A Rare Coexistence of Paraneoplastic Cerebellar Degeneration: Papillary Thyroid Carcinoma
Abstract
1. Introduction
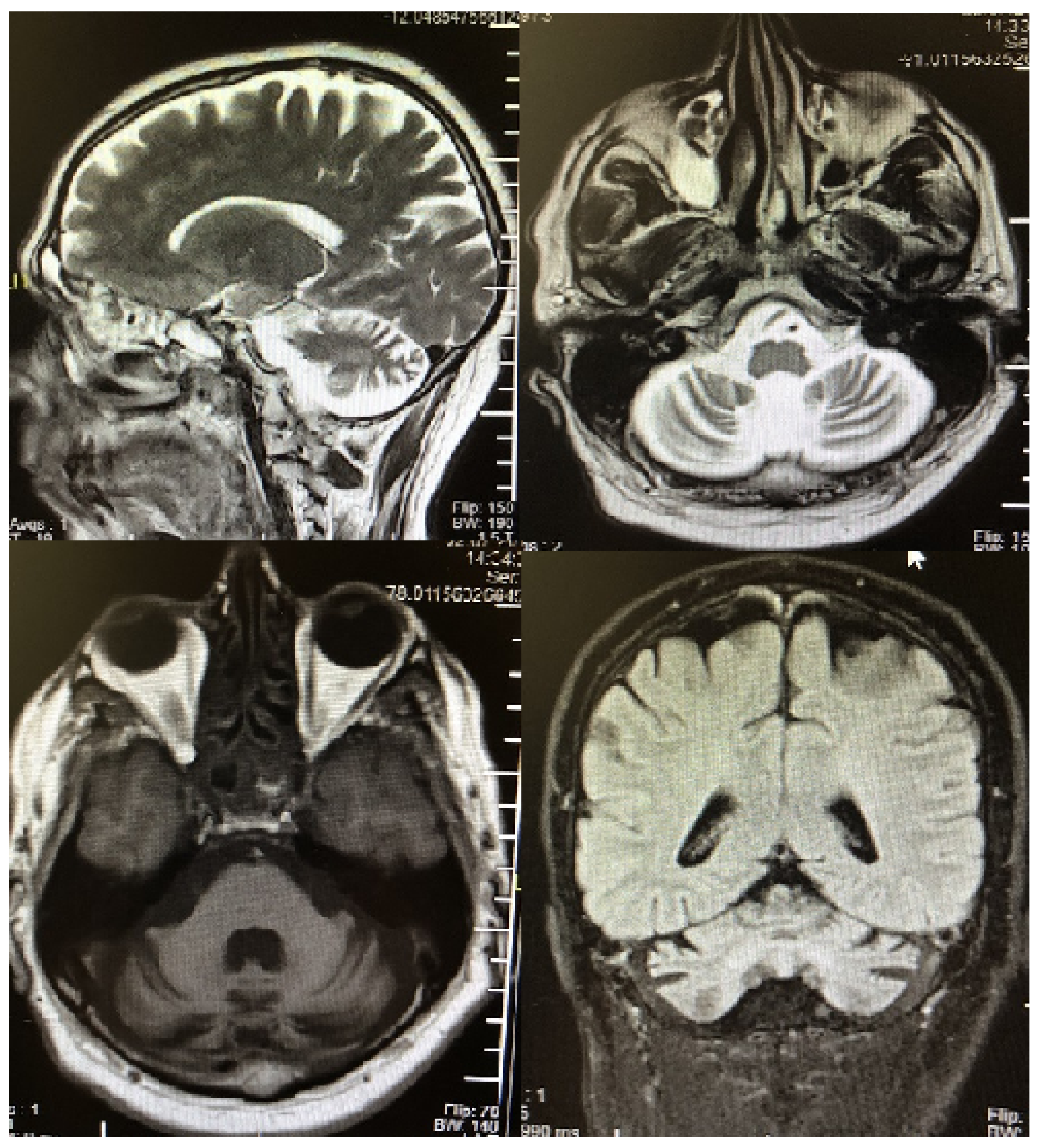
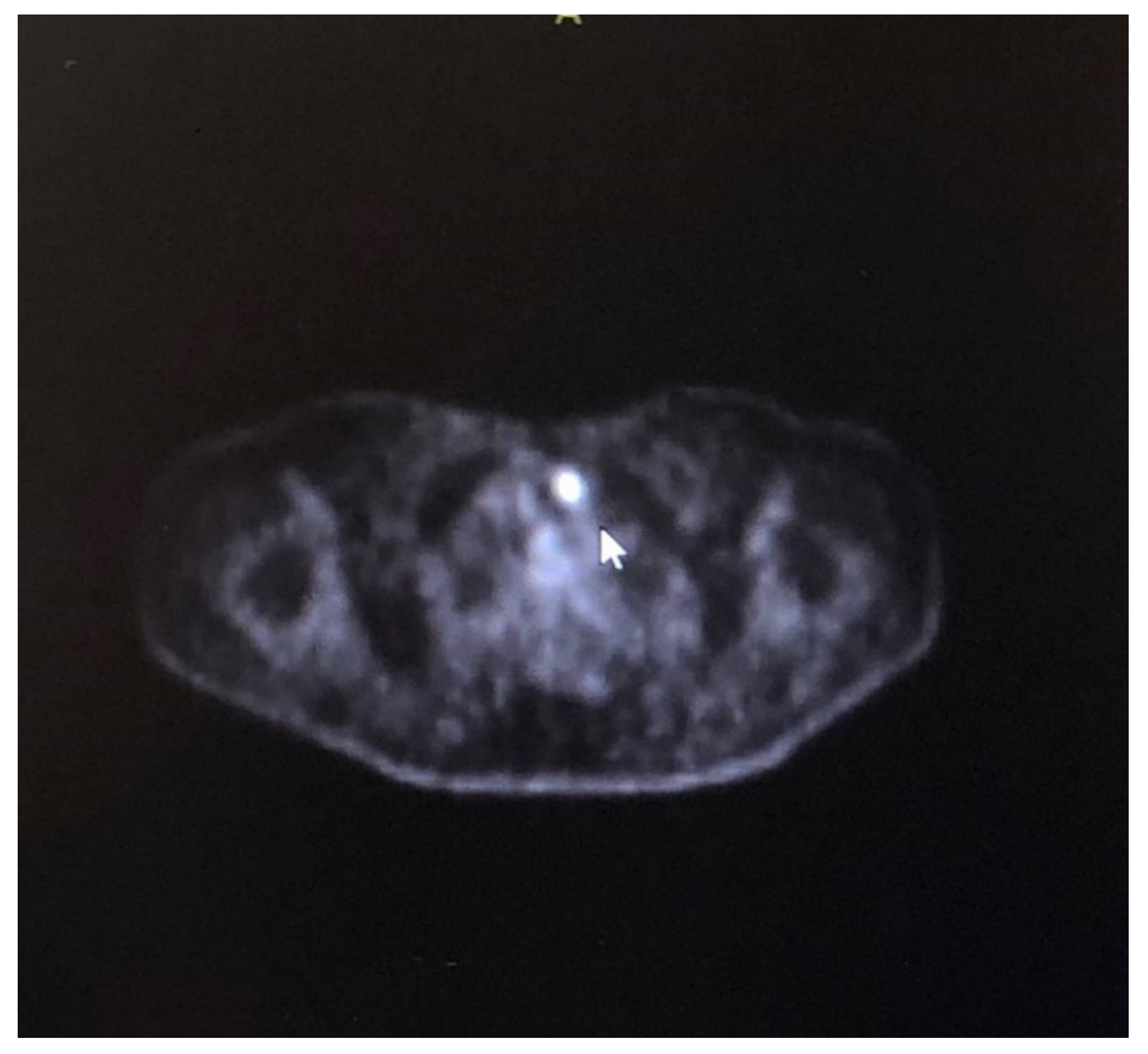
2. Case Description
3. Discussion
Author Contributions
Funding
Ethics Informed Consent
Conflicts of Interest
References
- Anderson, N.E.; Rosenblum, M.K.; Posner, J.B. Paraneoplastic cerebellar degeneration: Clinical-immunological correlations. Ann. Neurol. 1988, 24, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Graus, F.; Delattre, J.Y.; Antoine, J.C.; Dalmau, J.; Giometto, B.; Grisold, W.; Honnorat, J.; Smitt, P.S.; Vedeler, C.H.; Verschuuren, J.J.; et al. Recommended diagnostic criteria for paraneoplastic neurological syndromes. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1135–1140. [Google Scholar] [CrossRef] [PubMed]
- Papanicolaou Society of Cytopathology Recommendations for Thyroid Fine Needle Aspiration. Available online: www.papsociety.org/guidelines.html (accessed on 27 July 2007).
- Odell, W.D. Paraneoplastic Syndromes. In Holland-Frei Cancer Medicine; Bast, R.C., Jr., Kufe, D.W., Pollock, R.E., Weichselbaum, R.R., Holland, J.F., Frei, E., III, Eds.; BC Decker: Hamilton, ON, Canada, 2003. [Google Scholar]
- Yshii, L.M.; Gebauer, C.M.; Pignolet, B.; Maure, E.; Queriault, C.; Pierau, M.; Saito, H.; Suzuki, N.; Brunner-Weinzierl, M.; Bauer, J.; et al. CTLA4 blockade elicits paraneoplastic neurological disease in a mouse model. Brain 2016, 139, 2923–2934. [Google Scholar] [CrossRef] [PubMed]
- Panja, D.; Vedeler, C.A.; Schubert, M. Paraneoplastic cerebellar degeneration: Yo antibody alters mitochondrial calcium buffering capacity. Neuropathol. Appl. Neurobiol. 2019, 45, 141–156. [Google Scholar] [CrossRef] [PubMed]
- Kroiss, A.; Uprimny, C.; Virgolini, I.J. Thyroid carcinoma detected by incidental (18)F-FDG uptake in a patient with progressive cerebellar syndrome. Endocrine 2016, 51, 199–200. [Google Scholar] [CrossRef] [PubMed]
- Gratwicke, J.P.; Alli, A.; Rollin, M.; Vaz, F.; Rees, J.; Vincent, A.; Edwards, M. Paraneoplastic cerbellar syndrome and sensory ganglionopathy with papillary thyroid carcinoma. J. Neurol. Sci. 2014, 15341, 183–184. [Google Scholar] [CrossRef] [PubMed]
- Vogrig, A.; Bernardini, A.; Gigli, G.L.; Corazza, E.; Marini, A.; Segatti, S.; Fabris, M.; Honnorat, J.; Valente, M. Stroke-like presentation of paraneoplastic cerebellar degeneration: A single-center experience and review of the literature. Cerebellum 2019, 18, 976–982. [Google Scholar] [CrossRef] [PubMed]
- Hezer, S.; Descargues, G.; Vignals, C.; Imbert, Y. Paraneoplastic cerebellar degeneration and primary fallopian cancer. Gynecol. Obstet. Fertil. Senol. 2019, 47, 610–612. [Google Scholar] [PubMed]
- Le May, M.; Dent, S. Anti-Yo antibody-mediated paraneoplastic cerebellar degeneration associated with cognitive affective syndrome in a patient with breast cancer: A case report and literature review. Curr. Oncol. 2018, 25, e585–e591. [Google Scholar] [CrossRef] [PubMed]
- Shams’ili, S.; Grefkens, J.; de Leeuw, B.; van den Bent, M.; Hooijkaas, H.; van der Holt, B.; Vecht, C.; Sillevis Smitt, P. Paraneoplastic cerebellar degeneration associated with antineuronal antibodies: Analysis of 50 patients. Brain 2003, 126, 1409–1418. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, D.M.; Natale, R.B.; Cody, R.L. Breast cancer and paraneoplastic cerebellar degeneration. Cancer 1991, 68, 1835–1841. [Google Scholar]
- Hens, M.J.; Camacho, B.; Maestre, A.; Gonzalez, V.; Villegas, I.; Magana, M. Renal cell carcinoma presenting as paraneoplastic neurological dysfuntion. Actas Urol. Esp. 2008, 32, 645–648. [Google Scholar] [CrossRef]
- Darnell, R.B. Onconeural antigens and the paraneoplastic neurologic disorders: At the intersection of cancer, immunity, and the brain. Proc. Natl. Acad. Sci. USA 1996, 93, 4529–4545. [Google Scholar] [CrossRef] [PubMed]
- Rojas, I.; Graus, F.; Keime-Guibert, F.; Rene, R.; Delattre, J.Y.; Ramon, J.M.; Dalmau, J.; Posner, J.B. Long-term clinical outcome of paraneoplastic cerebellar degeneration and anti-Yo antibodies. Neurology 2000, 55, 713–715. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ayas, Z.Ö.; Uncu, G. A Rare Coexistence of Paraneoplastic Cerebellar Degeneration: Papillary Thyroid Carcinoma. Curr. Oncol. 2021, 28, 560-564. https://doi.org/10.3390/curroncol28010055
Ayas ZÖ, Uncu G. A Rare Coexistence of Paraneoplastic Cerebellar Degeneration: Papillary Thyroid Carcinoma. Current Oncology. 2021; 28(1):560-564. https://doi.org/10.3390/curroncol28010055
Chicago/Turabian StyleAyas, Zeynep Özözen, and Gülgün Uncu. 2021. "A Rare Coexistence of Paraneoplastic Cerebellar Degeneration: Papillary Thyroid Carcinoma" Current Oncology 28, no. 1: 560-564. https://doi.org/10.3390/curroncol28010055
APA StyleAyas, Z. Ö., & Uncu, G. (2021). A Rare Coexistence of Paraneoplastic Cerebellar Degeneration: Papillary Thyroid Carcinoma. Current Oncology, 28(1), 560-564. https://doi.org/10.3390/curroncol28010055




