Implantable Cardioverter-Defibrillators in Poland Compared with other European Countries from the Patient’s Perspective: Insights from the EHRA Patient Survey
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
3.1. Patient Population
3.2. Quality of Life
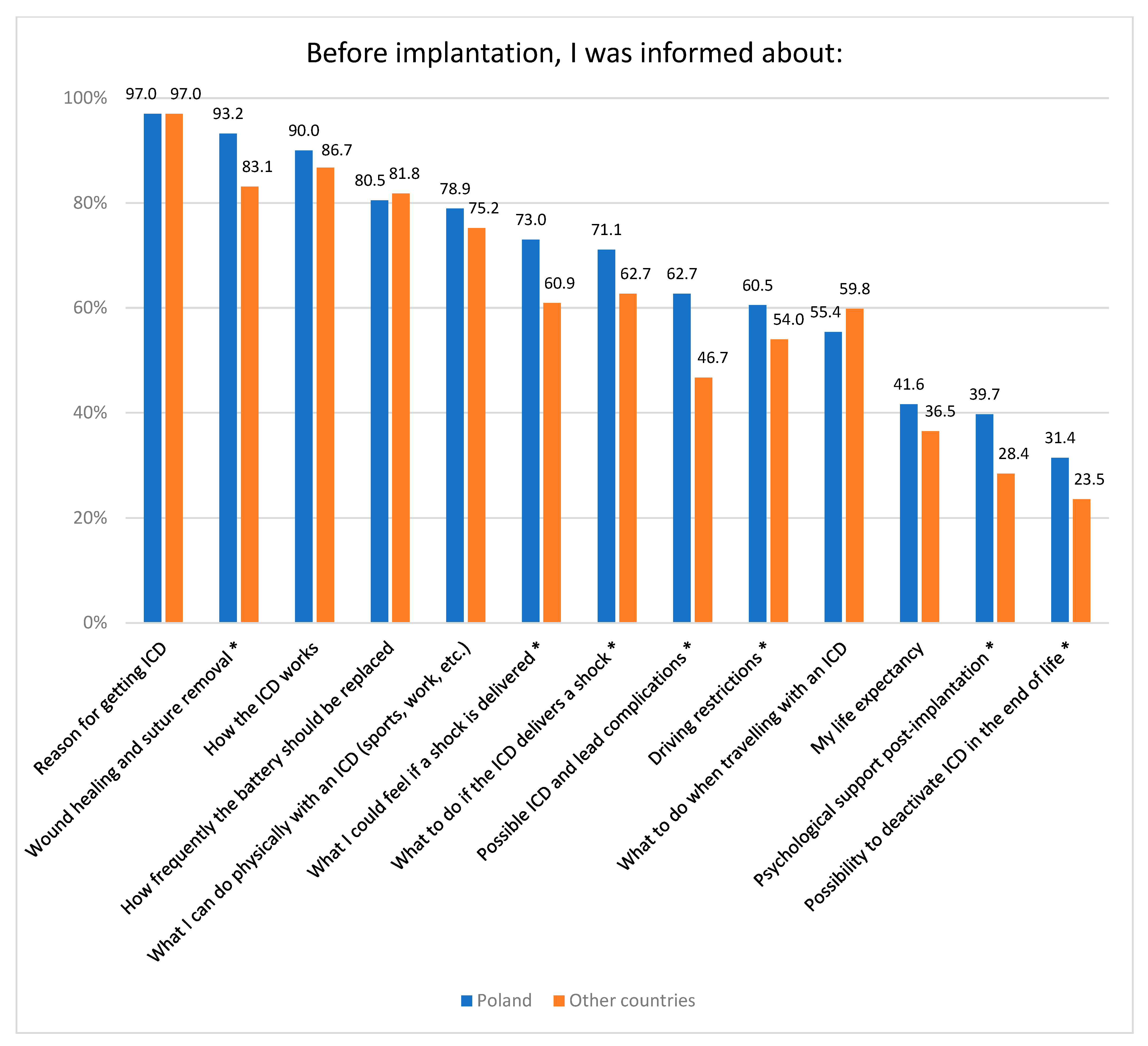
3.3. Information Provided to Patients before Implantation
3.4. Patients’ Needs
3.5. End of Life
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zeppenfeld, K.; Tfelt-Hansen, J.; de Riva, M.; Winkel, B.G.; Behr, E.R.; Blom, N.A.; Charron, P.; Corrado, D.; Dagres, N.; de Chillou, C.; et al. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur. Heart J. 2022, 43, 3997–4126. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, S.S.; Nielsen, J.C.; Wehberg, S.; Jørgensen, O.D.; Riahi, S.; Haarbo, J.; Philbert, B.T.; Larsen, M.L.; Johansen, J.B. New onset anxiety and depression in patients with an implantable cardioverter defibrillator during 24 months of follow-up (data from the national DEFIB-WOMEN study). Gen. Hosp. Psychiatry 2021, 72, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Krauze, N.; Smolis-Bąk, E.; Kowalik, I.; Sterliński, M. Patient-Reported Quality of Life, Depression, Anxiety, and Physical Activity in Patients Receiving an Implantable Cardioverter-Defibrillator for Primary versus Secondary Prevention: A Single-Centre, Prospective, Observational Cohort Study. Int. J. Environ. Res. Public Health 2022, 19, 12830. [Google Scholar] [CrossRef] [PubMed]
- Karczewska, O.; Młynarska, A. Factors That Cause Concerns after Cardioverter Defibrillator Implantation. Int. J. Environ. Res. Public Health 2021, 18, 6095. [Google Scholar] [CrossRef] [PubMed]
- Obrova, J.; Sovova, E.; Ivanova, K.; Furstova, J.; Taborsky, M. Let It Beat: How Lifestyle and Psychosocial Factors Affect the Risk of Sudden Cardiac Death—A 10-Year Follow-Up Study. Int. J. Environ. Res. Public Health 2022, 19, 2627. [Google Scholar] [CrossRef] [PubMed]
- MedTech Europe. Statistics for Cardiac Rhythm Management Products. Available online: https://www.medtecheurope.org/wp-content/uploads/2016/03/crm-charts-2020.pdf (accessed on 17 July 2022).
- Pedersen, S.B.; Farkas, D.K.; Hjortshøj, S.P.; Bøtker, H.E.; Johansen, J.B.; Philbert, B.T.; Haarbo, J.; Thomsen, R.W.; Nielsen, J.C. Significant regional variation in use of implantable cardioverter-defibrillators in Denmark. Eur. Heart J. Qual. Care Clin. Outcomes 2019, 5, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Matlock, D.D.; Peterson, P.N.; Heidenreich, P.A.; Lucas, F.L.; Malenka, D.J.; Wang, Y.; Curtis, J.P.; Kutner, J.S.; Fisher, E.S.; Masoudi, F.A. Regional variation in the use of implantable cardioverter-defibrillators for primary prevention: Results from the National Cardiovascular Data Registry. Circ. Cardiovasc. Qual. Outcomes 2011, 4, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Januszkiewicz, Ł.; Barra, S.; Providencia, R.; Conte, G.; de Asmundis, C.; Chun, J.K.R.; Farkowski, M.; Guerra, J.; Marijon, E.; Boveda, S. Long-term quality of life and acceptance of implantable cardioverter-defibrillator therapy: Results of the European Heart Rhythm Association survey. Europace 2022, 24, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Hindricks, G.; Taborsky, M.; Glikson, M.; Heinrich, U.; Schumacher, B.; Katz, A.; Brachmann, J.; Lewalter, T.; Goette, A.; Block, M.; et al. Implant-based multiparameter telemonitoring of patients with heart failure (IN-TIME): A randomised controlled trial. Lancet 2014, 384, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Tajstra, M.; Sokal, A.; Gadula-Gacek, E.; Kurek, A.; Wozniak, A.; Niedziela, J.; Adamowicz-Czoch, E.; Rozentryt, P.; Milewski, K.; Jachec, W.; et al. Remote Supervision to Decrease Hospitalization Rate (RESULT) study in patients with implanted cardioverter-defibrillator. Europace 2020, 22, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Glikson, M.; Nielsen, J.C.; Kronborg, M.B.; Michowitz, Y.; Auricchio, A.; Barbash, I.M.; Barrabés, J.A.; Boriani, G.; Braunschweig, F.; Brignole, M.; et al. 2021 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy. Eur. Heart J. 2021, 42, 3427–3520. [Google Scholar] [CrossRef] [PubMed]
- Slotwiner, D.; Varma, N.; Akar, J.G.; Annas, G.; Beardsall, M.; Fogel, R.I.; Galizio, N.O.; Glotzer, T.V.; Leahy, R.A.; Love, C.J.; et al. HRS Expert Consensus Statement on remote interrogation and monitoring for cardiovascular implantable electronic devices. Heart Rhythm 2015, 12, e69–e100. [Google Scholar] [CrossRef] [PubMed]
- Piotrowicz, R.; Krzesiński, P.; Balsam, P.; Piotrowicz, E.; Kempa, M.; Lewicka, E.; Główczyńska, R.; Grabowski, M.; Kołtowski, Ł.; Peller, M.; et al. Telemedicine solutions in cardiology: A joint expert opinion by the Information Technology and Telemedicine Committee of the Polish Cardiac Society, the Section of Noninvasive Electrocardiology and Telemedicine of the Polish Cardiac Society, and the Clinical Research Committee of the Polish Academy of Sciences (short version, 2021). Kardiol. Pol. 2021, 79, 227–241. [Google Scholar] [PubMed]
- Clinical Standards and Guidelines for the Follow up of Cardiac Implantable Electronic Devices (CIEDs) for Cardiac Rhythm Management. Available online: https://bhrs.com/wp-content/uploads/2022/06/BHRS-CIED-FU-Standards-June22.pdf (accessed on 12 November 2022).
- Maciąg, A.; Mitkowski, P.; Mazurek, M.; Kaźmierczak, J.; Nowak, K.; Grabowski, M.; Oręziak, A.; Kempa, M.; Bacior, B.; Gepner, K.; et al. Patient perspective and safety of remote monitoring of implantable cardioverter-defibrillators in the Polish Nationwide Multicenter Registry: The Medtronic CareLink network evaluation. Kardiol. Pol. 2020, 78, 1115–1121. [Google Scholar] [CrossRef] [PubMed]
- Simovic, S.; Providencia, R.; Barra, S.; Kircanski, B.; Guerra, J.M.; Conte, G.; Duncker, D.; Marijon, E.; Anic, A.; Boveda, S. The use of remote monitoring of cardiac implantable devices during the COVID-19 pandemic: An EHRA physician survey. Europace 2022, 24, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Mairesse, G.H.; Braunschweig, F.; Klersy, K.; Cowie, M.R.; Leyva, F. Implementation and reimbursement of remote monitoring for cardiac implantable electronic devices in Europe: A survey from the health economics committee of the European Heart Rhythm Association. Europace 2015, 17, 814–818. [Google Scholar] [CrossRef] [PubMed]
- Tajstra, M.; Wojtaszczyk, A.; Sterliński, M.; Świerżyńska, E.; Szumowski, Ł.; Tomasiuk, M.; Grabowski, M.; Januszkiewicz, Ł.J.; Romanek, J.; Romanek, J.; et al. Patients with heart failure and an implanted cardioverter-defibrillator during the coronavirus disease 2019 pandemic: Insights from a multicenter registry in Poland. Kardiol. Pol. 2021, 79, 562–565. [Google Scholar] [PubMed]
- Boriani, G.; Burri, H.; Svennberg, E.; Imberti, J.F.; Merino, J.L.; Leclercq, C. Current status of reimbursement practices for remote monitoring of cardiac implantable electrical devices across Europe. Europace 2022, 24, 1875–1880. [Google Scholar] [CrossRef] [PubMed]
- Tajstra, M.; Dyrbuś, M.; Grabowski, M.; Rokicki, J.K.; Nowak, M.; Gąsior, M. The use of remote monitoring of patients with cardiac implantable electronic devices in Poland. Kardiol. Pol. 2022, 80, 479–481. [Google Scholar] [CrossRef] [PubMed]
- Regulation of the Minister of Health Regarding Remote Monitoring Reimbursement in Poland. Available online: https://dziennikustaw.gov.pl/D2022000264101.pdf (accessed on 6 March 2023).
- Kim, J.S.; Park, H.W.; An, M.; Shim, J.L. Exploring Advance Directive Perspectives and Associations with Preferences for End-of-Life Life-Sustaining Treatments among Patients with Implantable Cardioverter-Defibrillators. Int. J. Environ. Res. Public Health 2020, 17, 4257. [Google Scholar] [CrossRef] [PubMed]

| N (%) Or Median, IQR | Whole Cohort 1809 (100) | Poland 410 (22.7) | Other Countries 1399 (77.3) | p-Value |
|---|---|---|---|---|
| Age | <0.001 | |||
| 0-20 | 7 (0.4) | 2 (0.5) | 5 (0.4) | |
| 21-40 | 271 (15.0) | 88 (21.5) | 183 (13.1) | |
| 41-60 | 682 (37.7) | 128 (31.2) | 554 (39.6) | |
| 61-80 | 784 (43.3) | 174 (42.4) | 610 (43.6) | |
| ≥81 | 65 (3.6) | 18 (4.4) | 47 (3.4) | |
| Sex (% female) | 624 (34.5) | 164 (40.0) | 460 (32.9) | 0.008 |
| Education level | <0.001 | |||
| Primary school | 147 (8.1) | 45 (11.0) | 102 (7.3) | |
| Secondary school | 608 (33.6) | 234 (57.1) | 374 (26.7) | |
| College | 381 (21.1) | 36 (8.8) | 345 (24.7) | |
| University | 673 (37.2) | 95 (23.2) | 578 (41.3) | |
| Employment status | <0.001 | |||
| Student | 21 (1.2) | 4 (1.0) | 17 (1.2) | |
| Employed | 715 (39.5) | 150 (36.6) | 565 (40.4) | |
| Not employed | 186 (10.3) | 44 (10.7) | 142 (10.2) | |
| Retired | 887 (49.0) | 212 (51.7) | 675 (48.2) | |
| Marital status | 0.006 | |||
| Married or living with a partner | 1351 (74.7) | 317 (77.3) | 1034 (73.9) | |
| Single | 320 (17.7) | 51 (12.4) | 269 (19.2) | |
| Widower or widow | 87 (4.8) | 29 (7.1) | 58 (4.1) | |
| Living at home (as a child) | 8 (0.4) | 2 (0.5) | 6 (0.4) | |
| Living alone with children | 43 (2.4) | 11 (2.7) | 32 (2.3) | |
| Time from first ICD implantation | 5 (2-10) | 5 (2-9) | 5 (2-10) | 0.620 |
| Device type | 0.573 | |||
| ICD-VR, ICD-DR | 829 (45.8) | 181 (44.1) | 648 (46.3) | |
| S-ICD | 563 (31.1) | 124 (30.2) | 439 (31.4) | |
| CRT-D | 281 (15.5) | 70 (17.1) | 211 (15.1) | |
| Do not know | 136 (7.5) | 35 (8.5) | 101 (7.2) | |
| Remote monitoring | <0.001 | |||
| Yes | 1021 (56.4) | 86 (21.0) | 935 (66.8) | |
| No | 675 (37.3) | 267 (65.1) | 408 (29.2) | |
| Do not know | 113 (6.2) | 57 (13.9) | 56 (4.0) | |
| ICD indication | ||||
| Post-cardiac arrest (secondary prevention) | 583 (32.2) | 136 (33.2) | 447 (32.0) | 0.642 |
| Prevention of sudden death | 808 (44.7) | 147 (35.9) | 661 (47.2) | <0.001 |
| Heart failure symptoms | 665 (36.8) | 197 (48.0) | 468 (33.5) | <0.001 |
| Do not know | 54 (3.0) | 14 (3.4) | 40 (2.9) | 0.561 |
| Complications | ||||
| None | 1404 (77.6) | 308 (75.1) | 1096 (78.3) | 0.169 |
| Inappropriate shocks | 209 (11.6) | 58 (14.1) | 151 (10.8) | 0.062 |
| Malfunctioning lead | 204 (11.3) | 52 (12.7) | 152 (10.9) | 0.306 |
| Unplanned re-operations | 128 (7.1) | 31 (7.6) | 97 (6.9) | 0.663 |
| N (%) | Whole Cohort 1809 (100) | Poland 410 (22.7) | Other Countries 1399 (77.3) | p-Value |
|---|---|---|---|---|
| Quality of life after ICD implantation | 0.041 | |||
| Improved | 829 (45.8) | 209 (51.0) | 620 (44.3) | |
| Unchanged | 675 (37.3) | 135 (32.9) | 540 (38.6) | |
| Worsened | 183 (10.1) | 45 (11.0) | 138 (9.9) | |
| I am not sure | 122 (6.7) | 21 (5.1) | 101 (7.2) | |
| Feeling of safety with an ICD | 1302 (80.3) | 299 (80.8) | 1003 (80.2) | 0.787 |
| Feeling of global depression since ICD implantation | 0.837 | |||
| Not at all | 928 (57.2) | 207 (55.9) | 721 (57.6) | |
| Slightly | 362 (22.3) | 88 (23.8) | 274 (21.9) | |
| Moderately | 235 (14.5) | 51 (13.8) | 184 (14.7) | |
| Very | 66 (4.1) | 15 (4.1) | 51 (4.1) | |
| Extremely | 31 (1.9) | 9 (2.4) | 22 (1.8) | |
| Acceptance of ICD limitations | 1119 (69.2) | 255 (68.9) | 864 (69.3) | 0.893 |
| Significant lifestyle change after ICD implantation | 563 (34.8) | 126 (34.1) | 437 (35.1) | 0.718 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Januszkiewicz, Ł.; Grabowski, M.; Farkowski, M.M.; Życiński, P.; Jędrychowski, T.; Pytkowski, M.; Chun, J.K.R.; Guerra, J.M.; Conte, G.; Barra, S.; et al. Implantable Cardioverter-Defibrillators in Poland Compared with other European Countries from the Patient’s Perspective: Insights from the EHRA Patient Survey. Int. J. Environ. Res. Public Health 2023, 20, 5045. https://doi.org/10.3390/ijerph20065045
Januszkiewicz Ł, Grabowski M, Farkowski MM, Życiński P, Jędrychowski T, Pytkowski M, Chun JKR, Guerra JM, Conte G, Barra S, et al. Implantable Cardioverter-Defibrillators in Poland Compared with other European Countries from the Patient’s Perspective: Insights from the EHRA Patient Survey. International Journal of Environmental Research and Public Health. 2023; 20(6):5045. https://doi.org/10.3390/ijerph20065045
Chicago/Turabian StyleJanuszkiewicz, Łukasz, Marcin Grabowski, Michał Mirosław Farkowski, Paweł Życiński, Tomasz Jędrychowski, Mariusz Pytkowski, Julian K. R. Chun, Jose M. Guerra, Giulio Conte, Sérgio Barra, and et al. 2023. "Implantable Cardioverter-Defibrillators in Poland Compared with other European Countries from the Patient’s Perspective: Insights from the EHRA Patient Survey" International Journal of Environmental Research and Public Health 20, no. 6: 5045. https://doi.org/10.3390/ijerph20065045
APA StyleJanuszkiewicz, Ł., Grabowski, M., Farkowski, M. M., Życiński, P., Jędrychowski, T., Pytkowski, M., Chun, J. K. R., Guerra, J. M., Conte, G., Barra, S., Boveda, S., & Kempa, M. (2023). Implantable Cardioverter-Defibrillators in Poland Compared with other European Countries from the Patient’s Perspective: Insights from the EHRA Patient Survey. International Journal of Environmental Research and Public Health, 20(6), 5045. https://doi.org/10.3390/ijerph20065045






