Comparison of the Quality of Life of Patients with Breast or Colon Cancer with an Arm Vein Port (TIVAD) Versus a Peripherally Inserted Central Catheter (PICC)
Abstract
1. Introduction
2. Methods
2.1. Subject Selection
2.2. Central Venous Catheter Devices
2.3. Subject Surveys and Clinical Follow-Up
2.4. Quality of Life Constructs
2.5. Statistical Analysis
3. Results
3.1. Patients and Device Characteristics
3.2. Survey Results
3.3. Complications
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Canadian Cancer Statistics. Available online: https://www.cancer.ca/~/media/cancer.ca/CW/cancer%20information/cancer%20101/Canadian%20cancer%20statistics/Canadian-Cancer-Statistics-2015-EN.pdf (accessed on 2 July 2020).
- Ahmed, S.; Sami, A.; Xiang, J. HER2-directed therapy: Current treatment options for HER2-positive breast cancer. Breast Cancer 2015, 22, 101–116. [Google Scholar]
- Ahmed, S.; Johnson, K.; Ahmed, O.; Iqbal, N. Advances in the management of colorectal cancer: From biology to treatment. Int. J. Colorectal. Dis. 2014, 29, 1031–1042. [Google Scholar] [CrossRef] [PubMed]
- Burbridge, B.; Chan, I.; Bryce, R.; Lim, H.; Stoneham, G.; Haggag, H.; Roh, C. Satisfaction and Quality of Life Related to Chemotherapy with an Arm Port: A Pilot Study. CARJ 2016, 67, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Burbridge, B.; Plewes, C.; Stoneham, G.; Szkup, P.; Otani, R.; Babyn, P.; Bryce, R. Randomized Clinical Trial Evaluating Complications and Complication-Related Removal of Arm-Situated Power-Injectable and Non-Power-Injectable Totally Implanted Venous Access Devices among Cancer Patients. JVIR 2018, 29, 648–659. [Google Scholar] [CrossRef]
- Sansivero, G. The Microintroducer Technique for Peripherally Inserted Central Catheter Placement. J. Infus. Nurs. 2000, 23, 345–351. [Google Scholar]
- Marcy, P.Y.; Mari, V.; Figl, A.; Ben-Taarit, I.; Fouche, Y.; Peyrade, F.; Follana, P.; Michel, C.; Chamorey, E. Patient Satisfaction with and Acceptance of Their Totally-Implanted Central Venous Catheter: Construction and First Validation of a Questionnaire. J. Cancer Ther. 2014, 7, 706–716. [Google Scholar] [CrossRef][Green Version]
- Liu, Y.; Xu, L.; Jiang, M.; Chen, B.; Jing, L.; Zhang, L.; Su, P.; Jin, F.; Mao, X. Chinesization of the quality of life assessment, venous device-port, and it reliability and validity tests for patients with breast cancer. JVA 2020. [Google Scholar] [CrossRef]
- Marcy, P.; Chamorey, E.; Amoretti, N. A comparison between distal and proximal port device insertion in head and neck cancer. Eur. J. Surg. Oncol. 2008, 34, 1262–1269. [Google Scholar] [CrossRef]
- Marcy, P.; Magné, N.; Castadot, P.; Italiano, A. Is radiologic placement of an arm port mandatory in oncology patients? Cancer 2007, 110, 2331–2338. [Google Scholar] [CrossRef]
- Bodner, L.J.; Nosher, J.L.; Patel, K.M.; Siegel, R.L.; Biswal, R.; Gribbin, C.E.; Tokarz, R. Peripheral venous access ports: Outcomes analysis in 109 patients. Cardiovasc. Intervent. Radiol. 2000, 23, 187–193. [Google Scholar] [CrossRef]
- Burbridge, B.; Krieger, E.; Stoneham, G. Arm placement of the Cook Titanium Petite Vital-Port: Results of radiologic placement in 125 patients with cancer. CARJ 2000, 51, 163. [Google Scholar]
- Lenhart, M.; Chegini, M.; Gmeinwieser, J.; Manke, C. Radiologic implantation of central venous portal systems in the forearm. Rofo 1998, 169, 189–194. [Google Scholar] [CrossRef]
- Deppe, G.; Kahn, M.L.; Malviya, V.K.; Malone, J.M.; Christensen, C.W. Experience with the PAS Port venous access device in patients with gynecologic malignancies. Gynecol. Oncol. 1996, 62, 340–343. [Google Scholar] [CrossRef]
- Fayers, P.M.; Aaronson, N.K.; Bjordal, K.; Groenvold, M.; Curran, D.; Bottomley, A. The EORTC QLQ-C30 Scoring Manual, on Behalf of the EORTC Quality of Life Group, 3rd ed.; European Organisation for Research and Treatment of Cancer: Brussels, Belgium, 2001. [Google Scholar]
- Luo, L.; Jing, X.M.; Wang, G.R.; Qin, Y.; Liang, H.X.; Liu, S.S. Peripherally inserted central catheter-related upper extremity venous thrombosis in oncology patients: A prospective study based on Doppler sonography. J. Ultrasound Med. 2016, 35, 1759–1763. [Google Scholar] [CrossRef] [PubMed]
- Patel, G.S.; Jain, K.; Kumar, R.; Strickland, A.H.; Pellegrini, L.; Slavotinek, J.; Eaton, M.; McLeay, L.; Price, T.; Ly, M. Comparison of peripherally inserted central venous catheters (PICC) versus subcutaneously implanted port-chamber catheters by complication and cost for patients receiving chemotherapy for non-haematological malignancies. Support Care Cancer 2014, 22, 121–128. [Google Scholar] [CrossRef]
- Qi, F.; Cheng, H.; Yuan, X.; Zhang, L. Comparison of PICC and TIVAP in chemotherapy for patients with thyroid cancer. Oncol. Lett. 2020, 20, 1657–1662. [Google Scholar] [CrossRef]
- Marcy, P.Y.; Magne, N.; Castadot, P.; Bailet, C.; Macchiavello, J.-C.; Namer, M.; Gallard, J.-C. Radiological and surgical placement of port devices: A 4-year institutional analysis of procedure performance, quality of life and cost in breast cancer patients. Breast Cancer Res. Treat 2005, 92, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Johansson, E.; Hammarskjöld, F.; Lundberg, D.; Arnlind, M.H. Advantages and disadvantages of peripherally inserted central venous catheters (PICC) compared to other central venous lines: A systematic review of the literature. Acta Oncologica 2013, 52, 886–892. [Google Scholar] [CrossRef]
- Taxbro, K.; Hammarskjöld, F.; Thelin, B.; Lewin, F.; Hagman, H.; Hanberger, H.; Berg, S. Clinical impact of peripherally inserted central catheters vs implanted port catheters in patients with cancer: An open-label, randomized, two-centre trial. Br. J. Anaesth. 2019, 122, 734–741. [Google Scholar] [CrossRef] [PubMed]
- Schummer, C.; Sakr, Y.; Steenbeck, J.; Gugel, M.; Reinhart, K.; Schummer, W. Risk of extravasation after power injection of contrast media via the proximal port of multilumen central venous catheters: Case report and review of the literature. Rofo 2010, 182, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Marcy, P.Y.; Thariat, J.; Figl, A. Power injection via a venous port: A new challenge for radiologists. Rofo 2010, 182, 536. [Google Scholar] [CrossRef] [PubMed]
| QoL Constructs | 0–1 Months | p-Value | 3 Months | p-Value | ||
|---|---|---|---|---|---|---|
| PICC | Port | PICC | Port | |||
| Global Health Status | 59.1 ± 21.1 | 57.1 ± 23.8 | 0.68 | 60.8 ± 22.5 | 66.7 ± 24.5 | 0.39 |
| Functional Scale | ||||||
| Physical functioning | 85.2 ± 13.7 | 82.8 ± 16.3 | 0.52 | 84.7 ± 13.1 | 84.4 ± 15.4 | 0.92 |
| Role functioning | 62.5 ± 35.7 | 72.7 ± 27.3 | 0.18 | 70.8 ± 33.1 | 71.7 ± 26.8 | 0.92 |
| Emotional functioning | 76.4 ± 18.7 | 71.5 ± 22.7 | 0.33 | 78.5 ± 18.0 | 79.7 ± 15.5 | 0.80 |
| Cognitive functioning | 67.1 ± 28.3 | 74.2 ± 21.7 | 0.25 | 83.3 ± 16.3 | 76.8 ± 24.0 | 0.28 |
| Social functioning | 58.8 ± 28.6 | 55.6 ± 31.1 | 0.65 | 66.7 ± 26.5 | 65.9 ± 23.3 | 0.92 |
| Symptoms Scale | ||||||
| Fatigue | 42.9 ± 28.3 | 44.8 ± 23.8 | 0.76 | 51.4 ± 21.9 | 40.6 ± 26.3 | 0.13 |
| Nausea and vomiting | 16.2 ± 15.2 | 13.1 ± 22.7 | 0.52 | 11.8 ± 17.4 | 4.4 ± 7.5 | 0.06 |
| Pain | 22.2 ± 27.3 | 25.3 ± 25.4 | 0.63 | 27.1 ± 29.8 | 13.0 ± 22.5 | 0.07 |
| Dyspnea | 16.7 ± 21.8 | 19.2 ± 20.5 | 0.62 | 15.3 ± 24.0 | 24.6 ± 25.1 | 0.20 |
| Insomnia | 47.2 ± 33.2 | 43.4 ± 33.8 | 0.64 | 36.1 ± 29.4 | 40.6 ± 34.8 | 0.63 |
| Appetite loss | 27.8 ± 28.2 | 21.2 ± 31.0 | 0.36 | 34.7 ± 36.1 | 14.5 ± 24.3 | 0.03 |
| Constipation | 27.8 ± 24.6 | 24.2 ± 29.2 | 0.59 | 22.2 ± 27.2 | 20.3 ± 29.7 | 0.82 |
| Diarrhea | 22.2 ± 30.9 | 34.3 ± 40.4 | 0.16 | 25.0 ± 31.5 | 18.8 ± 24.3 | 0.52 |
| Financial Difficulties | 22.2 ± 30.9 | 34.3 ± 40.4 | 0.16 | 19.4 ± 27.7 | 24.6 ± 27.0 | 0.52 |
| QLVAD Constructs | 0–1 Months | p-Value | 3 Months | p-Value | ||
|---|---|---|---|---|---|---|
| PICC | Port | PICC | Port | |||
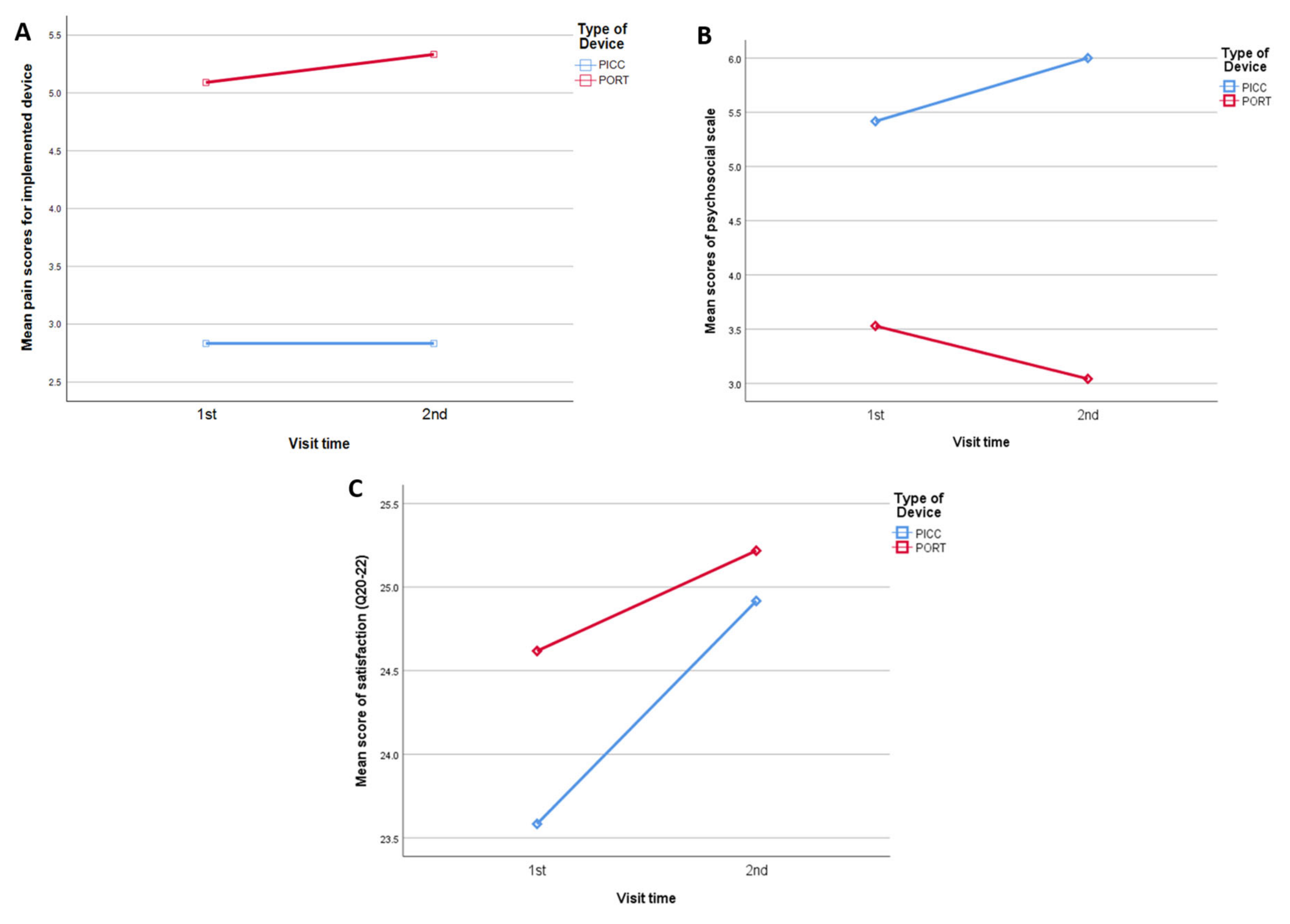
| How painful was it to have your device implanted? * | 2.8 ± 2.2 | 5.1 ± 2.9 | 0.001 | 2.8 ± 2.3 | 5.3 ± 2.9 | 0.002 |
| Rate the degree of discomfort you experience during needle insertion into the device * | 1.3 ± 0.9 | 3.0 ± 2.5 | 0.001 | 1.3 ± 0.75 | 3.5 ± 2.3 | <0.001 |
| Rate the degree of discomfort you experience during treatment infusion * | 1.4 ± 1.3 | 2.1 ± 1.8 | 0.10 | 1.5 ± 0.9 | 2.0 ± 1.4 | 0.12 |
| Rate the degree of discomfort you experience during needle removal after treatment * | 1.3 ± 0.9 | 1.6 ± 1.1 | 0.21 | 1.2 ± 0.5 | 1.8 ± 1.3 | 0.03 |
| Do you believe that insertion of your device was a good thing to have done? * | 8.5 ± 2.0 | 8.5 ± 2.4 | 0.92 | 9.3 ± 1.9 | 8.8 ± 1.8 | 0.36 |
| Assess your degree of satisfaction with your device * | 7.5 ± 2.3 | 8.4 ± 2.3 | 0.12 | 8.0 ± 2.3 | 8.3 ± 1.9 | 0.74 |
| If you had to have a device implanted for another session of treatment during your life, would you have another? * | 7.6 ± 3.0 | 7.8 ± 2.9 | 0.77 | 7.6 ± 3.2 | 8.3 ± 2.4 | 0.38 |
| QLVAD Constructs | 1 Month % * | OR (95% CI) p-Values | 3 Months % * | OR (95% CI) p-Values | ||
|---|---|---|---|---|---|---|
| PICC | Port | PICC | Port | |||
| Do you find it easy to present your device for treatment or blood sampling? | 97.2 | 82.4 | 7.5 (0.85–66.0) 0.052 | 100 | 91.7 | 1.1 (0.97–1.23) 0.49 |
| Do you feel that your device is too visible? | 19.4 | 11.8 | 1.8 (0.47–6.8) 0.37 | 37.5 | 16.7 | 3.0 (0.77–11.6) 0.10 |
| Do you feel that your device is unsightly or ugly? | 27.8 | 11.8 | 2.9 (0.81–10.3) 0.09 | 41.7 | 16.7 | 3.6 (0.93–13.7) 0.05 |
| Do you feel that you have changed the way you dress due to your device? | 61.1 | 17.6 | 7.3 (2.4–22.2) <0.001 | 66.7 | 33.3 | 4.0 (1.2–13.3) 0.02 |
| Have people commented on your device when they see it? | 33.3 | 14.7 | 2.9 (0.89–9.4) 0.07 | 41.7 | 12.5 | 5.0 (1.2–21.5) 0.02 |
| Do you try to cover your device with clothing? | 55.6 | 38.2 | 2.02 (0.19–1.3) 0.15 | 62.5 | 37.5 | 2.80 (0.11–1.3) 0.08 |
| Does your device make you feel anxious (worried)? | 19.4 | 8.8 | 2.50 (0.60–10.6) 0.30 | 16.7 | 4.2 | 4.6 (0.47–44.6) 0.34 |
| Are you worried that your device might become damaged? | 50.0 | 29.4 | 2.4 (0.90–6.4) 0.07 | 41.7 | 29.2 | 1.7 (0.52–5.7) 0.36 |
| Are you worried that your device might become blocked? | 44.4 | 32.4 | 1.7 (0.63–4.4)0.30 | 45.8 | 33.3 | 1.7 (0.52–5.4) 0.37 |
| Are you worried that your device might become infected? | 58.3 | 35.3 | 2.6 (0.97–6.7) 0.054 | 45.8 | 8.3 | 9.3 (1.8–48.7) 0.003 |
| Does your device bother you when performing your work-related activities? | 25.0 | 26.5 | 0.92 (0.31–2.7) 0.88 | 33.3 | 12.5 | 3.5 (0.80–15.3) 0.08 |
| Does your device bother you when you shower, bathe, or perform personal hygiene? | 75.9 | 24.1 | 6.1 (2.1–17.6) 0.001 | 88.2 | 11.8 | 18.3 (3.5–97.1) <0.0001 |
| Does your device bother you when you engage in sports or exercise? | 38.9 | 17.6 | 3.0 (1.0–9.0) 0.049 | 29.2 | 25.0 | 1.2 (0.34–4.4) 0.74 |
| Does your device bother you during social activities? | 16.7 | 11.8 | 1.5 (0.38–5.9) 0.55 | 12.5 | 4.2 | 3.2 (0.32–34.0) 0.60 |
| Does your device bother you when you are lying down in bed? | 50.0 | 35.3 | 1.8 (0.70–4.8) 0.21 | 41.7 | 33.3 | 1.4 (0.44–4.6) 0.55 |
| Does your device hurt? | 36.1 | 32.4 | 1.2 (0.44–3.2) 0.74 | 20.8 | 20.8 | 1.0 (0.24–4.0) 0.999 |
| Is device uncomfortable to touch? | 11.1 | 20.6 | 0.48 (0.13–1.8) 0.28 | 8.3 | 16.7 | 0.44 (0.08–2.8) 0.66 |
| QoL Constructs | 1 Month | p-Value | 3 Months | p-Value | ||
|---|---|---|---|---|---|---|
| PICC | Port | PICC | Port | |||
| Procedural scale a (pain and discomfort) | 2.8 ± 2.2 | 5.1 ± 2.9 | 0.001 | 2.8 ± 2.3 | 5.3 ± 2.9 | 0.001 |
| Psychosocial scale b (device causing trouble) | 5.4 ± 3.1 | 3.5 ± 3.3 | 0.016 | 6.0 ± 4.1 | 3.0 ± 2.7 | 0.005 |
| Treatment scale c | 2.2 ± 2.1 | 3.0 ± 2.1 | 0.13 | 2.1 ± 1.9 | 2.6 ± 2.0 | 0.39 |
| Satisfaction with device d | 23.6 ± 5.9 | 24.6 ± 7.1 | 0.51 | 25.0 ± 6.6 | 25.2 ± 5.9 | 0.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burbridge, B.; Lim, H.; Dwernychuk, L.; Le, H.; Asif, T.; Sami, A.; Ahmed, S. Comparison of the Quality of Life of Patients with Breast or Colon Cancer with an Arm Vein Port (TIVAD) Versus a Peripherally Inserted Central Catheter (PICC). Curr. Oncol. 2021, 28, 1495-1506. https://doi.org/10.3390/curroncol28020141
Burbridge B, Lim H, Dwernychuk L, Le H, Asif T, Sami A, Ahmed S. Comparison of the Quality of Life of Patients with Breast or Colon Cancer with an Arm Vein Port (TIVAD) Versus a Peripherally Inserted Central Catheter (PICC). Current Oncology. 2021; 28(2):1495-1506. https://doi.org/10.3390/curroncol28020141
Chicago/Turabian StyleBurbridge, Brent, Hyun Lim, Lynn Dwernychuk, Ha Le, Tehmina Asif, Amer Sami, and Shahid Ahmed. 2021. "Comparison of the Quality of Life of Patients with Breast or Colon Cancer with an Arm Vein Port (TIVAD) Versus a Peripherally Inserted Central Catheter (PICC)" Current Oncology 28, no. 2: 1495-1506. https://doi.org/10.3390/curroncol28020141
APA StyleBurbridge, B., Lim, H., Dwernychuk, L., Le, H., Asif, T., Sami, A., & Ahmed, S. (2021). Comparison of the Quality of Life of Patients with Breast or Colon Cancer with an Arm Vein Port (TIVAD) Versus a Peripherally Inserted Central Catheter (PICC). Current Oncology, 28(2), 1495-1506. https://doi.org/10.3390/curroncol28020141






