Benefit from Adjuvant TKIs Versus TKIs Plus Chemotherapy in EGFR-Mutant Stage III-pN2 Lung Adenocarcinoma
Abstract
1. Background
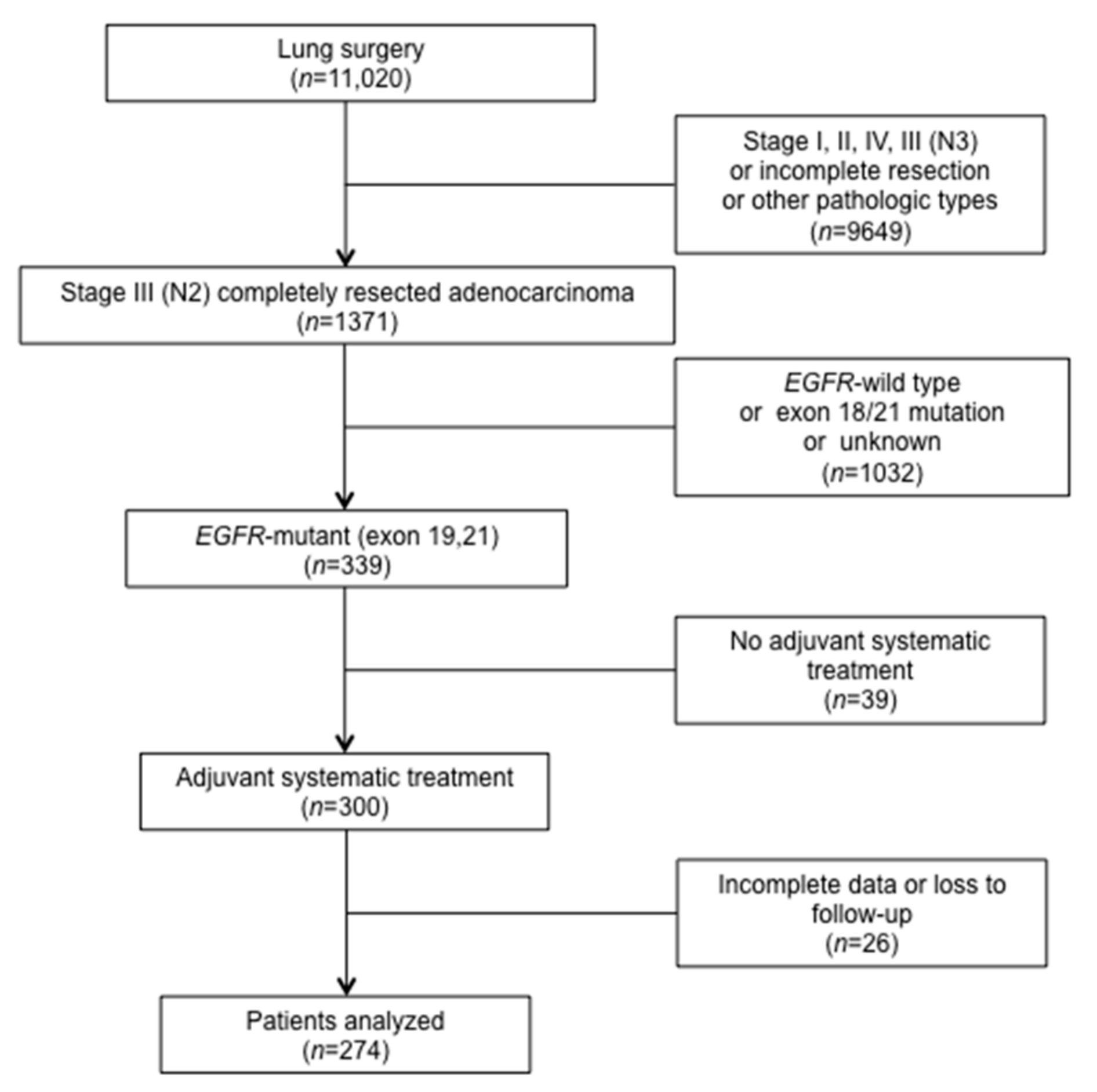
2. Methods
2.1. Study Population
2.2. N Sub-Staging
2.3. EGFR Genotyping
2.4. Pathologic Examination
2.5. Treatments
2.6. Follow-Up
2.7. Statistical Methods
3. Results
3.1. Patterns of Recurrence
3.2. Distant Metastasis and Prognostic Factors
3.3. Survival and Prognostic Factors
3.4. Additional Analysis
3.5. Toxicities
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Han, B.; Tjulandin, S.; Hagiwara, K.; Normanno, N.; Wulandari, L.; Laktionov, K.; Hudoyo, A.; He, Y.; Zhang, Y.P.; Wang, M.Z.; et al. EGFR mutation prevalence in Asia-Pacific and Russian patients with advanced NSCLC of adenocarcinoma and non-adenocarcinoma histology: The IGNITE study. Lung Cancer 2017, 113, 37–44. [Google Scholar] [CrossRef]
- Kelsey, C.R.; Marks, L.B.; Hollis, D.; Hubbs, J.L.; Ready, N.E.; D’Amico, T.A.; Boyd, J.A. Local recurrence after surgery for early stage lung cancer: An 11-year experience with 975 patients. Cancer 2009, 115, 5218–5227. [Google Scholar] [CrossRef]
- Walker, W.S.; Codispoti, M.; Soon, S.Y.; Stamenkovic, S.; Carnochan, F.; Pugh, G. Long-term outcomes following VATS lobectomy for non-small cell bronchogenic carcinoma. Eur J. Cardio-Thorac. Surg. 2003, 23, 397–402. [Google Scholar] [CrossRef]
- Cruz, C.; Afonso, M.; Oliveiros, B.; Pego, A. Recurrence and Risk Factors for Relapse in Patients with Non-Small Cell Lung Cancer Treated by Surgery with Curative Intent. Oncology 2017, 92, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Nelson, R.A.; Bogardus, A.; Grannis, F.W., Jr. Five-year lung cancer survival: Which advanced stage nonsmall cell lung cancer patients attain long-term survival? Cancer 2010, 116, 1518–1525. [Google Scholar] [CrossRef] [PubMed]
- Pignon, J.P.; Tribodet, H.; Scagliotti, G.V.; Douillard, J.Y.; Shepherd, F.A.; Stephens, R.J.; Dunant, A.; Torri, V.; Rosell, R.; Seymour, L.; et al. Lung adjuvant cisplatin evaluation: A pooled analysis by the LACE Collaborative Group. J. Clin. Oncol. 2008, 26, 3552–3559. [Google Scholar] [CrossRef]
- Goss, G.D.; O’Callaghan, C.; Lorimer, I.; Tsao, M.S.; Masters, G.A.; Jett, J.; Edelman, M.J.; Lilenbaum, R.; Choy, H.; Khuri, F.; et al. Gefitinib versus placebo in completely resected non-small-cell lung cancer: Results of the NCIC CTG BR19 study. J. Clin. Oncol. 2013, 31, 3320–3326. [Google Scholar] [CrossRef]
- Kelly, K.; Altorki, N.K.; Eberhardt, W.E.; OBrien, M.E.; Spigel, D.R.; Crinò, L.; Tsai, C.M.; Kim, J.H.; Cho, E.K.; Hoffman, P.C.; et al. Adjuvant Erlotinib Versus Placebo in Patients With Stage IB-IIIA Non-Small-Cell Lung Cancer (RADIANT): A Randomized, Double-Blind, Phase III Trial. J. Clin. Oncol. 2015, 33, 4007–4014. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.Z.; Wang, Q.; Mao, W.M.; Xu, S.T.; Wu, L.; Shen, Y.; Liu, Y.Y.; Chen, C.; Cheng, Y.; Xu, L.; et al. Gefitinib versus vinorelbine plus cisplatin as adjuvant treatment for stage II-IIIA (N1-N2) EGFR-mutant NSCLC (ADJUVANT/CTONG1104): A randomised, open-label, phase 3 study. Lancet Oncol. 2018, 19, 139–148. [Google Scholar] [CrossRef]
- Yue, D.; Xu, S.; Wang, Q.; Li, X.; Shen, Y.; Zhao, H.; Chen, C.; Mao, W.; Liu, W.; Liu, J.; et al. Erlotinib versus vinorelbine plus cisplatin as adjuvant therapy in Chinese patients with stage IIIA EGFR mutation-positive non-small-cell lung cancer (EVAN): A randomised, open-label, phase 2 trial. Lancet Respir. Med. 2018, 6, 863–873. [Google Scholar] [CrossRef]
- Wu, Y.L.; Tsuboi, M.; He, J.; John, T.; Grohe, C.; Majem, M.; Goldman, J.W.; Laktionov, K.; Kim, S.W.; Kato, T.; et al. Osimertinib in Resected EGFR-Mutated Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Giaccone, G.; Herbst, R.S.; Manegold, C.; Scagliotti, G.; Rosell, R.; Miller, V.; Natale, R.B.; Schiller, J.H.; Von Pawel, J.; Pluzanska, A.; et al. Gefitinib in combination with gemcitabine and cisplatin in advanced non-small-cell lung cancer: A phase III trial—INTACT 1. J. Clin. Oncol. 2004, 22, 777–784. [Google Scholar] [CrossRef]
- Herbst, R.S.; Giaccone, G.; Schiller, J.H.; Natale, R.B.; Miller, V.; Manegold, C.; Scagliotti, G.; Rosell, R.; Oliff, I.; Reeves, J.A.; et al. Gefitinib in combination with paclitaxel and carboplatin in advanced non-small-cell lung cancer: A phase III trial--INTACT 2. J. Clin. Oncol. 2004, 22, 785–794. [Google Scholar] [CrossRef]
- Herbst, R.S.; Prager, D.; Hermann, R.; Fehrenbacher, L.; Johnson, B.E.; Sandler, A.; Kris, M.G.; Tran, H.T.; Klein, P.; Li, X.; et al. TRIBUTE: A phase III trial of erlotinib hydrochloride (OSI-774) combined with carboplatin and paclitaxel chemotherapy in advanced non-small-cell lung cancer. J. Clin. Oncol. 2005, 23, 5892–5899. [Google Scholar] [CrossRef]
- Wen, M.; Xia, J.; Sun, Y.; Wang, X.; Fu, X.; Zhang, Y.; Zhang, Z.; Zhou, Y.; Li, X. Combination of EGFR-TKIs with chemotherapy versus chemotherapy or EGFR-TKIs alone in advanced NSCLC patients with EGFR mutation. Biologics 2018, 12, 183–190. [Google Scholar] [CrossRef]
- Oizumi, S.; Sugawara, S.; Minato, K.; Harada, T.; Inoue, A.; Fujita, Y.; Maemondo, M.; Watanabe, S.; Ito, K.; Gemma, A.; et al. Updated survival outcomes of NEJ005/TCOG0902: A randomised phase II study of concurrent versus sequential alternating gefitinib and chemotherapy in previously untreated non-small cell lung cancer with sensitive EGFR mutations. ESMO Open 2018, 3, e000313. [Google Scholar] [CrossRef]
- Edge, S.B.; Byrd, D.R.; Compton, C.C.; Fritz, A.G.; Greene, F.L.; Trotti, A. (Eds.) AJCC Cancer Staging Manual, 7th ed.; Springer: Chicago, IL, USA, 2009. [Google Scholar]
- Robinson, L.A.; Ruckdeschel, J.C.; Wagner, H.; Stevens, C.W. On behalf of the American College of Chest Physicians. Treatment of non-small cell lung cancer-stage IIIA—ACCP evidence-based clinical practice guidelines (2nd edition). Chest 2007, 132, 243S–265S. [Google Scholar] [CrossRef]
- Liu, S.R.; Qiu, B.; Yang, H.; Liang, Y.; Wang, F.; Liu, S.L.; Chen, Z.L.; Zhang, L.; Liu, M.Z.; Wang, S.Y.; et al. Epidermal Growth Factor Receptor Mutation Status and Treatment Outcome for R0-Resected Patients with Stage 3 Non-small Cell Lung Cancer. Ann. Surg. Oncol. 2016, 23, 2115–2122. [Google Scholar] [CrossRef]
- Ni, J.; Guo, T.; Li, Y.; Yang, X.; Li, Y.; Zou, L.; Chu, L.; Chu, X.; Li, S.; Ye, L.; et al. Patterns and risks of postoperative recurrence in completely resected EGFR-mutant non-small cell lung cancer: Prognostic significance of routine immunohistochemical markers. Transl. Lung Cancer Res. 2019, 8, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Wang, Z.; Liu, X.; Feng, S.; Dong, X.; Shi, X.; Wang, H.; Wu, H.; Xiong, G.; Wang, H.; et al. Differential effects of adjuvant EGFR tyrosine kinase inhibitors in patients with different stages of non-small-cell lung cancer after radical resection: An updated meta-analysis. Cancer Manag. Res. 2019, 11, 2677–2690. [Google Scholar] [CrossRef]
- Wu, Y.L.; Herbst, R.S.; Mann, H.; Rukazenkov, Y.; Marotti, M.; Tsuboi, M. ADAURA: Phase III, Double-blind, Randomized Study of Osimertinib Versus Placebo in EGFR Mutation-positive Early-stage NSCLC After Complete Surgical Resection. Clin. Lung Cancer 2018, 19, e533-6. [Google Scholar] [CrossRef]
- Laurila, N.; Koivunen, J.P. EGFR inhibitor and chemotherapy combinations for acquired TKI resistance in EGFR-mutant NSCLC models. Med. Oncol. 2015, 32, 205. [Google Scholar] [CrossRef]
- Gatzemeier, U.; Pluzanska, A.; Szczesna, A.; Kaukel, E.; Roubec, J.; De Rosa, F.; Milanowski, J.; Karnicka-Mlodkowski, H.; Pesek, M.; Serwatowski, P.; et al. Phase III study of erlotinib in combination with cisplatin and gemcitabine in advanced non-small-cell lung cancer: The Tarceva Lung Cancer Investigation Trial. J. Clin. Oncol. 2007, 25, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Yao, W.; Chu, Q.; Han, R.; Wang, Y.; Sun, J.; Wang, D.; Wang, Y.; Cao, M.; He, Y. Synergistic effects of metformin in combination with EGFR-TKI in the treatment of patients with advanced non-small cell lung cancer and type 2 diabetes. Cancer Lett. 2015, 369, 97–102. [Google Scholar] [CrossRef]
- Li, K.L.; Li, L.; Zhang, P.; Kang, J.; Wang, Y.B.; Chen, H.Y.; He, Y. A Multicenter Double-blind Phase II Study of Metformin With Gefitinib as First-line Therapy of Locally Advanced Non-Small-cell Lung Cancer. Clin. Lung Cancer 2017, 18, 340–343. [Google Scholar] [CrossRef]
- Seto, T.; Kato, T.; Nishio, M.; Goto, K.; Atagi, S.; Hosomi, Y.; Yamamoto, N.; Hida, T.; Maemondo, M.; Nakagawa, K.; et al. Erlotinib alone or with bevacizumab as first-line therapy in patients with advanced non-squamous non-small-cell lung cancer harbouring EGFR mutations (JO25567): An open-label, randomised, multicentre, phase 2 study. Lancet Oncol. 2014, 15, 1236–1244. [Google Scholar] [CrossRef]
- Park, C.; Lee, I.J.; Jang, S.H.; Lee, J.W. Factors affecting tumor recurrence after curative surgery for NSCLC: Impacts of lymphovascular invasion on early tumor recurrence. J. Thorac. Dis. 2014, 6, 1420–1428. [Google Scholar] [CrossRef]
- Brandt, W.S.; Bouabdallah, I.; Tan, K.S.; Park, B.J.; Adusumilli, P.S.; Molena, D.; Bains, M.S.; Huang, J.; Isbell, J.M.; Bott, M.J.; et al. Factors associated with distant recurrence following R0 lobectomy for pN0 lung adenocarcinoma. J. Thorac. Cardiovasc. Surg. 2018, 155, 1212–1224. [Google Scholar] [CrossRef]
- Yilmaz, A.; Duyar, S.S.; Cakir, E.; Aydin, E.; Demirag, F.; Karakaya, J.; Yazici, U.; Erdogan, Y. Clinical impact of visceral pleural, lymphovascular and perineural invasion in completely resected non-small cell lung cancer. Eur J. Cardiothorac. Surg. 2011, 40, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Luchini, C.; Veronese, N.; Nottegar, A.; Cheng, M.; Kaneko, T.; Pilati, C.; Tabbo, F.; Stubbs, B.; Pea, A.; Bagante, F.; et al. Extranodal extension of nodal metastases is a poor prognostic moderator in non-small cell lung cancer: A meta-analysis. Virchows Arch. 2018, 472, 939–947. [Google Scholar] [CrossRef]

| Characteristics | No. (%) |
|---|---|
| N = 274 (100) | |
| Age | |
| ≥60 | 116 (42.3) |
| <60 | 158 (57.7) |
| Sex | |
| Male | 101 (36.9) |
| Female | 173 (63.1) |
| KPS | |
| 90–100 | 270 (98.5) |
| 80 | 4 (1.5) |
| EGFR mutation | |
| Exon 19 | 145 (52.9) |
| Exon 21 | 129 (47.1) |
| Smoking | |
| Yes | 74 (27.0) |
| No | 200 (73.0) |
| N2 classification | |
| IIIA1-3 | 248 (90.5) |
| IIIA4 | 26 (9.5) |
| Surgery type | |
| Lobectomy | 164 (94.3) |
| Pneumonectomy | 5 (2.9) |
| Sublobectomy | 5 (2.9) |
| Missing data | 100 (/) |
| Visceral pleural invasion | |
| Yes | 78 (29.4) |
| No | 187 (70.6) |
| Missing data | 9 (/) |
| Lymphovascular invasion | |
| Yes | 90 (36.3) |
| No | 158 (63.7) |
| Missing data | 26 (/) |
| Perineural invasion | |
| Yes | 12 (5.2) |
| No | 219 (94.8) |
| Missing data | 43 (/) |
| Extranodal extension | |
| Yes | 80 (33.6) |
| No | 158 (66.4) |
| Missing data | 36 (/) |
| pT | |
| T1-2 | 246 (89.8) |
| T3-4 | 28 (10.2) |
| Adjuvant systemic therapy | |
| Adjuvant TKIs+chemotherapy | 23 (8.4) |
| Adjuvant TKIs | 52 (19.0) |
| Adjuvant chemotherapy | 199 (72.6) |
| PORT | |
| Yes | 23 (8.4) |
| No | 251 (91.6) |
| Regimen | No. (%) |
|---|---|
| Chemotherapy alone | 199 (100) |
| Pemetrexed+cisplatin/carboplatin | 165 (82.9) |
| Paclitaxel+carboplatin | 7 (3.5) |
| Docetaxel+cisplatin/nedaplatin | 7 (3.5) |
| Vinorelbine detartrate+cisplatin | 20 (10.1) |
| TKIs alone | 52 (100) |
| Gefitinib | 20 (38.5) |
| Erlotinib | 22 (42.3) |
| Afatinib | 1 (1.9) |
| Icotinib | 9 (17.3) |
| Combination treatment | 23 (100) |
| Chemotherapy | |
| Pemetrexed+cisplatin/carboplatin | 21 (91.3) |
| Paclitaxel+carboplatin | 1 (4.3) |
| Docetaxel+cisplatin/nedaplatin | 1 (4.3) |
| TKIs | |
| Gefitinib | 12 (52.2) |
| Erlotinib | 7 (30.4) |
| Icotinib | 4 (17.4) |
| Variable | DMFS | DFS | OS | ||||
|---|---|---|---|---|---|---|---|
| Univariable Analysis | Multivariable Analysis | Univariable Analysis | Multivariable Analysis | Univariable Analysis | |||
| p | HR (95% CI) | p | p | HR (95% CI) | p | p | |
| Age (≥60 vs. < 60) | 0.130 | 0.182 | 0.786 | ||||
| Sex (male vs. female) | 0.256 | 0.232 | 0.448 | ||||
| KPS (90–100 vs. 80) | 0.719 | 0.958 | 0.425 | ||||
| EGFR mutation (exon 19 vs. 21) | 0.134 | 0.025 | 0.90 (0.62–1.30) | 0.576 | <0.001 | ||
| Smoking (yes vs. no) | 0.505 | 0.522 | 0.374 | ||||
| N2 classification (IIIA1-3 vs. IIIA4) | 0.586 | 0.912 | 0.257 | ||||
| Surgery type (lobectomy vs. pneumonectomy vs. sublobectomy) | 0.520 | 0.526 | 0.198 | ||||
| Visceral pleural invasion (yes vs. no) | 0.066 | 1.14 (0.70–1.85) | 0.597 | 0.089 | 1.02 (0.61–1.71) | 0.940 | 0.534 |
| Lymphovascular invasion (yes vs. no) | <0.001 | 2.22 (1.38–3.57) | 0.001 | 0.013 | 1.44 (0.95–2.18) | 0.086 | 0.319 |
| Perineural invasion (yes vs. no) | 0.387 | 0.555 | 0.593 | ||||
| Extranodal extension (yes vs. no) | 0.005 | 1.35 (0.85–2.14) | 0.198 | 0.004 | 1.45 (0.96–2.19) | 0.078 | 0.550 |
| pT (T1-2 vs. T3-4) | 0.390 | 0.723 | 0.410 | ||||
| Adjuvant systemic therapy | 0.006 | 0.016 | <0.001 | 0.002 | 0.413 | ||
| TKIs vs. TKIs+chemotherapy | 0.008 | 0.40 (0.17–0.94) | 0.036 | 0.009 | 0.42 (0.19–0.94) | 0.034 | 0.602 |
| TKIs vs. chemotherapy | 0.002 | 0.38 (0.20–0.73) | 0.004 | <0.001 | 0.33 (0.18–0.61) | <0.001 | 0.187 |
| PORT (Yes vs. No) | 0.288 | 0.356 | |||||
| Grade 3–4 Toxicity | Chemotherapy Alone (N = 199) | TKIs Alone (N = 52) | Chemotherapy+TKIs (N = 23) |
|---|---|---|---|
| No. (%) | No. (%) | No. (%) | |
| Any Grade 3–4 toxicity | 50 (25.1) | 5 (9.6) | 4 (17.4) |
| Anemia | 9 (4.5) | 1 (1.9) | 0 (0) |
| Leukopenia | 33 (16.6) | 0 (0) | 3 (13.0) |
| Neutropenia | 20 (10.1) | 0 (0) | 1 (4.3) |
| Thrombocytopenia | 13 (6.5) | 0 (0) | 1 (4.3) |
| Elevated ALT | 2 (1.0) | 1 (1.9) | 1 (4.3) |
| Elevated AST | 2 (1.0) | 1 (1.9) | 0 (0) |
| Rash | 2 (1.0) | 2 (3.8) | 0 (0) |
| Diarrhea | 2 (1.0) | 1 (1.9) | 1 (4.3) |
| Vomiting | 8 (4.3) | 0 (0) | 0 (0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Ma, L.; Qiu, B.; Wen, Y.; Liang, W.; Hu, W.; Chen, N.; Zhang, T.; Xu, S.; Chen, L.; et al. Benefit from Adjuvant TKIs Versus TKIs Plus Chemotherapy in EGFR-Mutant Stage III-pN2 Lung Adenocarcinoma. Curr. Oncol. 2021, 28, 1424-1436. https://doi.org/10.3390/curroncol28020135
Li Q, Ma L, Qiu B, Wen Y, Liang W, Hu W, Chen N, Zhang T, Xu S, Chen L, et al. Benefit from Adjuvant TKIs Versus TKIs Plus Chemotherapy in EGFR-Mutant Stage III-pN2 Lung Adenocarcinoma. Current Oncology. 2021; 28(2):1424-1436. https://doi.org/10.3390/curroncol28020135
Chicago/Turabian StyleLi, Qiwen, Li Ma, Bo Qiu, Yuzhi Wen, Wenhua Liang, Wanming Hu, Naibin Chen, Tian Zhang, Shuangbing Xu, Lingjuan Chen, and et al. 2021. "Benefit from Adjuvant TKIs Versus TKIs Plus Chemotherapy in EGFR-Mutant Stage III-pN2 Lung Adenocarcinoma" Current Oncology 28, no. 2: 1424-1436. https://doi.org/10.3390/curroncol28020135
APA StyleLi, Q., Ma, L., Qiu, B., Wen, Y., Liang, W., Hu, W., Chen, N., Zhang, T., Xu, S., Chen, L., Guo, M., Zhao, Y., Liu, S., Guo, J., Wang, J., Wang, S., Wang, X., Pang, Q., Long, H., & Liu, H. (2021). Benefit from Adjuvant TKIs Versus TKIs Plus Chemotherapy in EGFR-Mutant Stage III-pN2 Lung Adenocarcinoma. Current Oncology, 28(2), 1424-1436. https://doi.org/10.3390/curroncol28020135





