Effects of Music-Based Interventions on Motor and Non-Motor Symptoms in Patients with Parkinson’s Disease: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Search Strategy
2.2. Eligibility Criteria
- Participants: Patients of any age, gender, or disease stage who have been diagnosed with PD. However, studies including atypical PD and other diagnoses (e.g., Alzheimer’s Disease) were excluded.
- Interventions and comparisons: The inclusion criteria for the interventions in the experimental group were any music-based intervention or other interventions combined with music, but the studies which used music as a secondary medium were excluded. Other modalities of intervention including dance and movement therapy using music as a secondary medium of treatment were also excluded. Studies which examined the immediate effect of interventions were also excluded. The comparison groups included those who received non-music interventions, usual care, or no intervention. The intervention format (individual/group), setting (clinic/hospital/home), duration, and frequency were not limited.
- Outcomes: There was no limit to the dependent variable that music-based interventions affected. Therefore, the outcomes of interest were both motor (e.g., gait, mobility) and non-motor symptoms (e.g., executive function and quality of life). All outcomes were measured using validated tools including the timed up-and-go test (TUG), the trail making test (TMT) and the Stroop color and word test (SCWT).
- Study design: The study design included in this review were randomized controlled trials (RCTs) and controlled clinical trials (CCTs). This study was limited to articles published in English and peer-reviewed journals. The following types of research were ruled out: master’s theses, doctoral dissertations, conference papers, case studies, descriptive studies, literature research, and any review studies.
2.3. Study Selection and Data Extraction
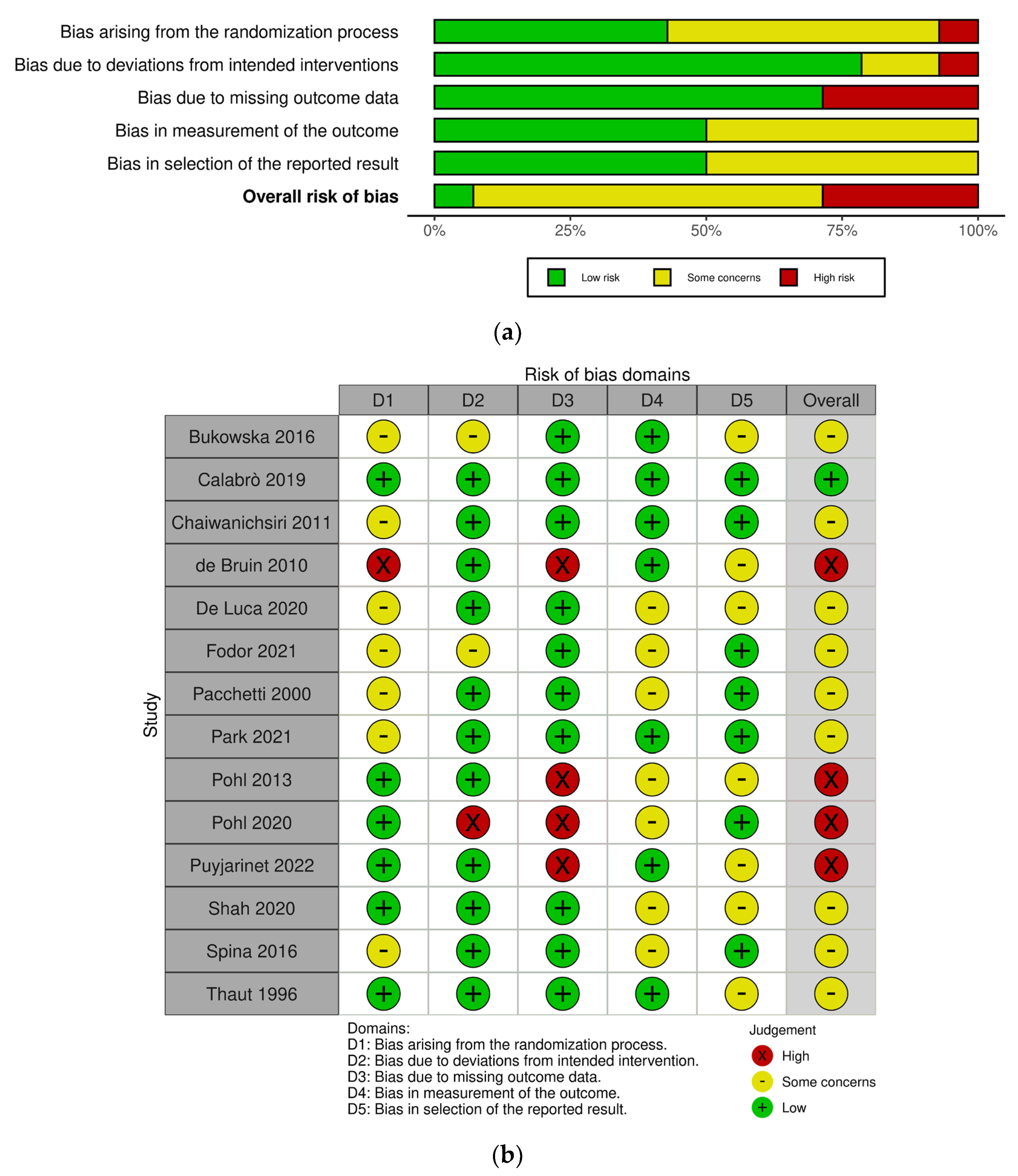
2.4. Risk-of-Bias Assessment
2.5. Data Synthesis
3. Results
3.1. Study Selection
3.2. Summary of Risk of Bias
3.3. Study Characteristics
3.3.1. Characteristics of Participants
3.3.2. Characteristics of Interventions
3.4. Effects of the Interventions on Motor Outcomes
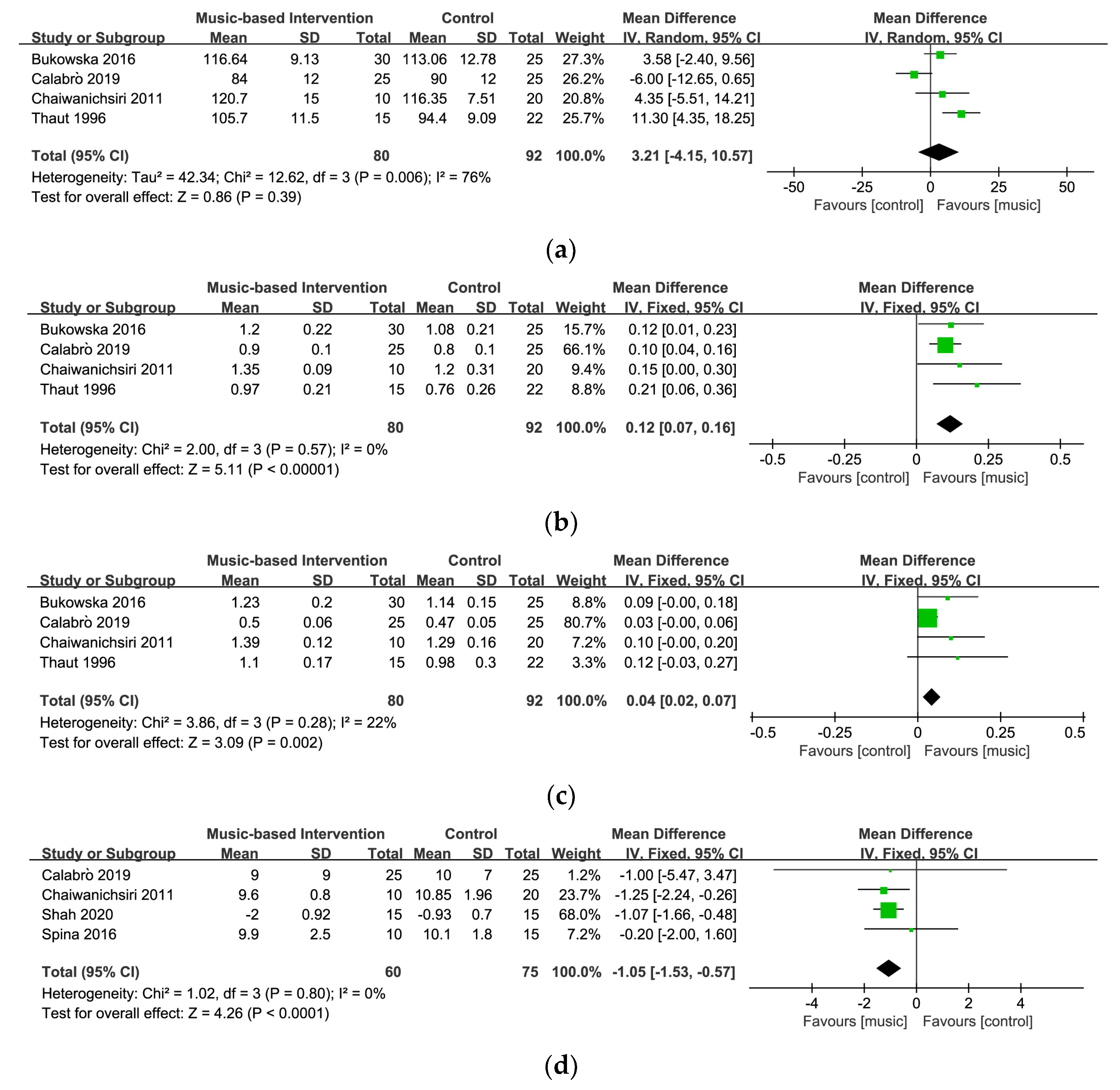
3.4.1. Cadence
3.4.2. Velocity
3.4.3. Stride Length
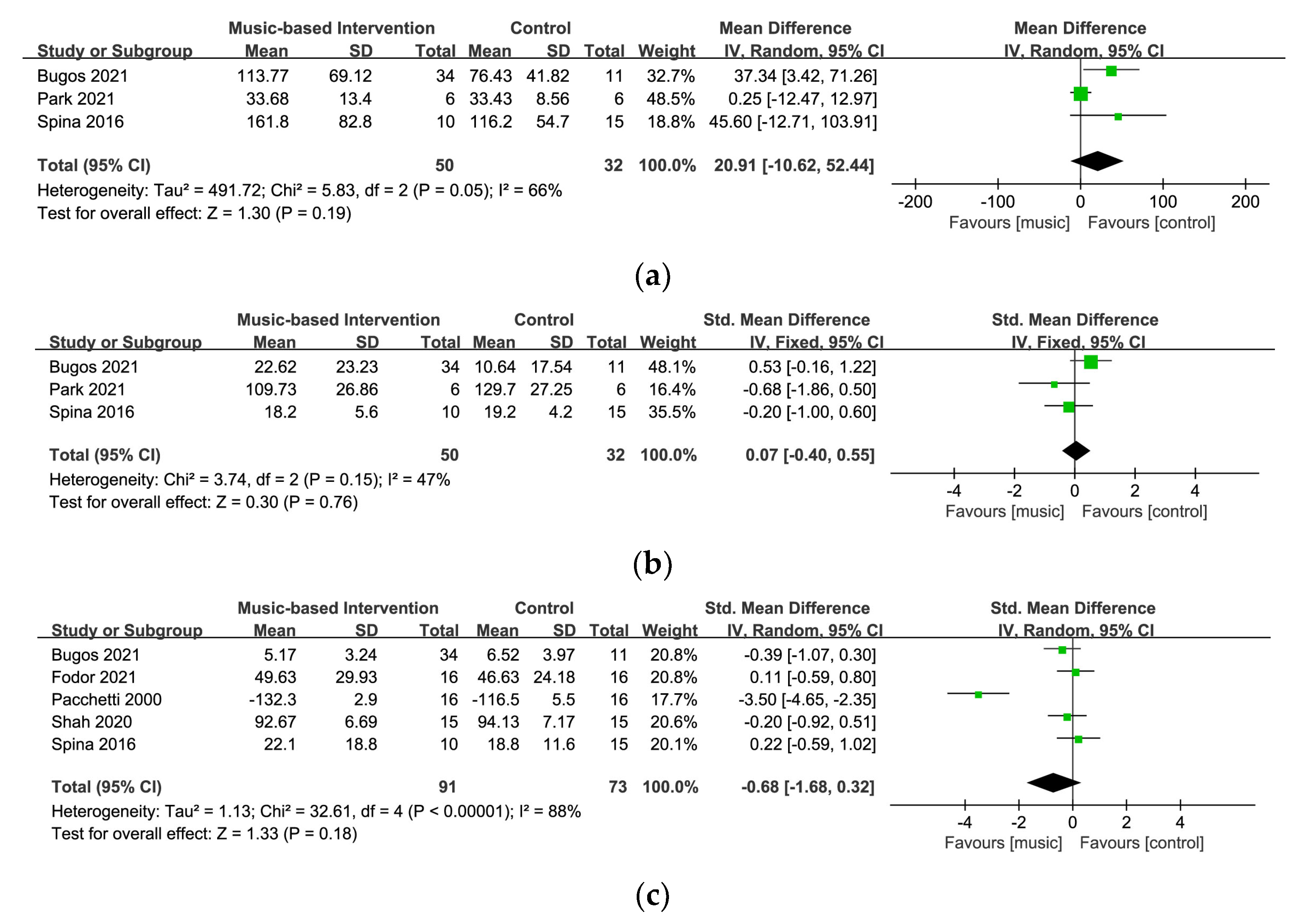
3.4.4. Mobility
3.5. Effects of the Intervention on Non-Motor Outcomes
3.5.1. Cognitive Flexibility
3.5.2. Inhibition
3.5.3. Quality of Life
3.6. Sensitivity Analysis
3.7. Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Lau, L.M.; Breteler, M.M. Epidemiology of Parkinson disease. Lancet Neurol. 2006, 5, 525–535. [Google Scholar] [CrossRef]
- Freeman, J.S.; Cody, F.W.J.; Schady, W. The influence of external timing cues upon the rhythm of voluntary movements in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1993, 56, 1078–1084. [Google Scholar] [CrossRef] [PubMed]
- Jankovic, J. Parkinson’s disease: Clinical features and diagnosis. J. Neurol. Neurosurg. Psychiatry 2008, 79, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.E.; Menz, H.B.; McGinley, J.L.; Watts, J.J.; Huxham, F.E.; Murphy, A.T.; Danoudis, M.; Lansek, R. A randomized controlled trial to reduce falls and improve mobility in people with Parkinson’s disease. Neurorehabil. Neural. Repair 2015, 29, 777–785. [Google Scholar] [CrossRef]
- Bloem, B.R.; de Vries, N.M.; Ebersbach, G. Nonpharmacological treatments for patient with Parkinson’s disease. Mov. Disord. 2015, 30, 1504–1520. [Google Scholar] [CrossRef]
- Magee, M.; Copland, D.A.; Vogel, A.P. Motor speech and non-motor language endophenotypes of Parkinson’s disease. Expert. Rev. Neurother. 2019, 19, 1191–1200. [Google Scholar] [CrossRef] [PubMed]
- Wirdefeldt, K.; Adami, H.O.; Cole, P.; Trichopoulos, D.; Mandel, J. Epidemiology and etiology of Parkinson’s disease: A review of the evidence. Eur. J. Epidemiol. 2011, 26, S1–S58. [Google Scholar] [CrossRef]
- Titova, N.; Chaudhuri, K.R. Non-motor Parkinson disease: New concepts and personalized management. Med. J. Aust. 2018, 208, 404–409. [Google Scholar] [CrossRef]
- De Dreu, M.J.; van der Wilk, A.S.; Poppe, E.; Kwakkel, G.; van Wegen, E.E. Rehabilitation, exercise therapy and music in patients with Parkinson’s disease: A meta-analysis of the effects of music-based movement therapy on walking ability, balance and quality of life. Park. Relat. Disord. 2012, 18, S114–S119. [Google Scholar] [CrossRef]
- Borgohain, R.; Kandadai, R.M.; Jabeen, A.; Kabbikannan, M.A. Nonmotor outcomes in Parkinson’s disease: Is deep brain stimulation better than dopamine replacement therapy? Ther. Adv. Neurol. Disord. 2012, 5, 23–41. [Google Scholar] [CrossRef]
- Borovac, J.A. Side effects of a dopamine agonist therapy for Parkinson’s disease: A mini-review of clinical pharmacology. Yale J. Boil. Med. 2016, 89, 37–47. [Google Scholar]
- Müller, T. Current and investigational non-dopaminergic agents for management of motor symptoms (including motor complications) in Parkinson’s disease. Expert Opin. Pharmacother. 2017, 18, 1457–1465. [Google Scholar] [CrossRef] [PubMed]
- Jenner, P. Treatment of the later stages of Parkinson’s disease—Pharmacological approaches now and in the future. Transl. Neurodegener. 2015, 4, 3. [Google Scholar] [CrossRef]
- Zafar, M.; Bozzorg, A.; Hackney, M.E. Adapted Tango improves aspects of participation in older adults versus individuals with Parkinson’s disease. Disabil. Rehabil. 2016, 39, 2294–2301. [Google Scholar] [CrossRef] [PubMed]
- Pacchetti, C.; Aglieri, R.; Mancini, F.; Martignoni, E.; Nappi, G. Active music therapy and Parkinson’s disease: Methods. Funct. Neurol. 1998, 13, 57–67. [Google Scholar] [PubMed]
- Pacchetti, C.; Mancini, F.; Aglieri, R.; Fundaro, C.; Martignoni, E.; Nappi, G. Active music therapy in Parkinson’s disease: An integrative method for motor and emotional rehabilitation. Psychosom. Med. 2000, 62, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Amengual, J.L.; Rojo, N.; Veciana de Las Heras, M.; Marco-Pallarés, J.; Grau-Sánchez, J.; Schneider, S.; Vaquero, L.; Juncadella, M.; Montero, J.; Mohammadi, B.; et al. Sensorimotor plasticity after music-supported therapy in chronic stroke patients revealed by transcranial magnetic stimulation. PLoS ONE 2013, 8, e61883. [Google Scholar] [CrossRef]
- Tsoi, K.K.F.; Chan, J.Y.C.; Ng, Y.M.; Lee, M.M.Y.; Kwok, T.C.Y.; Wong, S.Y.S. Receptive music therapy is more effective than interactive music therapy to relieve behavioral and psychological symptoms of dementia: A systematic review and meta-analysis. J. Am. Med. Dir. Assoc. 2018, 19, 568–576. [Google Scholar] [CrossRef]
- Phol, P.; Didzar, N.; Hallert, E. The Ronnie Gardiner rhythm and music method -a feasibility study in Parkinson’s disease. Disabil. Rehabil. 2013, 35, 2197–2204. [Google Scholar]
- Thaut, M.H.; Rice, R.R.; Braun Janzen, T.; Hurt-Thaut, C.P.; McIntosh, G.C. Rhythmic auditory stimulation for reduction of falls in Parkinson’s disease: A randomized controlled study. Clin. Rehabil. 2019, 33, 34–43. [Google Scholar] [CrossRef]
- Song, J.H.; Yang, J.J.; Li, Y.; Zhang, G.B.; Wang, P.Q. Rhythmic auditory stimulation with treadmill training on motor function in people with Parkinson’s disease. J. Apoplexy Nerv. Dis. 2018, 35, 242–244. [Google Scholar]
- Li, P. Analysis of the effect of rhythmic auditory stimulation combined with exercise training on the motor function of patients with Parkinson’s disease. Modern. Diag. Treat. 2020, 31, 1129–1131. [Google Scholar]
- Harro, C.C.; Shoemaker, M.J.; Frey, O.J.; Gamble, A.C.; Harring, K.B.; Karl, K.L.; McDonald, J.D.; Murray, C.J.; Tomassi, E.M.; Van Dyke, J.M.; et al. The effects of speed-dependent treadmill training and rhythmic auditory-cued overground walking on gait function and fall risk in individuals with idiopathic Parkinson’s disease: A randomized controlled trial. NeuroRehabilitation 2014, 34, 557–572. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.J.; Liu, J.; Liu, S. Influence of rhythmic auditory attention stimulation combined with intensive muscle strength training on lower limb function and balance function in elderly patients with Parkinson’s disease. Chin. Nurs. Res. 2018, 32, 229–233. [Google Scholar]
- Bella, S.D.; Benoit, C.E.; Farrugia, N.; Schwartze, M.; Kotz, S.A. Effects of musically cued gait training in Parkinson’s disease: Beyond a motor benefit. Ann. N. Y. Acad. Sci. 2015, 1337, 77–85. [Google Scholar] [CrossRef]
- Calabrò, R.S.; Naro, A.; Filoni, S.; Pullia, M.; Billeri, L.; Tomasello, P.; Portaro, S.; Di Lorenzo, G.; Tomaino, C.; Bramanti, P. Walking to your right music: A randomized controlled trial on the novel use of treadmill plus music in Parkinson’s disease. J. Neuroeng. Rehabil. 2019, 16, 68. [Google Scholar] [CrossRef] [PubMed]
- Bukowska, A.A.; Krężałek, P.; Mirek, E.; Bujas, P.; Marchewka, A. Neurologic music therapy training for mobility and stability rehabilitation with Parkinson’s disease—A pilot study. Front. Hum. Neurosci. 2016, 9, 710. [Google Scholar] [CrossRef]
- Chomiak, T.; Watts, A.; Meyer, N.; Pereira, F.V.; Bin, H. A training approach to improve stepping automaticity while dual-tasking in Parkinson’s disease: A prospective pilot study. Medicine 2017, 96, 5. [Google Scholar] [CrossRef]
- Bugos, J.A.; Lesiuk, T.; Nathani, S. Piano training enhances Stroop performance and musical self-efficacy in older adults with Parkinson’s disease. Psychol. Music. 2021, 49, 615–630. [Google Scholar] [CrossRef]
- Spina, E.; Barone, P.; Mosca, L.L.; Lombardi, A.; Longo, K.; Lavarone, A.; Amboni, M. Music therapy for motor and nonmotor symptoms of Parkinson’s disease: A prospective, randomized, controlled, single-blinded study. J. Am. Geriatr. Soc. 2016, 64, e36–e38. [Google Scholar] [CrossRef]
- Stegemöller, E.L.; Hurt, T.R.; O’Connor, M.C.; Camp, R.D.; Green, C.W.; Pattee, J.; Williams, E.K. Experiences of persons with parkinson’s disease engaged in group therapeutic singing. J. Music. Ther. 2017, 54, 405–431. [Google Scholar] [CrossRef] [PubMed]
- Stegemöller, E.L.; Zaman, A.; Shelley, M.; Patel, B.; Kouzi, A.E.; Shirtcliff, E.A. The effects of group therapeutic singing on cortisol and motor symptoms in persons with Parkinson’s disease. Front. Hum. Neurosci. 2021, 15, 703382. [Google Scholar] [CrossRef] [PubMed]
- Tamplin, J.; Morris, M.E.; Marigliani, C.; Baker, F.A.; Vogel, A.P. ParkinSong: A Controlled Trial of Singing-Based Therapy for Parkinson’s Disease. Neurorehabilit. Neural Repair. 2019, 33, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Tamplin, J.; Vogel, A.; Marigliani, C.; Baker, F.; Morris, M. A controlled trial of ParkinSong singing groups to improve communication and wellbeing in Parkinson’s disease. Mov. Disord. 2018, 33 (Suppl. 2), S138. [Google Scholar]
- Matthews, R.; Purdy, S.; Tippett, L. Acoustic, respiratory, cognitive and wellbeing comparisons of two groups of people with Parkinson’s disease participating in voice and choral singing group therapy (VCST) versus music appreciation activity [abstract]. Mov. Disord. 2018, 33 (Suppl. 2), S406. [Google Scholar]
- Fodor, D.M.; Breda, X.M.; Valean, D.; Marta, M.M.; Perju-Dumbrava, L. Music as add-on therapy in the rehabilitation program of parkinson’s disease patients—A Romanian pilot study. Brain Sci. 2021, 11, 569. [Google Scholar] [CrossRef] [PubMed]
- Pantelyat, A.; Syres, C.; Reichwein, S.; Willis, A. DRUIM-PD: The use of a drum circle to improve the symptoms and signs of Parkinson’s disease (PD). Mov. Disord. Clin. Pract. 2016, 3, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Pohl, P.; Wressle, E.; Lundin, F.; Enthoven, P.; Dizdar, N. Group-based music intervention in Parkinson’s disease—Findings from a mixed-methods study. Clini. Rehabil. 2020, 34, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Shah, V.; Yeole, U.; Purandare, S.S.; Vishwakarma, K.R. Effect of physical therapy with music therapy on gait, balance, and quality of life in Parkinson’s disease. Indian J. Public Health Res. Dev. 2020, 11, 1064–1069. [Google Scholar]
- Tamplin, J.; Baker, F.A.; Morris, M.E.; Marigliani, C.; Noffs, G.; Vogel, A.P. ParkinSong: Outcomes of a 12-month controlled trial of therapeutic singing groups in Parkinson’s disease. J. Parkinson’s Dis. 2020, 10, 1217–1230. [Google Scholar] [CrossRef] [PubMed]
- Raglio, A. Music therapy interventions in Parkinson’s disease: The state-of-the-art. Front. Neurol. 2015, 6, 1865. [Google Scholar] [CrossRef] [PubMed]
- Machado Sotomayor, M.J.; Arufe-Giráldez, V.; Ruíz-Rico, G.; Navarro-Patón, R. Music therapy and Parkinson’s disease: A systematic review from 2015–2020. Int. J. Environ. Res. Public Health 2021, 18, 11618. [Google Scholar] [CrossRef]
- Ghai, S.; Ghai, I.; Schmitz, G.; Effenberg, A.O. Effect of rhythmic auditory cueing on parkinsonian gait: A systematic review and meta-analysis. Sci. Rep. 2018, 8, 506. [Google Scholar] [CrossRef]
- Wang, L.; Peng, J.; Ou-Yang, J.; Gan, L.; Zeng, S.; Wang, H.; Zuo, G.; Qiu, L. Effects of rhythmic auditory stimulation on gait and motor function in Parkinson’s disease: A systematic review and meta-analysis of clinical randomized controlled studies. Front. Neurol. 2022, 13, 818559. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, D.; Ye, D.; Li, H.; Chen, F. Can music-based movement therapy improve motor dysfunction in patients with Parkinson’s disease? Systematic review and meta-analysis. Neurol. Sci. 2017, 38, 1629–1636. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhou, R.; Wei, W.; Luan, R.; Li, K. Effects of music-based movement therapy on motor function, balance, gait, mental health, and quality of life for patients with parkinson’s disease: A systematic review and meta-analysis. Clin. Rehabil. 2021, 35, 937–951. [Google Scholar] [CrossRef] [PubMed]
- Barnish, M.S.; Barran, S.M. A systematic review of active group-based dance, singing, music therapy and theatrical interventions for quality of life, functional communication, speech, motor function and cognitive status in people with Parkinson’s disease. BMC Neurol. 2020, 20, 371. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.; Brennan, S.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef]
- Da Costa Santos, C.M.; de Mattos Pimenta, C.A.; Nobre, M.R. The PICO strategy for the research question construction and evidence search. Rev. Lat. Am. Enferm. 2007, 15, 508–511. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomized trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.C.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomized studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Savović, J.; Page, M.J.; Elbers, R.G.; Sterne, J.A.C. Chapter 8: Assessing risk of bias in a randomized trial. In Cochrane Handbook for Systematic Reviews of Interventions; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Version 6.2 (Updated February 2021); John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2021. [Google Scholar]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- De Luca, R.; Latella, D.; Maggio, M.G.; Leonardi, S.; Sorbera, C.; Di Lorenzo, G.; Balletta, T.; Cannavò, A.; Naro, A.; Impellizzeri, F.; et al. Do patients with PD benefit from music- assisted therapy plus treadmill-based gait training? An exploratory study focused on behavioral outcomes. Int. J. Neurosci. 2020, 130, 933–940. [Google Scholar] [CrossRef]
- Thaut, M.H.; McIntosh, G.C.; Rice, R.R.; Miller, R.A.; Rathbun, J.; Brault, J.M. Rhythmic auditory stimulation in gait training for Parkinson’s disease patients. Mov. Disord. 1996, 11, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Chaiwanichsiri, D.; Wangmo, W.; Kitisomprayoonkul, W.; Bhidayasiri, R. Treadmill training with music cueing: A new approach for Parkinson’ gait facilitation. Asian Biomed. 2011, 5, 649–654. [Google Scholar]
- Park, J.; Kim, S. Dual-task-based drum playing with rhythmic cueing on motor and attention control in patients with Parkinson’s disease: A preliminary randomized study. Int. J. Environ. Res. Public Health 2021, 18, 10095. [Google Scholar] [CrossRef] [PubMed]
- De Bruin, N.; Doan, J.B.; Turnbull, G.; Suchowersky, O.; Bonfield, S.; Hu, B.; Brown, L.A. Walking with music is a safe and viable tool for gait training in Parkinson’s Disease: The effect of a 13-week feasibility study on single and dual task walking. Parkinsons Dis. 2010, 2010, 483530. [Google Scholar] [CrossRef]
- Puyjarinet, F.; Bégel, V.; Geny, C.; Geny, C.; Driss, V.; Cuartero, M.C.; Pinto, S.; De Cock, V.C.; Dalla Bella, S. At-home training with a rhythmic video game for improving orofacial, manual, and gait abilities in Parkinson’s Disease: A pilot study. Front. Neurosci. 2022, 16, 874032. [Google Scholar] [CrossRef]
- Mollinedo, I.; Cancela, J.M. Evaluation of the psychometric properties and clinical applications of the Timed Up and Go Test in Parkinson disease: A systematic review. J. Exerc. Rehabil. 2020, 16, 302–312. [Google Scholar] [CrossRef]
- Mollà-Casanova, S.; Inglés, M.; Muñoz-Gómez, E.; Aguilar-Rodríguez, M.; Sempere-Rubio, N.; Serra-Añó, P.; Pedrero-Sánchez, J.; López-Pascual, J. Impact of Parkinson’s disease on functional mobility at different stages. Front. Aging. Neurosci. 2022, 14, 935841. [Google Scholar] [CrossRef]
- Koshimori, Y.; Thaut, M.H. Future perspectives on neural mechanisms underlying rhythm and music based neurorehabilitation in Parkinson’s disease. Ageing Res. Rev. 2018, 47, 133–139. [Google Scholar] [CrossRef]
- Thaut, M.H.; McIntosh, K.W.; McIntosh, G.C.; Hoemberg, V. Auditory rhythmicity enhances movement and speech motor control in patients with Parkinson’s disease. Funct. Neurol. 2001, 16, 163–172. [Google Scholar] [PubMed]
- McIntosh, G.M.; Brown, S.H.; Rice, R.R. Rhythmic auditory-motor facilitation of gait patterns in patients with Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1997, 62, 22–26. [Google Scholar] [CrossRef]
- Rubinstein, T.C.; Giladi, N.; Hausdorff, J.M. The power of cueing to circumvent dopamine deficits: A review of physical therapy treatment of gait disturbances in Parkinson’s disease. Mov. Disord. 2002, 17, 1148–1160. [Google Scholar] [CrossRef]
- Thaut, M.H. The future of music in therapy and medicine. Ann. N. Y. Acad. Sci. 2005, 1060, 303–308. [Google Scholar] [CrossRef]
- De Dreu, M.; Kwakkel, G.; van Wegen, E. Rhythmic auditory stimulation (RAS) in gait rehabilitation for patients with Parkinson’s disease: A research perspective. In Handbook of Neurologic Music Therapy; Thaut, M.H., Hoemberg, V., Eds.; Oxford University Press: Oxford, UK, 2014; pp. 69–93. [Google Scholar]
- Zanardi, A.P.J.; da Silva, E.S.; Costa, R.R.; Passos-Monteiro, E.; dos Santos, I.O.; Kruel, L.F.M.; Peyré-Tartaruga, L.A. Gait parameters of Parkinson’s disease compared with healthy controls: A systematic review and meta-analysis. Sci. Rep. 2021, 11, 752. [Google Scholar] [CrossRef] [PubMed]
- Lesuik, T.; Bugos, J.A.; Murakami, B. A rationale for music training to enhance executive functions in Parkinson’s disease: An Overview of the problem. Healthcare 2018, 6, 35. [Google Scholar] [CrossRef]
- Bradt, J. Randomized controlled trials in music therapy: Guidelines for design and implementation. J. Music Ther. 2012, 49, 120–149. [Google Scholar] [CrossRef] [PubMed]


| First Author | Country | Study | Sample | Age | Gender | Disease Duration (Years) | UPDRS | Hoehn and Yahr Score | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (Year) | Types | Ex | Co | Ex | Co | Ex | Co | Ex | Co | Ex | Co | Ex | Co | |
| Bugos (2021) [29] | USA | CCT | 34 | 11 | 65.79 ± 8.38 | 67.55 ± 7.29 | M11/F18 | M6/F5 | N/A | N/A | N/A | N/A | 1 or 2 a | 1 or 2 b |
| Bukowska (2016) [27] | Poland | RCT | 30 | 25 | 63.4 ± 10.61 | 63.44 ± 9.67 | M15/F15 | M15/F10 | N/A | N/A | N/A | N/A | 2 or 3 | 2 or 3 |
| Calabrò (2019) [26] | Italy | RCT | 25 | 25 | 70 ± 8 | 73 ± 8 | M11/F9 | M14/F6 | 10 ± 3 | 9.3 ± 3 | 29 ± 3 | 31 ± 5 | 3 ± 1 | 3 ± 1 |
| Chaiwanichsiri (2011) [56] | Thailand | RCT | 10 | 10/ 10 | 67.1 ± 4.0 | 67.9 ± 6.3/ 68.6 ± 5.2 | N/A | N/A | 3.7 ± 4.1 | 7.4 ± 3.4 4.4 ± 2.3 | N/A | N/A | 2, 2.5, 3 | 2, 2.5, 3 |
| Chomiak (2017) [28] | Canada | CCT | 5 | 6 | 70.8 ± 5.6 | 69.0 ± 5.7 | M5/F0 | M4/F2 | 15.4 ± 5.4 | 11.2 ± 5.0 | 18.2 ± 6.4 (Part III) | 20.3 ± 4.7 (Part III) | 2.5 ± 0.50 | 2.7 ± 0.41 |
| De Luca (2020) [54] | Italy | RCT | 20 | 20 | 63.2 ± 8.4 | 66.5 ± 6.2 | M10/F10 | M7/F13 | N/A | N/A | N/A | N/A | 1.5 ± 0.53 | 1.7 ± 0.59 |
| Fodor (2021) [36] | Romania | RCT | 16 | 16 | 67.1 ± 5.9 | 65.6 ± 5.5 | M4/F12 | M6/F10 | N/A | N/A | N/A | N/A | 1~3 | 1~3 |
| Pacchetti (2000) [16] | Italy | RCT | 16 | 16 | 62.5 ± 5 | 63.2 ± 5 | M12/F4 | M11/F5 | 4.8 ± 3 | 5.2 ± 2 | 40.2 ± 7.7 (Part III) | 40.7 ± 7 (Part III) | 2 or 3 | 2 or 3 |
| Pantelyat (2016) [37] | USA | CCT | 8 | 10 | 72 ± 8 | 71 ± 7 | F% 63 | F% 60 | N/A | N/A | 22 ± 6 (MDS Part III) | 25 ± 11 (MDS Part III) | 2.2 ± 0.46 | 2.2 ± 0.24 |
| Park (2021) [57] | Korea | RCT | 6 | 6 | 61.6 ± 4.9 | 63.1 ± 10.1 | N/A | N/A | 5.6 ± 3.1 | 4.8 ± 1.4 | N/A | N/A | 2 or 3 | 2 or 3 |
| Shah (2020) [39] | India | RCT | 15 | 15 | 66.98 ± 5.4 | 68.78 ± 4.76 | M11/F4 | M12/F3 | 2 ± 1.51 | 3 ± 1.58 | N/A | N/A | 2, 2.5, 3 (modified) | 2, 2.5, 3 (modified) |
| Spina (2016) [30] | Italy | RCT | 10 | 15 | 68.3 ± 8.1 | 61.9 ± 6.8 | N/A | N/A | 79.2 ± 39.2 (months) | 80 ± 53.4 (months) | 18.5 ± 8.1 (MDS Part III) | 17.7 ± 8.3 (MDS Part III) | N/A | N/A |
| Thaut (1996) [55] | U.S.A. | RCT | 15 | 11/ 11 | 69 ± 8 | 74 ± 3/ 71 ± 8 | M10/F5 | M8/F3 M8/F3 | 7.2 ± 4 | 5.4 ± 3/ 8.5 ± 4 | N/A | N/A | 2, 2.5, 3 (modified) | 2, 2.5, 3 (modified) |
| First Author (Year) | Contents | Intervention | Control | Outcomes | Evaluation Methods | |
|---|---|---|---|---|---|---|
| Intensity (Duration; Frequency; Time) | Type | |||||
| Bugos (2021) [29] | Intensive piano training | 10 days; daily each morning; 3 h/session | Group | No treatment | ⑦⑨⑩ | PASAT, cued color–word Stroop, digit coding, symbol search, TMT part B, D-KEFS: verbal fluency subtest; PDQ-39; GSE |
| Bukowska (2016) [27] | The combination of three NMT sensorimotor techniques: TIMP, PSE, and RAS for daily life activities, balance, pre-gait, and gait pattern | 4 weeks; 4 times/week; 45 min/session | Individual | Stay active and perform daily life activities b/t measures | ①② | Computerized dynamic posturography CQ stab; optoelectric 3D movement analysis, system BTS Smart |
| Calabrò (2019) [26] | Treadmill gait training using GaitTrainer3 with RAS | 8 weeks; 5 times/week; 20 min/session | Individual | Treadmill gait training using GaitTrainer3 without RAS (same intensity) | ①②⑩ | UPDRS, BBS, TUG; A single wireless inertial sensor (GSensor, BTS Bioengineering), 10 m walking test, FGA, gait quality index; Tinetti FES |
| Chaiwanichsiri (2011) [56] | Treadmill with music cue 3 days/week and home walking program 3 days/week | 8 weeks; 6 days/week; 30 min/session | Individual | Control 1. Treadmill training 3 days/week and home walking program 3 days/week Control 2. Home walking program 6 days/week | ①② | TUG, single leg stance test; expanded TUG, six-minute walk test |
| Chomiak (2017) [28] | In-home music-contingent stepping-in-place training | 4 weeks; minimum of 3/week; 10 to 20 min/session | Individual | In-home auditory-contingent stepping-in-place training using a podcast (auditory feedback) (same intensity) | ①⑧⑩ | FOG-Q; MoCA; FES-International |
| De Luca (2020) [54] | Gait training via a treadmill and RAS | 8 weeks; 3 times/week; about 30 min/session | Individual | Traditional over-ground gait training (same intensity) | ①②⑧ ⑨⑩ | FIM: motor, TUG; 10 m walking test; FIM: cognitive; PGWBI; Brief COPE |
| Fodor (2021) [36] | A multimodal rehabilitation program centered on physical therapy combined with listening to music | 14 days; daily; 2.5 h | Group | A multimodal rehabilitation program centered on physical therapy (same intensity) | ⑨ | PDQ-39 |
| Pacchetti (2000) [16] | Music therapy, choral singing, voice exercise, rhythmic and free body movements, and improvisation | 13 weeks; weekly; 2 h/session | Group | PT, stretching exercises, specific motor tasks, and balance and gait training (intensity: 13 weeks; weekly; 1.5 h/session) | ①⑨⑩ | UPDRS-ADL, UPDRS-MS; PDQL; happiness measure |
| Pantelyat (2016) [37] | West African drum circle | 6 weeks; twice-weekly; 45–60 min/session | Group | No treatment | ⑧⑨⑩ | MoCA; PDQ-39; GDS-15 |
| Park (2021) [57] | Drum playing with rhythmic cueing (DPRC intervention) | 12 weeks; weekly; 50 min/session | Group | Regular program: gait rehabilitation and speech therapy | ①③⑦ | Nine-hole peg test; K-TMT-e: part A; K-TMT-e: part B, Korean Stroop test |
| Shah (2020) [39] | PT with MT: standing and marching activity, gait training, tap dance, and balance training | 6 weeks; 4 times per week; 60 min/session | Group | Conventional PT | ①②⑨ | TUG; dynamic gait index; PDQ-39 |
| Spina (2016) [30] | Active music therapy—production of music, singing, and dancing | 24 weeks; once a week; 90 min/session | Group | No treatment | ①③④⑤⑥⑦⑨ | New FOG-Q, TUG, MDS-UPDRS; TMT part A; Rey immediate recall, Rey delayed recall; clock drawing test; names denomination, verbal denomination; FAB, phonemic verbal fluency, Raven test, TMT part B, Stroop test; PDQ-39 |
| Thaut (1996) [55] | RAS; home-based training | 3 weeks; daily; 30 min/session | Individual | Control 1. Self (internally)-paced group: same exercise program but without the aid of RAS Control 2. No-training group: normal daily activities | ①② | EMG; computerized foot-switch recording system |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.; Ko, B. Effects of Music-Based Interventions on Motor and Non-Motor Symptoms in Patients with Parkinson’s Disease: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2023, 20, 1046. https://doi.org/10.3390/ijerph20021046
Lee H, Ko B. Effects of Music-Based Interventions on Motor and Non-Motor Symptoms in Patients with Parkinson’s Disease: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2023; 20(2):1046. https://doi.org/10.3390/ijerph20021046
Chicago/Turabian StyleLee, Hyunjung, and Bumsuk Ko. 2023. "Effects of Music-Based Interventions on Motor and Non-Motor Symptoms in Patients with Parkinson’s Disease: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 20, no. 2: 1046. https://doi.org/10.3390/ijerph20021046
APA StyleLee, H., & Ko, B. (2023). Effects of Music-Based Interventions on Motor and Non-Motor Symptoms in Patients with Parkinson’s Disease: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 20(2), 1046. https://doi.org/10.3390/ijerph20021046