cancer 2000, 88, 640–644. [Google Scholar] [CrossRef]
- Kotnik, T.; Pucihar, G.; Miklavcic, D. Induced transmembrane voltage and its correlation with electroporation-mediated molecular transport. J. Membr. Biol. 2010, 236, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Fini, M.; Tschon, M.; Ronchetti, M.; Cavani, F.; Bianchi, G.; Mercuri, M.; Alberghini, M.; Cadossi, R. Ablation of bone cells by electroporation. J. Bone Jt. Surg. Br. 2010, 92, 1614–1620. [Google Scholar] [CrossRef]
- Giardino, R.; Fini, M.; Bonazzi, V.; Cadossi, R.; Nicolini, A.; Carpi, A. Electrochemotherapy a novel approach to the treatment of metastatic nodules on the skin and subcutaneous tissues. Biomed. Pharmacother. 2006, 60, 458–462. [Google Scholar] [CrossRef]
- Zhou, Y.-F. High intensity focused ultrasound in clinical tumor ablation. World J. Clin. Oncol. 2011, 2, 8–27. [Google Scholar] [CrossRef]
- Cornelis, F.H.; Ben Ammar, M.; Nouri-Neuville, M.; Matton, L.; Benderra, M.A.; Gligorov, J.; Fallet, V.; Mir, L.M. Percutaneous image-guided electrochemotherapy of spine metastases: Initial experience. Cardiovasc. Interv. Radiol. 2019, 42, 1806–1809. [Google Scholar] [CrossRef]
- Campanacci, L.; Bianchi, G.; Cevolani, L.; Errani, C.; Ciani, G.; Facchini, G.; Spinnato, P.; Tognù, A.; Massari, L.; Cornelis, F.H.; et al. Operating procedures for electrochemotherapy in bone metastases: Results from a multicenter prospective study on 102 patients. Eur. J. Surg. Oncol. 2021. [Google Scholar] [CrossRef]
- Simon, C.J.; Dupuy, D.E.; Mayo-Smith, W.W. Microwave ablation: Principles and applications. Radiographics 2005, 25 (Suppl. 1), S69–S83. [Google Scholar] [CrossRef] [PubMed]
- Lynn, J.G.; Zwemer, R.L.; Chick, A.J.; Miller, A.E. A new method for the generation and use of focused ultrasound in experimental biology. J. Gen. Physiol. 1942, 26, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Bazzocchi, A.; Napoli, A.; Sacconi, B.; Battista, G.; Guglielmi, G.; Catalano, C.; Albisinni, U. MRI-guided focused ultrasound surgery in musculoskeletal diseases: The hot topics. Br. J. Radiol. 2016, 89, 20150358. [Google Scholar] [CrossRef]
- Fry, W.J.; Barnard, J.W.; Fry, E.J.; Krumins, R.F.; Brennan, J.F. Ultrasonic lesions in the mammalian central nervous system. Science 1955, 122, 517–518. [Google Scholar] [CrossRef]
- Lynn, J.G.; Putnam, T.J. Histology of cerebral lesions produced by focused ultrasound. Am. J. Pathol. 1944, 20, 637–649. [Google Scholar]
- Chamberlain, M.C.; Kormanik, P.A. Epidural spinal cord compression: A single institution’s retrospective experience. Neuro Oncol. 1999, 1, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, M.D.; Ghanouni, P.; Kanaev, S.V.; Iozeffi, D.; Gianfelice, D.; Fennessy, F.M.; Kuten, A.; Meyer, J.E.; LeBlang, S.D.; Roberts, A.; et al. Magnetic resonance-guided focused ultrasound for patients with painful bone metastases: Phase III trial results. J. Natl. Cancer Inst. 2014, 106, dju082. [Google Scholar] [CrossRef]
- Scipione, R.; Anzidei, M.; Bazzocchi, A.; Gagliardo, C.; Catalano, C.; Napoli, A. HIFU for bone metastases and other musculoskeletal applications. Semin. Interv. Radiol. 2018, 35, 261–267. [Google Scholar] [CrossRef]
- Tsai, Y.-C.; Lee, H.-L.; Kuo, C.-C.; Chen, C.-Y.; Hsieh, K.L.-C.; Wu, M.-H.; Wen, Y.-C.; Yu, H.-W.; Hsu, F.-C.; Tsai, J.-T.; et al. Prognostic and predictive factors for clinical and radiographic responses in patients with painful bone metastasis treated with magnetic resonance-guided focused ultrasound surgery. Int. J. Hyperth. 2019, 36, 932–937. [Google Scholar] [CrossRef] [PubMed]
- Liberman, B.; Gianfelice, D.; Inbar, Y.; Beck, A.; Rabin, T.; Shabshin, N.; Chander, G.; Hengst, S.; Pfeffer, R.; Chechick, A.; et al. Pain palliation in patients with bone metastases using MR-guided focused ultrasound surgery: A multicenter study. Ann. Surg. Oncol. 2009, 16, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Napoli, A.; Anzidei, M.; Marincola, B.C.; Brachetti, G.; Ciolina, F.; Cartocci, G.; Marsecano, C.; Zaccagna, F.; Marchetti, L.; Cortesi, E.; et al. Primary pain palliation and local tumor control in bone metastases treated with magnetic resonance-guided focused ultrasound. Investig. Radiol. 2013, 48, 351–358. [Google Scholar] [CrossRef]
- Lee, H.-L.; Kuo, C.-C.; Tsai, J.-T.; Chen, C.-Y.; Wu, M.-H.; Chiou, J.-F. Magnetic resonance-guided focused ultrasound versus conventional radiation therapy for painful bone metastasis: A matched-pair study. J. Bone Jt. Surg. Am. 2017, 99, 1572–1578. [Google Scholar] [CrossRef]
- Westhoff, P.G.; de Graeff, A.; Monninkhof, E.M.; Pomp, J.; van Vulpen, M.; Leer, J.W.H.; Marijnen, C.A.M.; van der Linden, Y.M.; Dutch Bone Metastasis Study Group. Quality of Life in relation to pain response to radiation therapy for painful bone metastases. Int. J. Radiat. Oncol. Biol. Phys. 2015, 93, 694–701. [Google Scholar] [CrossRef]
- Huisman, M.; ter Haar, G.; Napoli, A.; Hananel, A.; Ghanouni, P.; Lövey, G.; Nijenhuis, R.J.; van den Bosch, M.A.A.J.; Rieke, V.; Majumdar, S.; et al. International consensus on use of focused ultrasound for painful bone metastases: Current status and future directions. Int. J. Hyperth. 2015, 31, 251–259. [Google Scholar] [CrossRef]

| Primary Tumor | Clinical Profile |
|---|---|
| Breast—positive 1 | Favorable |
| Breast—unknown 2 | Favorable |
| Kidney—solitary metastasis | Favorable |
| Thyroid | Favorable |
| Prostate | Moderate |
| Kidney—multiple metastases | Moderate |
| Soft tissue sarcoma | Moderate |
| Breast—triple-negative 3 | Moderate |
| Kidney—unknown 4 | Moderate |
| Endometrial carcinoma | Moderate |
| Osteosarcoma | Moderate |
| Ewing sarcoma | Moderate |
| Ovary | Moderate |
| Lung | Unfavorable |
| Colorectal | Unfavorable |
| Unknown primary | Unfavorable |
| Esophagus | Unfavorable |
| Bladder | Unfavorable |
| Melanoma | Unfavorable |
| Head and neck cancer | Unfavorable |
| Liver and/or pancreas | Unfavorable |
| Stomach | Unfavorable |
| Variables | Prediction of Prognosis | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Favorable | Moderate | Unfavorable | |||||||||
| 80–100 | ≤70 | 80–100 | ≤70 | 80–100 | ≤70 | ||||||
| No | Yes | No | Yes | No | Yes | No | Yes | No | Yes | No | Yes |
| Category | A | A | A | B | B | B | C | C | C | C | D | D |
| Median survival | 21.9 months | 10.5 months | 4.6 months | 2.2 months | ||||||||
| Model | Development | Variables to Input | Patient Population | Prediction of Survival | External Validation |
|---|---|---|---|---|---|
| PathFx model 1.0 | Forsberg et al. [20], 2011 | Not available | 189 patients treated for metastatic bone disease between 1999 and 2003 | Survival at 1, 3, 6, 12, 18, and 24 months | Forsberg et al. [10], 2012; Piccioli et al. [9], 2015; Forsberg et al. [12], 2017; Ogura et al. [11], 2017; Meares et al. [14], 2019 |
| PathFx model 2.0 | Overmann et al. [21], 2020 | Not available | 189 patients treated for metastatic bone disease between 1999 and 2003 | Survival at 1, 3, 6, 12, 18, and 24 months | Overmann et al. [21], 2020 |
| PathFx model 3.0 (https://www.pathfx.org) | Anderson et al. [13], 2020 | Age, sex, oncologic diagnosis, pathological fracture, ECOG performance status, hemoglobin level, absolute lymphocyte count, number of skeletal metastases, organ metastases. In case of prostate cancer, PSA, patient age at metastases, treatment, race, comorbidity, alkaline phosphatase velocity. | 208 patients treated for metastatic bone disease between 2015 and 2018 | Survival at 1, 3, 6, 12, 18, and 24 months | Anderson et al. [13], 2020 |
| Machine Learning Algorithm (https://sorg-apps.shinyapps.io/extremitymetssurvival/) | Thio et al. [19], 2020 | Age, primary tumor type, visceral metastases, brain metastases, previous systemic therapy, hemoglobin level, white blood cell count, platelet count, absolute lymphocyte count, absolute neutrophil count, albumin level, alkaline phosphatase level, calcium, creatinine, sodium. | 1090 patients treated surgically for long bone metastases between 1999 and 2017 | Survival at 3 and 12 months | None |
| SORG Machine Learning Algorithm (https://sorg-apps.shinyapps.io/spinemets/, accessed on 10 July 2021) | Karhade et al. [22], 2018 | White blood cell, albumin, American Society of Anesthesiologist Class, spine location, functional status, hematocrit, alkaline phosphatase. | 1790 patients treated surgically for spinal metastatic disease between 2000 and 2016 | Survival at 1 month | None |
| Machine Learning Algorithm (https://sorg-apps.shinyapps.io/spinemetssurvival/, accessed on 10 July 2021) | Karhade et al. [23], 2019 | BMI, other Charlson comorbidity, primary tumor histology, ECOG performance status, ASIA, number of spine metastases, visceral metastases, brain metastases, previous systemic therapy, hemoglobin, platelet, absolute lymphocyte, albumin, alkaline phosphatase, creatinine, INR. | 732 patients treated surgically for spinal metastatic disease between 2000 and 2016 | Survival at 3 and 12 months | None |
| Variable | Score | ||
|---|---|---|---|
| 1 Point | 2 Points | 3 Points | |
| Finding on imaging | Blastic | Mixed | Lytic |
| Size 1 | <1/3 | 1/3–2/3 | >2/3 |
| Site | Upper extremity | Lower extremity | Peritrochanteric |
| Pain | Mild | Moderate | Mechanical |
| Variable | Score |
|---|---|
| Location | |
| Junctional (occiput–C2, C7–T2, T11–L1, L5–S1) | 3 |
| Mobile spine (C3–C6, L2–L4) | 2 |
| Semi-rigid (T3–T10) | 1 |
| Rigid (S2–S5) | 0 |
| Pain | |
| Yes | 3 |
| Occasional pain but not mechanical | 1 |
| Pain-free lesion | 0 |
| Bone lesion | |
| Lytic | 2 |
| Mixed (lytic/blastic) | 1 |
| Blastic | 0 |
| Radiographic spinal alignment | |
| Subluxation/translation present | 4 |
| De novo deformity (kyphosis/scoliosis) | 2 |
| Normal alignment | 0 |
| Vertebral body collapse | |
| >50% collapse | 3 |
| <50% collapse | 2 |
| No collapse with >50% body involved | 1 |
| None of the above | 0 |
| Posterolateral involvement of spinal elements | |
| Bilateral | 3 |
| Unilateral | 1 |
| None of the above | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsukamoto, S.; Kido, A.; Tanaka, Y.; Facchini, G.; Peta, G.; Rossi, G.; Mavrogenis, A.F. Current Overview of Treatment for Metastatic Bone Disease. Curr. Oncol. 2021, 28, 3347-3372. https://doi.org/10.3390/curroncol28050290
Tsukamoto S, Kido A, Tanaka Y, Facchini G, Peta G, Rossi G, Mavrogenis AF. Current Overview of Treatment for Metastatic Bone Disease. Current Oncology. 2021; 28(5):3347-3372. https://doi.org/10.3390/curroncol28050290
Chicago/Turabian StyleTsukamoto, Shinji, Akira Kido, Yasuhito Tanaka, Giancarlo Facchini, Giuliano Peta, Giuseppe Rossi, and Andreas F. Mavrogenis. 2021. "Current Overview of Treatment for Metastatic Bone Disease" Current Oncology 28, no. 5: 3347-3372. https://doi.org/10.3390/curroncol28050290
APA StyleTsukamoto, S., Kido, A., Tanaka, Y., Facchini, G., Peta, G., Rossi, G., & Mavrogenis, A. F. (2021). Current Overview of Treatment for Metastatic Bone Disease. Current Oncology, 28(5), 3347-3372. https://doi.org/10.3390/curroncol28050290








