Occurrence of Hepatitis A Virus in Water Matrices: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol and Registration
2.2. Eligibility Criteria
2.3. Inclusion and Exclusion Criteria
2.4. Information about Searches
2.5. Data Extraction and Management
2.6. Quality Assessment
2.7. Statistical Analysis
3. Results
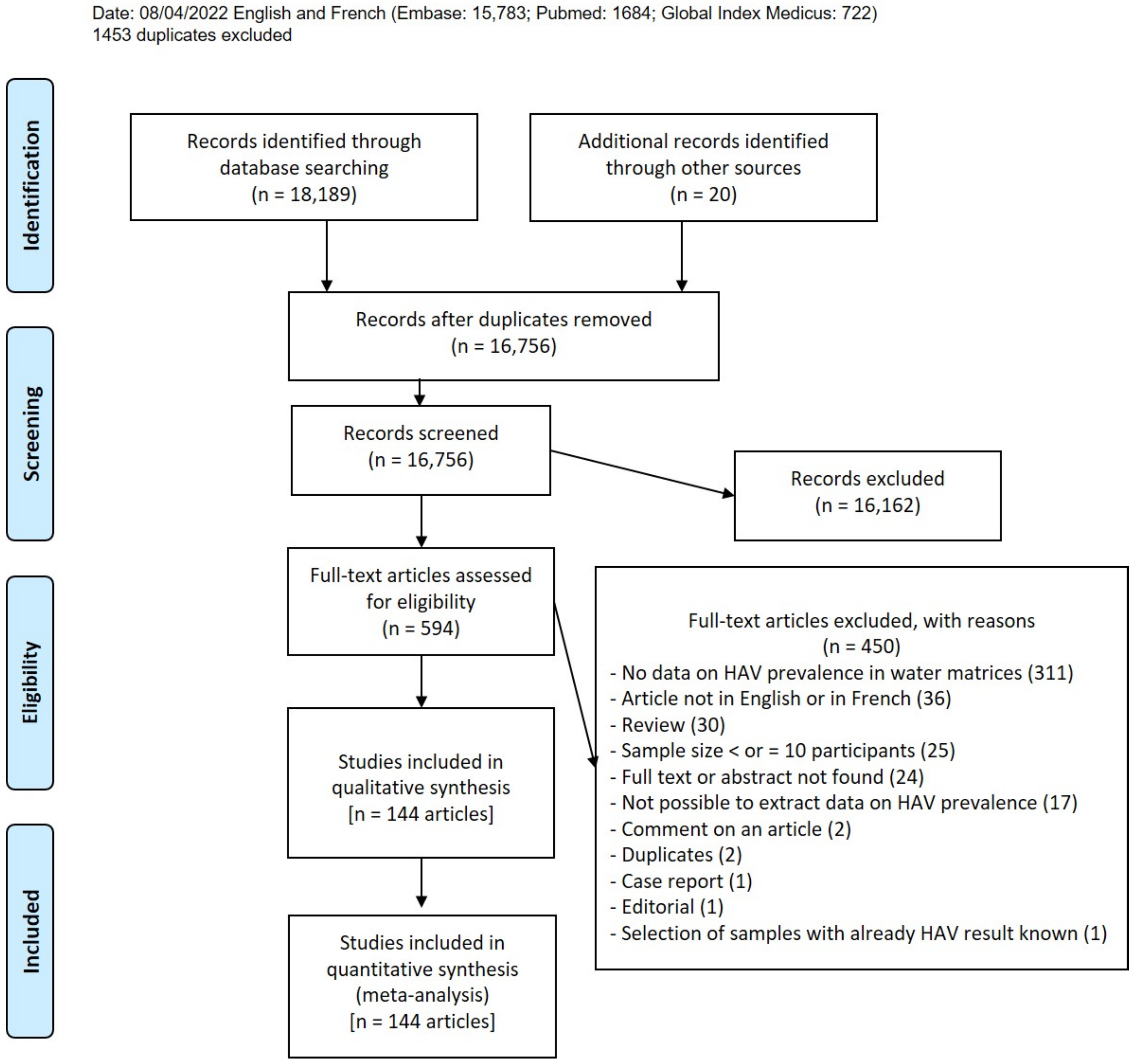
3.1. Study Selection
3.2. Study Characteristics
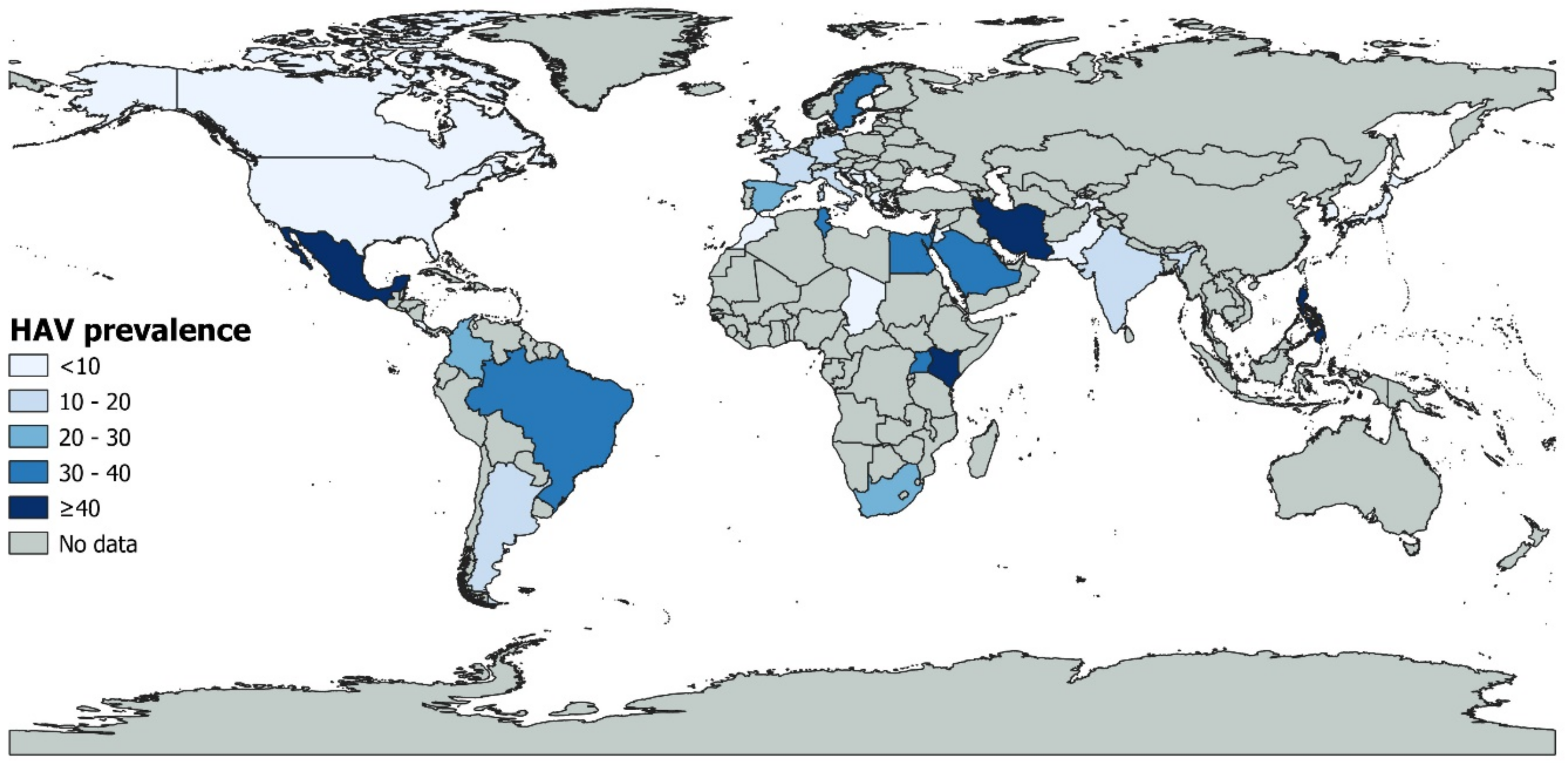
3.3. HAV Prevalence in Water Matrices
3.4. Heterogeneity, Publication Bias and Sensitivity Analysis
3.5. Subgroup Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cao, G.; Jing, W.; Liu, J.; Liu, M. The global trends and regional differences in incidence and mortality of hepatitis A from 1990 to 2019 and implications for its prevention. Hepatol. Int. 2021, 15, 1068–1082. [Google Scholar] [CrossRef] [PubMed]
- Chakravarti, A.; Bharara, T. Epidemiology of Hepatitis A: Past and Current Trends. In Hepatitis A and Other Associated Hepatobiliary Diseases; Costin, T.S., Cristin, C.V., Ion, R., Valeria, T., Silvia, L., Eds.; IntechOpen: London, UK, 2020. [Google Scholar]
- Amarapurkar, D.N.; Amarapurkar, A.D. Extrahepatic manifestations of viral hepatitis. Ann. Hepatol. 2002, 1, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.H.; Lee, H.S. Hepatitis A: Clinical manifestations and management. Intervirology 2010, 53, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Andani, A.; van Elten, T.M.; Bunge, E.M.; Marano, C.; Salgado, F.; Jacobsen, K.H. Hepatitis A epidemiology in Latin American countries: A 2020 view from a systematic literature review. Expert Rev. Vaccines 2020, 19, 795–805. [Google Scholar] [CrossRef] [PubMed]
- Hepatitis, A. Available online: https://www.who.int/news-room/fact-sheets/detail/hepatitis-a (accessed on 11 November 2022).
- Barrett, C.E.; Pape, B.J.; Benedict, K.M.; Foster, M.A.; Roberts, V.A.; Rotert, K.; Mattioli, M.C.; Yoder, J.S. Impact of Public Health Interventions on Drinking Water-Associated Outbreaks of Hepatitis A—United States, 1971–2017. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Bruni, R.; Taffon, S.; Equestre, M.; Cella, E.; Lo Presti, A.; Costantino, A.; Chionne, P.; Madonna, E.; Golkocheva-Markova, E.; Bankova, D.; et al. Hepatitis a virus genotypes and strains from an endemic area of Europe, Bulgaria 2012–2014. BMC Infect. Dis 2017, 17, 497. [Google Scholar] [CrossRef] [PubMed]
- Sinha, A.; Dutta, S. Waterborne & foodborne viral hepatitis: A public health perspective. Indian J. Med. Res. 2019, 150, 432–435. [Google Scholar] [CrossRef]
- Chatziprodromidou, I.P.; Dimitrakopoulou, M.E.; Apostolou, T.; Katopodi, T.; Charalambous, E.; Vantarakis, A. Hepatitis A and E in the Mediterranean: A systematic review. Travel Med. Infect. Dis. 2022, 47, 102283. [Google Scholar] [CrossRef]
- Hu, X.; Collier, M.G.; Xu, F. Hepatitis A Outbreaks in Developed Countries: Detection, Control, and Prevention. Foodborne Pathog. Dis. 2020, 17, 166–171. [Google Scholar] [CrossRef]
- Jacobsen, K.H. Globalization and the Changing Epidemiology of Hepatitis A Virus. Cold Spring Harb. Perspect. Med. 2018, 8. [Google Scholar] [CrossRef]
- Van der Poel, W.; Rzezutka, A.; Hepatitis, A. In Sanitation and Disease in the 21st Century---Part Three: Specific Excreted Pathogens: Environmental and Epidemiology Aspects (Global Water Pathogen Project), Rose, J.B., Jiménez-Cisneros, B., Eds. Available online: https://www.waterpathogens.org/toc (accessed on 11 November 2022).
- Chatziprodromidou, I.P.; Bellou, M.; Vantarakis, G.; Vantarakis, A. Viral outbreaks linked to fresh produce consumption: A systematic review. J. Appl. Microbiol. 2018, 124, 932–942. [Google Scholar] [CrossRef]
- Bellou, M.; Kokkinos, P.; Vantarakis, A. Shellfish-borne viral outbreaks: A systematic review. Food Environ. Virol. 2013, 5, 13–23. [Google Scholar] [CrossRef]
- Thébault, A.; Roque-Afonso, A.-M.; Kooh, P.; Cadavez, V.; Gonzales-Barron, U.; Pavio, N. Risk factors for sporadic hepatitis A infection: A systematic review and meta-analysis. Microb. Risk Anal. 2021, 17, 100155. [Google Scholar] [CrossRef]
- Gholipour, S.; Ghalhari, M.R.; Nikaeen, M.; Rabbani, D.; Pakzad, P.; Miranzadeh, M.B. Occurrence of viruses in sewage sludge: A systematic review. Sci. Total Environ. 2022, 824, 153886. [Google Scholar] [CrossRef]
- Boehm, A.B.; Silverman, A.I.; Schriewer, A.; Goodwin, K. Systematic review and meta-analysis of decay rates of waterborne mammalian viruses and coliphages in surface waters. Water Res. 2019, 164, 114898. [Google Scholar] [CrossRef]
- Kuodi, P.; Patterson, J.; Silal, S.; Hussey, G.D.; Kagina, B.M. Characterisation of the environmental presence of hepatitis A virus in low-income and middle-income countries: A systematic review and meta-analysis. BMJ Open 2020, 10, e036407. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- UN. UNSD—Methodology. Available online: https://unstats.un.org/unsd/methodology/m49/ (accessed on 8 April 2022).
- World Bank. World Bank Country and Lending Groups—World Bank Data Help Desk. Available online: https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups (accessed on 8 April 2022).
- Hoy, D.; Brooks, P.; Woolf, A.; Blyth, F.; March, L.; Bain, C.; Baker, P.; Smith, E.; Buchbinder, R. Assessing risk of bias in prevalence studies: Modification of an existing tool and evidence of interrater agreement. J. Clin. Epidemiol. 2012, 65, 934–939. [Google Scholar] [CrossRef]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.; Rothstein, H.R. A basic introduction to fixed-effect and random-effects models for meta-analysis. Res. Synth. Methods 2010, 1, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Schwarzer, G. Meta: An R Package for Meta-Analysis. Newsl. R Proj. 2007, 7, 40–45. [Google Scholar]
- Abbaszadegan, M.; Stewart, P.; LeChevallier, M. A strategy for detection of viruses in groundwater by PCR. Appl. Environ. Microbiol. 1999, 65, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Adefisoye, M.A.; Nwodo, U.U.; Green, E.; Okoh, A.I. Quantitative PCR Detection and Characterisation of Human Adenovirus, Rotavirus and Hepatitis A Virus in Discharged Effluents of Two Wastewater Treatment Facilities in the Eastern Cape, South Africa. Food Environ. Virol. 2016, 8, 262–274. [Google Scholar] [CrossRef]
- Ahmad, T.; Adnan, F.; Nadeem, M.; Kakar, S.J.; Anjum, S.; Saad, A.; Waheed, A.; Arshad, N. Assessment of the risk for human health of enterovirus and hepatitis a virus in clinical and water sources from three metropolitan cities of Pakistan. Ann. Agric. Environ. Med. 2018, 25, 708–713. [Google Scholar] [CrossRef]
- Ahmad, T.; Anjum, S.; Zaidi, N.S.S.; Ali, A.; Waqas, M.; Afzal, M.S.; Arshad, N. Frequency of hepatitis E and Hepatitis A virus in water sample collected from Faisalabad, Pakistan. Ann. Agric. Environ. Med. 2015, 22, 661–664. [Google Scholar] [CrossRef]
- Ahmad, T.; Arshad, N.; Adnan, F.; Zaidi, N.U.S.S.; Shahid, M.T.; Zahoor, U.; Afzal, M.S.; Anjum, S. Prevalence of rotavirus, adenovirus, hepatitis a virus and enterovirus in water samples collected from different region of Peshawar, Pakistan. Ann. Agric. Environ. Med. 2016, 23, 576–580. [Google Scholar] [CrossRef]
- Amdiouni, H.; Maunula, L.; Al-Shuwaikh, A.; Nourlil, J. Comparison of two virus concentration methods for enteric viruses detection in Moroccan wastewater and treated effluent. Iraqi JMS 2017, 15, 27–38. [Google Scholar] [CrossRef]
- Anastasi, P.; Bonanni, E.; Cecchini, G.; Divizia, M.; Donia, D.; Di Gianfilippo, F.; Gabrieli, R.; Petrinca, A.R.; Zanobini, A. Virus removal in conventional wastewater treatment process. Ig. Sanità Pubblica 2008, 64, 313–330. [Google Scholar]
- Arankalle, V.A.; Devi, K.L.S.; Lole, K.S.; Shenoy, K.T.; Verma, V.; Haneephabi, M. Molecular characteristics of hepatitis A virus from a large outbreak from Kerala, India. Indian J. Med. Res. 2006, 123, 760–769. [Google Scholar]
- Aw, T.G.; Gin, K.Y.H. Environmental surveillance and molecular characterization of human enteric viruses in tropical urban wastewaters. J. Appl. Microbiol. 2010, 109, 716–730. [Google Scholar] [CrossRef]
- Aw, T.G.; Gin, K.Y.H. Prevalence and genetic diversity of waterborne pathogenic viruses in surface waters of tropical urban catchments. J. Appl. Microbiol. 2011, 110, 903–914. [Google Scholar] [CrossRef]
- Bae, K.S.; Lee, S.; Lee, J.Y.; Kim, J.H.; Joo, Y.L.; Lee, S.H.; Chung, H.M.; You, K.A. Development of diagnostic systems for wide range and highly sensitive detection of two waterborne hepatitis viruses from groundwater using the conventional reverse transcription nested PCR assay. J. Virol. Methods 2022, 299, 114344. [Google Scholar] [CrossRef]
- Bahk, Y.Y.; Kim, M.H.; Kim, T.S.; Park, S.J.; Kim, J.M.; Rhee, O.J.; Lee, S.S. Occurrence of four waterborne viruses at five typical raw water resources in the Republic of Korea during August 2013 to February 2019. J. Microbiol. 2020, 58, 915–925. [Google Scholar] [CrossRef]
- Bai, H.; Shiota, T.; Yoshizaki, S.; Saito-Obata, M.; Malbas, F.F.; Lupisan, S.P.; Oshitani, H.; Takeda, N.; Muramatsu, M.; Wakita, T.; et al. Detection of subgenotype IA and IIIA hepatitis A viruses in rivers flowing through metro Manila, the Philippines. Jpn. J. Infect. Dis. 2019, 72, 53–55. [Google Scholar] [CrossRef]
- Barrella, K.M.; Garrafa, P.; Monezi, T.A.; Hársi, C.M.; Salvi, C.; Violante, P.A.B.C.; Mehnert, D.U. Longitudinal study on occurrence of adenoviruses and hepatitis A virus in raw domestic sewage in the city of Limeira, São Paulo. Braz. J. Microbiol. 2009, 40, 102–107. [Google Scholar] [CrossRef]
- Béji-Hamza, A.; Khélifi-Gharbi, H.; Hassine-Zaafrane, M.; Della Libera, S.; Iaconelli, M.; Muscillo, M.; Petricca, S.; Ciccaglione, A.R.; Bruni, R.; Taffon, S.; et al. Qualitative and Quantitative Assessment of Hepatitis A Virus in Wastewaters in Tunisia. Food Environ. Virol. 2014, 6, 246–252. [Google Scholar] [CrossRef]
- Bisseux, M.; Colombet, J.; Mirand, A.; Roque-Afonso, A.M.; Abravanel, F.; Izopet, J.; Archimbaud, C.; Peigue-Lafeuille, H.; Debroas, D.; Bailly, J.L.; et al. Monitoring human enteric viruses in wastewater and relevance to infections encountered in the clinical setting: A one-year experiment in central France, 2014 to 2015. Eurosurveillance 2018, 23. [Google Scholar] [CrossRef]
- Blanco, A.; Abid, I.; Al-Otaibi, N.; Pérez-Rodríguez, F.J.; Fuentes, C.; Guix, S.; Pintó, R.M.; Bosch, A. Glass Wool Concentration Optimization for the Detection of Enveloped and Non-enveloped Waterborne Viruses. Food Environ. Virol. 2019, 11, 184–192. [Google Scholar] [CrossRef]
- Borchardt, M.A.; Bertz, P.D.; Spencer, S.K.; Battigelli, D.A. Incidence of enteric viruses in groundwater from household wells in Wisconsin. Appl. Environ. Microbiol. 2003, 69, 1172–1180. [Google Scholar] [CrossRef] [PubMed]
- Borchardt, M.A.; Haas, N.L.; Hunt, R.J. Vulnerability of drinking-water wells in La Crosse, Wisconsin, to enteric-virus contamination from surface water contributions. Appl. Environ. Microbiol. 2004, 70, 5937–5946. [Google Scholar] [CrossRef] [PubMed]
- Carducci, A.; Battistini, R.; Rovini, E.; Verani, M. Viral removal by wastewater treatment: Monitoring of indicators and pathogens. Food Environ. Virol. 2009, 1, 85–91. [Google Scholar] [CrossRef]
- Carducci, A.; Casini, B.; Bani, A.; Rovini, E.; Verani, M.; Mazzoni, F.; Giuntini, A. Virological control of groundwater quality using biomolecular tests. Water Sci. Technol. 2003, 47, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Carducci, A.; Morici, P.; Pizzi, F.; Battistini, R.; Rovini, E.; Verani, M. Study of the viral removal efficiency in a urban wastewater treatment plant. Water Sci Technol. 2008, 58, 893–897. [Google Scholar] [CrossRef]
- Carducci, A.; Verani, M.; Battistini, R.; Pizzi, F.; Rovini, E.; Andreoli, E.; Casini, B. Epidemiological surveillance of human enteric viruses by monitoring of different environmental matrices. Water Sci. Technol. A J. Int. Assoc. Water Pollut. Res. 2006, 54, 239–244. [Google Scholar] [CrossRef]
- Chacón, L.; Barrantes, K.; Santamaría-Ulloa, C.; Solano, M.; Reyes, L.; Taylor, L.; Valiente, C.; Symonds, E.M.; Achí, R. A Somatic Coliphage Threshold Approach to Improve the Management of Activated Sludge Wastewater Treatment Plant Effluents in Resource-Limited Regions. Appl. Environ. Microbiol. 2020, 86, e00616–e00620. [Google Scholar] [CrossRef]
- Chacón, L.; Morales, E.; Valiente, C.; Reyes, L.; Barrantes, K. Wastewater-based epidemiology of enteric viruses and surveillance of acute gastrointestinal illness outbreaks in a resource-limited region. Am. J. Trop. Med. Hyg. 2021, 105, 1004–1012. [Google Scholar] [CrossRef]
- Chigor, V.N.; Okoh, A.I. Quantitative RT-PCR detection of hepatitis A virus, rotaviruses and enteroviruses in the Buffalo River and source water dams in the Eastern Cape Province of South Africa. Int. J. Environ. Res. Public Health 2012, 9, 4017–4032. [Google Scholar] [CrossRef]
- Chitambar, S.; Joshi, M.; Lole, K.; Walimbe, A.; Vaidya, S. Cocirculation of and coinfections with hepatitis A virus subgenotypes IIIA and IB in patients from Pune, western India. Hepatol. Res. Off. J. Jpn. Soc. Hepatol. 2007, 37, 85–93. [Google Scholar] [CrossRef]
- Cioffi, B.; Ianiro, G.; Iaccarino, D.; D’Apice, F.; Ferraro, A.; Race, M.; Spasiano, D.; Esposito, E.; Monini, M.; Serra, F.; et al. A potential risk assessment tool to monitor pathogens circulation in coastal waters. Environ. Res. 2021, 200, 111748. [Google Scholar] [CrossRef]
- Clemente-Casares, P.; Pina, S.; Buti, M.; Jardi, R.; Martín, M.; Bofill-Mas, S.; Girones, R. Hepatitis E virus epidemiology in industrialized countries. Emerg. Infect. Dis. 2003, 9, 448–454. [Google Scholar] [CrossRef]
- Corsi, S.R.; Borchardt, M.A.; Spencer, S.K.; Hughes, P.E.; Baldwin, A.K. Human and bovine viruses in the Milwaukee River watershed: Hydrologically relevant representation and relations with environmental variables. Sci. Total Environ. 2014, 490, 849–860. [Google Scholar] [CrossRef]
- De Giglio, O.; Caggiano, G.; Bagordo, F.; Barbuti, G.; Brigida, S.; Lugoli, F.; Grassi, T.; La Rosa, G.; Lucentini, L.; Uricchio, V.F.; et al. Enteric viruses and fecal bacteria indicators to assess groundwater quality and suitability for irrigation. Int. J. Environ. Res. Public Health 2017, 14, 558. [Google Scholar] [CrossRef]
- De Paula, V.S.; Diniz-Mendes, L.; Villar, L.M.; Luz, S.L.B.; Silva, L.A.; Jesus, M.S.; da Silva, N.M.V.S.; Gaspar, A.M.C. Hepatitis A virus in environmental water samples from the Amazon Basin. Water Res. 2007, 41, 1169–1176. [Google Scholar] [CrossRef]
- De Serres, G.; Cromeans, T.L.; Levesque, B.; Brassard, N.; Barthe, C.; Dionne, M.; Prud’Homme, H.; Paradis, D.; Shapiro, C.N.; Nainan, O.V.; et al. Molecular confirmation of hepatitis A virus from well water: Epidemiology and public health implications. J. Infect. Dis. 1999, 179, 37–43. [Google Scholar] [CrossRef]
- De Souza, F.G.; da Silva, F.P.; Staggemeier, R.; Rigotto, C.; Spilki, F.R. Low occurrence of hepatitis A virus in water samples from an urban area of southern Brazil. Rev. Inst. Med. Trop. Sao Paulo 2018, 60, e69. [Google Scholar] [CrossRef]
- Denis-Mize, K.; Fout, G.S.; Dahling, D.R.; Francy, D.S. Detection of human enteric viruses in stream water with RT-PCR and cell culture. J. Water Health 2004, 2, 37–47. [Google Scholar] [CrossRef]
- Dias, J.; Pinto, R.N.; Vieira, C.B.; de Abreu Corrêa, A. Detection and quantification of human adenovirus (HAdV), JC polyomavirus (JCPyV) and hepatitis A virus (HAV) in recreational waters of Niterói, Rio de Janeiro, Brazil. Mar. Pollut. Bull. 2018, 133, 240–245. [Google Scholar] [CrossRef]
- Divizia, M.; Morace, G.; Gabrieli, R.; Pisani, G.; Pana, A. Application of the PCR technique to the detection of hepatitis A virus in the environment. Water Sci. Technol. 1993, 27, 223–225. [Google Scholar] [CrossRef]
- Divizia, M.; Ruscio, V.; Degener, A.M.; Panà, A. Hepatitis A virus detection in wastewater by PCR and hybridization. New Microbiol. 1998, 21, 161–167. [Google Scholar]
- Dubrou, S.; Kopecka, H.; Lopez Pila, J.M.; Marechal, J.; Prevot, J. Detection of hepatitis A virus and other enteroviruses in wastewater and surface water samples by gene probe assay. Water Sci. Technol. 1991, 24, 267–272. [Google Scholar] [CrossRef]
- Elmahdy, E.M.; Fongaro, G.; Schissi, C.D.; Petrucio, M.M.; Barardi, C.R.M. Enteric viruses in surface water and sediment samples from the catchment area of Peri Lagoon, Santa Catarina State, Brazil. J. Water Health 2016, 14, 142–154. [Google Scholar] [CrossRef]
- Elmahdy, M.E.; Fongaro, G.; Magri, M.E.; Petruccio, M.M.; Barardi, C.R. Spatial distribution of enteric viruses and somatic coliphages in a Lagoon used as drinking water source and recreation in Southern Brazil. Int. J. Hyg. Environ. Health 2016, 219, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Farkas, K.; Cooper, D.M.; McDonald, J.E.; Malham, S.K.; de Rougemont, A.; Jones, D.L. Seasonal and spatial dynamics of enteric viruses in wastewater and in riverine and estuarine receiving waters. Sci. Total Environ. 2018, 634, 1174–1183. [Google Scholar] [CrossRef]
- Félix, J.L.; Fernandez, Y.C.; Velarde-Félix, J.S.; Torres, B.V.; Cháidez, C. Detection and phylogenetic analysis of hepatitis a virus and norovirus in marine recreational waters of Mexico. J. Water Health 2010, 8, 269–278. [Google Scholar] [CrossRef]
- Fernández-Molina, M.C.; Álvarez, A.; Espigares, M. Presence of hepatitis a virus in water and its relationship with indicators of fecal contamination. Water Air Soil Pollut. 2004, 159, 197–208. [Google Scholar] [CrossRef]
- Fongaro, G.; Nascimento, M.A.; Viancelli, A.; Tonetta, D.; Petrucio, M.M.; Barardi, C.R.M. Surveillance of human viral contamination and physicochemical profiles in a surface water lagoon. Water Sci. Technol. 2012, 66, 2682–2687. [Google Scholar] [CrossRef]
- Fongaro, G.; Viancelli, A.; dos Reis, D.A.; Santiago, A.F.; Hernández, M.; Michellon, W.; da Silva Lanna, M.C.; Treichel, H.; Rodríguez-Lázaro, D. Mineral Waste Containing High Levels of Iron from an Environmental Disaster (Bento Rodrigues, Mariana, Brazil) is Associated with Higher Titers of Enteric Viruses. Food Environ. Virol. 2019, 11, 178–183. [Google Scholar] [CrossRef]
- Formiga-Cruz, M.; Hundesa, A.; Clemente-Casares, P.; Albiñana-Gimenez, N.; Allard, A.; Girones, R. Nested multiplex PCR assay for detection of human enteric viruses in shellfish and sewage. J. Virol. Methods 2005, 125, 111–118. [Google Scholar] [CrossRef]
- Fout, G.S.; Martinson, B.C.; Moyer, M.W.N.; Dahling, D.R. A multiplex reverse transcription-PCR method for detection of human enteric viruses in groundwater. Appl. Environ. Microbiol. 2003, 69, 3158–3164. [Google Scholar] [CrossRef]
- Fumian, T.M.; Victoria, M.; Vieira, C.B.; Fioretti, J.M.; Rocha, M.S.; Prado, T.; Guimarães, F.R.; da Gama, N.P.; de Oliveira, J.M.; Mendes, A.C.O.; et al. Enteric viruses’ dissemination in a private reserve of natural heritage. Lett. Appl. Microbiol. 2018, 66, 313–320. [Google Scholar] [CrossRef]
- Gcilitshana, O.; Sibanda, T.; Zhou, L.; Okoh, A.I. Assessment of the prevalence of enteric viruses in the final effluents of two peri-urban wastewater treatment plants. Asian Pac. J. Trop. Dis. 2017, 7, 121–126. [Google Scholar] [CrossRef]
- Gersberg, R.M.; Rose, M.A.; Robles-Sikisaka, R.; Dhar, A.K. Quantitative detection of hepatitis A virus and enteroviruses near the United States-Mexico border and correlation with levels of fecal indicator bacteria. Appl. Environ. Microbiol. 2006, 72, 7438–7444. [Google Scholar] [CrossRef]
- Gharbi-Khelifi, H.; Abid, N.B.S.; Sdiri, K.; Harrath, R.; Beji, A.; Bhiri, L.; Billaudel, S.; Ferre, V.; Aouni, M. Characterization of outbreak hepatitis a isolates in five Tunisian childcare centers. Braz. J. Microbiol. 2011, 42, 1204–1212. [Google Scholar] [CrossRef]
- Girones, R.; Puig, M.; Allard, A.; Lucena, F.; Wadell, G.; Jofre, J. Detection of adenovirus and enterovirus by PCR amplification in polluted waters. Water Sci. Technol. 1995, 31, 351–357. [Google Scholar] [CrossRef]
- Gozlan, Y.; Bar-Or, I.; Volnowitz, H.; Asulin, E.; Rich, R.; Anis, E.; Shemer, Y.; Cohen, M.S.; Dahary, E.L.; Schreiber, L.; et al. Lessons from intensified surveillance of viral hepatitis a, Israel, 2017 and 2018. Eurosurveillance 2021, 26, 2000001. [Google Scholar] [CrossRef]
- Gozlan, Y.; Volnowitz, H.; Bar-or, I.; Rich, R.; Mendelson, E.; Ari, Z.B.; Mor, O. Circulation of hepatitis a genotypes in Israel 2017-2018: Environmental surveillance supports clinical findings. J. Hepatol. 2019, 70, e722. [Google Scholar] [CrossRef]
- Grabow, W.O.K.; Taylor, M.B.; De Villiers, J.C. New methods for the detection of viruses: Call for review of drinking water quality guidelines. Water Sci. Technol. 2001, 43, 1–8. [Google Scholar] [CrossRef]
- Guerrero-Latorre, L.; Carratala, A.; Rodriguez-Manzano, J.; Calgua, B.; Hundesa, A.; Girones, R. Occurrence of water-borne enteric viruses in two settlements based in Eastern Chad: Analysis of hepatitis E virus, hepatitis A virus and human adenovirus in water sources. J. Water Health 2011, 9, 515–524. [Google Scholar] [CrossRef]
- Hassine, M.; Sdiri, K.; Riabi, S.; Beji, A.; Aouni, Z.; Aouni, M. Detection of enteric viruses in wastewater of Monastir region by RT-PCR method. Tunis. Med. 2010, 88, 57–62. [Google Scholar]
- Hee, J.K.; Young, O.S.; Sang, H.K. Detection of enteroviruses and mammalian reoviruses in Korean environmental waters. Microbiol. Immunol. 2006, 50, 781–786. [Google Scholar] [CrossRef]
- Hernandez-Morga, J.; Leon-Felix, J.; Peraza-Garay, F.; Gil-Salas, B.G.; Chaidez, C. Detection and characterization of hepatitis a virus and norovirus in estuarine water samples using ultrafiltration—RT-PCR integrated methods. J. Appl. Microbiol. 2009, 106, 1579–1590. [Google Scholar] [CrossRef] [PubMed]
- Hot, D.; Legeay, O.; Jacques, J.; Gantzer, C.; Caudrelier, Y.; Guyard, K.; Lange, M.; Andréoletti, L. Detection of somatic phages, infectious enteroviruses and enterovirus genomes as indicators of human enteric viral pollution in surface water. Water Res. 2003, 37, 4703–4710. [Google Scholar] [CrossRef] [PubMed]
- Iaconelli, M.; Muscillo, M.; Libera, S.D.; Fratini, M.; Meucci, L.; De Ceglia, M.; Giacosa, D.; La Rosa, G. One-year Surveillance of Human Enteric Viruses in Raw and Treated Wastewaters, Downstream River Waters, and Drinking Waters. Food Environ. Virol. 2017, 9, 79–88. [Google Scholar] [CrossRef]
- Iaconelli, M.; Purpari, G.; Libera, S.D.; Petricca, S.; Guercio, A.; Ciccaglione, A.R.; Bruni, R.; Taffon, S.; Equestre, M.; Fratini, M.; et al. Hepatitis A and E Viruses in Wastewaters, in River Waters, and in Bivalve Molluscs in Italy. Food Environ. Virol. 2015, 7, 316–324. [Google Scholar] [CrossRef]
- Janahi, E.M.; Mustafa, S.; Parkar, S.F.D.; Naser, H.A.; Eisa, Z.M. Detection of enteric viruses and bacterial indicators in a sewage treatment center and shallow water bay. Int. J. Environ. Res. Public Health 2020, 17, 6483. [Google Scholar] [CrossRef]
- Jebri, S.; Jofre, J.; Barkallah, I.; Saidi, M.; Hmaied, F. Presence and fate of coliphages and enteric viruses in three wastewater treatment plants effluents and activated sludge from Tunisia. Environ. Sci. Pollut. Res. 2012, 19, 2195–2201. [Google Scholar] [CrossRef]
- Jehl-Pietri, C.; Hugues, B.; Andre, M.; Diez, J.M.; Bosch, A. Comparison of immunological and molecular hybridization detection methods for the detection of hepatitis A virus in sewage. Lett. Appl. Microbiol. 1993, 17, 162–166. [Google Scholar] [CrossRef]
- Jiang, S.C.; Chu, W. PCR detection of pathogenic viruses in southern California urban rivers. J. Appl. Microbiol. 2004, 97, 17–28. [Google Scholar] [CrossRef]
- Jones, T.H.; Brassard, J.; Topp, E.; Wilkes, G.; Lapen, D.R. Waterborne Viruses and F-Specific Coliphages in Mixed-Use Watersheds: Microbial Associations, Host Specificities, and Affinities with Environmental/Land Use Factors. Appl. Environ. Microbiol. 2017, 83, e02763-16. [Google Scholar] [CrossRef]
- Jothikumar, N.; Cliver, D.O.; Mariam, T.W. Immunomagnetic capture PCR for rapid concentration and detection of hepatitis A virus from environmental samples. Appl. Environ. Microbiol. 1998, 64, 504–508. [Google Scholar] [CrossRef]
- Jothikumar, N.; Paulmurugan, R.; Padmanabhan, P.; Balathiripura Sundari, R.; Kamatchiammal, S.; Subba Rao, K. Duplex RT-PCR for simultaneous detection of hepatitis A and hepatitis E virus isolated from drinking water samples. J. Environ. Monit. 2000, 2, 587–590. [Google Scholar] [CrossRef]
- Jung, J.H.; Yoo, C.H.; Koo, E.S.; Kim, H.M.; Na, Y.; Jheong, W.H.; Jeong, Y.S. Occurrence of norovirus and other enteric viruses in untreated groundwaters of Korea. J. Water Health 2011, 9, 544–555. [Google Scholar] [CrossRef]
- Kaas, L.; Gourinat, A.C.; Urbès, F.; Langlet, J. A 1-Year Study on the Detection of Human Enteric Viruses in New Caledonia. Food Environ. Virol. 2016, 8, 46–56. [Google Scholar] [CrossRef]
- Kamel, A.H.; Ali, M.A.; El-Nady, H.G.; Deraz, A.; Aho, S.; Pothier, P.; Belliot, G. Presence of enteric hepatitis viruses in the sewage and population of Greater Cairo. Clin. Microbiol. Infect. 2011, 17, 1182–1185. [Google Scholar] [CrossRef]
- Katukiza, A.Y.; Temanu, H.; Chung, J.W.; Foppen, J.W.A.; Lens, P.N.L. Genomic copy concentrations of selected waterborne viruses in a slum environment in Kampala, Uganda. J. Water Health 2013, 11, 358–370. [Google Scholar] [CrossRef]
- Kittigul, L.; Raengsakulrach, B.; Siritantikorn, S.; Kanyok, R.; Utrarachkij, F.; Diraphat, P.; Thirawuth, V.; Siripanichgon, K.; Pungchitton, S.; Chitpirom, K.; et al. Detection of poliovirus, hepatitis A virus and rotavirus from sewage and water samples. Southeast. Asian J. Trop. Med. Public Health 2000, 31, 41–46. [Google Scholar]
- Kittigul, L.; Uthaisin, A.; Ekchaloemkiet, S.; Utrarachkij, F.; Luksamijarulkul, P. Detection and characterization of hepatitis A virus in water samples in Thailand. J. Appl. Microbiol. 2006, 100, 1318–1323. [Google Scholar] [CrossRef]
- Kiulia, N.M.; Netshikweta, R.; Page, N.A.; Van Zyl, W.B.; Kiraithe, M.M.; Nyachieo, A.; Mwenda, J.M.; Taylor, M.B. The detection of enteric viruses in selected urban and rural river water and sewage in Kenya, with special reference to rotaviruses. J. Appl. Microbiol. 2010, 109, 818–828. [Google Scholar] [CrossRef]
- Kokkinos, P.; Filippidou, S.; Karlou, K.; Vantarakis, A. Molecular typing of enteroviruses, adenoviruses, and hepatitis a viruses in untreated and treated sewage of a biological treatment plant in Greece. Food Environ. Virol. 2010, 2, 89–96. [Google Scholar] [CrossRef]
- Kokkinos, P.; Karayanni, H.; Meziti, A.; Feidaki, R.; Paparrodopoulos, S.; Vantarakis, A. Assessment of the Virological Quality of Marine and Running Surface Waters in NW Greece: A Case Study. Food Environ. Virol. 2018, 10, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Kokkinos, P.; Ziros, P.; Meri, D.; Filippidou, S.; Kolla, S.; Galanis, A.; Vantarakis, A. Environmental surveillance. An additional/alternative approach for virological surveillance in Greece? Int. J. Environ. Res. Public Health 2011, 8, 1914–1922. [Google Scholar] [CrossRef]
- La Rosa, G.; Della Libera, S.; Iaconelli, M.; Ciccaglione, A.R.; Bruni, R.; Taffon, S.; Equestre, M.; Alfonsi, V.; Rizzo, C.; Tosti, M.E.; et al. Surveillance of hepatitis A virus in urban sewages and comparison with cases notified in the course of an outbreak, Italy 2013. BMC Infect. Dis. 2014, 14, 419. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, G.; Mancini, P.; Ferraro, G.B.; Iaconelli, M.; Veneri, C.; Paradiso, R.; De Medici, D.; Vicenza, T.; Proroga, Y.T.R.; Di Maro, O.; et al. Hepatitis A Virus Strains Circulating in the Campania Region (2015–2018) Assessed through Bivalve Biomonitoring and Environmental Surveillance. Viruses 2021, 13, 16. [Google Scholar] [CrossRef]
- La Rosa, G.; Sanseverino, I.; Della Libera, S.; Iaconelli, M.; Ferrero, V.E.V.; Barra Caracciolo, A.; Lettieri, T. The impact of anthropogenic pressure on the virological quality of water from the Tiber River, Italy. Lett. Appl. Microbiol. 2017, 65, 298–305. [Google Scholar] [CrossRef]
- Lazić, G.; Grubač, S.; Lupulović, D.; Bugarski, D.; Lazić, S.; Knežević, P.; Petrović, T. Presence of Human and Animal Viruses in Surface Waters in Vojvodina Province of Serbia. Food Environ. Virol. 2015, 7, 149–158. [Google Scholar] [CrossRef]
- Lee, C.H.; Kim, S.J. Molecular detection of human enteric viruses in urban rivers in Korea. J. Microbiol. Biotechnol. 2008, 18, 1156–1163. [Google Scholar]
- Lee, G.C.; Kim, M.J.; Nam, S.; Lee, C.H. Incidence and molecular characterization of hepatitis A viruses in Korean surface water between 2007 and 2010. Microbiol. Immunol. 2014, 58, 342–351. [Google Scholar] [CrossRef]
- Lenaker, P.L.; Corsi, S.R.; Borchardt, M.A.; Spencer, S.K.; Baldwin, A.K.; Lutz, M.A. Hydrologic, land cover, and seasonal patterns of waterborne pathogens in Great Lakes tributaries. Water Res. 2017, 113, 11–21. [Google Scholar] [CrossRef]
- Masachessi, G.; Pisano, M.B.; Prez, V.E.; Martínez, L.C.; Michelena, J.F.; Martínez-Wassaf, M.; Giordano, M.O.; Isa, M.B.; Pavan, J.V.; Welter, A.; et al. Enteric Viruses in Surface Waters from Argentina: Molecular and Viable-Virus Detection. Appl. Environ. Microbiol. 2018, 84, e02327-17. [Google Scholar] [CrossRef]
- Miagostovich, M.P.; Guimarães, F.R.; Vieira, C.B.; Fumian, T.M.; da Gama, N.P.; Victoria, M.; de Oliveira, J.M.; de Oliveira Mendes, A.C.; Gaspar, A.M.C.; Leite, J.P.G. Assessment of Water Quality in a Border Region between the Atlantic Forest and an Urbanised Area in Rio de Janeiro, Brazil. Food Environ. Virol. 2014, 6, 110–115. [Google Scholar] [CrossRef]
- Morace, G.; Pisani, G.; Divizia, M.; Panà, A. Detection of hepatitis A virus in concentrated river water by polymerase chain reaction. Zent. Für Hyg. Und Umweltmed. Int. J. Hyg. Environ. Med. 1993, 193, 521–527. [Google Scholar]
- Moresco, V.; Viancelli, A.; Nascimento, M.A.; Souza, D.S.M.; Ramos, A.P.D.; Garcia, L.A.T.; Simões, C.M.O.; Barardi, C.R.M. Microbiological and physicochemical analysis of the coastal waters of southern Brazil. Mar. Pollut. Bull. 2012, 64, 40–48. [Google Scholar] [CrossRef]
- Nasiri, M.; Ghalejoogh, Z.Y.; Ataei-Pirkooh, A.; Bokharaei-Salim, F.; Monavari, S.H.; Tavakoli, A.; Asadifar, B.; Esghaei, M.; Pasalari, H.; Samimi-Rad, K.; et al. Detection and Phylogenetic Analysis of Hepatitis A Virus in the Wastewater Treatment Plant of Ekbatan Town in Tehran, Iran. Hepat. Mon. 2021, 21, e121270. [Google Scholar] [CrossRef]
- Nasser, A.M.; Metcalf, T.G. An A-ELISA to detect hepatitis A virus in estuarine samples. Appl. Environ. Microbiol. 1987, 53, 1192–1195. [Google Scholar] [CrossRef]
- Ngaosuwankul, N.; Thippornchai, N.; Yamashita, A.; Vargas, R.E.M.; Tunyong, W.; Mahakunkijchareon, Y.; Ikuta, K.; Singhasivanon, P.; Okabayashi, T.; Leaungwutiwong, P. Detection and characterization of enteric viruses in flood water from the 2011 Thai flood. Jpn. J. Infect. Dis. 2013, 66, 398–403. [Google Scholar] [CrossRef]
- Novaković, T.; Agolli, B.; Maretić, Z.; Mijajlović, Z.; Cobeljić, M.; Zivanović-Marinković, V.; Krstić, L.; Birtasević, B. An epidemic of viral hepatitis A after consumption of mussels from polluted seawater. Vojnosanit. Pregl. Mil. Med. Pharm. Rev. 1983, 40, 163–167. [Google Scholar]
- O’Brien, E.; Nakyazze, J.; Wu, H.; Kiwanuka, N.; Cunningham, W.; Kaneene, J.B.; Xagoraraki, I. Viral diversity and abundance in polluted waters in Kampala, Uganda. Water Res. 2017, 127, 41–49. [Google Scholar] [CrossRef]
- Osuolale, O.; Okoh, A. Incidence of human adenoviruses and Hepatitis A virus in the final effluent of selected wastewater treatment plants in Eastern Cape Province, South Africa. Virol. J. 2015, 12, 98. [Google Scholar] [CrossRef]
- Ouardani, I.; Manso, C.F.; Aouni, M.; Romalde, J.L. Efficiency of hepatitis A virus removal in six sewage treatment plants from central Tunisia. Appl. Microbiol. Biotechnol. 2015, 99, 10759–10769. [Google Scholar] [CrossRef] [PubMed]
- Ouardani, I.; Turki, S.; Aouni, M.; Romalde, J.L. Detection and Molecular Characterization of Hepatitis A Virus from Tunisian Wastewater Treatment Plants with Different Secondary Treatments. Appl. Environ. Microbiol. 2016, 82, 3834–3845. [Google Scholar] [CrossRef] [PubMed]
- Peláez, D.; Guzmán, B.L.; Rodríguez, J.; Acero, F.; Nava, G. Presence of enteric viruses in water samples for consumption in Colombia: Challenges for supply systems. Biomed. Rev. Inst. Nac. Salud 2016, 36, 169–178. [Google Scholar] [CrossRef]
- Pellegrinelli, L.; Galli, C.; Binda, S.; Primache, V.; Tagliacarne, C.; Pizza, F.; Mazzini, R.; Pariani, E.; Romanò, L. Molecular Characterization and Phylogenetic Analysis of Enteroviruses and Hepatitis A Viruses in Sewage Samples, Northern Italy, 2016. Food Environ. Virol. 2019, 11, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Petrinca, A.R.; Donia, D.; Pierangeli, A.; Gabrieli, R.; Degener, A.M.; Bonanni, E.; Diaco, L.; Cecchini, G.; Anastasi, P.; Divizia, M. Presence and environmental circulation of enteric viruses in three different wastewater treatment plants. J. Appl. Microbiol. 2009, 106, 1608–1617. [Google Scholar] [CrossRef]
- Pietri, C.; Hugues, B.; Puel, D. Immune electron microscopy in the detection of viruses other than enteroviruses on cell culture in untreated sewage. Zent. Fur Bakteriol. Mikrobiol. Hygiene Ser. B Umwelthyg. Krankenh. Arb. Prav. Med. 1988, 186, 67–72. [Google Scholar]
- Pina, S.; Buti, M.; Jardí, R.; Clemente-Casares, P.; Jofre, J.; Girones, R. Genetic analysis of hepatitis A virus strains recovered from the environment and from patients with acute hepatitis. J. Gen. Virol. 2001, 82, 2955–2963. [Google Scholar] [CrossRef]
- Pina, S.; Puig, M.; Lucena, F.; Jofre, J.; Girones, R. Viral pollution in the environment and in shellfish: Human adenovirus detection by PCR as an index of human viruses. Appl. Environ. Microbiol. 1998, 64, 3376–3382. [Google Scholar] [CrossRef]
- Pintó, R.M.; Alegre, D.; Domíngueza, A.; El-Senousy, W.M.; Sánchez, G.; Villena, C.; Costafreda, M.I.; Aragonès, L.; Bosch, A. Hepatitis A virus in urban sewage from two Mediterranean countries. Epidemiol. Infect. 2007, 135, 270–273. [Google Scholar] [CrossRef]
- Prado, T.; Barbosa, M.R.F.; Araújo, R.S.; Garcia, S.C.; Melo, A.J.; Galvani, A.T.; Brandão, C.J.; Silva, R.L.O.; Sato, M.I.Z. Hepatitis A Outbreaks and Environmental Circulation of Genotype IA Strains in the São Paulo City, 2017–2018. Food Environ. Virol. 2021, 13, 520–527. [Google Scholar] [CrossRef]
- Prado, T.; Fumian, T.M.; Miagostovich, M.P.; Gaspar, A.M.C. Monitoring the hepatitis A virus in urban wastewater from Rio de Janeiro, Brazil. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 104–109. [Google Scholar] [CrossRef]
- Prado, T.; Silva, D.M.; Guilayn, W.C.; Rose, T.L.; Gaspar, A.M.C.; Miagostovich, M.P. Quantification and molecular characterization of enteric viruses detected in effluents from two hospital wastewater treatment plants. Water Res. 2011, 45, 1287–1297. [Google Scholar] [CrossRef]
- Prevost, B.; Lucas, F.S.; Goncalves, A.; Richard, F.; Moulin, L.; Wurtzer, S. Large scale survey of enteric viruses in river and waste water underlines the health status of the local population. Environ. Int. 2015, 79, 42–50. [Google Scholar] [CrossRef]
- Puntaric, D.; Cecuk, D.; Grce, M.; Vodopija, I.; Ljubicic, M.; Baklaic, Z. Human virus detection in drinking water in Zagreb from 1991 to 1994. Period. Biol. 1995, 97, 347–349. [Google Scholar]
- Purpari, G.; Macaluso, G.; Di Bella, S.; Gucciardi, F.; Mira, F.; Di Marco, P.; Lastra, A.; Petersen, E.; La Rosa, G.; Guercio, A. Molecular characterization of human enteric viruses in food, water samples, and surface swabs in Sicily. Int. J. Infect. Dis. 2019, 80, 66–72. [Google Scholar] [CrossRef]
- Pusch, D.; Oh, D.Y.; Wolf, S.; Dumke, R.; Schröter-Bobsin, U.; Höhne, M.; Röske, I.; Schreier, E. Detection of enteric viruses and bacterial indicators in German environmental waters. Arch. Virol. 2005, 150, 929–947. [Google Scholar] [CrossRef]
- Rachida, S.; Matsapola, P.N.; Wolfaardt, M.; Taylor, M.B. Genetic characterization of a novel hepatitis a virus strain in irrigation water in South Africa. J. Med. Virol. 2016, 88, 734–737. [Google Scholar] [CrossRef]
- Rachida, S.; Taylor, M.B. Potentially infectious novel hepatitis a virus strains detected in selected treated wastewater discharge sources, South Africa. Viruses 2020, 12, 1468. [Google Scholar] [CrossRef]
- Rigotto, C.; Victoria, M.; Moresco, V.; Kolesnikovas, C.K.; Corrêa, A.; Souza, D.S.M.; Miagostovich, M.P.; Simões, C.M.O.; Barardi, C.R.M. Assessment of adenovirus, hepatitis A virus and rotavirus presence in environmental samples in Florianopolis, South Brazil. J. Appl. Microbiol. 2010, 109, 1979–1987. [Google Scholar] [CrossRef]
- Rodriguez-Manzano, J.; Alonso, J.L.; Ferrús, M.A.; Moreno, Y.; Amorós, I.; Calgua, B.; Hundesa, A.; Guerrero-Latorre, L.; Carratala, A.; Rusiñol, M.; et al. Standard and new faecal indicators and pathogens in sewage treatment plants, microbiological parameters for improving the control of reclaimed water. Water Sci. Technol. 2012, 66, 2517–2523. [Google Scholar] [CrossRef]
- Rodriguez-Manzano, J.; Miagostovich, M.; Hundesa, A.; Clemente-Casares, P.; Carratala, A.; Buti, M.; Jardi, R.; Girones, R. Analysis of the evolution in the circulation of HAV and HEV in Eastern Spain by testing urban sewage samples. J. Water Health 2010, 8, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Saadoun, I.; Ababneh, Q.; Jaradat, Z. Genomic detection of waterborne enteric viruses as water quality indicators in Al-Zarqa River, Jordan. J. Water Health 2021, 19, 604–615. [Google Scholar] [CrossRef] [PubMed]
- Saïd, R.; Wolfaardt, M.; Taylor, M.B. Molecular characterisation of hepatitis A virus strains from water sources in South Africa. Water Sci. Technol. 2014, 69, 923–933. [Google Scholar] [CrossRef] [PubMed]
- Schlindwein, A.D.; Rigotto, C.; Simões, C.M.; Barardi, C.R. Detection of enteric viruses in sewage sludge and treated wastewater effluent. Water Sci. Technol. A J. Int. Assoc. Water Pollut. Res. 2010, 61, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Schvoerer, E.; Bonnet, F.; Dubois, V.; Cazaux, G.; Serceau, R.; Fleury, H.J.A.; Lafon, M.E. PCR detection of human enteric viruses in bathing areas, waste waters and human stools in southwestern France. Res. Microbiol. 2000, 151, 693–701. [Google Scholar] [CrossRef]
- Shaheen, M.N.F.; Elmahdy, E.M.; Chawla-Sarkar, M. Quantitative PCR-based identification of enteric viruses contaminating fresh produce and surface water used for irrigation in Egypt. Environ. Sci. Pollut. Res. Int. 2019, 26, 21619–21628. [Google Scholar] [CrossRef]
- Shin, H.; Park, H.; Seo, D.J.; Jung, S.; Yeo, D.; Wang, Z.; Park, K.H.; Choi, C. Foodborne Viruses Detected Sporadically in the Fresh Produce and Its Production Environment in South Korea. Foodborne Pathog. Dis. 2019, 16, 411–420. [Google Scholar] [CrossRef]
- Simmons, F.J.; Xagoraraki, I. Release of infectious human enteric viruses by full-scale wastewater utilities. Water Res. 2011, 45, 3590–3598. [Google Scholar] [CrossRef]
- Staggemeier, R.; Heck, T.M.S.; Demoliner, M.; Ritzel, R.G.F.; Röhnelt, N.M.S.; Girardi, V.; Venker, C.A.; Spilki, F.R. Enteric viruses and adenovirus diversity in waters from 2016 Olympic venues. Sci. Total Environ. 2017, 586, 304–312. [Google Scholar] [CrossRef]
- Steyer, A.; Gutiérrez-Aguirre, I.; Rački, N.; Glaser, S.B.; Humar, B.B.; Stražar, M.; Škrjanc, I.; Poljšak-Prijatelj, M.; Ravnikar, M.; Rupnik, M. The Detection Rate of Enteric Viruses and Clostridium difficile in a Waste Water Treatment Plant Effluent. Food Environ. Virol. 2015, 7, 164–172. [Google Scholar] [CrossRef]
- Stokdyk, J.P.; Firnstahl, A.D.; Walsh, J.F.; Spencer, S.K.; de Lambert, J.R.; Anderson, A.C.; Rezania, L.W.; Kieke, B.A., Jr.; Borchardt, M.A. Viral, bacterial, and protozoan pathogens and fecal markers in wells supplying groundwater to public water systems in Minnesota, USA. Water Res. 2020, 178, 115814. [Google Scholar] [CrossRef]
- Taylor, M.B.; Cox, N.; Vrey, M.A.; Grabow, W.O.K. The occurrence of hepatitis A and astroviruses in selected river and dam waters in South Africa. Water Res. 2001, 35, 2653–2660. [Google Scholar] [CrossRef]
- Thongprachum, A.; Fujimoto, T.; Takanashi, S.; Saito, H.; Okitsu, S.; Shimizu, H.; Khamrin, P.; Maneekarn, N.; Hayakawa, S.; Ushijima, H. Detection of nineteen enteric viruses in raw sewage in Japan. Infect. Genet. Evol. 2018, 63, 17–23. [Google Scholar] [CrossRef]
- Truchado, P.; Garre, A.; Gil, M.I.; Simón-Andreu, P.J.; Sánchez, G.; Allende, A. Monitoring of human enteric virus and coliphages throughout water reuse system of wastewater treatment plants to irrigation endpoint of leafy greens. Sci. Total Environ. 2021, 782, 146837. [Google Scholar] [CrossRef]
- Tucker, J.D.; Grasso-Knight, G. Environmental hepatitis A detection and awareness on a Native American reservation. Bull. World Health Organ. 2000, 78, 948. [Google Scholar]
- Vaidya, S.R.; Chitambar, S.D.; Arankalle, V.A. Polymerase chain reaction-based prevalence of hepatitis A, hepatitis E and TT viruses in sewage from an endemic area. J. Hepatol. 2002, 37, 131–136. [Google Scholar] [CrossRef]
- Van Zyl, W.B.; Zhou, N.A.; Wolfaardt, M.; Matsapola, P.N.; Ngwana, F.B.; Symonds, E.M.; Fagnant-Sperati, C.S.; Shirai, J.H.; Kossik, A.L.; Beck, N.K.; et al. Detection of potentially pathogenic enteric viruses in environmental samples from Kenya using the bag-mediated filtration system. Water Sci. Technol. Water Supply 2019, 19, 1668–1676. [Google Scholar] [CrossRef]
- Vantarakis, A.; Papapetropoulou, M. Detection of enteroviruses, adenoviruses and Hepatitis A viruses in raw sewage and treated effluents by nested-PCR. Water Air Soil Pollut. 1999, 114, 85–93. [Google Scholar] [CrossRef]
- Vantarakis, A.C.; Tsibouxi, A.; Venieri, D.; Komninou, G.; Athanassiadou, A.; Papapetropoulou, M. Evaluation of microbiological quality of coastal waters in Greece. J. Water Health 2005, 3, 371–380. [Google Scholar] [CrossRef]
- Venter, J.M.; van Heerden, J.; Vivier, J.C.; Grabow, W.O.; Taylor, M.B. Hepatitis A virus in surface water in South Africa: What are the risks? J. Water Health 2007, 5, 229–240. [Google Scholar] [CrossRef]
- Verma, V.; Arankalle, V.A. Hepatitis e virus-based evaluation of a virion concentration method and detection of enteric viruses in environmental samples by multiplex nested RT-PCR. J. Appl. Microbiol. 2010, 108, 1630–1641. [Google Scholar] [CrossRef] [PubMed]
- Villar, L.M.; De Paula, V.S.; Diniz-Mendes, L.; Guimarães, F.R.; Ferreira, F.F.M.; Shubo, T.C.; Miagostovich, M.P.; Lampe, E.; Gaspar, A.M.C. Molecular detection of hepatitis A virus in urban sewage in Rio de Janeiro, Brazil. Lett. Appl. Microbiol. 2007, 45, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Neyvaldt, J.; Enache, L.; Sikora, P.; Mattsson, A.; Johansson, A.; Lindh, M.; Bergstedt, O.; Norder, H. Variations among Viruses in Influent Water and Effluent Water at a Wastewater Plant over One Year as Assessed by Quantitative PCR and Metagenomics. Appl. Environ. Microbiol. 2020, 86, e02073-20. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Sikora, P.; Rutgersson, C.; Lindh, M.; Brodin, T.; Björlenius, B.; Larsson, D.G.J.; Norder, H. Differential removal of human pathogenic viruses from sewage by conventional and ozone treatments. Int. J. Hyg. Environ. Health 2018, 221, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Shin, H.; Jung, S.; Yeo, D.; Park, H.; Shin, S.; Seo, D.J.; Park, K.H.; Choi, C. Effects of Weather and Environmental Factors on the Seasonal Prevalence of Foodborne Viruses in Irrigation Waters in Gyeonggi Province, Korea. Microorganisms 2020, 8, 1224. [Google Scholar] [CrossRef]
- Wong, M.V.; Hashsham, S.A.; Gulari, E.; Rouillard, J.M.; Aw, T.G.; Rose, J.B. Detection and characterization of human pathogenic viruses circulating in community wastewater using multi target microarrays and polymerase chain reaction. J. Water Health 2013, 11, 659–670. [Google Scholar] [CrossRef]
- Yanez, L.A.; Lucero, N.S.; Barril, P.A.; Díaz, M.D.P.; Tenaglia, M.M.; Spinsanti, L.I.; Nates, S.V.; Isa, M.B.; Ré, V.E. Evidence of Hepatitis A virus circulation in central Argentina: Seroprevalence and environmental surveillance. J. Clin. Virol. 2014, 59, 38–43. [Google Scholar] [CrossRef]
- Jacobsen, K.H.; Koopman, J.S. The effects of socioeconomic development on worldwide hepatitis A virus seroprevalence patterns. Int. J. Epidemiol. 2005, 34, 600–609. [Google Scholar] [CrossRef]
- Nelson, N.P. Chapter 4: Travel-Related Infectious Diseases, Hepatitis A. In CDC Yellow Book; Oxford Universitiy Press: Oxford, UK, 2018; Volume 8, p. a031716. [Google Scholar]
- Costafreda, M.I.; Bosch, A.; Pintó, R.M. Development, evaluation, and standardization of a real-time TaqMan reverse transcription-PCR assay for quantification of hepatitis A virus in clinical and shellfish samples. Appl. Environ. Microbiol. 2006, 72, 3846–3855. [Google Scholar] [CrossRef]
- Carrillo-Santisteve, P.; Tavoschi, L.; Severi, E.; Bonfigli, S.; Edelstein, M.; Byström, E.; Lopalco, P. Seroprevalence and susceptibility to hepatitis A in the European Union and European Economic Area: A systematic review. Lancet. Infect. Dis. 2017, 17, e306–e319. [Google Scholar] [CrossRef]
- Desbois, D.; Couturier, E.; Mackiewicz, V.; Graube, A.; Letort, M.J.; Dussaix, E.; Roque-Afonso, A.M. Epidemiology and genetic characterization of hepatitis A virus genotype IIA. J. Clin. Microbiol. 2010, 48, 3306–3315. [Google Scholar] [CrossRef]
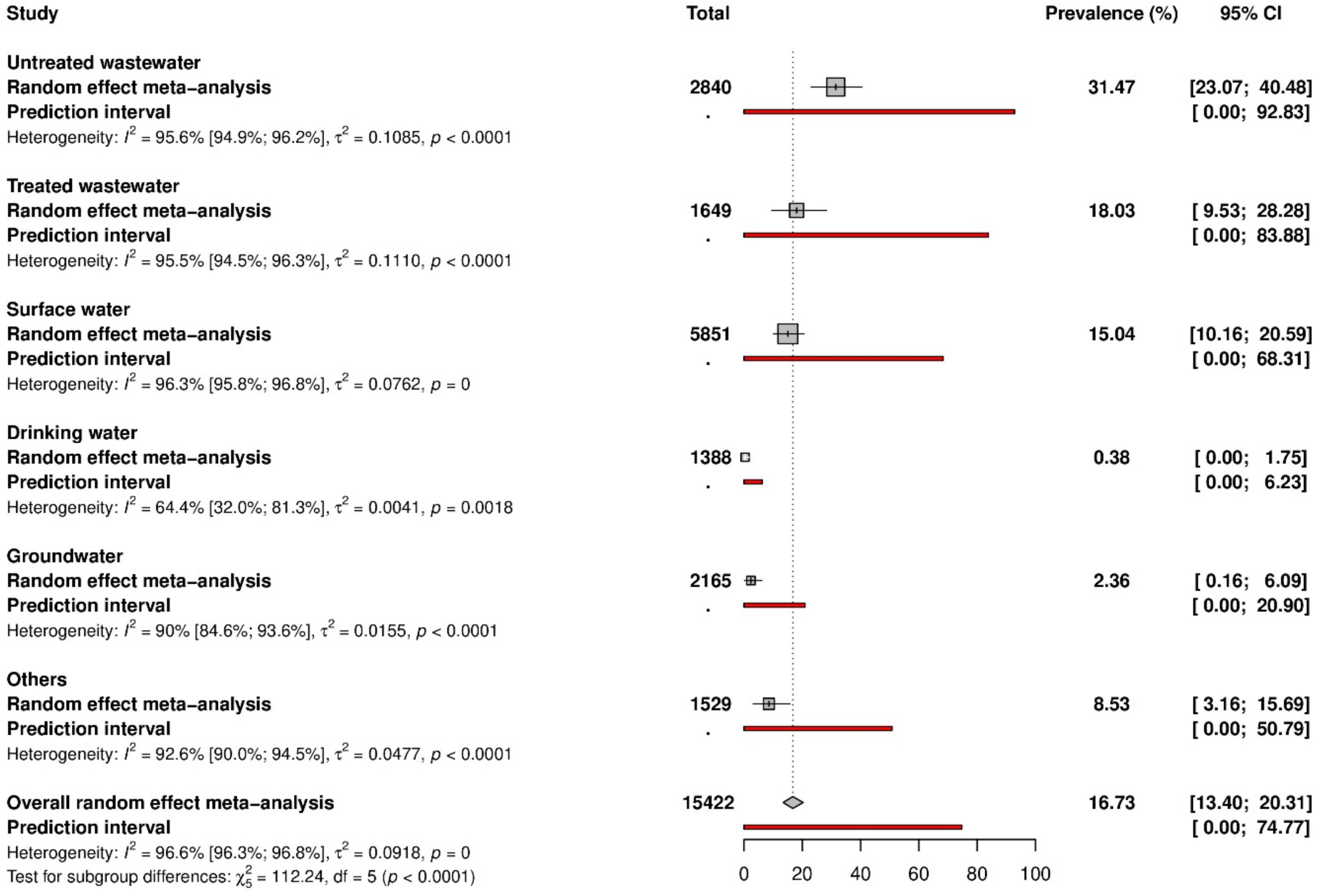
| Water Matrix | Prevalence (%) [95%CI] | 95% Prediction Interval | N° of Studies | N° of Samples | H [95%CI] | I2 [95%CI] | p Heterogeneity |
|---|---|---|---|---|---|---|---|
| Untreated wastewater | |||||||
| Overall | 31.5 [23.1–40.5] | [0–92.8] | 56 | 2840 | 4.8 [4.4–5.2] | 95.6 [94.9–96.2] | <0.001 |
| Low risk of bias | 8.5 [0.9–21] | [0–65] | 5 | 314 | 2.7 [1.8–4] | 86 [69.5–93.6] | <0.001 |
| Process control | 36.8 [18–57.8] | [0–100] | 15 | 1097 | 6.8 [6–7.7] | 97.9 [97.3–98.3] | <0.001 |
| Treated wastewater | |||||||
| Overall | 18.0 [9.5–28.3] | [0–83.9] | 34 | 1649 | 4.7 [4.3–5.2] | 95.5 [94.5–96.3] | <0.001 |
| Low risk of bias | 16.0 [0–53] | [0–100] | 3 | 92 | 3.5 [2.2–5.6] | 91.8 [79.3–96.8] | <0.001 |
| Process control | 18.9 [3.4–41.9] | [0–99.1] | 11 | 826 | 7 [6.1–8.1] | 98 [97.3–98.5] | <0.001 |
| Surface water | |||||||
| Overall | 15.0 [10.2–20.6] | [0–68.3] | 66 | 5851 | 5.2 [4.9–5.6] | 96.3 [95.8–96.8] | <0.001 |
| Low risk of bias | 28.6 [0–85.7] | [0–100] | 3 | 73 | 5.1 [3.5–7.5] | 96.2 [91.9–98.2] | <0.001 |
| Process control | 12.1 [5.9–19.9] | [0–68.4] | 33 | 2620 | 5.2 [4.7–5.7] | 96.3 [95.5–96.9] | <0.001 |
| Drinking water | |||||||
| Overall | 0.4 [0–1.8] | [0–6.2] | 11 | 1388 | 1.7 [1.2–2.3] | 64.4 [32–81.3] | 0.002 |
| Process control | 1.5 [0–17.7] | NA | 2 | 380 | 2.8 [1.4–5.6] | 87.4 [50.7–96.8] | 0.005 |
| Groundwater | |||||||
| Overall | 2.4 [0.2–6.1] | [0–20.9] | 12 | 2165 | 3.2 [2.5–3.9] | 90 [84.6–93.6] | <0.001 |
| Low risk of bias | 0.4 [0–3.3] | NA | 2 | 1158 | 3 [1.5–5.8] | 88.9 [58.1–97.1] | 0.003 |
| Process control | 1.2 [0.1–3.2] | [0–8.2] | 5 | 618 | 1.3 [1–2.1] | 38.9 [0–77.4] | 0.162 |
| Others | |||||||
| Overall | 8.5 [3.2–15.7] | [0–50.8] | 21 | 1529 | 3.7 [3.2–4.3] | 92.6 [90–94.5] | <0.001 |
| Process control | 0.4 [0–2.7] | [0–7.4] | 8 | 640 | 1.2 [1–1.9] | 34.2 [0–70.9] | 0.155 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takuissu, G.R.; Kenmoe, S.; Ebogo-Belobo, J.T.; Kengne-Ndé, C.; Mbaga, D.S.; Bowo-Ngandji, A.; Ndzie Ondigui, J.L.; Kenfack-Momo, R.; Tchatchouang, S.; Kenfack-Zanguim, J.; et al. Occurrence of Hepatitis A Virus in Water Matrices: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2023, 20, 1054. https://doi.org/10.3390/ijerph20021054
Takuissu GR, Kenmoe S, Ebogo-Belobo JT, Kengne-Ndé C, Mbaga DS, Bowo-Ngandji A, Ndzie Ondigui JL, Kenfack-Momo R, Tchatchouang S, Kenfack-Zanguim J, et al. Occurrence of Hepatitis A Virus in Water Matrices: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2023; 20(2):1054. https://doi.org/10.3390/ijerph20021054
Chicago/Turabian StyleTakuissu, Guy Roussel, Sebastien Kenmoe, Jean Thierry Ebogo-Belobo, Cyprien Kengne-Ndé, Donatien Serge Mbaga, Arnol Bowo-Ngandji, Juliette Laure Ndzie Ondigui, Raoul Kenfack-Momo, Serges Tchatchouang, Josiane Kenfack-Zanguim, and et al. 2023. "Occurrence of Hepatitis A Virus in Water Matrices: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 20, no. 2: 1054. https://doi.org/10.3390/ijerph20021054
APA StyleTakuissu, G. R., Kenmoe, S., Ebogo-Belobo, J. T., Kengne-Ndé, C., Mbaga, D. S., Bowo-Ngandji, A., Ndzie Ondigui, J. L., Kenfack-Momo, R., Tchatchouang, S., Kenfack-Zanguim, J., Lontuo Fogang, R., Zeuko’o Menkem, E., Kame-Ngasse, G. I., Magoudjou-Pekam, J. N., Veneri, C., Mancini, P., Bonanno Ferraro, G., Iaconelli, M., Orlandi, L., ... La Rosa, G. (2023). Occurrence of Hepatitis A Virus in Water Matrices: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 20(2), 1054. https://doi.org/10.3390/ijerph20021054







