Androgen Deprivation Therapy, Hypogonadism and Cardiovascular Toxicity in Men with Advanced Prostate Cancer
Abstract
1. Introduction
2. Cardiovascular Effects of Testosterone
2.1. Favourable Effects
2.2. Deleterious Effects
2.3. Controversial Effects
3. Clinical Cardiovascular Effects of Hypogonadism
3.1. Hypogonadism and Cardiovascular Risk Factors
3.2. Hypogonadism and QTc Interval on ECG
3.3. Hypogonadism and Cardiovascular Mortality
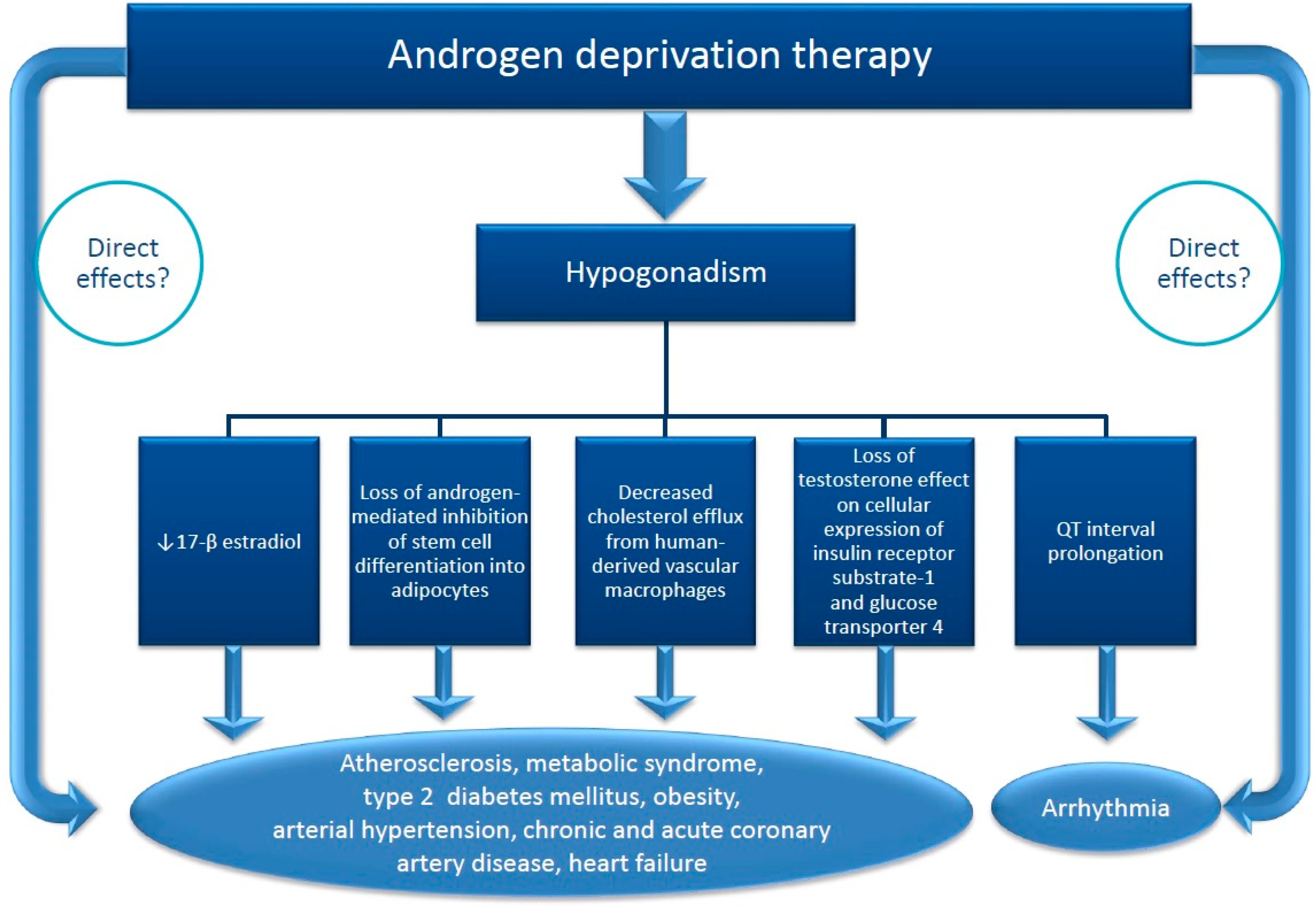
4. Cardiovascular Effects of ADT
4.1. Mechanisms of Cardiovascular Effects of ADT
4.2. Clinical Studies
4.2.1. Studies Showing an Additional Cardiovascular Risk in Patients on ADT
4.2.2. Studies Showing No Additional Cardiovascular Risk in Patients on ADT
4.2.3. Studies Comparing the Cardiovascular Risk of Various ADT
5. Therapeutic Consequences
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Humphrey, P.A. Cancer of the Male Reproductive Organs. In World Cancer Report 2014; Stewart, B.W., Wild, C.P., Eds.; The International Agency for Research on Cancer: Lyon, France, 2014; pp. 453–464. [Google Scholar]
- Wu, F.-C.; Tajar, A.; Beynon, J.M.; Pye, S.R.; Silman, A.J.; Finn, J.D.; O’Neill, T.W.; Bartfai, G.; Casanueva, F.F.; Forti, G.; et al. Identification of late-onset hypogonadism in middle-aged and elderly men. N. Engl. J. Med. 2010, 363, 123–135. [Google Scholar] [CrossRef]
- Gheorghe, A.C.D.; Ciobanu, A.; Hodorogea, A.S.; Nanea, I.T.; Gheorghe, G.S. Testosterone: Friend or foe for the cardiovascular system in men? Ann. Clin. Anal. Med. 2020, 11, 688–693. [Google Scholar]
- Ahmad, I.; Sansom, O.J.; Leung, H.Y. Advances in mouse models of prostate cancer. Expert Rev. Mol. Med. 2008, 10, e16. [Google Scholar] [CrossRef] [PubMed]
- Green, S.M.; Mostaghel, E.A.; Nelson, P.S. Androgen action and metabolism in prostate cancer. Mol. Cell. Endocrinol. 2012, 360, 3–13. [Google Scholar] [CrossRef]
- Parker, C.; Castro, E.; Fizazi, K.; Heidenreich, A.; Ost, P.; Procopio, G.; Tombal, B.; Gillessen, S.; on behalf of the ESMO Guidelines Committee. Prostate cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 1119–1134. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Tharp, D.L.; Masseau, I.; Ivey, J.; Ganjam, V.K.; Bowles, D.K. Endogenous testosterone attenuates neointima formation after moderate coronary balloon injury in male swine. Cardiovasc. Res. 2009, 82, 152–160. [Google Scholar] [CrossRef]
- Sedlak, T.; Shufelt, C.; Iribarren, C.; Merz, C.N.B. Sex Hormones and the QT Interval: A Review. J. Women’s Health 2012, 21, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Salem, J.-E.; Alexandre, J.; Bachelot, A.; Funck-Brentano, C. Influence of steroid hormones on ventricular repolarization. Pharmacol. Ther. 2016, 167, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.; Monami, M.; Rastrelli, G.; Aversa, A.; Sforza, A.; Lenzi, A.; Forti, G.; Mannucci, E.; Maggi, M. Type 2 diabetes mellitus and testosterone: A meta-analysis study. Int. J. Androl. 2011, 34, 528–540. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, M.; Fui, M.N.T.; Dupuis, P. Lowered testosterone in male obesity: Mechanisms, morbidity and management. Asian J. Androl. 2014, 16, 223–231. [Google Scholar] [CrossRef]
- Chazenbalk, G.; Singh, P.; Irge, D.; Shah, A.; Abbott, D.H.; Dumesic, D.A. Androgens inhibit adipogenesis during human adipose stem cell commitment to preadipocyte formation. Steroids 2013, 78, 920–926. [Google Scholar] [CrossRef]
- Rosen, E.D.; Hsu, C.H.; Wang, X.; Sakai, S.; Freeman, M.W.; Gonzalez, F.J.; Spiegelman, B.M. C/EBP induces adipogenesis through PPAR: A unified pathway. Genes Dev. 2002, 16, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Madsen, M.S.; Siersbæk, R.; Boergesen, M.; Nielsen, R.; Mandrup, S. Peroxisome Proliferator-Activated Receptor and C/EBP Synergistically Activate Key Metabolic Adipocyte Genes by Assisted Loading. Mol. Cell. Biol. 2014, 34, 939–954. [Google Scholar] [CrossRef]
- Hu, J.R.; Duncan, M.S.; Morgans, A.K.; Brown, J.D.; Meijers, W.C.; Freiberg, M.S.; Salem, J.E.; Beckman, J.A.; Moslehi, J.J. Cardiovascular Effects of Androgen Deprivation Therapy in Prostate Cancer Contemporary Meta-Analysis. Arterioscler. Thromb. Vasc. Biol. 2020, 40, e55–e64. [Google Scholar] [CrossRef]
- Zhang, K.; Guo, W.; Yang, Y. JAK2/STAT3 pathway is involved in the early stage of adipogenesis through regulating C/EBPβ transcription. J. Cell Biochem. 2011, 112, 488–497. [Google Scholar] [CrossRef]
- Rossouw, J.E. Hormones, genetic factors, and gender differences in cardiovascular disease. Cardiovasc. Res. 2002, 53, 550–557. [Google Scholar] [CrossRef]
- Holmegard, H.N.; Nordestgaard, B.G.; Schnohr, P.; Tybjaerg-Hansen, A.; Benn, M.; Tybjærg-Hansen, A. Endogenous sex hormones and risk of venous thromboembolism in women and men. J. Thromb. Haemost. 2014, 12, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Lin, J.; Fu, L.; Mei, Y.-F.; Peng, G.; Tan, X.; Wang, D.-M.; Wang, W.; Li, Y.-G. Physiological testosterone stimulates tissue plasminogen activator and tissue factor pathway inhibitor and inhibits plasminogen activator inhibitor type 1 release in endothelial cells. This paper is one of a selection of papers in this Special Issue, entitled International Symposium on Recent Advances in Molecular, Clinical, and Social Medicine, and has undergone the Journal’s usual peer-review process. Biochem. Cell Biol. 2007, 85, 246–251. [Google Scholar] [CrossRef]
- Oskui, P.M.; French, W.J.; Herring, M.J.; Mayeda, G.S.; Burstein, S.; Kloner, R.A. Testosterone and the Cardiovascular System: A Comprehensive Review of the Clinical Literature. J. Am. Hear. Assoc. 2013, 2, e000272. [Google Scholar] [CrossRef] [PubMed]
- Vikan, T.; Schirmer, H.; Njølstad, I.; Svartberg, J. Low testosterone and sex hormone-binding globulin levels and high estradiol levels are independent predictors of type 2 diabetes in men. Eur. J. Endocrinol. 2010, 162, 747–754. [Google Scholar] [CrossRef][Green Version]
- Ohlsson, C.; Barrett-Connor, E.; Bhasin, S.; Orwoll, E.; Labrie, F.; Karlsson, M.; Ljunggren, O.; Vandenput, L.; Mellstrom, D.; Tivesten, A. High serum testosterone is associated with reduced risk of cardiovascular events in elderly men. The MrOS (Osteoporotic fractures in men) study in Sweden. J. Am. Coll. Cardiol. 2011, 58, 1674–1682. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.C.W.; von Eckardstein, A. Androgens and Coronary Artery Disease. Endocr. Rev. 2003, 24, 183–217. [Google Scholar] [CrossRef] [PubMed]
- Tsai, E.; Boyko, E.; Leonetti, D.; Fujimoto, W. Low serum testosterone level as a predictor of increased visceral fat in Japanese-American men. Int. J. Obes. 2000, 24, 485–491. [Google Scholar] [CrossRef]
- Bai, C.X.; Kurokawa, J.; Tamagawa, M.; Nakaya, H.; Furukawa, T. No transcriptional regulation of cardiac repolarization currrents by testosterone. Circulation 2005, 112, 1701–1710. [Google Scholar] [CrossRef]
- Giraldi, F.P.; Toja, P.M.; Filippini, B.; Michailidis, J.; Scacchi, M.; Badiale, M.S.; Cavagnini, F. Increased prevalence of prolonged QT interval in males with primary or secondary hypogonadism: A pilot study. Int. J. Androl. 2009, 33, e132–e138. [Google Scholar] [CrossRef]
- Charbit, B.; Christin-Maître, S.; Démolis, J.-L.; Soustre, E.; Young, J.; Funck-Brentano, C. Effects of Testosterone on Ventricular Repolarization in Hypogonadic Men. Am. J. Cardiol. 2009, 103, 887–890. [Google Scholar] [CrossRef]
- Salem, J.-E.; Waintraub, X.; Courtillot, C.; Shaffer, C.M.; Gandjbakhch, E.; Maupain, C.; Moslehi, J.J.; Badilini, F.; Haroche, J.; Gougis, P.; et al. Hypogonadism as a Reversible Cause of Torsades de Pointes in Men. Circulation 2018, 138, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Haring, R.; Völzke, H.; Steveling, A.; Krebs, A.; Felix, S.B.; Schöfl, C.; Dörr, M.; Nauck, M.; Wallaschofski, H. Low serum testosterone levels are associated with increased risk of mortality in a population-based cohort of men aged 20-79. Eur. Hear. J. 2010, 31, 1494–1501. [Google Scholar] [CrossRef] [PubMed]
- Menke, A.; Guallar, E.; Rohrmann, S.; Nelson, W.G.; Rifai, N.; Kanarek, N.; Feinleib, M.; Michos, E.D.; Dobs, A.; Platz, E.A. Sex Steroid Hormone Concentrations and Risk of Death in US Men. Am. J. Epidemiol. 2010, 171, 583–592. [Google Scholar] [CrossRef]
- Vikan, T.; Schirmer, H.; Njølstad, I.; Svartberg, J. Endogenous sex hormones and the prospective association with cardiovascular disease and mortality in men: The Tromsø Study. Eur. J. Endocrinol. 2009, 161, 435–442. [Google Scholar] [CrossRef]
- Shores, M.M.; Matsumoto, A.M.; Sloan, K.L.; Kivlahan, D.R. Low Serum Testosterone and Mortality in Male Veterans. Arch. Intern. Med. 2006, 166, 1660–1665. [Google Scholar] [CrossRef]
- Tivesten, A.; Vandenput, L.; Labrie, F.; Karlsson, M.K.; Ljunggren, Ö.; Mellström, D.; Ohlsson, C. Low Serum Testosterone and Estradiol Predict Mortality in Elderly Men. J. Clin. Endocrinol. Metab. 2009, 94, 2482–2488. [Google Scholar] [CrossRef]
- Khaw, K.-T.; Dowsett, M.; Folkerd, E.; Bingham, S.; Wareham, N.; Luben, R.; Welch, A.; Day, N. Endogenous Testosterone and Mortality Due to All Causes, Cardiovascular Disease, and Cancer in Men. Circulation 2007, 116, 2694–2701. [Google Scholar] [CrossRef]
- Keating, N.L.; Liu, M.P.-H.; O’Malley, A.J.; Freedland, S.J.; Smith, M.R. Androgen-deprivation Therapy and Diabetes Control Among Diabetic Men with Prostate Cancer. Eur. Urol. 2014, 65, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, X.; Huang, H.-Y.; Li, X.; Lin, J.-F. Effects of testosterone on insulin receptor substrate-1 and glucose transporter 4 expression in cells sensitive to insulin. Zhonghua Yi Xue Za Zhi 2006, 86, 1474–1477. [Google Scholar] [PubMed]
- Barber, M.; Nguyen, L.S.; Wassermann, J.; Spano, J.-P.; Funck-Brentano, C.; Salem, J.-E. Cardiac arrhythmia considerations of hormone cancer therapies. Cardiovasc. Res. 2019, 115, 878–894. [Google Scholar] [CrossRef] [PubMed]
- Gheorghe, A.C.D.; Ciobanu, A.; Hodorogea, A.S.; Radavoi, D.; Jinga, V.; Nanea, I.T.; Gheorghe, G.S. Evolution of Electrocardiographic Repolarization Parameters during Antiandrogen Therapy in Patients with Prostate Cancer and Hypogonadism. Cardiovasc. Toxicol. 2020, 20, 390–400. [Google Scholar] [CrossRef]
- Mason, M.D.; Lester, J.F. Cardiovascular effects of hormone therapy for prostate cancer. Drug Health Patient Saf. 2015, 7, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Ziaran, S.; Goncalves, F.M.; Breza, J., Sr. Patients with prostate cancer treated by ADT have significantly higher fibrinogenemia than healthy control. World J. Urol. 2013, 31, 289–292. [Google Scholar] [CrossRef] [PubMed]
- Fajemiroye, J.O.; Da Cunha, L.C.; Saavedra-Rodríguez, R.; Rodrigues, K.L.; Naves, L.M.; Mourão, A.A.; Da Silva, E.F.; Williams, N.E.E.; Martins, J.L.R.; Sousa, R.B.; et al. Aging-Induced Biological Changes and Cardiovascular Diseases. BioMed Res. Int. 2018, 2018, 1–14. [Google Scholar] [CrossRef]
- Mitcheson, J.S. hERG Potassium Channels and the Structural Basis of Drug-Induced Arrhythmias. Chem. Res. Toxicol. 2008, 21, 1005–1010. [Google Scholar] [CrossRef] [PubMed]
- Melong, N.; Steele, S.; Macdonald, M.; Holly, A.; Collins, C.C.; Zoubeidi, A.; Berman, J.N.; Dellaire, G. Enzalutamide inhibits testosterone-induced growth of human prostate cancer xenografts in zebrafish and can induce bradycardia. Sci. Rep. 2017, 7, 14698. [Google Scholar] [CrossRef]
- Koczor, C.A.; Ludlow, I.; Fields, E.; Jiao, Z.; Ludaway, T.; Russ, R.; Lewis, W. Mytochondrial polymerase gamma dysfunction and aging cause cardiac nuclear DNA methylation changes. Physiol. Genom. 2016, 48, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.W.; Stansfield, B.K. Genetic and Epigenetic Regulation of Aortic Aneurysms. BioMed Res. Int. 2017, 2017, 1–12. [Google Scholar] [CrossRef]
- Tabas, I.; Garcia-Cardena, G.; Owens, G.K. Recent insight into the cellular biology of atherosclerosis. Int. J. Cell Biol. 2015, 209, 13–22. [Google Scholar] [CrossRef]
- Shalev, I.; Entringer, S.; Wadhwa, P.D.; Wolkowitz, O.M.; Puterman, E.; Lin, J.; Epel, E.S. Stress and telomere biology: A lifespan perspective. Psychoneuroendocrinology 2013, 38, 1835–1842. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, A.V.; Chen, M.-H.; ARenshaw, A.; Loffredo, M.; Kantoff, P. Long-Term Follow-Up of a Randomized Trial of Radiation with or without Androgen Deprivation Therapy for Localized Prostate Cancer. JAMA 2015, 314, 1291–1293. [Google Scholar] [CrossRef] [PubMed]
- Morgans, A.K.; Fan, K.H.; Koyama, T.; Albertsen, P.C.; Goodman, M.; Hamilton, A.S.; Hoffman, R.M.; Stanford, J.L.; Stroup, A.M.; Resnick, M.J.; et al. Influence of age on incident diabetes and cardiovascular disease in prostate cancer survivors receiving androgen deprivation therapy. J. Urol. 2015, 193, 1226–1231. [Google Scholar] [CrossRef]
- Yamazaki, H.; Masui, K.; Suzuki, G.; Nakamura, S.; Aibe, N.; Shimizu, D.; Yamada, K.; Okihara, K.; Shiraishi, T.; Kotsuma, T.; et al. Effect of Androgen Deprivation Therapy on Other-Cause of Mortality in Elderly Patients with Clinically Localized Prostate Cancer Treated with Modern Radiotherapy: Is There a Negative Impact? J. Clin. Med. 2019, 8, 338. [Google Scholar] [CrossRef]
- O’Farrell, S.; Garmo, H.; Holmberg, L.; Adolfsson, J.; Stattin, P.; Van Hemelrijck, M. Risk and Timing of Cardiovascular Disease after Androgen-Deprivation Therapy in Men with Prostate Cancer. J. Clin. Oncol. 2015, 33, 1243–1251. [Google Scholar] [CrossRef]
- Keating, N.L.; O’Malley, A.J.; Smith, M.R. Diabetes and Cardiovascular Disease during Androgen Deprivation Therapy for Prostate Cancer. J. Clin. Oncol. 2006, 24, 4448–4456. [Google Scholar] [CrossRef]
- D’Amico, A.V.; Denham, J.W.; Crook, J.; Chen, M.-H.; Goldhaber, S.Z.; Lamb, D.S.; Joseph, D.; Tai, K.-H.; Malone, S.; Ludgate, C.; et al. Influence of Androgen Suppression Therapy for Prostate Cancer on the Frequency and Timing of Fatal Myocardial Infarctions. J. Clin. Oncol. 2007, 25, 2420–2425. [Google Scholar] [CrossRef]
- Tsai, H.K.; D’Amico, A.V.; Sadetsky, N.; Chen, M.-H.; Carroll, P.R. Androgen Deprivation Therapy for Localized Prostate Cancer and the Risk of Cardiovascular Mortality. J. Natl. Cancer Inst. 2007, 99, 1516–1524. [Google Scholar] [CrossRef]
- Alibhai, S.M.; Duong-Hua, M.; Sutradhar, R.; Fleshner, N.E.; Warde, P.; Cheung, A.M.; Paszat, L.F. Impact of Androgen Deprivation Therapy on Cardiovascular Disease and Diabetes. J. Clin. Oncol. 2009, 27, 3452–3458. [Google Scholar] [CrossRef]
- Nanda, A.; Chen, M.-H.; Braccioforte, M.H.; Moran, B.J.; D’Amico, A.V. Hormonal Therapy Use for Prostate Cancer and Mortality in Men with Coronary Artery Disease–Induced Congestive Heart Failure or Myocardial Infarction. JAMA 2009, 302, 866–873. [Google Scholar] [CrossRef] [PubMed]
- Bolla, M.; de Reijke, T.M.; Van Tienhoven, G.; Van den Bergh, A.; Oddens, J.; Poortmans, P.M.P.; Gez, E.; Kil, P.; Akdas, A.; Soete, G.; et al. For the EORTC Radiation Oncology Group and Genito-Urinary Tract Cancer Group Duration of Androgen Suppression in the Treatment of Prostate Cancer. N. Engl. J. Med. 2009, 360, 2516–2527. [Google Scholar] [CrossRef] [PubMed]
- Punnen, S.; Cooperberg, M.R.; Sadetsky, N.; Carroll, P.R. Androgen Deprivation Therapy and Cardiovascular Risk. J. Clin. Oncol. 2011, 29, 3510–3516. [Google Scholar] [CrossRef] [PubMed]
- Efstathiou, J.A.; Bae, K.; Shipley, W.U.; Hanks, G.E.; Pilepich, M.V.; Sandler, H.M.; Smith, M.R. Cardiovascular Mortality and Duration of Androgen Deprivation for Locally Advanced Prostate Cancer: Analysis of RTOG 92-02. Eur. Urol. 2008, 54, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Zhu, J.; Chen, L.; Xu, Y.; Yang, Y.; Hu, R.; Zhang, W.; Song, Y.; Lu, Y.; Ou, N.; et al. Is androgen deprivation therapy for prostate cancer associated with cardiovascular disease? A meta-analysis and systematic review. Andrology 2019, 8, 559–574. [Google Scholar] [CrossRef]
- Shore, N.D.; Saad, F.; Cookson, M.S.; George, D.J.; Saltzstein, D.R.; Tutrone, R.; Akaza, H.; Bossi, A.; van Veenhuyzen, D.F.; Selby, B.; et al. For the HERO Study Investigators Oral relugolix for androgen-deprivation therapy in advanced prostate cancer. N. Engl. J. Med. 2020, 382, 2187–2196. [Google Scholar] [CrossRef] [PubMed]
- Moreira, R.B.; Debiasi, M.; Francini, E.; Nuzzo, P.V.; De Velasco, G.; Maluf, F.C.; Fay, A.P.; Bellmunt, J.; Choueiri, T.K.; Schutz, F.A. Differential side effects profile in patients with mCRPC treated with abiraterone or enzalutamide: A meta-analysis of randomized controlled trials. Oncotarget 2017, 8, 84572–84578. [Google Scholar] [CrossRef]
- Margel, D.; Peer, A.; Ber, Y.; Shavit-Grievink, L.; Tabachnik, T.; Sela, S.; Witberg, G.; Baniel, J.; Kedar, D.; Duivenvoorden, W.C.M.; et al. Cardiovascular Morbidity in a Randomized Trial Comparing GnRH Agonist and GnRH Antagonist among Patients with Advanced Prostate Cancer and Preexisting Cardiovascular Disease. J. Urol. 2019, 202, 1199–1208. [Google Scholar] [CrossRef]
- Zhang, K.-W.; Reimers, M.A.; Calaway, A.C.; Fradley, M.G.; Ponsky, L.; Garcia, J.A.; Cullen, J.; Baumann, B.C.; Addison, D.; Campbell, C.M.; et al. Cardiovascular Events in Men with Prostate Cancer Receiving Hormone Therapy: An Analysis of the FDA Adverse Event Reporting System (FAERS). J. Urol. 2021, 206, 613–622. [Google Scholar] [CrossRef]
- Levine, G.N.; D’Amico, A.V.; Berger, P.; Clark, P.E.; Eckel, R.H.; Keating, N.L. Androgen-Deprivation Therapy in Prostate Cancer and Cardiovascular Risk a Science Advisory from the American Heart Association, American Cancer Society, and American Urological Association. Circulation 2010, 121, 833–840. [Google Scholar] [CrossRef]
- Gheorghe, A.C.D.; Ciobanu, A.; Hodorogea, A.S.; Radavoi, G.D.; Jinga, V.; Rascu, A.S.C.; Nanea, I.T.; Gheorghe, G.S. Subclinical left ventricular dysfunction in men under androgen deprivation therapy for prostate cancer, revealed by speckle-tracking-derived parameters, repolarization, and myocardial injury markers. Echocardiography 2021, 38, 632–640. [Google Scholar] [CrossRef]
- Iacovelli, R.; Ciccarese, C.; Bria, E.; Romano, M.; Fantinel, E.; Bimbatti, D.; Muraglia, A.; Porcaro, A.B.; Siracusano, S.; Brunelli, M.; et al. The Cardiovascular Toxicity of Abiraterone and Enzalutamide in Prostate Cancer. Clin. Genitourin. Cancer 2018, 16, e645–e653. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.L.; Chen, M.-H.; Beckman, J.; Beard, C.J.; Martin, N.E.; Choueiri, T.K.; Hu, J.C.; Hoffman, K.E.; Dosoretz, D.E.; Moran, B.J.; et al. Influence of Androgen Deprivation Therapy on All-Cause Mortality in Men With High-Risk Prostate Cancer and a History of Congestive Heart Failure or Myocardial Infarction. Int. J. Radiat. Oncol. 2012, 82, 1411–1416. [Google Scholar] [CrossRef]
- Bhatia, N.; Santos, M.; Jones, L.W.; Beckman, J.A.; Penson, D.F.; Morgans, A.K.; Moslehi, J. Cardiovascular Effects of Androgen Deprivation Therapy for the Treatment of Prostate Cancer ABCDE Steps to Reduce Cardiovascular Disease in Patients with Prostate Cancer. Circulation 2016, 133, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Harshman, L.C.; Wang, X.; Nakabayashi, M.; Xie, W.; Valenca, L.B.; Werner, L.; Yu, Y.; Kantoff, A.M.; Sweeney, C.J.; Mucci, L.A.; et al. Statin Use at the Time of Initiation of Androgen Deprivation Therapy and Time to Progression in Patients with Hormone-Sensitive Prostate Cancer. JAMA Oncol. 2015, 1, 495–504. [Google Scholar] [CrossRef]
- Culos-Reed, S.N.; Robinson, J.L.; Lau, H.; O’Connor, K.; Keats, M.R. Benefits of a Physical Activity Intervention for Men with Prostate Cancer. J. Sport Exerc. Psychol. 2007, 29, 118–127. [Google Scholar] [CrossRef] [PubMed]
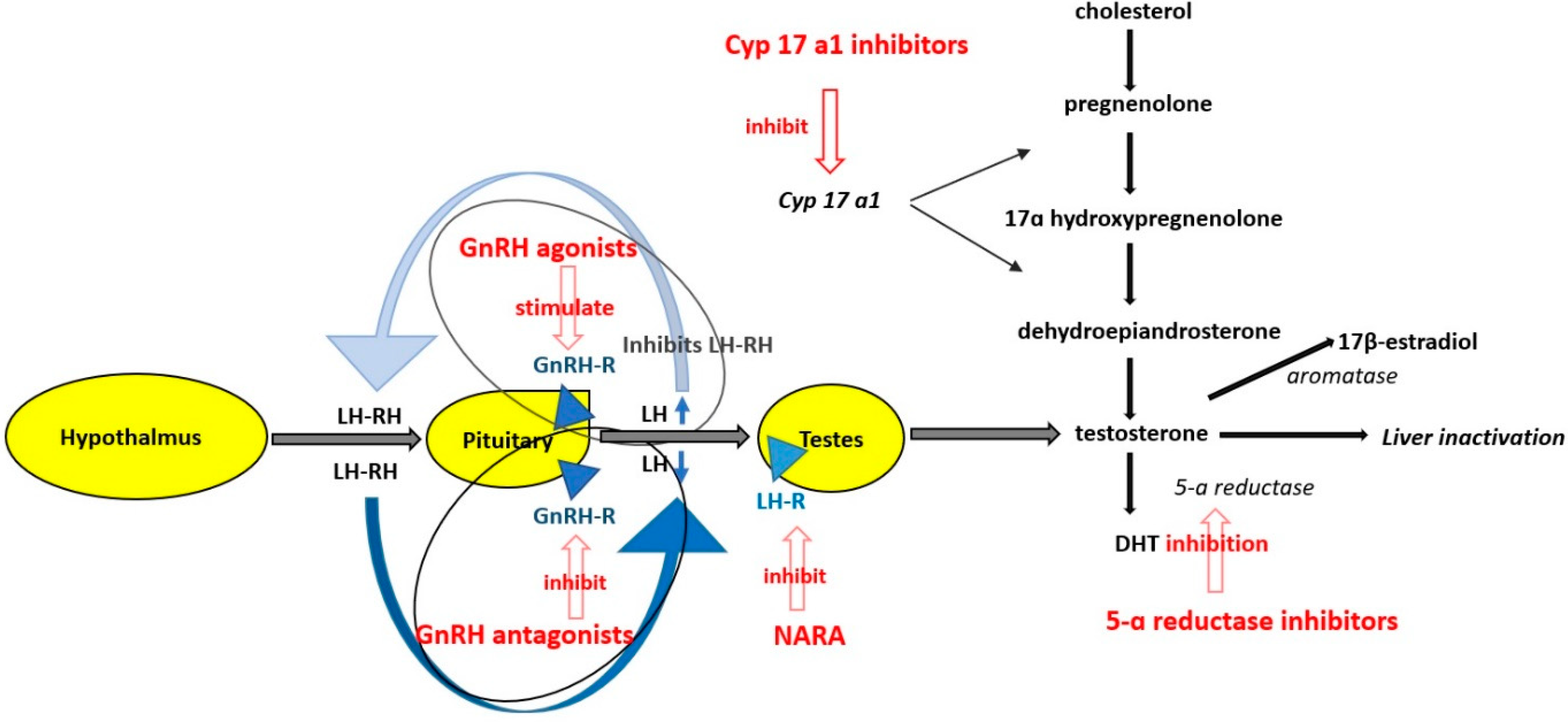
| Class of Drugs | Examples |
|---|---|
| GnRH agonist a | Leuprolide, Buserelin, Goserelin, Triptorelin |
| GnRH antagonist b | Degarelix, Relugorix |
| Cytochrome P450-17 A1 inhibitor | Abiraterone |
| Nonsteroidal androgen receptor antagonists | Bicalutamide, Flutamide, Nilutamide, Enzalutamide, Apalutamide, Darolutamide |
| 5α-reductase inhibitors | Finasteride, Dutasteride |
| Population | Adult Men with Prostate Cancer |
|---|---|
| Intervention | Androgen deprivation therapy |
| Comparisons | Placebo, no therapy, radiotherapy, orchiectomy, watchful waiting/active surveillance, local therapy |
| Outcomes | Hypertension, myocardial infarction, stroke, arrhythmia, heart failure, venous thromboembolism, QT interval prolongation, sudden cardiac death, cardiovascular mortality |
| First Author [Reference] | Year | Study Type | Pts Enrolled | Type of ADT/No pts | Treatment in the Control Group/No pts | Median Age | Follow-Up | Results |
|---|---|---|---|---|---|---|---|---|
| Studies Showing an Additional Cardio-Vascular Risk in Patients on ADT | ||||||||
| Keating [53] | 2006 | Observational SEER program 11 population-based cancer registries | 73,193 | GnRH agonists 25,570 | No therapy/orchiectomy 47,623 | 74.2 ± 5.8 | 4.5 years | Increased risk of DM, CAD, MI, SCD, in the GnRH agonist group versus non therapy group Increased risk of DM in orchiectomy group versus non therapy group |
| D’Amico [54] | 2007 | Randomized study | 1372 | GnRH agonist + flutamide + RT | RT | 70.88 | 3–8 months | Fatal MI occurs earlier in men older than 65 years who received 6 months of ADT than in those without ADT and in men younger than 65 years. No significant difference (p = 0.97) was observed in the time to fatal MIs in men age 65 years or older who received 6 to 8 months of ADT compared with 3 months of ADT |
| Tsai [55] | 2007 | Observational study | 4881 | 3262 pts on radical prostatectomy + ADT in 1051 pts | RT 1630 pts | 63 | 3.8 years | The use of ADT appears to be associated with an increased risk of death from cardiovascular causes in patients undergoing radical prostatectomy for localized prostate cancer |
| O’Farrell [52] | 2015 | Retrospective study | 229,147 | 41,362 pts on GnRH agonist ± non steroidian antiandrogen | 187,785 cancer free pts | 75 | 6 years | CVD risk was highest during the first 6 months of ADT in men who experienced two or more cardiovascular events before therapy |
| Alibhai [56] | 2009 | Cohort study | 116769 | 46,995 on ADT | No ADT 69,774 | 75 ± 6.3 | 6.47 years | ADT use for at least 6 months in older men is associated with an increased risk of diabetes and fragility fracture but not MI or sudden cardiac death. |
| Studies Showing No Additional Cardio-Vascular Risk in Patients on ADT | ||||||||
| Nanda [57] | 2009 | Retrospective study | 5077 | 1521 pts on GnRH agonist+ non steroidian antiandrogen | 3556 RT | 69.5 | 2 years | ADT is significantly associated with an increased risk of all-cause mortality among men with a history of CAD-induced HF or MI but not among men with no comorbidity or a single CAD risk factor. |
| Bolla [58] | 2009 | Randomized study | 970 | 487 pts on RT + 2.5 years GnRH agonists | 483 pts on RT + 6 months GnRH agonists | 69 | 6.4 | There was no significant difference in the cumulative incidence of fatal cardiac events at 5 years: 4.0% in the short-term group and 3.0% in the long-term group |
| Punnen [59] | 2011 | Retrospective study of CaPSURE registry | 7248 | ADT (1086 pts) ADT + local therapy (485 pts) | Local therapy (5170 pts), watchful waiting/active surveillance (506 pts) | >65 | 47.6–57 months | A propensity-matching algorithm in a subset of 1391 patients was unable to find a significant difference in cardiovascular mortality between those who did or did not receive ADT. |
| Efstathiou [60] | 1987–1992 | randomized | 945 | RT + goserelin (477) | RT (468) | 70 | 9 years | GnRH agonists do not seem to increase cardiovascular mortality in men with locally advanced prostate cancer |
| Studies That Compared Different Type of ADT Regarding Their Cardiotoxicity | ||||||||
| Liang [61] | 2009–2017 | Meta analysis | 5 studies | ADT 63,258 | Non ADT 209,403 | 69–75 | Abiraterone and enzalutamide increased risk of AMI, CAD, in contrast, this association is not detected in SCD. | |
| Shore [62] | 2017–2018 | Randomized phase III trial (HERO) | 930 pts | 622 pts relugolix | 308 pts leuprolide | 71 | 1 year | Relugolix achieved rapid, sustained suppression of testosterone levels that was superior to that with leuprolide, with a 54% lower risk of major adverse cardiovascular events |
| Moreira [63] | 2011–2014 | Meta-analysis | 4 studies/5183 pts | Abiraterone-prednisone/prednisone 1333/936 | Enzalutamid/placebo 1672/1244 | 46–95 | 5 years | Abiraterone was associated with cardiovascular adverse effects; enzalutamide was associated with fatigue |
| Margel [64] | 2019 | Phase II randomized study included patients with pre-existing cardio-vascular disease; primary endpoint endothelial dysfunction | 80 pts | GnRH antagonist/41 pts | Gn agonist/39 pts | 71–72 | 1 year | 20% randomized to GnRH agonist experienced a major cardiovascular and cerebrovascular event compared to 3% of those on GnRH antagonist |
| Zhang [65] | 2000–2020 | Analysis of FDA Adverse Event Reporting System (FAERS} | 6231 hormone monotherapy | 1793 combined therapy | ≥18 years | 20 years | GnRH antagonists were associated with fewer cardiovascular adverse events than GnRH agonists as monotherapy and combination therapy, especially in men ≥ 60 years | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gheorghe, G.S.; Hodorogea, A.S.; Ciobanu, A.; Nanea, I.T.; Gheorghe, A.C.D. Androgen Deprivation Therapy, Hypogonadism and Cardiovascular Toxicity in Men with Advanced Prostate Cancer. Curr. Oncol. 2021, 28, 3331-3346. https://doi.org/10.3390/curroncol28050289
Gheorghe GS, Hodorogea AS, Ciobanu A, Nanea IT, Gheorghe ACD. Androgen Deprivation Therapy, Hypogonadism and Cardiovascular Toxicity in Men with Advanced Prostate Cancer. Current Oncology. 2021; 28(5):3331-3346. https://doi.org/10.3390/curroncol28050289
Chicago/Turabian StyleGheorghe, Gabriela Silvia, Andreea Simona Hodorogea, Ana Ciobanu, Ioan Tiberiu Nanea, and Andrei Cristian Dan Gheorghe. 2021. "Androgen Deprivation Therapy, Hypogonadism and Cardiovascular Toxicity in Men with Advanced Prostate Cancer" Current Oncology 28, no. 5: 3331-3346. https://doi.org/10.3390/curroncol28050289
APA StyleGheorghe, G. S., Hodorogea, A. S., Ciobanu, A., Nanea, I. T., & Gheorghe, A. C. D. (2021). Androgen Deprivation Therapy, Hypogonadism and Cardiovascular Toxicity in Men with Advanced Prostate Cancer. Current Oncology, 28(5), 3331-3346. https://doi.org/10.3390/curroncol28050289




