Early Reassessment of Total Metabolic Tumor Volume on FDG-PET/CT in Advanced Melanoma Patients Treated with Pembrolizumab Predicts Long-Term Outcome
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patient Population
2.2. Acquisition and Image Analysis
2.3. Clinical Response Evaluation
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Outcome
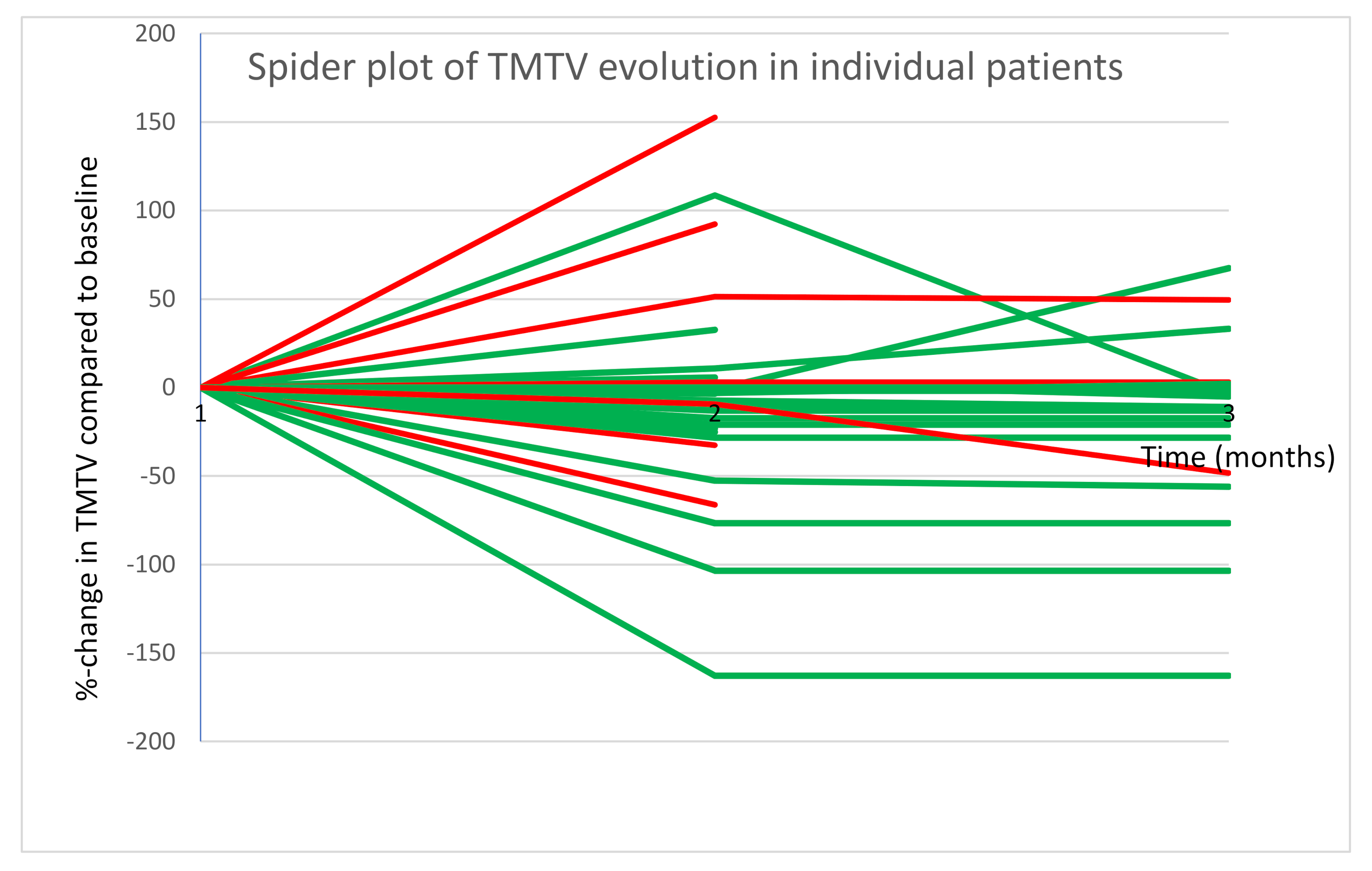
3.3. Response Evaluation Based on PFS and OS Compared to TMTV Change
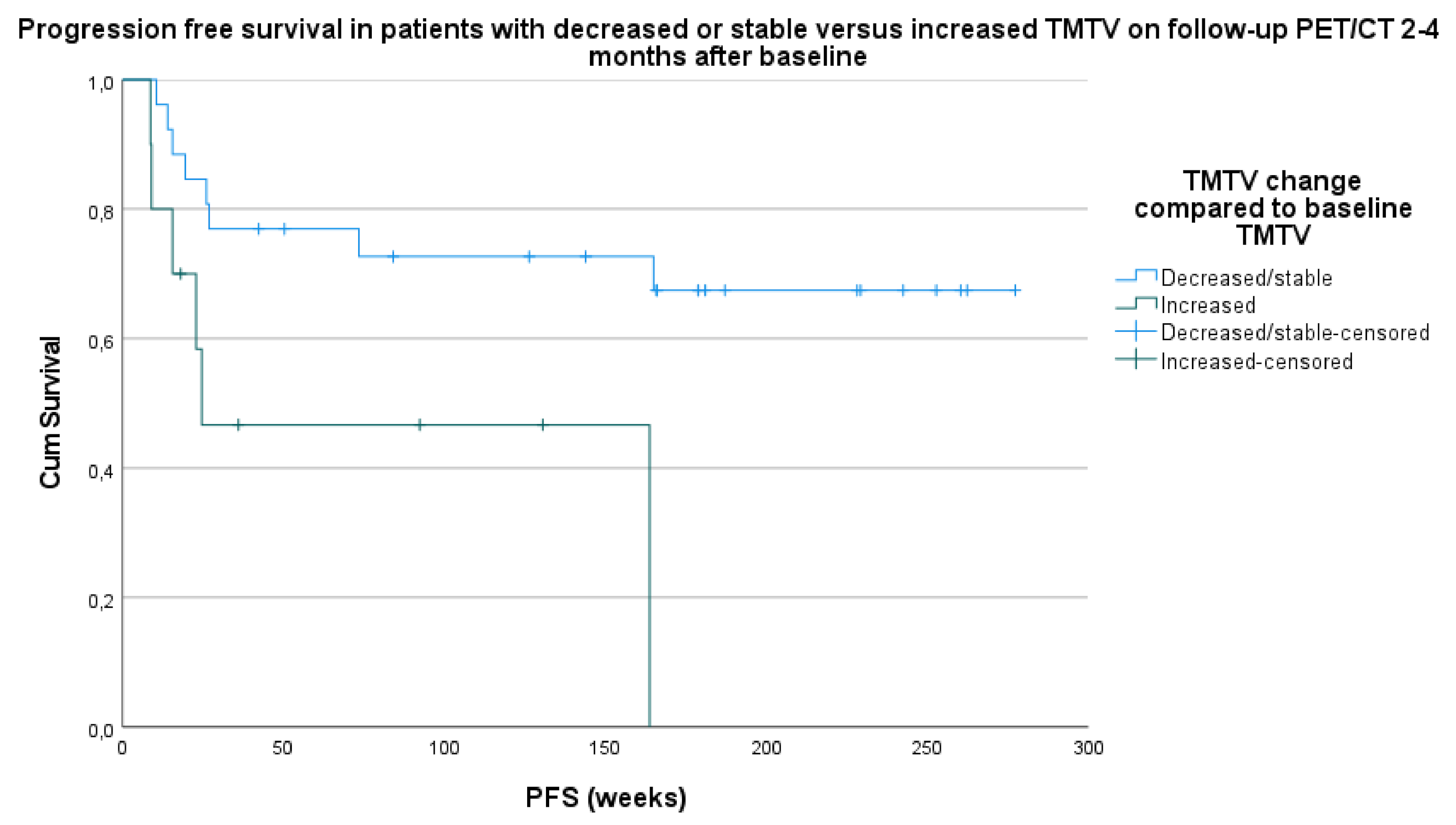
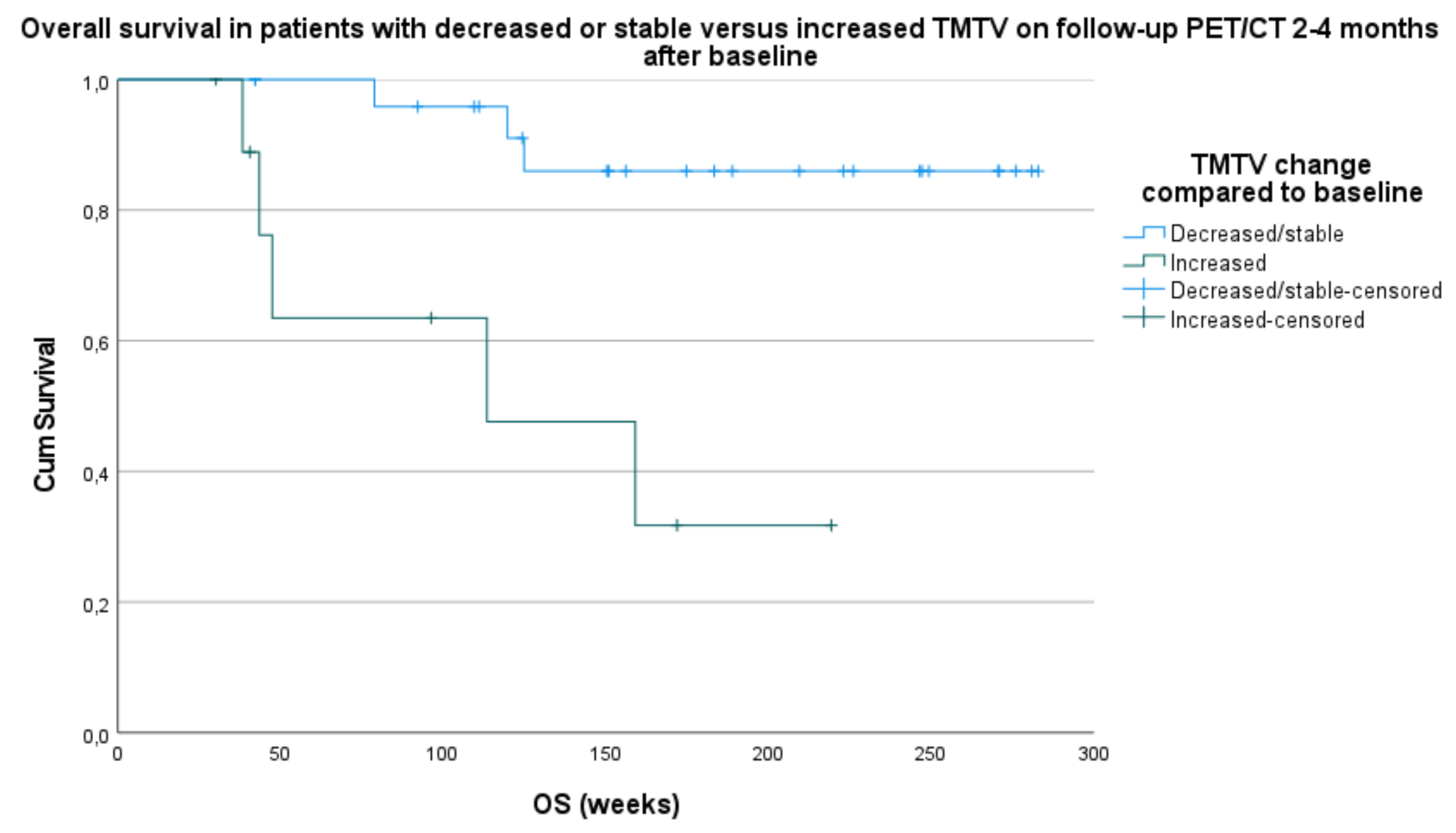
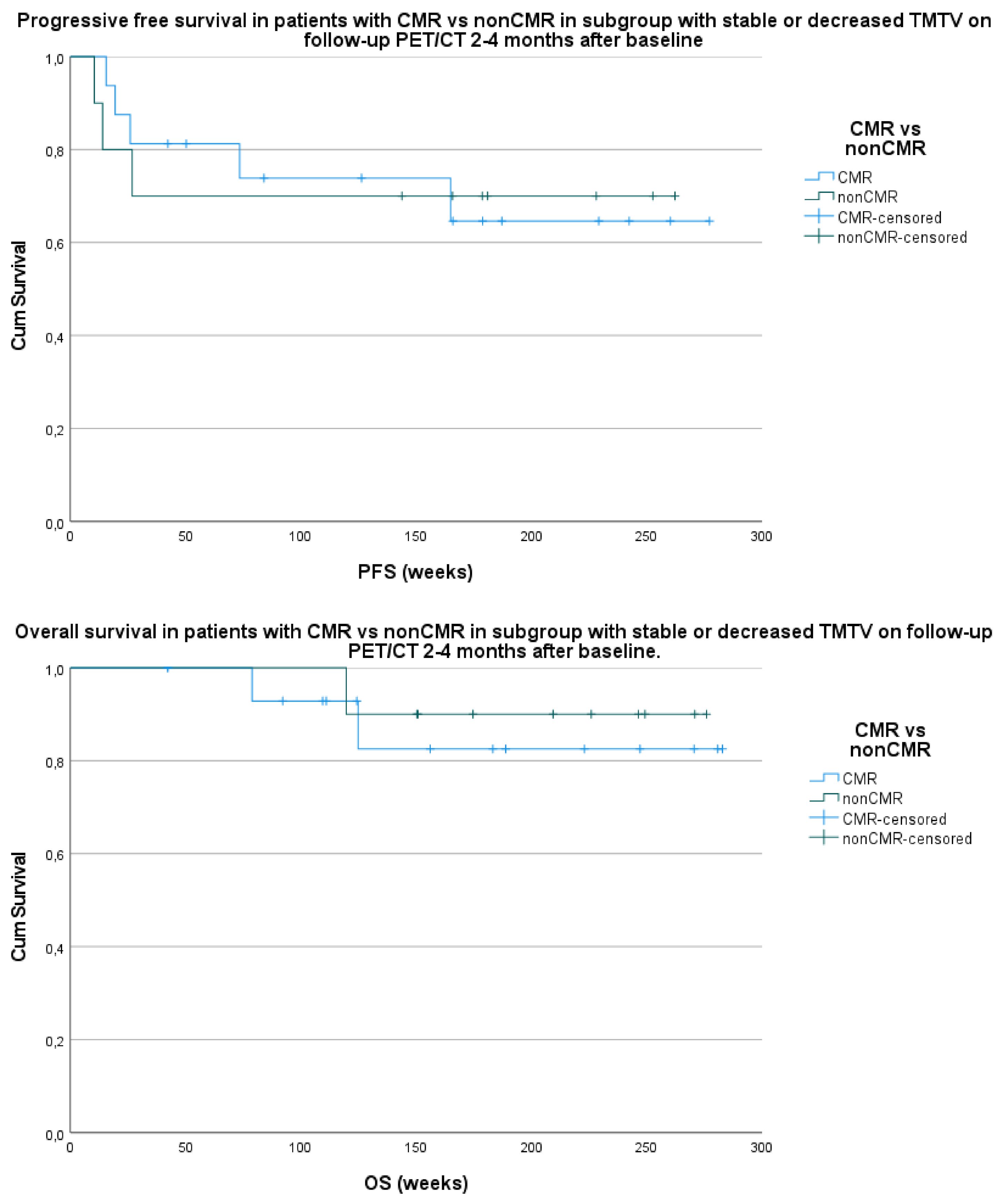
3.3.1. PFS and OS Compared to TMTV Change between Baseline PET/CT and Follow-Up PET/CT after 2–4 Months
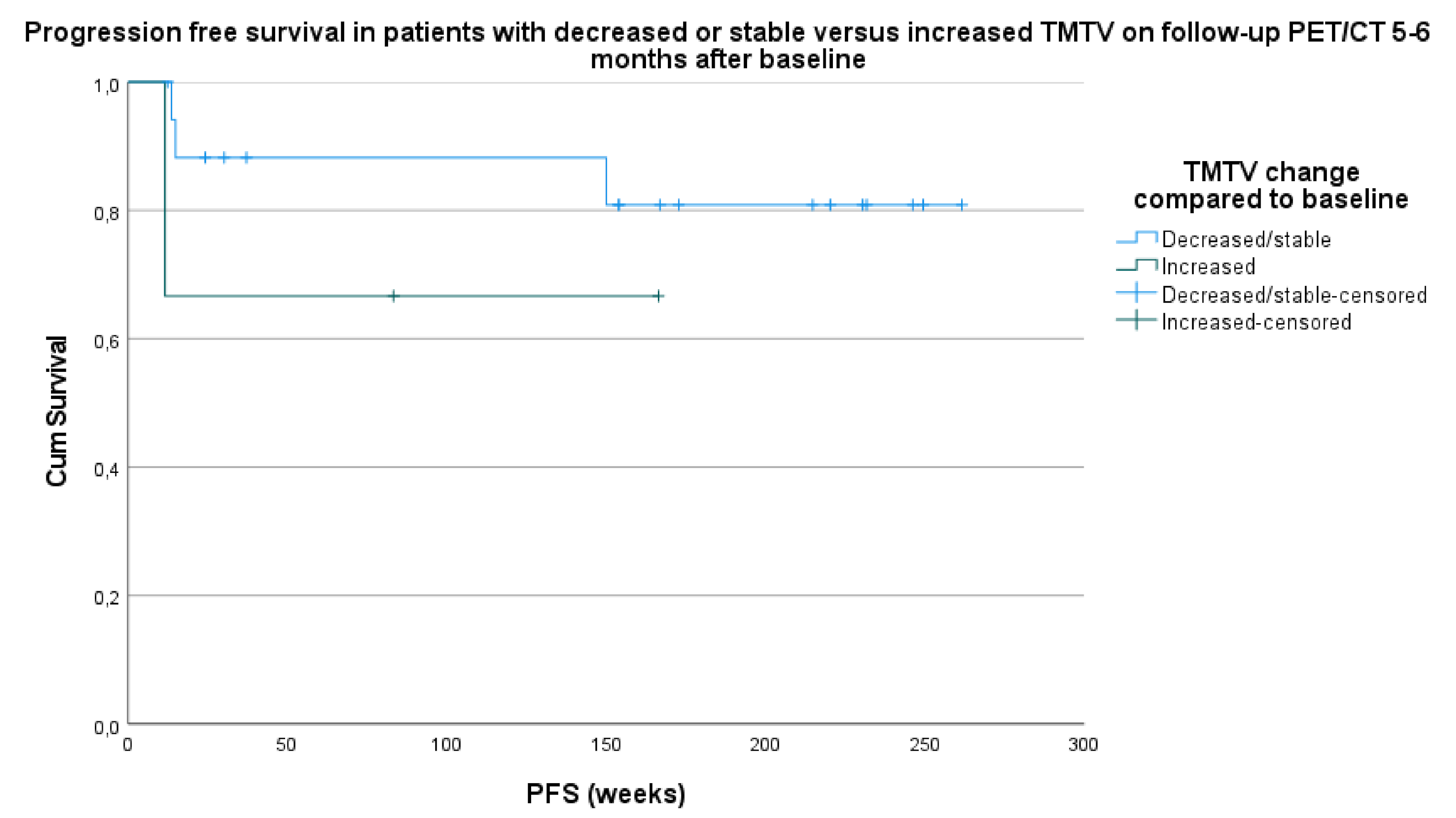
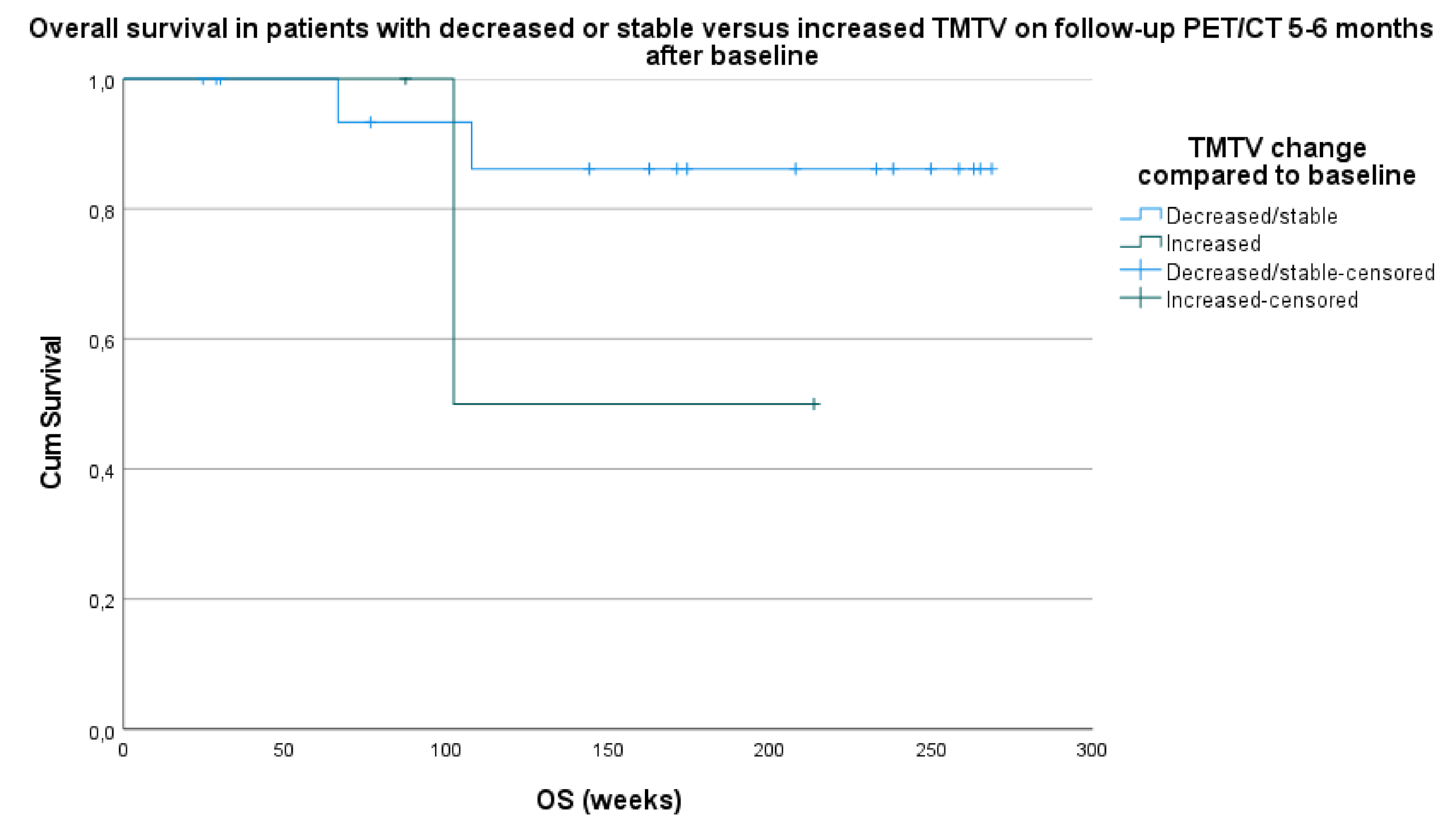
3.3.2. PFS and OS Compared to TMTV Changes between Baseline PET/CT and Follow-Up PET/CT after 5–6 Months
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tan, A.C.; Emmett, L.; Lo, S.; Liu, V.; Kapoor, R.; Carlino, M.S.; Guminski, A.D.; Long, G.V.; Menzies, A.M. FDG-PET response and outcome from anti-PD-1 therapy in metastatic melanoma. Ann. Oncol. 2018, 29, 2115–2120. [Google Scholar] [CrossRef] [PubMed]
- Sachpekidis, C.; Anwar, H.; Winkler, J.K.; Kopp-Schneider, A.; Larribere, L.; Haberkorn, U.; Hassel, J.C.; Dimitrakopoulou-Strauss, A. Longitudinal studies of the 18F-FDG kinetics after ipilimumab treatment in metastatic melanoma patients based on dynamic FDG PET/CT. Cancer Immunol. Immunother. 2018, 67, 1261–1270. [Google Scholar] [CrossRef] [PubMed]
- Amrane, K.; Le Goupil, D.; Quere, G.; Delcroix, O.; Gouva, S.; Schick, U.; Salaun, P.Y.; Abgral, R.; Alavi, Z.; Keromnes, N.; et al. Prediction of response to immune checkpoint inhibitor therapy using 18F-FDG PET/CT in patients with melanoma. Medicine 2019, 98, e16417. [Google Scholar] [CrossRef] [PubMed]
- Ralli, M.; Botticelli, A.; Visconti, I.C.; Angeletti, D.; Fiore, M.; Marchetti, P.; Lambiase, A.; de Vincentiis, M.; Greco, A. Immunotherapy in the Treatment of Metastatic Melanoma: Current Knowledge and Future Directions. J. Immunol. Res. 2020, 28, 9235638. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Paeng, J.C.; Cheon, G.J.; Lee, D.S.; Chung, J.K.; Kang, K.W. Recurrence of Melanoma After Initial Treatment: Diagnostic Performance of FDG PET in Posttreatment Surveillance. Nucl. Med. Mol. Imaging 2018, 52, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Hamid, O.; Robert, C.; Daud, A.; Hodi, F.S.; Hwu, W.J.; Kefford, R.; Wolchok, J.D.; Hersey, P.; Joseph, R.; Weber, J.S.; et al. Five-year survival outcomes for patients with advanced melanoma treated with pembrolizumab in KEYNOTE-001. Ann. Oncol. 2019, 30, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Kitao, T.; Hirata, K.; Shima, K.; Hayashi, T.; Sekizawa, M.; Takei, T.; Ichimura, W.; Harada, M.; Kondo, K.; Tamaki, N. Reproducibility and uptake time dependency of volume-based parameters on FDG-PET for lung cancer. BMC Cancer 2016, 16, 576. [Google Scholar] [CrossRef] [PubMed]
- Seban, R.D.; Nemer, J.S.; Marabelle, A.; Yeh, R.; Deutsch, E.; Ammari, S.; Moya-Plana, A.; Mokrane, F.Z.; Gartrell, R.D.; Finkel, G.; et al. Prognostic and theranostic 18F-FDG PET biomarkers for anti-PD1 immunotherapy in metastatic melanoma: Association with outcome and transcriptomics. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2298–2310. [Google Scholar] [CrossRef] [PubMed]
- Awada, G.; Jansen, Y.; Schwarze, J.K.; Tijtgat, J.; Hellinckx, L.; Gondry, O.; Vermeulen, S.; Warren, S.; Schats, K.; van Dam, P.J.; et al. A Comprehensive Analysis of Baseline Clinical Characteristics and Biomarkers Associated with Outcome in Advanced Melanoma Patients Treated with Pembrolizumab. Cancers 2021, 13, 168. [Google Scholar] [CrossRef] [PubMed]
- Wolchok, J.D.; Hoos, A.; O’Day, S.; Weber, J.S.; Hamid, O.; Lebbé, C.; Maio, M.; Binder, M.; Bohnsack, O.; Nichol, G.; et al. Guidelines for the evaluation of immune therapy activity in solid tumors: Immune-related response criteria. Clin. Cancer Res. 2009, 15, 7412–7420. [Google Scholar] [CrossRef] [PubMed]
- Young, H.; Baum, R.; Cremerius, U.; Herholz, K.; Hoekstra, O.; Lammertsma, A.A.; Pruim, J.; Price, P. Measurement of clinical and subclinical tumour response using [18F]-fluorodeoxyglucose and positron emission tomography: Review and 1999 EORTC recommendations. European Organization for Research and Treatment of Cancer (EORTC) PET Study Group. Eur. J. Cancer 1999, 35, 1773–1782. [Google Scholar] [CrossRef]
- Kluger, H.M.; Chiang, V.; Mahajan, A.; Zito, C.R.; Sznol, M.; Tran, T.; Weiss, S.A.; Cohen, J.V.; Yu, J.; Hegde, U.; et al. Long-Term Survival of Patients With Melanoma With Active Brain Metastases Treated With Pembrolizumab on a Phase II Trial. J. Clin. Oncol. 2019, 37, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Seban, R.D.; Moya-Plana, A.; Antonios, L.; Yeh, R.; Marabelle, A.; Deutsch, E.; Schwartz, L.H.; Gómez, R.G.H.; Saenger, Y.; Robert, C.; et al. Prognostic 18F-FDG PET biomarkers in metastatic mucosal and cutaneous melanoma treated with immune checkpoint inhibitors targeting PD-1 and CTLA-4. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2301–2312. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, R.; Zaba, L.C.; Rosenberg, J.; Reddy, S.A.; Nobashi, T.W.; Davidzon, G.; Aparici, C.M.; Nguyen, J.; Moradi, F.; Iagaru, A.; et al. Prognostic value of volumetric PET parameters at early response evaluation in melanoma patients treated with immunotherapy. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2787–2795. [Google Scholar] [CrossRef] [PubMed]
- Iravani, A.; Osman, M.M.; Weppler, A.M.; Wallace, R.; Galligan, A.; Lasocki, A.; Hunter, M.O.; Akhurst, T.; Hofman, M.S.; Lau, P.K.H.; et al. FDG PET/CT for tumoral and systemic immune response monitoring of advanced melanoma during first-line combination ipilimumab and nivolumab treatment. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2776–2786. [Google Scholar] [CrossRef]
- Decazes, P.; Bohn, P. Immunotherapy by Immune Checkpoint Inhibitors and Nuclear Medicine Imaging: Current and Future Applications. Cancers 2020, 12, 371. [Google Scholar] [CrossRef]
- Reinert, C.P.; Gatidis, S.; Sekler, J.; Dittmann, H.; Pfannenberg, C.; la Fougère, C.; Nikolaou, K.; Forschner, A. Clinical and prognostic value of tumor volumetric parameters in melanoma patients undergoing 18F-FDG-PET/CT: A comparison with serologic markers of tumor burden and inflammation. Cancer Imaging 2020, 20, 44. [Google Scholar] [CrossRef] [PubMed]
- Lewin, J.; Sayers, L.; Kee, D.; Walpole, I.; Sanelli, A.; Te Marvelde, L.; Herschtal, A.; Spillane, J.; Gyorki, D.; Speakman, D.; et al. Surveillance imaging with FDG-PET/CT in the post-operative follow-up of stage 3 melanoma. Ann. Oncol. 2018, 29, 1569–1574. [Google Scholar] [CrossRef] [PubMed]
- Bisschop, C.; de Heer, E.C.; Brouwers, A.H.; Hospers, G.A.P.; Jalving, M. Rational use of 18F-FDG PET/CT in patients with advanced cutaneous melanoma: A systematic review. Crit. Rev. Oncol. Hematol. 2020, 153, 103044. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.N.M.; McArthur, G.A.; Hofman, M.S.; Hicks, R.J. The Advantages and Challenges of Using FDG PET/CT for Response Assessment in Melanoma in the Era of Targeted Agents and Immunotherapy. Eur. J. Nucl. Med. Mol. Imaging 2017, 44 (Suppl. 1), 67–77. [Google Scholar] [CrossRef]
| Baseline Patient Characteristics | Patients with Baseline FDG-PET/CT | Patients with Follow-Up FDG-PET/CT 2-4 Months after Baseline | Patients with Follow-Up FDG-PET/CT 5-6 Months after Baseline |
|---|---|---|---|
| n = 77 | n = 36 | n = 21 | |
| Subpopulation | Baseline | Group 1 | Group 2 |
| Age | |||
| Median | 66 | 65 | 62 |
| Range | 31–98 | 31–89 | 31–82 |
| Sex | |||
| Male | 43 (55.8%) | 18 (50.0%) | 11 (52.4%) |
| Female | 34 (44.2%) | 18 (50.0%) | 10 (47.6%) |
| Melanoma subtype | |||
| Cutaneous | 69 (89.6%) | 33 (91.7%) | 20 (95.2%) |
| Mucosal | 3 (3.9%) | 0 (0%) | 0 (0%) |
| Unknown primary | 5 (6.5%) | 3 (8.3%) | 1 (4.8%) |
| Baseline WHO PS | |||
| 0 | 49 (63.6%) | 29 (80.6%) | 15 (71.4%) |
| 1 | 19 (24.7%) | 5 (13.9%) | 4 (19.1%) |
| 2 | 9 (11.7%) | 2 (5.6%) | 2 (9.5%) |
| Tumor Stage | |||
| IIIB | 1 (1.3%) | 1 (2.8%) | 0 (0%) |
| IIIC | 8 (10.4%) | 7 (19.4%) | 4 (19.1%) |
| IV-M1a | 5 (6.5%) | 3 (8.3%) | 2 (9.5%) |
| IV-M1b | 12 (15.6%) | 6 (16.7%) | 2 (9.5%) |
| IV-M1c | 38 (49.4%) | 16 (44.4%) | 11 (52.4%) |
| IV-M1d | 13 (16.9%) | 3 (8.3%) | 2 (9.5%) |
| Brain metastases | |||
| Active | 6 (7.8%) | 0 (0%) | 0 (0%) |
| Inactive | 7 (9.1%) | 3 (8.3%) | 2 (9.5%) |
| Number of affected organs | |||
| 1 | |||
| 2 | 21 (27.3%) | 15 (41.7%) | 8 (38.1%) |
| 3 | 19 (24.7%) | 10 (27.8%) | 6 (28.6%) |
| >3 | 16 (20.8%) | 6 (16.7%) | 5 (23.8%) |
| 21 (27.3%) | 5 (13.9%) | 2 (9.5%) | |
| Number of prior therapies | |||
| 0 | |||
| 1 | 24 (31.2%) | 14 (38.9%) | 8 (38.1%) |
| 2 | 30 (39.0%) | 12 (33.3%) | 5 (23.8%) |
| 3 | 13 (16.9%) | 6 (16.7%) | 4 (19.1%) |
| >3 | 4 (5.2%) | 1 (2.8%) | 1 (4.8%) |
| 6 (7.8%) | 3 (8.3%) | 3 (14.3%) | |
| LDH level | |||
| <ULN | 54 (70.1%) | 27 (75.0%) | 17 (81.0%) |
| ≥ULN | 23 (29.9%) | 9 (25.0%) | 4 (19.1%) |
| BRAFV600 status | |||
| Mutant | 34 (44.2%) | 12 (33.3%) | 8 (38.1%) |
| Wild type | 43 (55.8%) | 24 (66.7%) | 13 (61.9%) |
| TMTV | |||
| 0 | 0 (0.0%) | 16 (44.4%) | 15 (71.4%) |
| >0–50 | 52 (67.5%) | 15 (41.7%) | 4 (19.1%) |
| (median, range) | (15.8, 1.0–49.5) | (10.7, 0.8–49.9) | (6.8, 3.8–10.8) |
| >50–100 | 9 (11.7%) | 2 (5.6%) | 1 (4.8%) |
| (median, range) | (66.2, 56.0–85.9) | (54.2, 50.5–58.0) | (52.0) |
| ≥100 | 16 (20.8%) | 3 (8.3%) | 1 (4.8%) |
| (median, range) | (283.5, 103.5–1341.5) | (201.8, 116.4–336.4) | (126.7) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vermeulen, S.; Awada, G.; Keyaerts, M.; Neyns, B.; Everaert, H. Early Reassessment of Total Metabolic Tumor Volume on FDG-PET/CT in Advanced Melanoma Patients Treated with Pembrolizumab Predicts Long-Term Outcome. Curr. Oncol. 2021, 28, 1630-1640. https://doi.org/10.3390/curroncol28030152
Vermeulen S, Awada G, Keyaerts M, Neyns B, Everaert H. Early Reassessment of Total Metabolic Tumor Volume on FDG-PET/CT in Advanced Melanoma Patients Treated with Pembrolizumab Predicts Long-Term Outcome. Current Oncology. 2021; 28(3):1630-1640. https://doi.org/10.3390/curroncol28030152
Chicago/Turabian StyleVermeulen, Sim, Gil Awada, Marleen Keyaerts, Bart Neyns, and Hendrik Everaert. 2021. "Early Reassessment of Total Metabolic Tumor Volume on FDG-PET/CT in Advanced Melanoma Patients Treated with Pembrolizumab Predicts Long-Term Outcome" Current Oncology 28, no. 3: 1630-1640. https://doi.org/10.3390/curroncol28030152
APA StyleVermeulen, S., Awada, G., Keyaerts, M., Neyns, B., & Everaert, H. (2021). Early Reassessment of Total Metabolic Tumor Volume on FDG-PET/CT in Advanced Melanoma Patients Treated with Pembrolizumab Predicts Long-Term Outcome. Current Oncology, 28(3), 1630-1640. https://doi.org/10.3390/curroncol28030152





