Significance of Concurrent Chemoradiotherapy as Primary Treatment in Patients with Metastatic Cervical Cancer
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- International Agency for Research on Cancer GLOBOCAN 2018. Available online: https://www.uicc.org/news/global-cancer-data-globocan-2018 (accessed on 5 December 2020).
- Vaccarella, S.; Lortet-Tieulent, J.; Plummer, M.; Franceschi, S.; Bray, F. Worldwide trends in cervical cancer incidence: Impact of screening against changes in disease risk factors. Eur. J. Cancer 2013, 49, 3262–3273. [Google Scholar] [CrossRef] [PubMed]
- Drolet, M.; Benard, E.; Perez, N.; Brisson, M. Group HPVVIS Population-level impact and herd effects following the introduction of human papillomavirus vaccination programmes: Updated systematic review and meta-analysis. Lancet 2019, 394, 497–509. [Google Scholar] [CrossRef]
- Utada, M.; Chernyayskiy, P.; Lee, W.J.; Franceschi, S.; Sauvaget, C.; De Gonzalez, A.B.; Withrow, D.R. Increasing risk of uterine cervical cancer among young Japanese women: Comparison of incidence trends in Japan, South Korea and Japanese-Americans between 1985 and 2012. Int. J. Cancer 2019, 144, 2144–2152. [Google Scholar] [CrossRef] [PubMed]
- Koh, W.J.; Abu-Rustum, N.R.; Bean, S.; Bradley, K.; Campos, S.M.; Cho, K.R.; Chon, H.S.; Chu, C.; Clark, R.; Cohn, D.; et al. Cervical Cancer, Version 3.2019, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2019, 17, 64–84. [Google Scholar] [CrossRef] [PubMed]
- Khoury-Collado, F.; Bowes, R.J.; Jhamb, N.; Aghajanian, C.; Abu-Rustum, N.R. Unexpected long-term survival without evidence of disease after salvage chemotherapy for recurrent metastatic cervical cancer: A case series. Gynecol. Oncol. 2007, 105, 823–825. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Kim, T.; Lee, E.S.; Kim, H.J.; Chung, H.H.; Kim, J.W.; Song, Y.S.; Park, N.H. Impact of Chemoradiation on Prognosis in Stage IVB Cervical Cancer with Distant Lymphatic Metastasis. Cancer Res. Treat. 2013, 45, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Berek, J.S.; Matsuo, K.; Grubbs, B.H.; Gaffney, D.K.; Lee, S.I.; Kilcoyne, A.; Cheon, G.J.; Yoo, C.W.; Li, L.; Shao, Y.; et al. Multidisciplinary perspectives on newly revised 2018 FIGO staging of cancer of the cervix uteri. J. Gynecol. Oncol. 2019, 30, e40. [Google Scholar] [CrossRef]
- Stehman, F.B.; Bundy, B.N.; Disaia, P.J.; Keys, H.M.; Larson, J.E.; Fowler, W.C. Carcinoma of the Cervix Treated with Radiation Therapy-I—A Multi-Variate Analysis of Prognostic Variables in the Gynecologic Oncology Group. Cancer 1991, 67, 2776–2785. [Google Scholar] [CrossRef]
- Rose, P.G.; Java, J.; Whitney, C.W.; Stehman, F.B.; Lanciano, R.; Thomas, G.M.; DiSilvestro, P.A. Nomograms Predicting Progression-Free Survival, Overall Survival, and Pelvic Recurrence in Locally Advanced Cervical Cancer Developed From an Analysis of Identifiable Prognostic Factors in Patients From NRG Oncology/Gynecologic Oncology Group Randomized Trials of Chemoradiotherapy. J. Clin. Oncol. 2015, 33, 2136–2142. [Google Scholar]
- Kim, K.; Cho, S.Y.; Kim, B.J.; Kim, M.H.; Choi, S.C.; Ryu, S.Y. The type of metastasis is a prognostic factor in disseminated cervical cancer. J. Gynecol. Oncol. 2010, 21, 186–190. [Google Scholar] [CrossRef]
- Hwang, J.H.; Lim, M.C.; Seo, S.S.; Kang, S.; Park, S.Y.; Kim, J.Y. Outcomes and toxicities for the treatment of stage IVB cervical cancer. Arch. Gynecol. Obstet. 2012, 285, 1685–1693. [Google Scholar] [CrossRef] [PubMed]
- Horn, L.C.; Fischer, U.; Raptis, G.; Bilek, K.; Hentschel, B. Tumor size is of prognostic value in surgically treated FIGO stage II cervical cancer. Gynecol. Oncol. 2007, 107, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.H.; Tsai, C.S.; Lai, C.H.; Chang, T.C.; Wang, C.C.; Chou, H.H.; Lee, S.P.; Lee, C.C.; Tang, S.G.; Hsueh, S. Risk stratification of patients with advanced squamous cell carcinoma of cervix treated by radiotherapy alone. Int. J.Radiat. Oncol. Biol. Phys. 2005, 63, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Takashima, A.; Kasamatsu, T.; Nakamura, K.; Mizusawa, J.; Nakanishi, T.; Takeshima, N.; Kamiura, S.; Onda, T.; Sumi, T.; et al. Clinical tumor diameter and prognosis of patients with FIGO stage IB1 cervical cancer (JCOG0806-A). Gynecol. Oncol. 2015, 137, 34–39. [Google Scholar] [CrossRef]
- Wagner, A.E.; Pappas, L.; Ghia, A.J.; Gaffney, D.K. Impact of tumor size on survival in cancer of the cervix and validation of stage IIA1 and IIA2 subdivisions. Gynecol. Oncol. 2013, 129, 517–521. [Google Scholar] [CrossRef]
- Bhatla, N.; Aoki, D.; Sharma, D.N.; Sankaranarayanan, R. Cancer of the cervix uteri. Int. J. Gynaecol. Obstet. 2018, 143, 22–36. [Google Scholar] [CrossRef]
- Nishio, S.; Katsumata, N.; Matsumoto, K.; Tanabe, H.; Yonemori, K.; Kohno, T.; Shimizu, C.; Ando, M.; Kasamatsu, T.; Fujiwara, Y. Analysis of the clinicopathological prognosis of stage IVb cervical carcinoma. Oncol. Rep. 2008, 19, 497–503. [Google Scholar] [CrossRef]
- Sasano, T.; Mabuchi, S.; Kuroda, H.; Takahashi, R.; Kozasa, K.; Isohashi, F.; Yoshioka, Y.; Ogawa, K.; Kimura, T. Predictors of Survival in Patients With FIGO Stage IVB Cervical Cancer. Int. J. Gynecol. Cancer 2016, 26, 528–533. [Google Scholar] [CrossRef]
- Im, J.H.; Yoon, H.I.; Kim, S.; Nam, E.J.; Kim, S.W.; Yim, G.W.; Keum, K.C.; Kim, Y.T.; Kim, G.E.; Kim, Y.B. Tailored radiotherapeutic strategies for disseminated uterine cervical cancer patients. Radiat. Oncol. 2015, 10, 77. [Google Scholar] [CrossRef]
- Ferrandina, G.; Palluzzi, E.; Gallotta, V.; Gambacorta, M.A.; Autorino, R.; Turco, L.C.; Macchia, G.; Cosentino, F.; Gui, B.; Mattoli, M.V.; et al. Neo-adjuvant platinum-based chemotherapy followed by chemoradiation and radical surgery in locally advanced cervical cancer (Lacc) patients: A phase II study. Eur. J. Surg. Oncol. 2018, 44, 1062–1068. [Google Scholar] [CrossRef]
- Carey, M.S.; Bacon, M.; Tu, D.; Butler, L.; Bezjak, A.; Stuart, G.C. The prognostic effects of performance status and quality of life scores on progression-free survival and overall survival in advanced ovarian cancer. Gynecol. Oncol. 2008, 108, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Kemeny, N.; Braun, D.W. Prognostic Factors in Advanced Colorectal-Carcinoma. Importance of Lactic-Dehydrogenase Level, Performance Status, and White Blood-Cell Count. Am. J. Med. 1983, 74, 786–794. [Google Scholar] [CrossRef]
- Reck, M.; Thatcher, N.; Smit, E.F.; Lorigan, P.; Szutowicz-Zielinska, E.; Liepa, A.M.; Winfree, K.B.; Peterson, P.; Guba, S.C.; Socinski, M.A. Baseline quality of life and performance status as prognostic factors in patients with extensive-stage disease small cell lung cancer treated with pemetrexed plus carboplatin vs. etoposide plus carboplatin. Lung Cancer 2012, 78, 276–281. [Google Scholar] [CrossRef] [PubMed]
| Variable | All Cases (n = 73) | IIIC2 Group (n = 28) | IVB Group (n = 45) | ||||
|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | p-Value | |
| Age, years | |||||||
| mean ± SD | 55.6 ± 12.6 | 53.3 ± 11.8 | 57.0 ± 13.1 | 0.2363 | |||
| ≤55 | 34 | 46.6 | 16 | 57.1 | 18 | 40.0 | 0.1529 |
| >55 | 39 | 53.4 | 12 | 42.9 | 27 | 60.0 | |
| Preoperative BMI | |||||||
| mean ± SD | 20.8 ± 3.6 | 21.3 ± 3.8 | 20.5 ± 3.5 | 0.3929 | |||
| ≤22 | 50 | 68.5 | 20 | 71.4 | 30 | 66.7 | 0.6691 |
| >22 | 23 | 31.5 | 8 | 28.6 | 15 | 33.3 | |
| Histology | |||||||
| SCC | 49 | 67.1 | 19 | 67.8 | 30 | 66.6 | 0.5124 |
| AC | 19 | 26.0 | 6 | 21.4 | 13 | 28.8 | |
| Others | 5 | 6.8 | 3 | 10.7 | 2 | 4.4 | |
| TNM classification | |||||||
| cT | |||||||
| 1 | 7 | 9.5 | 1 | 3.6 | 6 | 13.3 | 0.0021 |
| 2 | 32 | 43.8 | 20 | 71.4 | 12 | 26.7 | |
| 3 | 22 | 30.1 | 6 | 21.4 | 16 | 35.6 | |
| 4 | 11 | 15.0 | 1 | 3.6 | 9 | 20.0 | |
| NA | 1 | 1.3 | 0 | 0.0 | 2 | 4.4 | |
| N | |||||||
| 0 | 8 | 10.9 | 0 | 0.0 | 8 | 17.8 | 0.00035 |
| 1 | 64 | 87.6 | 28 | 100.0 | 36 | 80.0 | |
| NA | 1 | 1.3 | 0 | 0.0 | 1 | 2.2 | |
| Tumor size, mm | |||||||
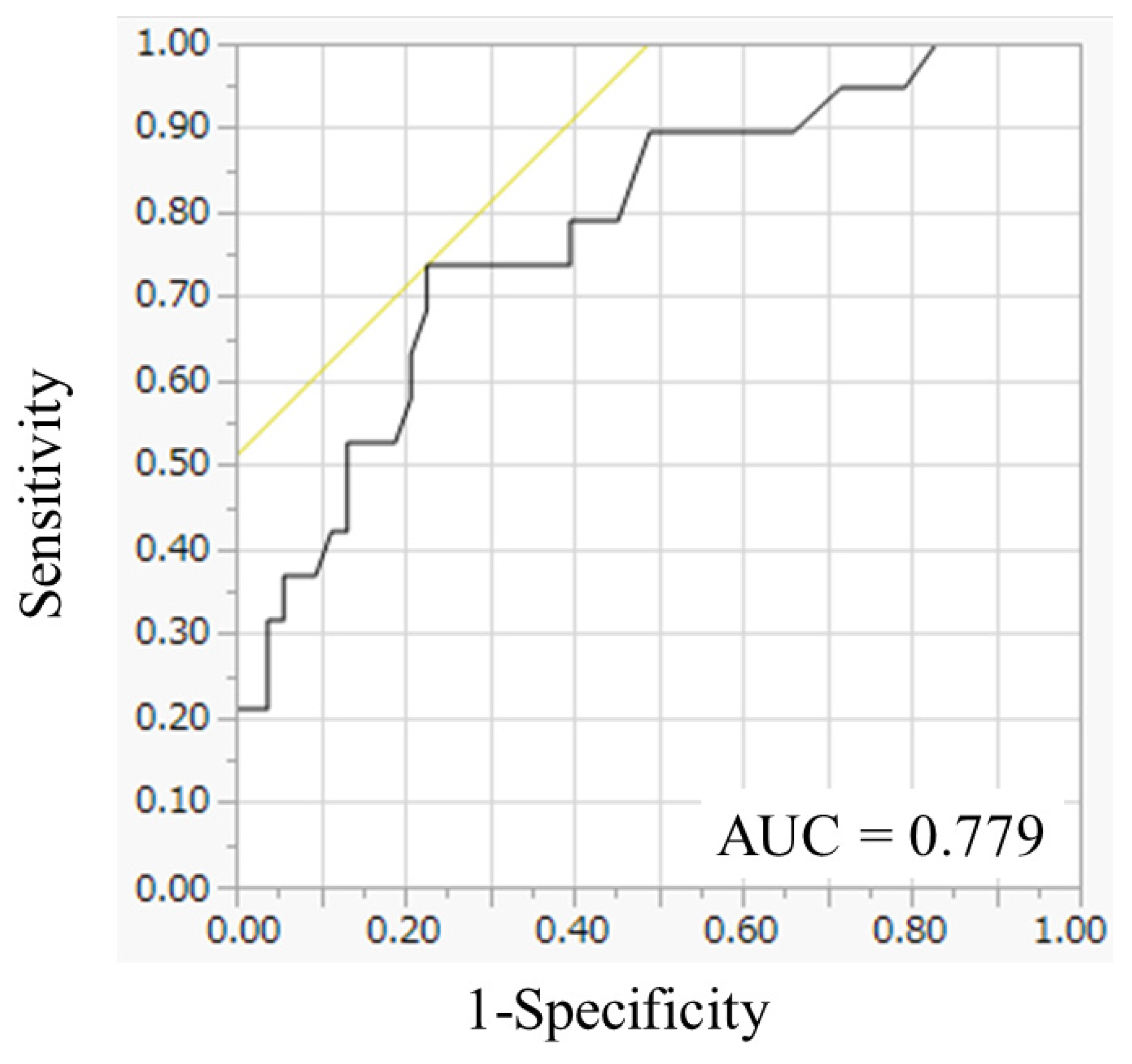
| mean ± SD | 64.4 ± 23.9 | 56.6 ± 4.4 | 69.4 ± 3.5 | 0.0266 | |||
| ≤69 | 45 | 61.6 | 21 | 75.0 | 24 | 53.3 | 0.0603 |
| >69 | 28 | 38.4 | 7 | 25.0 | 21 | 46.7 | |
| SCC antigen value, ng/mL | |||||||
| median (range) | 14.4 (0.6–465.6) | 15.4 (1–349.6) | 11.5 (0.6–465.6) | ||||
| ≤10 or NA | 37 | 50.7 | 13 | 46.4 | 24 | 53.3 | 0.566 |
| >10 | 36 | 49.3 | 15 | 53.6 | 21 | 46.7 | |
| CA125, U/mL | |||||||
| median (range) | 59.4 (3.4–19761) | 84.7 (3.4–1018) | 48.3 (9.3–19761) | ||||
| ≤100 or NA | 51 | 69.9 | 19 | 67.9 | 32 | 71.1 | 0.7688 |
| >100 | 22 | 30.1 | 9 | 32.1 | 13 | 28.9 | |
| Primary Treatment | |||||||
| CCRT | 55 | 75.3 | 23 | 82.1 | 32 | 71.1 | 0.28 |
| Others | 18 | 24.7 | 5 | 17.9 | 13 | 28.9 | |
| Surgery | 4 | (22.2) | 3 | (60.0) | 1 | (7.7) | |
| Chemotherapy | 1 | (5.6) | 0 | (0.0) | 1 | (7.7) | |
| Radiation | 13 | (72.2) | 2 | (40.0) | 11 | (84.6) | |
| Tumor Size (cm) at Diagnosis | All Cases (n = 73) | IIIC2 Group (n = 28) | IVB Group (n = 45) | p-Value | |||
|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | ||
| ≤4 | 9 | 12.3 | 4 | 14.3 | 5 | 11.1 | 0.6904 |
| >4 | 64 | 87.6 | 24 | 85.7 | 40 | 88.9 | |
| ≤5 | 24 | 32.8 | 13 | 46.4 | 11 | 24.4 | 0.0533 |
| >5 | 49 | 67.1 | 15 | 53.6 | 34 | 75.6 | |
| ≤6 | 38 | 52.1 | 20 | 71.4 | 18 | 40.0 | 0.0081 |
| >6 | 35 | 47.9 | 8 | 28.6 | 27 | 60.0 | |
| ≤6.9 | 45 | 61.6 | 21 | 75.0 | 24 | 53.3 | 0.0603 |
| >6.9 | 28 | 38.4 | 7 | 25.0 | 21 | 46.7 | |
| Mean (SD) | 6.44 (5.88–7.00) | 5.66 (4.98–6.34) | 6.94 (6.14–7.74) | 0.0266 | |||
| Median (range) | 5.85 (3.0–14.2) | 5.20 (3.0–10.4) | 6.55 (3.1–14.2) | ||||
| Variable | Category | All Patients | p-Value | IIIC2 Group | p-Value | IVB Group | p-Value |
|---|---|---|---|---|---|---|---|
| HR (95% CI) | HR (95% CI) | HR (95% CI) | |||||
| Univariate Analysis | |||||||
| Tumor size (cm) | >4 vs. ≤4 | 1.35 (0.53–4.54) | 0.5539 | ||||
| >5 vs. ≤ 5 | 2.19 (1.07–4.92) | 0.0291 | |||||
| >6 vs. ≤6 | 2.19 (1.15–4.25) | 0.0163 | |||||
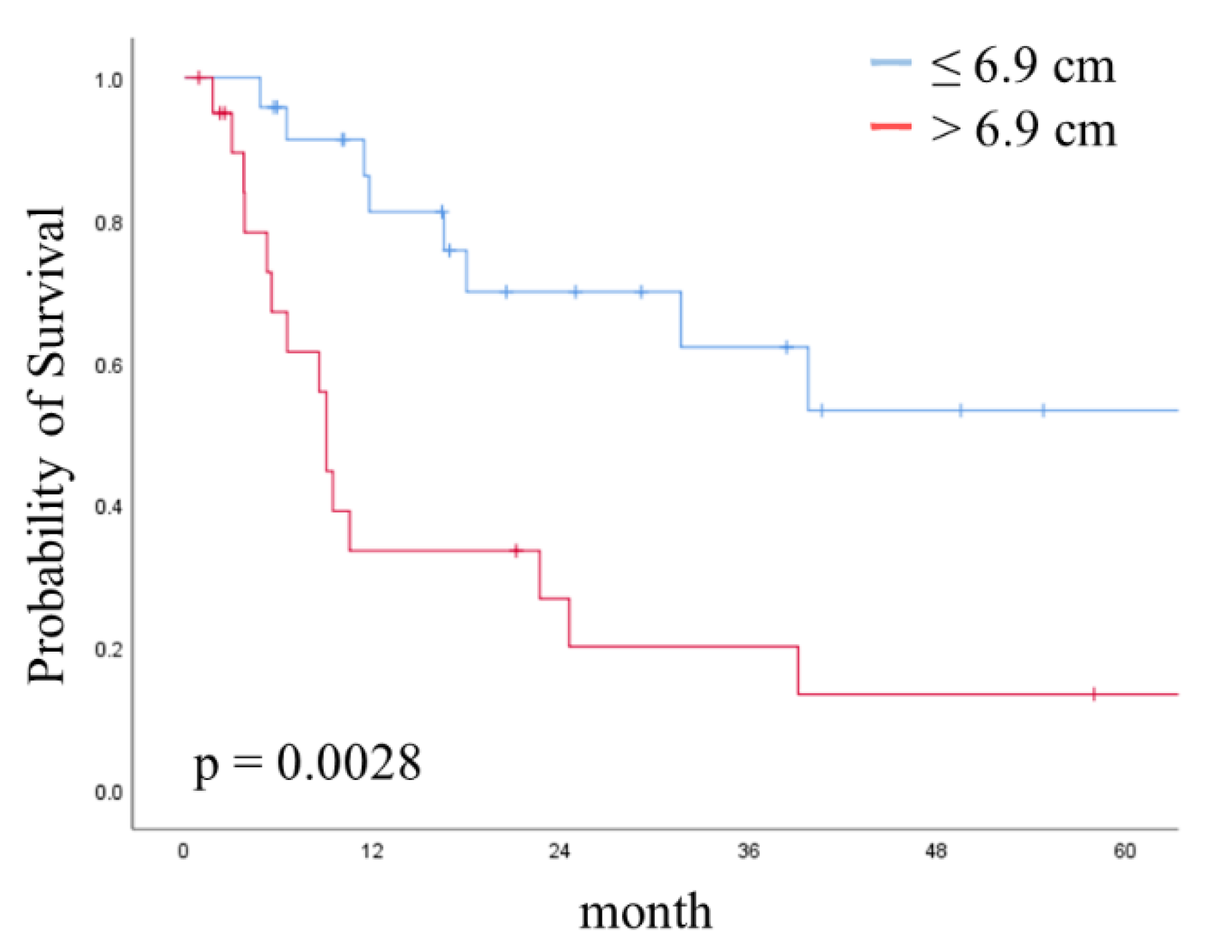
| >6.9 vs. ≤6.9 | 2.48 (1.30–4.73) | 0.006 | 1.21 (0.33–3.66) | 0.7443 | 3.47 (1.53–8.33) | 0.0028 | |
| Metastatic status | IVB vs. IIIC2 | 1.69 (0.88–3.38) | 0.1115 | NA | NA | NA | NA |
| Age at diagnosis | >55 vs. ≤55 | 0.96 (0.50–1.83) | 0.9178 | 0.46 (0.12–1.40) | 0.1785 | 1.34 (0.58–3.26) | 0.4857 |
| Preoperative BMI | >22 vs. ≤22 | 0.65 (0.29– 1.30) | 0.2369 | 0.29 (0.06–0.97) | 0.0444 | 0.99 (0.38–2.31) | 0.9935 |
| Histology | AC or others vs. SCC | 1.40 (0.70–2.68) | 0.3282 | 2.51 (0.85–7.42) | 0.0921 | 0.90 (0.34–2.10) | 0.8238 |
| cT | 3–4 vs. 1–2 | 1.35 (0.70–2.57) | 0.3547 | 0.91 (0.20–2.95) | 0.8904 | 1.38 (0.61–3.23) | 0.4381 |
| N | 1 vs. 0 or NA | 0.89 (0.40–2.38) | 0.8118 | NA | NA | 1.15 (0.48–3.19) | 0.7587 |
| SCC | >10 vs. ≤10 | 1.21 (0.64–2.31) | 0.5433 | 0.84 (0.27–2.44) | 0.7594 | 1.67 (0.73–3.92) | 0.2194 |
| CA125 | >100 vs. ≤100 | 0.90 (0.40–1.86) | 0.8021 | 1.49 (0.45–4.35) | 0.4868 | 0.64 (0.18–1.72) | 0.4017 |
| Primary Treatment | Others vs. CCRT | 3.69 (1.69–7.57) | 0.0015 | 10.98 (2.11–80.01) | 0.0049 | 3.12 (1.17–7.51) | 0.024 |
| Multivariate Analysis | |||||||
| Tumor size | >6.9 vs. ≤6.9 | 3.54 (1.67–7.68) | 0.001 | 4.90 (0.69–37.80) | 0.1079 | 4.79 (1.83–14.16) | 0.0012 |
| Metastatic status | Distant vs. PaLN only | 1.87 (0.87–4.06) | 0.1036 | NA | NA | NA | NA |
| Age at diagnosis | >55 vs. ≤55 | 1.08 (0.50–2.27) | 0.8279 | 0.10 (0.007–0.85) | 0.0345 | 1.63 (0.62– 4.40) | 0.3143 |
| Preoperative BMI | >22 vs. ≤22 | 0.53 (0.21–1.19) | 0.1297 | 0.11 (0.008–0.80) | 0.0274 | 0.63 (0.20– 1.73) | 0.3806 |
| Histology | AC or others vs. SCC | 2.01 (0.73–5.75) | 0.174 | 4.88 (0.82–41.21) | 0.0818 | 1.48 (0.38– 6.00) | 0.5687 |
| cT | 3–4 vs. 1–2 | 0.95 (0.40–2.22) | 0.9201 | 3.09 (0.38–25.21) | 0.2738 | 1.12 (0.38– 3.33) | 0.8315 |
| N | 1 vs. 0 or NA | 1.56 (0.58–4.74) | 0.3795 | NA | NA | 1.32 (0.48– 4.16) | 0.5999 |
| SCC | >10 vs. ≤10 | 1.49 (0.63–3.71) | 0.3659 | 7.28 (0.80–201.09) | 0.1038 | 1.41 (0.44– 4.95) | 0.5636 |
| CA125 | >100 vs. ≤100 | 0.87 (0.33–2.10) | 0.7695 | 0.36 (0.07–1.54) | 0.1713 | 0.97 (0.25– 3.11) | 0.9654 |
| Primary Treatment | Others vs. CCRT | 3.80 (1.55–9.14) | 0.0039 | 2.95 (0.36–28.34) | 0.3053 | 5.24 (1.65– 16.59) | 0.0056 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hattori, S.; Yoshikawa, N.; Mogi, K.; Yoshida, K.; Yoshihara, M.; Tamauchi, S.; Ikeda, Y.; Yokoi, A.; Nishino, K.; Niimi, K.; et al. Significance of Concurrent Chemoradiotherapy as Primary Treatment in Patients with Metastatic Cervical Cancer. Curr. Oncol. 2021, 28, 1663-1672. https://doi.org/10.3390/curroncol28030155
Hattori S, Yoshikawa N, Mogi K, Yoshida K, Yoshihara M, Tamauchi S, Ikeda Y, Yokoi A, Nishino K, Niimi K, et al. Significance of Concurrent Chemoradiotherapy as Primary Treatment in Patients with Metastatic Cervical Cancer. Current Oncology. 2021; 28(3):1663-1672. https://doi.org/10.3390/curroncol28030155
Chicago/Turabian StyleHattori, Satomi, Nobuhisa Yoshikawa, Kazumasa Mogi, Kosuke Yoshida, Masato Yoshihara, Satoshi Tamauchi, Yoshiki Ikeda, Akira Yokoi, Kimihiro Nishino, Kaoru Niimi, and et al. 2021. "Significance of Concurrent Chemoradiotherapy as Primary Treatment in Patients with Metastatic Cervical Cancer" Current Oncology 28, no. 3: 1663-1672. https://doi.org/10.3390/curroncol28030155
APA StyleHattori, S., Yoshikawa, N., Mogi, K., Yoshida, K., Yoshihara, M., Tamauchi, S., Ikeda, Y., Yokoi, A., Nishino, K., Niimi, K., Suzuki, S., & Kajiyama, H. (2021). Significance of Concurrent Chemoradiotherapy as Primary Treatment in Patients with Metastatic Cervical Cancer. Current Oncology, 28(3), 1663-1672. https://doi.org/10.3390/curroncol28030155





