A Case of Pathological Complete Response and Resolution of Dermatomyositis Following Neoadjuvant Chemotherapy in HER2-Positive Early Breast Cancer
Abstract
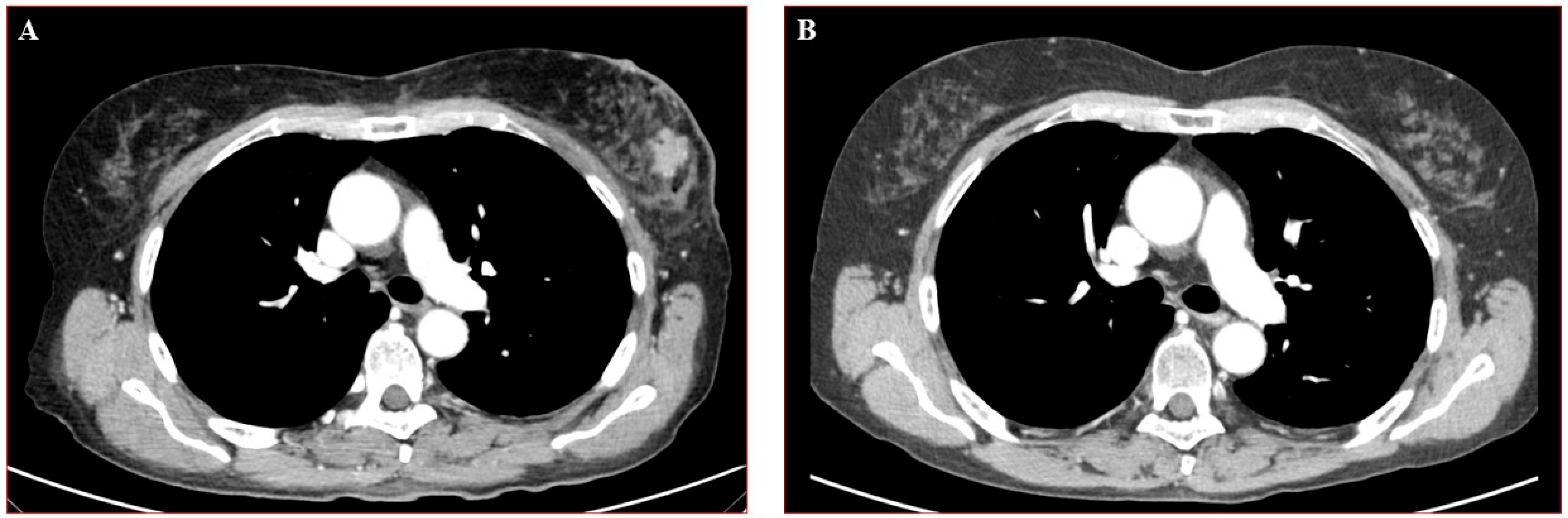
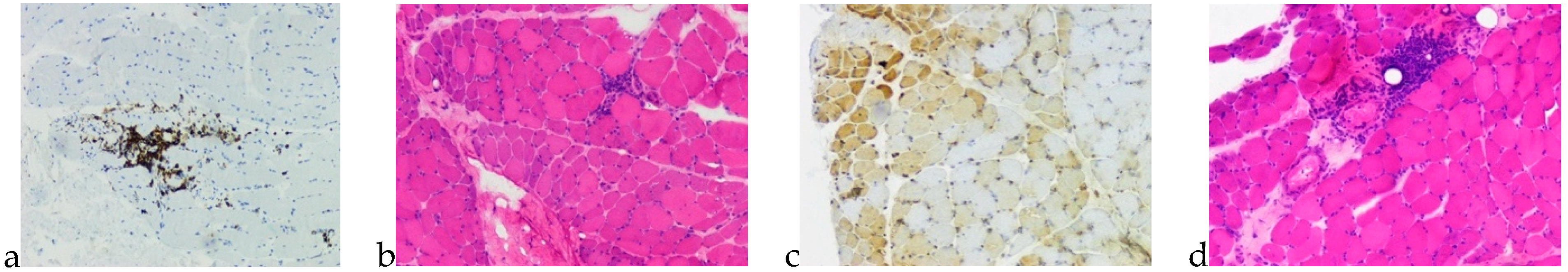
1. Case Presentation
2. Discussion
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dias, L.P.N.; Faria, A.L.A.; Scandiuzzi, M.M.; Inhaia, C.L.d.S.; Shida, J.Y.; Gebrim, L.H. A rare case of severe myositis as paraneoplastic syndrome on breast cancer. World J. Surg. Oncol. 2015. [Google Scholar] [CrossRef]
- Ahuja, S.; Makkar, P.; Gupta, S.; Vigoda, I. Paraneoplastic syndrome and underlying breast cancer: A worsening rash despite initiation of chemotherapy. J. Community Support. Oncol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Trallero-Araguás, E.; Rodrigo-Pendás, J.Á.; Selva-O’Callaghan, A.; Martínez-Gõmez, X.; Bosch, X.; Labrador-Horrillo, M.; Grau-Junyent, J.M.; Vilardell-Tarrés, M. Usefulness of anti-p155 autoantibody for diagnosing cancer-associated dermatomyositis: A systematic review and meta-analysis. Arthritis Rheum. 2012. [Google Scholar] [CrossRef] [PubMed]
- Kubeček, O.; Soukup, T.; Paulík, A.; Kopecký, J. Dermatomyositis with anti-TIF-1γ antibodies as a presenting symptom of underlying triple-negative breast cancer: A case report. BMC Cancer 2016. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, N.; Yamaguchi, Y.; Kanaoka, M.; Ototake, Y.; Akita, A.; Watanabe, T.; Aihara, M. Clinical significance of serum levels of anti-transcriptional intermediary factor 1-γ antibody in patients with dermatomyositis. J. Dermatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Luu, X.; Leonard, S.; Joseph, K.-A. Dermatomyositis presenting as a paraneoplastic syndrome with resolution of symptoms following surgical management of underlying breast malignancy. J. Surg. Case Rep. 2015. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.L.; Zhang, Y.; Sigurgeirsson, B.; Pukkala, E.; Mellemkjaer, L.; Airio, A.; Evans, S.R.; Felson, D.T. Frequency of specific cancer types in dermatomyositis and polymyositis: A population-based study. Lancet 2001. [Google Scholar] [CrossRef]
- Hendren, E.; Vinik, O.; Faragalla, H.; Haq, R. Breast cancer and dermatomyositis: A case study and literature review. Curr. Oncol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Osako, T.; Ito, Y.; Morimatsu, A.; Jinnin, M.; Tada, K.; Sakurai, N.; Takahashi, S.; Akiyama, F.; Sakamoto, G.; Iwase, T.; et al. Flare-up of dermatomyositis along with recurrence of breast cancer. Breast J. 2007. [Google Scholar] [CrossRef]
- Cardoso, F.; Kyriakides, S.; Ohno, S.; Penault-Llorca, F.; Poortmans, P.; Rubio, I.T.; Zackrisson, S.; Senkus, E. Early breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019. [Google Scholar] [CrossRef]
- Lamquami, S.; Errarhay, S.; Mamouni, N.; Bouchikhi, C.; Banani, A. Dermatomyositis revealing breast cancer: Report of a case. Pan Afr. Med. J. 2015. [Google Scholar] [CrossRef]
- Khoo, H.Y.; Tan, W.J.; Cheong, Y.T. Breast cancer with dermatomyositis as initial presentation. Med. J. Malays. 2018, 73, 44–45. [Google Scholar]
- Otsuka, Y.; Watanabe, H.; Kano, Y.; Tatebe, N.; Sunahori-Watanabe, K.; Kawabata, T.; Sada, K.E.; Wada, J. Occurrence of dermatomyositis immediately after mastectomy subsequent to severe chemotherapeutic drug eruption. Intern. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, N.P.; Zakaria, S.; Degnim, A.C.; Boughey, J.C. Dermatomyositis presenting as a paraneoplastic syndrome due to underlying breast cancer. BMJ Case Rep. 2011. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.J.; Wu, Y.F.; Fan, T. Dermatosis as the initial manifestation of malignant breast tumors: Retrospective analysis of 4 cases. Breast Care 2010. [Google Scholar] [CrossRef] [PubMed]
- Pectasides, D.; Koumpou, M.; Gaglia, A.; Pectasides, M.; Lambadiari, V.; Lianos, E.; Papaxoinis, G.; Economopoulos, T. Dermatomyositis associated with breast cancer. Anticancer Res. 2006, 26, 2329–2332. [Google Scholar]
- Inaguma, G.; Shimada, A.; Tsunoda, J.; Matsuzaki, T.; Nishi, T.; Seki, H.; Matsumoto, H. Inflammatory breast cancer associated with amyopathic dermatomyositis: A case report. Surg. Case Rep. 2020. [Google Scholar] [CrossRef] [PubMed]
- Allouch, A.; Zaatarikahale, T.B.; Moussa, M.K.; Jounblat, Y.; Bitar, N. Dermatomyositis: A Presenting Clinical Vignette in a Patient with Breast Cancer. Cureus 2020. [Google Scholar] [CrossRef]
- Pellegrino, B.; Mazzaschi, G.; Madeddu, D.; Mori, C.; Lagrasta, C.A.M.; Missale, G.; Quaini, F.; Musolino, A. Clinico-Immunological Profile of a 67-Year-Old Woman Affected by HER2-Positive Breast Cancer and Autoimmune Dermatomyositis. Front. Oncol. 2020. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piras, M.; Panebianco, M.; Garibaldi, M.; Roberto, M.; Merlonghi, G.; Pellegrini, P.; Marchetti, P. A Case of Pathological Complete Response and Resolution of Dermatomyositis Following Neoadjuvant Chemotherapy in HER2-Positive Early Breast Cancer. Curr. Oncol. 2021, 28, 1957-1961. https://doi.org/10.3390/curroncol28030182
Piras M, Panebianco M, Garibaldi M, Roberto M, Merlonghi G, Pellegrini P, Marchetti P. A Case of Pathological Complete Response and Resolution of Dermatomyositis Following Neoadjuvant Chemotherapy in HER2-Positive Early Breast Cancer. Current Oncology. 2021; 28(3):1957-1961. https://doi.org/10.3390/curroncol28030182
Chicago/Turabian StylePiras, Marta, Martina Panebianco, Matteo Garibaldi, Michela Roberto, Gioia Merlonghi, Patrizia Pellegrini, and Paolo Marchetti. 2021. "A Case of Pathological Complete Response and Resolution of Dermatomyositis Following Neoadjuvant Chemotherapy in HER2-Positive Early Breast Cancer" Current Oncology 28, no. 3: 1957-1961. https://doi.org/10.3390/curroncol28030182
APA StylePiras, M., Panebianco, M., Garibaldi, M., Roberto, M., Merlonghi, G., Pellegrini, P., & Marchetti, P. (2021). A Case of Pathological Complete Response and Resolution of Dermatomyositis Following Neoadjuvant Chemotherapy in HER2-Positive Early Breast Cancer. Current Oncology, 28(3), 1957-1961. https://doi.org/10.3390/curroncol28030182





